Abstract
Transfer of information about food source characteristics within insect societies is essential to colony-foraging success. The food odor communicated within honeybee hives has been shown to be important for food source exploitation. When successful foragers return to the nest and transfer the collected nectar to hive mates through mouth-to-mouth contacts (trophallaxis), potential recruits receiving these samples learn the food odor by associative learning. The food then becomes rapidly distributed among colony members, which is mainly a consequence of the numerous trophallaxes between hive-mates of all ages during food processing. We tested whether the distribution of food among hive mates causes a propagation of olfactory information within the hive. Using the proboscis extension response paradigm, we show that large proportions of bees of the age groups representing the main worker castes, 4 to 9-day-old bees (nurse-aged bees), 12 to 16-day-old bees (food processor-aged bees), and actual foragers (about 17+ day old bees) associatively learn the food odor in the course of processing food that has been collected by only a few foragers. Results further suggest that the information is shared more or less equally between bees of the three age groups. This shows that olfactory information about the flower species exploited by foragers is distributed within the entire colony and is acquired by bees of all age groups, which may influence many behaviors inside and outside the hive.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Information acquisition and transfer among individuals in an insect society is crucial for adaptive colony-level responses to relevant ecological parameters in a variable world; the better informed a colony, the better it can adjust its behavior to meet the demands of its environment (Seeley 1995; Dall et al. 2005). Olfactory information transferred within the hive can help experienced foragers exploit known food sources (von Frisch 1923; Johnson 1967; Reinhard et al. 2004) and new recruits locate food sources advertised by dances (von Frisch 1923, 1967; Wenner et al. 1969). Recruits can perceive both the food odor clinging on the returning foragers’ bodies (von Frisch 1923, 1967) and that contained in the nectar transferred during mouth-to-mouth contacts (trophallaxis) (von Frisch 1923, 1967; Farina et al. 2005). The latter has been shown to be more important for recruitment (von Frisch 1923, 1967). Successful incoming foragers unload their nectar through several trophallactic contacts of different durations to their hive mates (von Frisch 1923; Farina and Wainselboim 2001). Receivers are either food processors (main unloading contacts) (Seeley et al. 1996, Pírez and Farina 2004) or other foragers (short contacts) (De Marco and Farina 2003). During short contacts, small samples of food can be transferred, thereby allowing the receivers to taste and/or smell the nectar (Farina and Wainselboim 2001, 2005). However, only recently, it has been shown that receiving foragers learn the odor/food association during trophallaxis within the hive by directly testing recruits in the laboratory using the proboscis extension response (PER) assay (Farina et al. 2005).
Trophallaxis plays a key role in the organization of food processing of many species of social insects as it links the various subtasks in a partitioned task, i.e., the sequential stages in the handling and processing of material (Wilson 1971; Ratnieks and Anderson 1999). Food processing involves honeybees belonging to the three main worker castes: nurses, food processors, and foragers (Nixon and Ribbands 1952; Seeley 1995), which are bees of three different age groups (Lindauer 1952; Seeley 1982). While foraging is normally performed by bees older than about 20 days, food receiving and processing (distribution and storage) is performed by middle-aged bees (between 11–20 days old) (Rösch 1925; Seeley 1982). Nurse bees are young bees, normally 3–11 days old, that are largely responsible for preparing nutrients from pollen and distribute the nutritionally valuable proteins produced by their hypopharyngeal glands, nectar, and honey to larvae and practically all hive mates (Crailsheim 1998; DeGrandi-Hoffman and Hagler 2000). The distribution of food within the hive has been shown to be rapid and extensive, i.e., a large proportion of all hive bees comes into contact with the nectar within a few hours (Nixon and Ribbands 1952; DeGrandi-Hoffman and Hagler 2000). The circulation of food within the hive via trophallaxis not only serves nutritional purposes, but is also considered to have informational importance, even though there exists little direct evidence (Crailsheim 1998). It is not yet known, for example, whether young hive bees, e.g., bees performing nurse tasks, also learn the odor of the incoming nectar. However, information cues present in the circulating nectar may provide the colony with global information, which means that the information leads to a response in most colony members and provides them with information about the current foraging opportunities (Pankiw et al. 2004).
