Abstract
Purpose
We evaluated whether synovial fluid (SF) leptin concentrations correlate with pain severity in patients with hip or knee endstage osteoarthritis (OA) and whether they mediate the association between increased joint pain and (1) female gender and (2) obesity.
Methods
We conducted a cross-sectional study including patients with primary hip and knee OA undergoing joint replacement between January and December 2010. SF leptin concentrations obtained on the day of surgery were assessed. Main outcome was pain severity measured pre-operatively using WOMAC and VAS pain scales.
Results
A total of 219 patients were included, 123 hip and 96 knee arthroplasties. Mean age was 72 years, 59 % were women. Mean SF leptin levels were 22.9 (±25.6) ng/ml in women and 5.4 (±5.9) ng/ml in men. Levels >19.6 ng/ml (highest quartile) were significantly associated with increased pain on both WOMAC (mean difference −9.6, 95 % CI −15.1 to −4.0) and VAS scale (mean difference 0.8, 95 % CI 0.2–1.3). Associations remained unchanged after adjusting for age, co-morbidities, contra-lateral arthritic joint, OA site, and disability. The associations observed between increased pain and female gender or obesity were substantially reduced after adjusting for SF leptin.
Conclusion
Joint pain is associated with SF leptin concentrations. Increased pre-operative pain observed in women and obese may be related to high intra-articular leptin levels.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Joint pain is the key clinical feature of osteoarthritis (OA) [1–3] and plays a major role in the decision for a total joint arthroplasty, but the causes of OA pain are not well established [4] and correlation between pain and structural joint damage is weak [5]. A possible link between joint pain and the adipokine leptin, which is considered as a key mediator in the well established association between obesity and osteoarthritis (OA) [6–13], has been indirectly suggested in studies on weight loss. Weight reduction consistently resulted in decreased joint pain in patients with OA [14, 15] and simultaneously decreased blood leptin levels [16–18]. Furthermore, leptin is particularly elevated in women and in obese patients [10, 19], and in these two patient groups, higher OA pain levels have been reported [20–26]. Studies from outside the musculoskeletal field have lent further support to a possible association between pain and leptin concentrations. Leptin levels in peritoneal fluid correlated positively with pain severity in endometriosis patients [27]. Furthermore, recent studies in rodents [28–30] suggested that spinal leptin might be involved in the pathogenesis of neuropathic pain.
So far two clinical studies have evaluated the association between adipokines and OA pain. Gandhi et al. [31] did not find a clear association between intra-articular leptin concentrations and pre-operative pain, but they reported that the synovial fluid (SF) adiponectin/leptin ratio predicted pain. Massengale et al. [32] showed that leptin serum concentration was associated with the intensity of pain in patients with chronic hand OA.
The objective of this study was to investigate if leptin may influence joint pain in OA patients. We hypothesized that SF leptin concentrations correlate with pain severity, and mediate the association between joint pain and (1) female gender and (2) obesity.
Materials and methods
Study design and patient population
We conducted a cross-sectional study including patients with end-stage OA of the hip and knee who presented for a total joint arthroplasty at a large orthopaedic centre between January and December 2010. Only patients with primary hip or knee OA were eligible. The study was approved by the local ethics committee and informed consent was obtained from all patients. Overall, synovial fluid (SF) was collected from 250 patients. Of those 219 had completed the pain scores and they were included in the final analysis.
Exposures of interest
Exposure of interest was the intra-articular (SF) concentration of leptin obtained at the time of joint replacement surgery (hip or knee). It was measured by a sandwich ELISA technique using the DuoSet ELISA Development Systems (R&D Systems, Abingdon, UK) according to the manufacturer’s protocols. The minimum detectable cytokine concentration for these assays was estimated to be 31 pg/ml for leptin.
Sex and body mass index (BMI) were the clinical factors of interest. BMI was analysed as continuous and dichotomised variable (BMI < 30 kg/m2 = non-obese vs. ≥ 30 kg/m2 = obese).
