Abstract
Locked plating attempts to improve mechanical stability via better anchorage of the screws in the bone. In 22 paired osteoporotic humeri an AO/ASIF 11-B 1 fracture was created. Locked and conventional plating using the same device of the latest generation was performed. Torsional loading around three axes (x = varus/valgus, y = flexion/extension, z = axial rotation) with an increasing moment (2, 3.5, 5 and 7.5 N·m) was applied. Interfragmentary motion within the locked group was lower for all three axes with higher cumulative survival rates (p < 0.05). The typical mode of failure was loss of fixation in the humeral head occurring earlier in the conventional group. The locking mechanism investigated provides more ultimate strength in an osteoporotic proximal humerus fracture model. Correlation with BMD suggests that this device may especially be suitable for use in osteoporotic bone.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Fractures of the proximal humerus are a typical injury of the elderly patient over 65 years of age [1–4]. The majority of these fractures are related to osteoporosis [5]. Stable fixation is hard to achieve in osteoporotic bone stock because anchoring of screws is difficult in these cases, leading to high rates of fixation failure, especially when conventional fixation techniques are used [6–8]. Fixed-angle devices have been designed to enhance fracture stability by using a locked screw plate interface [9–11]. Typically, this is realised by threaded screw heads which are either fixed in a thread in the plate requiring an exact angle of the screw, or by a thread in the screw cutting itself into the plate in an angle determined by the surgeon [12]. Biomechanical studies comparing locked and conventional implants in proximal humerus fracture models have shown that locked systems are biomechanically favourable in this anatomical region [13–15]. Clinical results, which increasingly report good functional results for these devices reflect the biomechanical advantages [16, 17]. However, most biomechanical studies compare different devices, thus different sizes and shapes of implants [5–7]. If, and to what extent, this influences the results remains unclear. In our study, locked and conventional plating with the same plate and screw geometry in an osteoporotic proximal humerus fracture model were compared. A second generation locking plate (NCB-PH®, Zimmer Inc.) which uses a new concept of locking, i.e. secondary insertion of a locking cap in combination with a conventionally implanted screw, was used. The purpose of this study was to investigate biomechanically this new concept of locking, and thus to reject or confirm the hypothesis that it provides more ultimate strength in a poor quality bone stock.
Materials and methods
Specimens
After consent from the local ethics committee board had been obtained (67/06, Ethics Committee Board, Ulm University), 11 pairs (n = 22 humeri) of fresh-frozen cadaveric bones were harvested (Southeast Tissue Alliance Inc, Florida, USA). Six pairs came from women and five pairs from men. The age of the donors ranged between 48 and 95 years with an average age of 79 years. For each specimen anthropometric measurements of the total length (head to distal condyles), head circumference, and metaphyseal shaft circumference were made [18]. Radiographs of each bone were taken to ensure a healthy bone. Radiographs were taken using high resolution X-ray (Faxitron 43805 N, Hewlett-Packard, Palo Alto, USA) with 65 kV and an exposure time of one minute.
Bone mineral density
A peripheral Quantitative Computed Tomography (pQCT) scanner (XCT 960, Stratec, Pforzheim, Germany) was used to evaluate bone mineral density (BMD) of each humeral head. After scanning the whole specimen, BMD was evaluated at the greatest transverse diameter of the humeral head (horizontal plane parallel to the humeral shaft) along three parallel sections with a 1-mm distance to each other (Fig. 1). The square region of interest was positioned over the cross-sectional area of the bone slice in such a way that every edge of the square reached the subcortical shell of the humeral head, measuring cancellous bone only [19]. The mean of the three measurements was calculated. CT scans had a slice thickness of 1 mm, an in-plane pixel size of 0.590 mm, and a matrix size of 128 × 128 pixels. After every 50 scans, the calibration of the scanner was checked with a hydroxyapatite phantom of known density (262.5 mg/cm3).