In a recent attempt to test olfactory experiences made within the hive, it was shown that the PER assay offers a powerful method to test associations established between odor and sugar present in the liquid solution transferred amongst colony members (Farina et al. 2005). Bees reflexively extend their proboscis to drink solution when the antennae are touched with sucrose solution (unconditioned stimulus; US). In classical conditioning in the laboratory, an odor as a conditioned stimulus (CS) is paired with the US, which causes the odor itself to become capable of eliciting proboscis extension as a conditioned response (Kuwabara 1957; Bitterman et al. 1983). During trophallaxis, the solution transferred functions as unconditioned stimulus (US), while the food odor functions as CS (Gil and De Marco 2005).
We hypothesized that the distribution of food bearing a floral scent also distributes olfactory information among workers of different age groups performing different tasks within their caste. The nectar distribution amongst workers during the course of food processing could result in many olfactory conditioning events within the hive. To test our hypothesis, we fed foragers with scented solution and then captured four groups of bees belonging to three different age groups to test their proboscis extension response on the first presentation of the solution odor and a novel odor in the laboratory (proboscis extension response; PER). The four groups were (1) 4–9 days old bees, normally performing nurse tasks at this age, (2) 12–16 days old bees, normally performing food-processor tasks at this age, (3) a group of randomly captured foragers, and (4) foragers recruited to the scented food source.
In this study, the PER assay allowed us for the first time to (1) measure the propagation of information about a food source characteristic (floral scent) within the different age groups, (2) measure the speed of information acquisition day by day during our experimental periods, and (3) compare the proportions of bees that learned the food odor between the age groups.
Materials and methods
Study site and animals
The experiment was performed at the end of the nectar flow season (March–April 2005) at the experimental field of the University of Buenos Aires. We used two two-frame observation hives (H1 and H2) containing a colony of about 3,800 European honeybees (Apis mellifera ligustica) each (Table 1). Colonies had a queen, brood, and reserves.
A group of bees was trained to collect 1 M unscented sucrose solution at an artificial feeder located 160 m from the hive. Bees were marked individually and a number of about five to 10 foragers was maintained throughout the experiment by training new foragers whenever necessary.
Experimental procedure
At the beginning of the experiment, combs with preemerging brood from several hives housed in the apiary of the University of Buenos Aires were obtained and maintained in an incubator (temperature: 32°C, relative humidity 55%). On the day of emergence, bees were color-marked and introduced to H1 and H2. Honeybee colonies readily accept newly emerged bees (Breed et al. 2004). Using a different color every 2 days made it possible to determine the age of the marked bees in H1 and H2.
The experiment consisted of two parts. The trained and marked foragers (that were trained to the 1 M unscented sucrose solution) of the experimental hive (EH) collected scented solution during 6 days (part 1) and 7 days (part 2) for 3 h each day at a feeder offering 2 M sucrose solution (about 70 μl/min flow rate). The solutions were scented with 50 μl pure odor per liter. The control hive (CH) had no access to solution offered at an artificial feeder. All the nectar that entered the CH was collected by foragers foraging at natural food sources. In part 1, H1 was used as experimental hive (EH) and H2 as control hive (CH). In part 2, the situation was reversed. Information about the odors used, population sizes of both colonies, number of collecting bees per day and amount of solution collected per day can be found in Table 1.
Bee capture
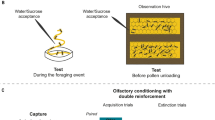
In the experiment, three groups of bees (foragers, 4–9 days old bees and 12–16 days old bees) were captured each from EH and CH. An additional group of bees (recruits) was captured from the EH (bees recruited to the experimental feeder are very likely to have interacted with our numbered foragers, von Frisch 1923, 1967). During part 1, bees were captured on days 0, 2, 4, and 6 from the EH and on days 3 and 5 from the CH. During part 2, bees were captured on days 0, 1, 3, 5, and 7 in the EH and on days 2, 4, and 6 in the CH. This sampling schedule was used to reduce time effects on learning performance that could cause differences in learning performance between EH bees and CH bees. The interval between part 1 and part 2 was 6 days.