Outcome of interest
One outcome of interest was the pre-operative pain level measured with the Western Ontario McMaster Universities Osteoarthritis Index (WOMAC) pain score [33]. We employed the 5-point Likert scale version of the WOMAC 3.0. We analysed the score both as continuous (0–100, 100 = no pain) and as dichotomized variable (> 25 vs. ≤ 25 = quartile with greatest pain). We also used the visual analog scale (VAS) for pain assessment. The scale was evaluated as continuous (0–10, 0 = no pain) and as a dichotomized variable (< 7 vs. ≥ 8 = quartile with greatest pain). Both patient-assessed instruments are validated and widely used. In addition, the physician assessed pain item of the Harris hip score (HHS) (0–44, 44 = no pain) [34] was evaluated in patients with hip OA, and the physician-assessed pain item of the American Knee Society score (AKSS) (0–50, 50 = no pain) [35] was evaluated in patients with knee OA.
Co-variates and potential confounders
The following variables were assessed: (a) age; (b) American Society of Anesthesiologists (ASA) score investigated as binary variable (ASA < 3 vs. ≥ 3); (c) OA site (hip vs. knee); (d) presence of diabetes; (e) Medical Outcomes Study Short Form-12 (SF-12), a patient-administered generic health-related quality of life measure comprising a mental (mcs) and physical component score (pcs) [36]; the summary measures range from 0 to 100 (100 = best); (f) presence of a contra-lateral arthritic or operated joint; (g) Charnley disability category C for the hip and AKSS disability category C for the knee; the category C is defined as presence of multiple joint disease or other disabilities leading to difficulties in ambulation [35, 37]; (h) radiological severity of OA (Kellgren-Lawrence grades 0–4 and presence of osteophytes, subchondral sclerosis and minimal joint space width [minJSW]) [38]; (i) serum (blood) leptin concentration in ng/ml; and (j) synovial fluid adiponectin concentration in μg/ml.
Data collection
Synovial fluid (1 ml) was taken during the intervention by the operating surgeon by direct aspiration through the joint capsule, after skin incision, in order to minimize blood contamination. Blood samples (5 ml) were obtained during the pre-operative examination, at the same time as the pre-operative blood draw. A medical secretary was responsible for SF and blood sample transport. Patients’ samples were made anonymous prior to transport to the laboratory. All blood and SF samples were immediately centrifuged, aliquoted and frozen at −80 °C, until they were measured.
Since 1996 all patients undergoing total hip arthroplasty (THA) and since 1998 all those undergoing total knee arthroplasty (TKA) in the Geneva University Hospitals have been routinely enrolled in a prospective hospital-based cohort. The patients included in this study are part of the two cohorts. Information about baseline characteristics including medical and orthopaedic co-morbidities is routinely documented on specifically designed data collection forms. Pre-operative score and questionnaire assessment is routinely performed for all patients undergoing joint arthroplasty surgery. Harris hip score in case of hip OA and AKSS score in knee OA were assessed by the orthopaedic surgeon in charge of the patient the day before surgery. The WOMAC and the SF-12 were sent to all patients seven to ten days prior to surgery. The medical secretary was involved in sending and collecting scores and questionnaires and in data entry. The assessment of the radiographic severity of OA and identification of specific OA features was performed on pre-operative radiographs by two orthopaedic surgeons (GP, DS) blinded to the patient’s laboratory and clinical results.
Statistical analysis
The distribution of leptin concentrations (synovial fluid and serum) was right-skewed. To allow for comparison with previous literature on leptin and OA, we reported both mean (standard deviation, ± SD) and median (interquartile range, IQR) concentrations.
The association between SF leptin and preoperative pain levels was evaluated (1) as continuous variable using the Spearman’s correlation coefficient and (2) as categorical variable using quartiles. In particular, we compared pain levels in the 4th quartile (> 19.6 ng/ml) to those in the combined 1st to 3rd quartiles (≤ 19.6 ng/ml). First, we evaluated the percentage of patients with severe pain (defined as WOMAC ≤ 25 and VAS ≥ 8) in the various leptin categories and computed relative risks (RR) of severe pain by leptin category. Second, we calculated mean pain score differences using linear regression analysis. Because differences in pain levels could be related to other factors (confounding factors), we adjusted the analysis for age, diabetes, ASA score, contra-lateral arthritic joint, OA site and disability category C. In a second model we additionally adjusted for the SF-12 mental component score. This was done to separately show the influence of mediating psychological factors (mental health) on pain expression. We first analysed the data separately in patients with hip and knee OA, but decided the priority was to combine the results in the absence of effect modification by OA joint. Baseline characteristics are presented according to leptin quartiles.