Preparation and osteotomy
Before preparation and osteotomy, specimens were stored overnight at 4°C. All soft tissues were stripped off and the bones were shortened to a total length of 19 cm. A noncomminuted 11-B1 fracture according to the AO/ASIF classification system was simulated using an oscillating saw [15]. First, an osteotomy line was created at the surgical neck. A second osteotomy was performed from the lateral aspect of the bicipital groove to just inferior to the flare of the tuberosity to separate the greater tuberosity from the humeral head (Fig. 1).
Operating technique
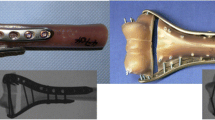
The right and left humerus of each donor were randomised for fixation either with locked or non-locked plating using the same implant (non-contact-bridging plate for the proximal humerus [NCB-PH], Zimmer, Inc.). The plate is made of titanium, and is anatomically precontoured (Fig. 2a). The system uses conventional screws which are locked to the plate by secondary insertion of a locking cap (Fig. 2b). Without the locking cap, screws can act as plate compression screws for fracture reduction against the plate. The locking mechanism therefore allows the application of the NCB-PH in both locked and conventional or non-locked mode. Solid self-tapping cancellous (Ø 4.5 mm) and cortical (Ø 4.0 mm) screws are used, which are predrilled using a Ø 3.3 mm drill. A targeting device, which is designed for minimally-invasive application of the plate, was used for screw placement, since it provides standardised (divergent) direction of screw placement [20].The procedure was performed according to the manufacture’s manual and as described elsewhere [21]. In all the specimens (n = 22) three screws were placed for humeral head fixation (locked or non-locked depending on randomisation to the respective group) as provided by the number of plate holes suitable for placement of the locking cap. To standardise subchondral screw placement in the humeral head a K-wire was inserted until its tip penetrated the chondral layer. Length measurement was performed via the wire using a depth gauge, and 5 mm were subtracted. Radiographs were taken after the procedure to check the correct position of the implant. The bones were then embedded in a special metal pot using bone cement (Technovit 3040, Haereus, Kulzer, Switzerland) and stored at −20°C.
Mechanical testing
Before testing, the specimens were thawed overnight at 4°C and then stored at room temperature for at least three hours before testing. Mechanical testing was performed in a three-dimensional spinal loading simulator [22]. By fixing the proximal part to a cardan device containing integrated stepper motors, pure moments around three axes (x = varus/valgus, y = flexion/extension, z = axial rotation) can be introduced (Fig. 3). An ultrasound-based three-dimensional motion analysis system (CMS, 70 P, Isny, Germany) was mounted on the specimens. The measuring principle is based on transmission time measurement of ultrasonic pulses and allows the registration of movements in all degrees of freedom with an accuracy of 0.1°. The sensory array consisted of a transmitter and a receiver (Fig. 3). Interfragmentary motion of the head (moving point = 2 cm proximal to the surgical neck osteotomy line and 1 cm medial to the greater tuberosity osteotomy line) against the shaft (9 cm distal to the moving point) was analysed (Fig. 1). Measurements were taken of angular displacements around the three axes (x, y and z). The data were recorded by the manufacturer’s software (WinBioMechanics, Version 0.1.2, Zebris, Isny, Germany).
The testing protocol was established in a pre-experimental set-up. Cycling loading with a total of 100 cycles of rotation around all the three axes at 2, 3.5, 5 and 7.5 N·m and with a speed of 0.5°/s was applied. The order of rotation around the axes was randomised by the spinal simulator software ensuring that the same number of cycles of rotation around each axis was applied within 100 respective cycles. After 0, 10, 20, 30, 50, 70 and 100 cycles with the respective moment, three cycles (one cycle of rotation around each of the three axes) were recorded to measure interfragmentary motion. The order of measurement (x, y and z) was again randomised. Failure was defined when angular displacement >30◦occurred, and the number of load cycles applied was recorded.
Statistical analysis
Statistics were performed using the SPSS Software 16.0 (SPSS Inc., Chicago, IL, USA). Descriptive analysis was performed by calculating the mean and standard deviations for both groups. Explorative statistical analysis used the Mann-Whitney U-Test for independent samples (non-locked vs. locked). Results with p < 0.05 were considered statistically significant. The survival rate was calculated according to Kaplan-Meier.