During the 3 h when the numbered bees collected scented solution, recruits arriving at the feeder (EH only) were captured with plastic tubes on the feeding platform before they touched the scented solution, otherwise they were killed with alcohol. Captured bees were fed a drop of 2 M unscented sucrose solution.
About 60 min after the 3-h sampling period, we started to capture bees of the following three groups.
-
–
Foragers. A group of random foragers was captured with plastic tubes while feeding from a small plate (about 8 cm Ø), filled with 2 M unscented sucrose solution at a distance of some centimeter from the entrance. Foragers leaving the hive and passing the plate will normally accept the offered solution at this time of the year. If they had color marks, their age was determined (about 10–20% of the captured bees had marks). There was no difference in forager age between the two colonies (ANOVA: F 1,69=0.49, P=0.826, mean: 23.9±5.97 and 24.2±5.03 days old, H1 and H2, respectively).
-
–
Four to nine days old bees (nurse aged bees) and 12–16 days old bees (food processor aged bees). Bees were identified by their color mark and captured from the hive. The windows of the observation hive were movable acrylic walls (movable horizontally). In the middle of these walls were rectangular holes (the height of the holes almost equaled the height of the walls). The rectangles contained acrylic slides that were movable vertically (up and down). The acrylic slides contained an aperture (3.5 cm width) that allowed to capture the bees with a plastic tube. This device allowed us to scan the whole comb area.
None of the captured bees had contact with the scented solution at the feeding station and associations could therefore only have been established within the hive. Captured bees of all groups were harnessed in plastic tubes so that they could move freely their mouthparts and antennae (Bitterman et al. 1983). They were fed 1.8 M unscented sucrose solution ad libitum and kept overnight in an incubator (25°C, 55% relative humidity, darkness). This guaranteed that bees had similar motivation levels when they were tested the next morning.
PER testing
The morning after harnessing, we tested the bees once for their responses to the solution odor (LIO in part 1, PHE in part 2) and once for their spontaneous responses to a novel odor, which was 2-octanol (2-OCT) in both parts. The interval between the two presentations lasted about 15 min. A device that delivered a continuous air flow was used for odorant application. We used 4 μl of pure odor impregnated on a filter paper (about 30×3 mm), which was placed inside a syringe. When odor was presented, the air flow produced by our setup passed through this syringe. Only bees that showed the unconditioned response (the reflexive extension of the proboscis after applying a 1.8 M sucrose solution to the antennae) and that did not respond to the mechanical air flow stimulus were used (this was tested when the harnessed bees were put in the continuous air flow before presenting the odors. Less than 1% of all tested bees did respond to the air flow). Test trials lasted for 46 s and consisted of 20 s of air flow, 6 s of odor (CS), and 20 s of air flow. The three odors are natural components of flower odors (Knudsen et al. 1993) and were obtained from Sigma-Aldrich, Steinheim, Germany.
Statistical analyses
G-tests were used to compare PER frequencies between groups. We used the Williams’ correction for adjustment of G-values, except in cases were we had 0 responses. In these cases, the more conservative Yates’ correction was used (see Sokal and Rohlf 1981 for discussion of the two corrections). For statistical analysis, we pooled the data of control hive (CH) bees for all four groups in part 1 and separately in part 2. Forager age comparison and the relation between percentage of PER and amount of scented solution (general linear model, GLM, with odor as a fixed factor and amount of scented solution as a covariate to explain the percentage of PER) that entered the colony, were done using parametric statistics as assumptions were met (Grafen and Hails 2002). The descriptive statistics are given in mean±SE.