In the second study objective, we explored whether leptin could mediate the association between pain and (1) female gender and (2) obesity in patients with OA. We first assessed crude pain differences by gender and by BMI categories and then we examined if adjusting for SF leptin concentration in a multivariate regression model would mitigate these univariate associations.
Results
Overall, 219 patients were evaluated, 123 with hip (56.2 %) and 96 with knee OA. Mean age was 72 (±9) years, and 59 % were women (n = 130). Mean BMI was 28.0 (±4.9) kg/m2, 27.3 kg/m2 in the hip group and 29.1 kg/m2 in the knee group. The median SF leptin concentration was 8.7 (IQR 3.0, 19.6) ng/ml and the mean concentration 15.8 (±21.8) ng/ml. In addition, mean SF leptin concentrations were 22.9 (±25.6) ng/ml in women compared to 5.4 (±5.9) ng/ml in men, and they were 12.4 (± 15.6) ng/ml in patients with hip OA compared to 20.1 (± 27.2) ng/ml in those with knee OA.
SF leptin concentrations were divided into quartiles with the highest quartile corresponding to levels > 19.6 ng/ml. The proportion of women increased strongly with rising leptin levels, up to 94 % in the highest quartile (Table 1). Similar results were seen for obesity, with the greatest proportion (59 %) of obese patients in the highest quartile. In addition, patients in the highest leptin category had less often diabetes, and more often knee OA, a contralateral arthritic joint, as well as an ASA score of 3–4. The SF-12 mcs and pcs scores were the lowest in this group. The radiographic severity of hip and knee OA did not differ substantially according to SF leptin quartiles (Table 1).
Synovial fluid leptin and pain
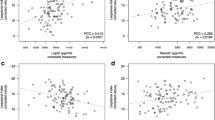
There was a weak correlation between SF leptin concentrations (continuous) and WOMAC pain (Spearman’s correlation coefficient r = −0.182) as well as between leptin and VAS pain (r = 0.115). Patients with high SF leptin concentrations (highest quartile: >19.6 ng/ml) reported higher pain levels on both the WOMAC and VAS pain scales. In the highest leptin quartile, 45 % of patients had a WOMAC pain score ≤ 25 compared to 20 % in the other quartiles (RR 2.3, 95 % CI 1.5; 3.5), and 42 % had a VAS pain score ≥ 8 compared to 21 % in the other quartiles (RR 2.0, 95 % CI 1.3–3.0) (Figs. 1 and 2). This association between SF leptin levels and pain (shown for WOMAC as percentage of patients ≤ 25; highest quartile vs. others) was apparent both in women (46 vs. 21 %) and men (33 vs. 19 %), obese (45 vs. 23 %) and non-obese patients (46 vs. 19 %), and in hip (57 vs. 24 %) and knee OA patients (37 vs. 14 %) (Figs. 3, 4 and 5).
The unadjusted mean score difference between patients in the highest leptin quartile compared to those in the other quartiles was −9.6 (95 % CI −15.1; −4.0; standardized effect size 0.54) for the WOMAC and 0.8 (95 % CI 0.2; 1.3; standardized effect size 0.44) for the VAS (Table 2). The association remained virtually unchanged after adjusting for potential confounders such as age, ASA score, contra-lateral arthritic joint, OA site, disability category C and diabetes (WOMAC adjusted mean difference −9.1 [95 % CI −14.8 to −3.5] and VAS adjusted mean difference 0.7 [95 % CI 0.2–1.3]). Additional adjustment for the SF-12 mental component score (WOMAC adjusted mean difference −6.8 (95 % CI −12.2 to −1.3) and VAS adjusted mean difference 0.6 [95 % CI 0–1.2]) slightly lowered the estimates, but the association remained significant. A greater degree of pain was seen in patients with high leptin levels as assessed by both the patient-assessed and the joint-specific physician-assessed scores HHS and AKSS (Table 2).