Results
Anthropometric measurements
No statistical differences could be detected for total length, head and metaphyseal shaft circumference between the two groups (Table 1).
Bone mineral density
For the humeri fixed with the conventional plate, BMD was 64 mg/cm3 (standard deviation [SD] 31 mg/cm3, range 14–125 mg/cm3). For the locked group BMD was 61 mg/cm3 (SD 30 mg/cm3, range 29–130 mg/cm3). There was no statistically significant difference between the two implant groups. BMD for both groups combined was 63 mg/cm3 (SD 30 mg/cm3) representing osteoporotic bone quality [23, 24].
Mechanical testing
The number of applicable load cycles showed a great variance (Table 2). Analysis based on the maximum moment survived takes, on the one hand, only a limited number of specimens into account and, on the other hand, ignores if, for example, a specimen failed after ten or 100 cycles of loading with the respective moment. Therefore, the analysis was performed based on the total sum of cycles applied to each specimen, i.e. 100 cycles of 2 Nm + 50 cycles of 3.5 Nm = 375 cycles. For all three axes there was statistical significance between the locked and the non locked groups (Fig. 4a–c).
Regression analysis of BMD vs. the total sum of cycles applied to the specimens showed that the regression coefficient for the non-locked group in all three axes was greater than 0.5 (x and y = 0.51, z = 0.53). For the locked group this was 0.45 in all three axes (Fig. 5).
The typical mode of failure was a loss of screw fixation in the humeral head with the greater tuberosity remaining reduced (Fig. 6a). Additionally, in the conventional group, backing out of screws occurred in highly osteoporotic humeri (Fig. 6b).
Discussion
In this study the locking concept of the NCB system was biomechanically investigated in an osteoporotic proximal humerus fracture model. The results showed a statistically significant higher survival rate of the locked group when exposed to cyclic torsional loading, thus confirming the hypothesis that locked plating with the NCB system provides more strength in this anatomical region. The insertion of the locking cap combined with conventional screws leading to “relative angular stability” according to Erhardt et al. effectively provides stability similar to first generation devices using threaded screws which are directly locked to the plate [5, 14, 25]. The correlation of BMD and overall sum of cycles survived as shown in our study may qualify the use of this specific plate, especially in osteoporotic bone quality, from a biomechanical point of view.
Locked plating acts as a load carrying lever beam, clinically also referred to as an “internal fixator”. The favourable mechanical behaviour is due to an evenly distributed load transfer from the bone to the implant over a perfectly stable screw. In contrast, non-locked plating presses the devices to the bone creating high focal loads on the bone plate contact area [12]. This and the possibility of tilting of the non-locked screws lead to early loss of fixation and loosening of screws as seen in our study.
The fracture model chosen in this study is rather simple but suitable for comparison purposes [15]. In clinical practice, the more complex fractures require the most stable fixation to allow functional treatment of the patient. Since these types of fractures are hard to simulate, making the comparison of implants difficult, the simple model in our study was chosen on purpose. We assume that the results of our study can be, at least in part, transferred to more complex fracture patterns, i.e. improved mechanical strength by locked plating. As with any kind of experimental set-up, the question remains as to what extent it can be transferred to the physiological situation in a patient. This is highlighted by the fact that we did not see displacement of the greater tuberosity which is a common problem in the clinical situation. In our view, this is due to the quite large size of the greater tuberosity fragment as artificially created by saw osteotomy; while in vivo these fragments are frequently small and/or multiple. Additionally, the tendon force of the supraspinatus muscle, which has a major impact on greater tuberosity displacement, was not simulated. Biomechanics of the shoulder is particularly difficult to transfer to the in vitro situation [6, 13, 14]. Most recent studies therefore use telemeterised shoulder implants to measure contact forces and moments in vivo when performing activities of daily living [26]. The authors report a maximum peak resultant moment of 4.5 N·m (combing, setting down a 2 kg weight on a board at head height) and a minimum of 0.17 N·m (holding a 10 kg weight passively). The moments applied in our study are within this range, thus comparable to the in vivo situation. Cyclic loading in our view seems to be reasonable, since activities of daily living or postoperative rehabilitation exercises are usually performed repetitively.