Results
Proboscis extension response frequencies for the solution odor (bees responding only to the solution odor) increased over experimental time and reached values between 30% for 4–9 days old bees (nurse-aged bees) and 56% for recruits on the last day of part 1 (day 6 using linalool; Fig. 1a, Table 1), and between 30% for foragers and 43% for 12–16 days old bees (processor-aged bees) on the last day of part 2 (day 7 using phenylacetaldehyde; Fig. 2a, Table 1). PER frequencies of control bees for the solution odor were low during both parts (Figs. 1b and 2b). Neither in part 1 (comparison between day 3 and 5: 4–9 days old bees: G-test, G adj<0.001, df=1, P=0.99; 12–16 days old bees: G-test, G adj=0.027, df=1, P=0.87; foragers: G-test, G adj=1.61, df=1, P=0.20; Fig. 1b) nor in part 2 (comparison between days 2, 4, and 6: 4–9 days old bees: G-test, G adj=0.34, df=2, P=0.84; 12–16 days old bees: G-test, G adj=0.58, df=2, P=0.75; foragers: G-test, G adj=1.15, df=2, P=0.56; Fig. 2b), did we find significant differences in PER frequencies for the solution odor (bees responding to the solution odor only). There is also no reason why the response frequencies for the solution odor should change during the course of the experiment in CH bees, as these bees never had access to the offered scented solution. For further statistical analysis, we pooled the data obtained on days 3 and 5 in part 1 and the data obtained on days 2, 4, and 6 in part 2 for the three groups of bees (4–9 days old bees, 12–16 days old bees, and foragers) separately. The differences between PER frequencies of experimental bees at the end of the experimental periods of part 1 and 2 and control bees were wide (between 23.3% for 4–9 day old bees (nurse-aged bees) and 56% for recruits (compared to foragers of the control group) in part 1 and between 26.2% for foragers and 39.3% for 12–16 days old bees (food processor-aged bees) in part 2; Figs. 1a and 2a). Therefore, we compared the PER frequencies of bees captured in the experimental hive on day 6 (i.e., at the end of conditioning) in part 1 with the PER frequencies of bees captured from the CH (days 3 and 5) in part 1 and on day 7 in part 2 with the PER frequencies of bees captured from the CH in part 2 (days 2, 4, and 6). We found significant differences in all cases, except for 4–9 days old bees (nurse-aged bees) during part 1 (Figs. 1a and 2a; Table 2). Unfortunately, the number of bees belonging to this category was very low, making the detection of significant differences difficult. (Table 2). Neither in part 1, nor in part 2, did we find significant differences in PER frequencies between the four groups (Table 3). To visualize the increase in PER frequencies in the entire colony and its relation to the amount of scented solution entering the hives, we summed the data of the different bee groups in Fig. 3a,b. There is a significant positive relationship between the amount of scented solution that entered the hive and the percentage of PER, but no difference between the odors (GLM: amount solution × percentage of PER: F 1,9=42.7, p=0.001; odor × percentage of PER: F 1,9=5.36, p=0.06).