Significant associations between increased joint pain as measured with WOMAC and (1) female gender and (2) obesity observed in univariate analyses were substantially reduced after adjusting for SF leptin concentrations. The mean difference in WOMAC for women vs. men decreased from 4.7 to 1.6 and from 7.0 to 4.1 for obese vs. non-obese after correcting for leptin concentrations, suggesting that these associations might partially be mediated by leptin (Table 3).
Correlation with serum leptin
Synovial fluid leptin levels strongly correlated with serum leptin levels (correlation coefficient r = 0.875). Overall, the median serum leptin concentration was 15.8 (IQR 5.7, 36.9) ng/ml and the mean concentration 27.9 (±34.3) ng/ml. In addition, mean serum leptin concentrations were 38.7 (±39.5) ng/ml in women compared to 11.6 (±13.2) ng/ml in men.
Discussion
We found that OA patients with high SF leptin concentrations reported substantially more OA pain. Furthermore, the increased levels of pain observed in women and in obese patients seemed to be associated with high intra-articular leptin concentrations. However, due to the cross-sectional design a causal relation between leptin and pain cannot be ascertained.
Pain is certainly the most important symptom of OA, but also the least well understood [39]. Recent models imply that OA pain consists of a nociceptive stimulation of the joint and of peripheral sensitization, which can be accompanied by spinal and central sensitization [2, 40–43]. Leptin may affect the OA pain response in at least two possible ways. First, leptin appears to be a mediator of the immuno-inflammatory response in different experimental models. The pro-inflammatory cytokine-like functions of leptin in OA [10, 44–47] could explain its role in peripheral sensitization. Second, animal models suggest an involvement of leptin in the pathogenesis of pain at the spinal level and a possible role in the development of neuropathic pain [28–30]. Our clinical results support these experimental findings in patients with lower limb OA.
Our findings are in accordance with Massengale et al. [32] who, in a cohort of patients with chronic hand OA, reported an association between serum leptin levels with pain intensity but not with radiographic severity. Ghandi et al. [31] have investigated the relation between leptin and pain. They did not find a clear association between intra-articular leptin concentrations and pain in a linear regression analysis, but reported that the synovial fluid (SF) adiponectin/leptin ratio predicted pain. We found that the association between intra-articular leptin concentration and pain intensity was not linear. Pain intensity was similar within the lower three quartiles, but substantially higher in the highest quartile of leptin concentration.
Clinical implications of these findings might be: (1) exercise and weight loss programs should be particularly targeted to women, who constitute the vast majority of the high leptin level group; and (2) prospective studies are warranted to test the clinical usefulness of serum leptin as a biomarker (as a proxy for intra-articular leptin level) to identify subgroups of patients, who would particularly benefit from exercise and weight loss programs, to detect the presence of neuropathic pain reported to be common in knee OA patients [48, 49], as well as to predict persistence of pain after total joint arthroplasty.
The main limitation of this analysis is its cross-sectional design, and therefore the study is not suited to establish a causal link between leptin and increased pain. Leptin is a pleiotropic cytokine with central and peripheral effects involved in the regulation of energy intake and expenditure, of bone metabolism and inflammatory responses as well as of stress response [50]. Despite the above mentioned evidence from experimental studies supporting a possible causal effect of leptin on pain, other explanations for our findings including inverse causation and residual confounding need to be explored in future studies.
Second, we evaluated pain with two widely used, patient-assessed validated instruments. Because physician evaluation of pain has been found to be less influenced by the presence of co-morbidities [51], we additionally included one hip- and one knee-specific physician-assessed pain sub-score. However, information about the quality of pain (pain on movement, pain on rest, morning stiffness) was not routinely recorded. Third, depression and comorbidities, in particular the presence of OA at multiple sites, have been identified as important factors influencing the perception and severity of pain [20, 52–54].