The locked group, like the conventional group, showed a considerable variance of the survival of the specimens. The earliest failure occurred after 20 cycles of loading at 2 N·m in a specimen of low (29.3 mg/cm3), but not the lowest, BMD within the study. Though providing more ultimate strength, locked plating does not seem to be able to provide sufficient stability in general when considering the result of this particular specimen before the background of the data by Bergmann et al. This mirrors most recent clinical studies reporting results after locked plating of proximal humerus fractures with complication rates up to 30%, among which fragment dislocation or loss of fixation is one of the most frequent problems [16, 17, 27]. Despite the clear in vitro mechanical advantages of locked plating as shown in ours and other studies with improved clinical results, these devices have not so far, proved to be a panacea. The consequence for the clinical situation should be that an individual decision on each patient regarding the postoperative treatment protocol has to be made based on fracture pattern and bone quality. To enhance stability in the future, further mechanisms, such as cement augmentation of locked screws, may have to be considered.
Apart from the limitations of the fracture model mentioned above, our study is further limited by the fact that pure moments were applied, which again fails to fully represent the complex mechanics of the shoulder joint.
Conclusion
Locked plating provided by a second generation locking device in an osteoporotic proximal humerus fracture model provides more mechanical strength when exposed to cyclic torsional loading. The new locking concept used in this study has proven to be as efficient as first-generation devices when compared to conventional plating. Correlation of BMD and survival of the specimen supports the conclusion that this device is especially beneficial in osteoporotic bone. However, in cases of severe osteoporosis early implant failure even occurred in locked plating. This supports the conclusion that not all problems in fixation of this fracture are fully resolved by this new generation of implants. For the future, additional mechanisms to enhance stability, i.e. augmentation of locked screws, may have to be considered.
References
Koval KJ, Blair B, Takei R et al (1996) Surgical neck fractures of the proximal humerus: a laboratory evaluation of ten fixation techniques. J Trauma 40:778–783
Lind T, Kroner TK, Jensen J (1989) The epidemiology of fractures of the proximal humerus. Arch Orthop Trauma Surg 108:285–287
Palvanen M, Kannus P, Niemi S et al (2006) Update in the epidemiology of proximal humeral fractures. Clin Orthop Rel Res 442:87–92
Rasmussen S, Hvass I, Dalsgaard J et al (1992) Displaced proximal humerus fractures: results of conservative treatment. Br J Accident Surg 23:1–3
Hessmann MH, Hansen WS, Krummenauer F et al (2005) Locked plate fixation and intramedullary nailing for proximal humerus fractures: a biomechanical evaluation. J Trauma 58(6):1194–1201
Kwon BK, Goertzen DJ, O’Brien PJ et al (2002) Biomechanical evaluation of proximal humeral fracture fixation supplemented with calcium phosphate cement. J Bone Joint Surg 84-A:951–961
Cofield RH (1988) Comminuted fractures of the proximal humerus. Clin Orthop 230:49–57
Esser RD (1994) Treatment of three and four part fractures of the proximal humerus with a modified cloverleaf plate. J Orthop Trauma 8:15–22
Fankenhauser F, Boldin C, Schnippinger G et al (2005) A new locking plate for unstable fractures of the proximal humerus. Clin Orthop Relat Res 430:176–181
Lill H, Hepp P, Rose T et al (2004) The angle stable Locking-Proximal-Humerus-Plate (LPHP®) for proximal humeral fractures using a small anterior-lateral-deltoid-splitting-approach-technique and first results. Zentralbl Chir 129:43–48 (in German)