Proboscis extension response (PER) frequencies for 4–9 days old bees, 12–16 days old bees, foragers, and recruits (EH) (the percentage of bees that extended the proboscis on the first presentation of the odor in the laboratory) for the solution odor (filled bars), the novel odor (crossed bars), or both odors (hatched bars). a EH: the PER frequencies measured on days 0, 2, 4, and 6 after starting to feed with scented solution (hive 1, H1, was used as experimental hive, EH). Linalool, LIO, was used as solution odor. b CH: the PER frequencies measured on days 3 and 5 (H2 was used as CH). Linalool, LIO, was used as solution odor. Number of tested bees above bars. Insert figure shows the quantity of scented sucrose solution, in milliliter (emptied circles), that entered the EH every day and the number of trained foragers (filled circles)
PER frequencies for 4–9 days old bees, 12–16 days old bees, foragers, and recruits (EH). a EH: the PER frequencies measured on days 0, 1, 3, 5, and 7 after starting to feed with scented solution (H2 was used as EH). Phenylacetaldehyde, PHE, was used as solution odor. b CH: the PER frequencies measured on days 2, 4, and 6 after starting to feed with scented solution (H1 was used as CH)
PER frequencies for bees of all four bee groups at different days (bars) and the amount of scented solution (ml) that entered the colonies until the given day (curve). a the PER frequencies measured on days 0, 2, 4, and 6 during part 1; Linalool, LIO, was used as solution odor. b the PER frequencies measured on days 0, 1, 3, 5, and 7 during part 2; phenylacetaldehyde, PHE, was used as solution odor
Discussion
Within a honeybee hive, transfer of information between bees allows the colony to adaptively respond to its changing environment. Previous studies suggested that the food odor transferred within the hive is an important information cue for foraging decisions of honeybees (von Frisch 1923, 1967; Wenner et al. 1969). However, extensive olfactory information propagation within honeybee colonies in the course of the processing food has never been reported. Our experiment allowed measuring the patterns of information propagation within the colony over time and comparing information acquisition between the different worker castes. The high PER frequencies for the solution odor, but not for the novel odor, observed in all tested groups and the low PER frequencies for the solution odor for CH bees demonstrates that propagation of olfactory information within the hive occurs. Results, therefore, clearly show that also very young hive bees (4–9 days old) having the age of nurse bees learn the odor of the incoming solution. The pattern of information propagation was different in the two colonies. While in part 1, PER frequencies on day 2 were above 25% for all groups, PER frequencies on day 3 in part 2 remained below 20%. The propagation pattern will be modified by many variables, e.g., hive population, the number of employed foragers, and the amount of solution entering the hive. A decrease of about 20% in colony size during part 2, combined with the low amount of solution that entered the EH in part 2 until day 3 and the increased nectar influx from day 4 on may explain to some extent the different pattern found in the second part and it shows a correlation between the speed of information propagation and the amount of solution entering the hive, and consequently the number of collecting foragers. The social structure of a colony (i.e., the age and the number of bees performing tasks) is also likely to influence the pattern of propagation of olfactory information. However, we found no difference in forager age between the two colonies and have no evidence of a difference in the social structure between the two colonies. Variables such as food odor characteristics (Smith 1991), previous olfactory experience of bees (Sandoz et al. 2000), sugar response thresholds (Scheiner et al. 2004), time of year (Ray and Ferneyhough 1997), and genotype (Bhagavan et al. 1994) are related to learning performance of honeybees and are therefore likely to influence the pattern of information propagation in yet unknown ways.
It is interesting that our results do not show consistent differences in PER frequencies between the four groups of bees. In neither part 1 nor part 2 did we find significant differences in PER frequencies between the different age groups. It seems that the information is shared more or less equally between bees of the three age groups. Differences in olfactory learning performances between bees of different age are also likely to influence the propagation of information within the hive. Earlier studies suggest that the acquisition process is similar in bees older than 8 days (Bhagavan et al. 1994; Laloi et al. 2001; Ichikawa and Sasaki 2003). For bees younger than 8 days, previous studies provided contradictory results (Ray and Ferneyhough 1997, Laloi et al. 2001; Ichikawa and Sasaki 2003). While some studies showed age-dependent learning abilities (Ray and Ferneyhough 1997; Ichikawa and Sasaki 2003), others did not (Bhagavan et al. 1994; Laloi et al. 2001).
Olfactory associative learning relies on the discrete pairing of odor and reward and cannot be accomplished by passive exposure to the scent in the hive, which has been shown to have inhibitory effects on associative learning (Menzel et al. 1993; Gerber et al. 1996; Sandoz et al. 2000). It is likely that the information propagation relied mostly on trophallaxis, rather than on individual feeding from the honey cells because the season and the small amount of scented food gathered by the foragers prevented an accumulation of scented solution within the colony. Food offerings via trophallaxis is a common behavior of returning foragers (von Frisch 1923, 1967), and it is often seen in nectar processors after receiving the incoming food (Pírez and Farina 2004). However, stored honey may be an important source of olfactory information, functioning like an “odor library” of previous and present food sources (Free 1969).