A depression-specific instrument was not used in our study, but we collected information on mental health status using the SF-12 mental component score. Moreover, specific information about OA in other joints except for the contra-lateral joint was not systematically available. However, we assessed (and adjusted for) the presence of multiple joint disease and other medical disabilities leading to difficulties in ambulation.
Conclusion
Our results suggest that high leptin concentrations may affect the level of joint pain in OA of the lower limbs. Furthermore, elevated leptin levels may potentially explain the well established association between increased pain and female sex or obesity. However, the causal relation between leptin and joint pain needs to be confirmed in a longitudinal study.
References
Creamer P (2004) Current perspectives on the clinical presentation of joint pain in human OA. Novartis Found Symp 260:64–74, discussion 74–8, 100–4, 277–9
Dieppe PA, Lohmander LS (2005) Pathogenesis and management of pain in osteoarthritis. Lancet 365(9463):965–973
Hunter DJ (2009) Insights from imaging on the epidemiology and pathophysiology of osteoarthritis. Radiol Clin North Am 47(4):539–551
Wenham CY, Conaghan PG (2009) Imaging the painful osteoarthritic knee joint: what have we learned? Nat Clin Pract Rheumatol 5(3):149–158
Bedson J, Croft PR (2008) The discordance between clinical and radiographic knee osteoarthritis: a systematic search and summary of the literature. BMC Musculoskelet Disord 9:116
Aspden RM, Scheven BA, Hutchison JD (2001) Osteoarthritis as a systemic disorder including stromal cell differentiation and lipid metabolism. Lancet 357(9262):1118–1120
Dumond H, Presle N, Terlain B, Mainard D et al (2003) Evidence for a key role of leptin in osteoarthritis. Arthritis Rheum 48(11):3118–3129
Figenschau Y, Knutsen G, Shahazeydi S, Johansen O et al (2001) Human articular chondrocytes express functional leptin receptors. Biochem Biophys Res Commun 287(1):190–197
Griffin TM, Huebner JL, Kraus VB, Guilak F (2009) Extreme obesity due to impaired leptin signaling in mice does not cause knee osteoarthritis. Arthritis Rheum 60(10):2935–2944
Simopoulou T, Malizos KN, Iliopoulos D, Stefanou N et al (2007) Differential expression of leptin and leptin’s receptor isoform (Ob-Rb) mRNA between advanced and minimally affected osteoarthritic cartilage; effect on cartilage metabolism. Osteoarthr Cartil 15(8):872–883
de Boer TN, van Spil WE, Huisman AM, Polak AA et al (2012) Serum adipokines in osteoarthritis; comparison with controls and relationship with local parameters of synovial inflammation and cartilage damage. Osteoarthr Cartil 20(8):846–853
Vuolteenaho K, Koskinen A, Moilanen T, Moilanen E (2012) Leptin levels are increased and its negative regulators, SOCS-3 and sOb-R are decreased in obese patients with osteoarthritis: a link between obesity and osteoarthritis. Ann Rheum Dis 71:1912–1913
Jacobsen S, Sonne-Holm S (2005) Increased body mass index is a predisposition for treatment by total hip replacement. Int Orthop 29(4):229–234
Christensen R, Astrup A, Bliddal H (2005) Weight loss: the treatment of choice for knee osteoarthritis? A randomized trial. Osteoarthr Cartil 13:20–27
Toda Y, Toda T, Takemura S, Wada T et al (1998) Change in body fat, but not body weight or metabolic correlates of obesity, is related to symptomatic relief of obese patients with knee osteoarthritis after a weight control program. J Rheumatol 25:2181–2186
Miller GD, Nicklas BJ, Loeser RF (2008) Inflammatory biomarkers and physical function in older, obese adults with knee pain and self-reported osteoarthritis after intensive weight-loss therapy. J Am Geriatr Soc 56:644–651
Monzillo LU, Hamdy O, Horton ES, Ledbury S et al (2003) Effect of lifestyle modification on adipokine levels in obese subjects with insulin resistance. Obes Res 11:1048–1054