Plecko M, Kraus A (2005) Internal fixation of proximal humerus fractures using the locking proximal humerus plate. Operat Orthop Traumatol 17:25–50 (in German)
Seide K, Triebe J, Faschingbauer M et al (2007) Locked vs. unlocked plate osteosynthesis of the proximal humerus—a biomechanical study. Clin Biomech 22(2):176–182
Chudik SC, Weinhold P, Dahners LE (2003) Fixed-angle plate fixation in simulated fractures of the proximal humerus: a biomechanical study of a new device. J Shoulder Elbow Surg 12(6):578–588
Siffri PC, Peindl RD, Coley ER et al (2006) Biomechanical analysis of blade plate versus locking plate fixation for a proximal humerus fracture: comparison using cadaveric and synthetic humeri. J Orthop Trauma 20(8):547–554
Weinstein DM, Bratton DR, Ciccone WJ et al (2006) Locking plate improves torsional resistance in the stabilization of three part proximal humeral fractures. J Shoulder Elbow Surg 15(2):239–243
Südkamp N, Bayer J, Hepp P et al (2009) Open reduction and internal fixation of proximal humeral fractures with use of the locking proximal humerus plate. Results of a prospective, multicenter, observational study. J Bone Joint Surg 91-A:1320–1328
Solberg BD, Moon CN, Franco DP et al (2009) Surgical treatment of three and four-part proximal humeral fractures. J Bone Joint Surg 91-A:1689–1697
Edwards S, Wilson N, Zhang L et al (2006) Two-part surgical neck fractures of the proximal humerus. A biomechanical evaluation of two fixation techniques. J Bone Joint Surg 88-A:2258–2264
Krischak GD, Augat P, Beck A et al (2007) Biomechanical comparison of two side plate fixation techniques in an unstable intertrochanteric osteotomy model: sliding hip screw and percutaneous compression plate. Clin Biomech 22(10):1112–1118
Röderer G, AbouElsoud M, Gebhard F et al (2007) Minimally-invasive application of the non-contact-bridging plate to the proximal humerus. An anatomical study. J Orthop Trauma 21(9):621–627
Röderer G, Gebhard F, Erhardt J et al (2007) The non-contact bridging plate: a new fixed angle device for the minimally-invasive treatment of fractures of the proximal humerus-technique and preliminary results. Unfallchirurg 110(6):505–512 (in German)
Wilke HJ, Claes L, Schmitt H et al (1994) A universal spine tester for in vitro experiments with muscle force simulation. Eur Spine J 3(2):91–97
Diederichs G, Korner J, Goldhahn J et al (2006) Assessment of bone quality in the proximal humerus by measurement of the contralateral site: a cadaveric analyze. Arch Orthop Trauma Surg 126(2):93–100
Tingart MJ, Bouxsein ML, Zurakowski D et al (2003) Three-dimensional distribution of bone density in the proximal humerus. Calcif Tissue Int 73(6):531–536
Erhardt JB, Röderer G, Grob K et al (2009) Early results in the treatment of proximal humeral fractures with a polyaxial locking plate. Arch Orthop Trauma Surg 129(10):1367–1374
Bergmann G, Graichen F, Bender A et al (2006) In vivo glenohumeral contact forces—measurements in the first patient 7 months postoperatively. J Biomech 40(10):2139–2145
Agueldo J, Schürmann M, Stahel P et al (2007) Analysis of efficacy and failure in proximal humerus fractures treated with locking plate. J Orthop Trauma 21(10):676–681
Acknowledgments
The authors would like to express their gratitude to Mrs. Karin Werner for expert technical assistance in performing the experiment.
Conflict of interest
The study was sponsored with a grant from Zimmer Inc. The study sponsor played no role in designing the study, in the collection, analysis and interpretation of data, in writing the manuscript or in the decision to submit the manuscript for publication.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Röderer, G., Gebhard, F., Krischak, G. et al. Biomechanical in vitro assessment of fixed angle plating using a new concept of locking for the treatment of osteoporotic proximal humerus fractures. International Orthopaedics (SICOT) 35, 535–541 (2011). https://doi.org/10.1007/s00264-010-1021-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00264-010-1021-9