Solution gathered by only 5–10 bees (about 1% of all foragers of our colonies, estimation after Seeley 1995) is likely to become mixed with other nectar samples during the course of food processing, which would cause different US and CS intensities experienced by bees of the different age groups involved at different stages of food processing. This affects the strength of the association made between odor and sucrose solution (Pelz et al. 1997; Gil and De Marco 2005) and therefore makes a simple relationship between number of trophallactic contacts and PER frequencies found in the three age groups unlikely. More studies are needed to unravel the food transmission pathways and functional identity of bees involved at the different stages of food processing.
It has recently been shown that the rate at which a receiver unloads nectar to another bee is positively correlated with the rate at which she received it from a food donor (Goyret and Farina 2005a). Therefore it could be that also quantitative aspects of trophallaxis are propagated during food distribution among honeybees. Propagation of chemosensory (olfactory and gustatory) information during the course of food processing is a highly economic information transfer that could influence many within-hive behaviors of young bees. It has been shown, for example, that experience with olfactory cues present in solution during trophallaxis would lead to the observed nonrandom occurrence of trophallaxis between foragers and receivers (Goyret and Farina 2005b). Transfer of information about floral scents also provides bees not involved in foraging with information about the food sources currently and recently exploited. It establishes associations in preforagers between nectar and floral odors they are likely to encounter some days later when they become foragers. In other words, bees leaving a hive for a foraging trip (independently of whether the bee is an experienced forager or a novice forager leaving for the first foraging trip) acquired information about foraging opportunities many times in the past during social interactions. This challenges the traditional scout-recruit concept, an important concept in honeybee foraging (Rösch 1925, Lindauer 1952; Seeley 1995; Anderson 2001; Biesmeijer and de Vries 2001; Dechaume-Moncharmont et al. 2005) in which foragers are divided into two groups: scouts that set out independently and recruits that use information provided by returning foragers (see also Biesmeijer and de Vries 2001 for a criticism). Even if foragers leave the hive without following dances that provide location information, they possess memorized information about food source characteristics acquired inside the nest. Therefore, the question seems not to be whether a bee should use information regarding foraging opportunities provided by other foragers (Dechaume-Moncharmont et al. 2005), but what kind of information she should use at any given moment. Foraging honeybees are capable of using more than one sensory cue to enhance their foraging efficiency in the field. While learned odors may help foragers to identify rewarding flowers when they come near to the food source, visual cues that are not learned inside the hive may attract their attention when they are still some distance away from the source (Giurfa et al. 1994, Galizia et al. 2004). Then, the notion of scout might be still valid when referred, for instance, to visual guidance. Adaptive decisions will then depend on the costs and benefits of using the different forms of information that are available (Dall et al. 2005).
Even if treated bees did not show a PER towards the solution odor, they may have experienced the combination of odor and solution previously (Menzel 1999) and as a consequence, learn the odor faster than bees without this experience (Farina et al. 2005). Olfactory learning by means of trophallaxis leads to long-term memory (Farina et al. 2005; Gil and De Marco 2005) and the behavior of bees and therefore the colony-foraging pattern could be affected for several days.