Sheu WH, Chang TM, Lee WJ, Ou HC et al (2008) Effect of weight loss on proinflammatory state of mononuclear cells in obese women. Obesity (Silver Spring) 16:1033–1038
Presle N, Pottie P, Dumond H, Guillaume C et al (2006) Differential distribution of adipokines between serum and synovial fluid in patients with osteoarthritis. Contribution of joint tissues to their articular production. Osteoarthr Cartil 14:690–695
Greenspan JD, Craft RM, LeResche L, Arendt-Nielsen L et al (2007) Studying sex and gender differences in pain and analgesia: a consensus report. Pain 132(Suppl 1):S26–S45
Creamer P, Lethbridge-Cejku M, Hochberg MC (1999) Determinants of pain severity in knee osteoarthritis: effect of demographic and psychosocial variables using 3 pain measures. J Rheumatol 26:1785–1792
Tonelli SM, Rakel BA, Cooper NA, Angstom WL et al (2011) Women with knee osteoarthritis have more pain and poorer function than men, but similar physical activity prior to total knee replacement. Biol Sex Differ 2:12
Cho HJ, Chang CB, Yoo JH, Kim SJ et al (2010) Gender differences in the correlation between symptom and radiographic severity in patients with knee osteoarthritis. Clin Orthop Relat Res 468:1749–1758
Holtzman J, Saleh K, Kane R (2002) Gender differences in functional status and pain in a Medicare population undergoing elective total hip arthroplasty. Med Care 40:461–470
Katz JN, Wright EA, Guadagnoli E, Liang MH et al (1994) Differences between men and women undergoing major orthopedic surgery for degenerative arthritis. Arthritis Rheum 37:687–694
Lubbeke A, Duc S, Garavaglia G, Finckh A et al (2009) BMI and severity of clinical and radiographic signs of hip osteoarthritis. Obesity (Silver Spring) 17:1414–1419
Bedaiwy MA, Falcone T, Goldberg JM, Sharma RK et al (2006) Peritoneal fluid leptin is associated with chronic pelvic pain but not infertility in endometriosis patients. Hum Reprod 21:788–791
Lim G, Wang S, Zhang Y, Tian Y et al (2009) Spinal leptin contributes to the pathogenesis of neuropathic pain in rodents. J Clin Invest 119:295–304
Maeda T, Kiguchi N, Kobayashi Y, Ikuta T et al (2009) Leptin derived from adipocytes in injured peripheral nerves facilitates development of neuropathic pain via macrophage stimulation. Proc Natl Acad Sci USA 106:13076–13081
Tian Y, Wang S, Ma Y, Lim G et al (2011) Leptin enhances NMDA-induced spinal excitation in rats: a functional link between adipocytokine and neuropathic pain. Pain 152:1263–1271
Gandhi R, Takahashi M, Smith H, Rizek R et al (2010) The synovial fluid adiponectin-leptin ratio predicts pain with knee osteoarthritis. Clin Rheumatol 29:1223–1228
Massengale M, Lu B, Pan JJ, Katz JN et al (2012) Adipokine hormones and hand osteoarthritis: radiographic severity and pain. PLoS One 7(10):e47860
Bellamy N, Buchanan WW, Goldsmith CH, Campbell J et al (1988) Validation study of WOMAC: a health status instrument for measuring clinically important patient relevant outcomes to antirheumatic drug therapy in patients with osteoarthritis of the hip or knee. J Rheumatol 15:1833–1840
Harris WH (1969) Traumatic arthritis of the hip after dislocation and acetabular fractures: treatment by mold arthroplasty. An end-result study using a new method of result evaluation. J Bone Joint Surg Am 51(4):737–755
Insall JN, Dorr LD, Scott RD, Scott WN (1989) Rationale of the Knee Society clinical rating system. Clin Orthop Relat Res 248:13–14
Ware J Jr, Kosinski M, Keller SD (1996) A 12-item short-form health survey: construction of scales and preliminary tests of reliability and validity. Med Care 34:220–233
Charnley J (1979) Numerical grading of clinical results. In: Charnley J (ed) Low friction arthroplasty of the hip: Theory and practice. Springer Verlag, Berlin, pp 20–24