References
Anderson C (2001) The adaptive value of inactive foragers and the scout-recruit system in honey bee (Apis mellifera) colonies. Behav Ecol 12:111–119
Biesmeijer JC, de Vries H (2001) Exploitation of food sources by social insect colonies: a revision of the scout-recruit concept. Behav Ecol Sociobiol 49:89–99
Bitterman ME, Menzel R, Fietz A, Schaefer S (1983) Classical conditioning of proboscis extension in honeybees. J Comp Psychol 97:107–119
Bhagavan S, Bentrar S, Cobey S, Smith BH (1994) Effect of genotype but not of age or caste on olfactory learning performance in the honey bee, Apis mellifera. Anim Behav 48:1357–1369
Breed MD, Perry S, Bjostad LB (2004) Testing the blank slate hypothesis: why honey bee colonies accept young bees. Insectes Soc 51:12–16
Crailsheim K (1998) Trophallactic interactions in the adult honeybee (Apis mellifera L.). Apidologie 29:97–112
Dall SRX, Giraldeau LA, Olsson O, McNamara JM, Stephens DW (2005) Information and its use by animals in evolutionary ecology. TREE 20:187–193
Dechaume-Moncharmont FX, Dornhaus A, Houston AI, McNamara JM, Collins EJ, Franks NR (2005) The hidden cost of information in collective foraging. Proc R Soc Lond B 272:1689–1695
DeGrandi-Hoffman G, Hagler J (2000) The flow of incoming nectar through a honey bee (Apis mellifera L.) colony as revealed by a protein marker. Insectes Soc 47:302–306
De Marco RJ, Farina WM (2003) Trophallaxis in forager honeybees (Apis mellifera): resource uncertainty enhances begging contacts? J Comp Physiol A 189:125–134
Farina WM, Wainselboim AJ (2001) Thermographic recordings show that honeybees may receive nectar from foragers even during short trophallactic contacts. Insectes Soc 48:360–362
Farina WM, Wainselboim AJ (2005) Trophallaxis within the dancing context: a behavioral and thermographic analysis in honeybees. Apidologie 36:43–47
Farina WM, Grüter C, Diaz PC (2005) Social learning of floral odours inside the honeybee hive. Proc R Soc Lond B 272:1923–1928
Free JB (1969) Influence of odour of a honeybee colonies food stores on behaviour of its foragers. Nature 222:778
Galizia CG, Kunze J, Gumbert A, Borg-Karlson A, Sachse S, Markl C, Menzel R (2004) Relationship of visual and olfactory signal parameters in a food-deceptive flower mimicry system. Behav Ecol 16:159–168
Gerber B, Gerberzahn N, Hellstern F, Klein J, Kowalsky O, Wustenberg D, Menzel R (1996) Honey bees transfer olfactory memories established during flower visits to a proboscis extension paradigm in the laboratory. Anim Behav 52:1079–1085
Gil M, De Marco RJ (2005) Olfactory learning by means of trophallaxis in Apis mellifera. J Exp Biol 208:671–680
Giurfa M, Núñez J, Backhaus W (1994) Odour and colour information in the foraging choice behaviour of the honeybee. J Comp Physiol A 175:773–779
Goyret J, Farina WM (2005a) Trophallactic chains in honeybees: a quantitative approach of the nectar circulation amongst workers. Apidologie 36:595–600
Goyret J, Farina WM (2005b) Non-random nectar unloading interactions between foragers and their receivers in the honeybee hive. Naturwissenschaften 92:440–443
Grafen A, Hails R (2002) Modern statistics for the life sciences, Oxford University Press, Oxford
Johnson DL (1967) Communication among honey bees with field experience. Anim Behav 15:487–492
Knudsen JT, Tollsten L, Bergström LG (1993) Floral scents: a checklist of volatile compounds isolated by head-space techniques. Phytochemistry 33:253–280
Kuwabara M (1957) Bildung des bedingten Reflexes von Pavlovs Typus bei der Honigbiene, Apis mellifera. J Fac Sci Hokkaido Univ Ser VI Zool 13:458–464
Ichikawa N, Sasaki M (2003) Importance of social stimuli for the development of learning capability in honeybees. Appl Entomol Zool 38(2):203–209
Laloi D, Gallois M, Roger B, Pham-Delègue MH (2001) Changes with age in olfactory conditioning performance of worker honey bees (Apis mellifera). Apidologie 32:231–242
Lindauer M (1952) Ein Beitrag zur Frage der Arbeitsteilung im Bienenstaat. Z vgl Physiol 34:299–345
Menzel R (1999) Memory dynamics in the honeybee. J Comp Physiol A 185:323–340
Menzel R, Greggers U, Hammer M (1993) Functional organization of appetitive learning and memory in a generalist pollinator, the honey bee. In: Papaj D, Lewis AC (eds) Insect Learning: Ecological and Evolutionary Perspectives. Chapman & Hall, New York, pp. 79–125
Nixon HL, Ribbands CR (1952) Food transmission within the honeybee community. Proc R Soc Lond B 140:43–50
Pankiw T, Nelson M, Page RE, Fondrk MK (2004) The communal crop: modulation of sucrose response thresholds of pre-foraging honey bees with incoming nectar quality. Behav Ecol Sociobiol 55:286–292
Pelz C, Gerber B, Menzel R (1997) Odorant intensity as a determinant for olfactory conditioning in honeybees: roles in discrimination, overshadowing and memory consolidation. J Exp Biol 200:837–847
Pírez N, Farina WM (2004) Nectar-receiver behavior in relation to the reward rate experienced by foraging honeybees. Behav Ecol Sociobiol 55:574–582
Ratnieks FLW, Anderson C (1999) Task partitioning in insect societies. Insectes Soc 46:95–108
Ray S, Ferneyhough B (1997) Seasonal variation of proboscis extension reflex conditioning in the honey bee (Apis mellifera). J Apic Res 36:108–110
Reinhard J, Srinivasan MV, Zhang SW (2004) Olfaction: scent-triggered navigation in honeybees. Nature 427:411
Rösch GA (1925) Untersuchungen über die Arbeitsteilung im Bienenstaat. Z vgl Physiol 2:571–631
Sandoz JC, Laloi D, Odoux JF, Pham-Delegue MH (2000) Olfactory information transfer in the honeybee: compared efficiency of classical conditioning and early exposure. Anim Behav 59:1025–1034
Scheiner R, Page RE, Erber J (2004) Sucrose responsiveness and behavioural plasticity in honey bees (Apis mellifera). Apidologie 35:133–142
Seeley TD (1982) Adaptive significance of the age polyethism schedule in honeybee colonies. Behav Ecol Sociobiol 11:287–293
Seeley TD (1995) The wisdom of the hive: the social physiology of honey bee colonies. Harvard University Press, Cambridge, Massachusetts
Seeley TD,·Kühnholz S, A Weidenmüller (1996) The honey bee’s tremble dance stimulates additional bees to function as nectar receivers. Behav Ecol Sociobiol 39:419–427
Smith BH (1991) The olfactory memory of the honeybee Apis mellifera. J Exp Biol 161:367–382
Sokal R, Rohlf F (1981) Biometry, 2nd edn. Freeman, New York
von Frisch K (1923) Über die Sprache der Bienen. Zool JB Physiol 40:1–186
von Frisch K (1967) The dance language and orientation of honeybees. Harvard University Press, Cambridge, Massachusetts
Wenner AM, Wells PH, Johnson DL (1969) Honey bee recruitment to food sources: olfaction or language? Science 164:84–86
Wilson EO (1971) The insect societies. Harvard University Press, Cambridge, Massachusetts
Acknowledgements
We are deeply indebted to A. Arenas for much thoughtful advice and support during the experiment and I. Hamilton and two anonymous referees for helpful comments on earlier versions of the manuscript. We also thank A. Arenas, and H. Verna for technical assistance. This study was supported by funds from ANPCYT (01-12319), CONICET (02049) and University of Buenos Aires (X 036) to WMF. CG is supported by the VDRB, Commission for travelgrants of the SANW, Dr. De Giacomi Stiftung, Basler Stiftung für biologische Forschung. When we carried out our experiments, we adhered to the legal requirements of the country.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by M. Giurfa
Rights and permissions
About this article
Cite this article
Grüter, C., Acosta, L.E. & Farina, W.M. Propagation of olfactory information within the honeybee hive. Behav Ecol Sociobiol 60, 707–715 (2006). https://doi.org/10.1007/s00265-006-0214-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00265-006-0214-0