Kellgren JH, Lawrence JS (1957) Radiological assessment of osteo-arthrosis. Ann Rheum Dis 16(4):494–502
Goldring MB (2009) The link between structural damage and pain in a genetic model of osteoarthritis and intervertebral disc degeneration: a joint misadventure. Arthritis Rheum 60(9):2550–2552
Hunter DJ, McDougall JJ, Keefe FJ (2008) The symptoms of osteoarthritis and the genesis of pain. Rheum Dis Clin North Am 34:623–643
Im HJ, Kim JS, Li X, Kotwal N et al (2010) Alteration of sensory neurons and spinal response to an experimental osteoarthritis pain model. Arthritis Rheum 62:2995–3005
Schaible HG, Ebersberger A, Von Banchet GS (2002) Mechanisms of pain in arthritis. Ann NY Acad Sci 966:343–354
Schaible HG, Richter F, Ebersberger A, Boettger MK et al (2009) Joint pain. Exp Brain Res 196:153–162
Bernotiene E, Palmer G, Gabay C (2006) The role of leptin in innate and adaptive immune responses. Arthritis Res Ther 8:217
Otero M, Lago R, Gomez R, Dieguez C et al (2006) Towards a pro-inflammatory and immunomodulatory emerging role of leptin. Rheumatol (Oxford) 45:944–950
Hoff P, Buttgereit F, Burmester GR, Jakstadt M et al (2013) Osteoarthritis synovial fluid activates pro-inflammatory cytokines in primary human chondrocytes. Int Orthop 37(1):145–151
Vuolteenaho K, Koskinen A, Kukkonen M, Nieminen R et al (2009) Leptin enhances synthesis of proinflammatory mediators in human osteoarthritic cartilage-mediator role of NO in leptin-induced PGE2, IL-6, and IL-8 production. Mediat Inflamm 2009:345838
Hochman JR, French MR, Bermingham SL, Hawker GA (2010) The nerve of osteoarthritis pain. Arthritis Care Res (Hoboken) 62:1019–1023
Hochman JR, Gagliese L, Davis AM, Hawker GA (2011) Neuropathic pain symptoms in a community knee OA cohort. Osteoarthr Cartil 19:647–654
Roubos EW, Dahmen M, Kozicz T, Xu L (2012) Leptin and the hypothalamo-pituitary-adrenal stress axis. Gen Comp Endocrinol 177(1):28–36
Dawson J, Fitzpatrick R, Murray D, Carr A (1996) The problem of ‘noise’ in monitoring patient-based outcomes: generic, disease-specific and site-specific instruments for total hip replacement. J Health Serv Res Policy 1:224–231
Hawker GA, Gignac MA, Badley E, French MR et al (2011) A longitudinal study to explain the pain-depression link in older adults with osteoarthritis. Arthritis Care Res (Hoboken) 63:1382–1390
Mallen CD, Peat G, Thomas E, Dunn KM et al (2007) Prognostic factors for musculoskeletal pain in primary care: a systematic review. Br J Gen Pract 57:655–661
Wylde V, Hewlett S, Learmonth ID, Dieppe P (2011) Persistent pain after joint replacement: prevalence, sensory qualities, and postoperative determinants. Pain 152:566–572
Acknowledgment
The authors would like to express their deep appreciation to all the orthopaedic surgeons, the personnel of the operating room and especially to Mme Carole Bandi and Mme Madeleine Vuillet for their efforts in data collection.
Funding
Internal institutional funding was received for this study.
Prof. Pierre Hoffmeyer received institutional financial support from Medacta, Johnson & Johnson, and Zimmer. The funding sources had no role in the collection, analysis, or interpretation of the data, in the preparation of the manuscript, or its submission for publication.
Conflicts of interest
None declared.
Ethics committee approval
The study was approved by the institutional review board of the Geneva University Hospitals and the patients’ informed consent was obtained (N° 09-215; NAC 09-072).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lübbeke, A., Finckh, A., Puskas, G.J. et al. Do synovial leptin levels correlate with pain in end stage arthritis?. International Orthopaedics (SICOT) 37, 2071–2079 (2013). https://doi.org/10.1007/s00264-013-1982-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00264-013-1982-6