Abstract
Purpose
The purpose of the study was to determine whether the pre-treated MR texture features of colorectal liver metastases (CRLMs) are predictive of therapeutic response after chemotherapy.
Methods
The study included twenty-six consecutive patients (a total of 193 liver metastasis) with unrespectable CRLMs at our institution from August 2014 to February 2016. Lesions were categorized into either responding group or non-responding group according to changes in size. Texture analysis was quantified on T2-weighted images by two radiologists with consensus on regions of interest which were manually drawn on the largest cross-sectional area of the lesions. Five histogram features (mean, variance, skewness, kurtosis, and entropy1) and five gray level co-occurrence matrix features (GLCM; angular second moment (ASM), entropy2, contrast, correlation, and inverse difference moment (IDM)) were extracted. The texture parameters were statistically analyzed to identify the differences between the two groups, and the potential predictive parameters to differentiate the responding group from the non-responding group were subsequently tested using multivariable logistic regression analysis.
Results
A total of 107 responding and 86 non-responding lesions were evaluated. A higher variance, entropy1, contrast, entropy2 and a lower ASM, correlation, IDM were independently (P < 0.05) associated with a good response to chemotherapy with the areas under the ROC curves (AUCs) of 0.602–0.784. Variance (P < 0.001) and ASM (P = 0.001) remained potential predictive values to discriminate responding lesions from non-responding lesions when tested using multivariable logistic regression analysis. The highest AUC of the predictors from the association of variance and ASM was 0.814.
Conclusion
MR texture features on pre-treated T2 images have the potential to predict the therapeutic response of colorectal liver metastases.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Colorectal cancer is one of the three most commonly diagnosed cancers worldwide [1]. Metastasis is associated with a poor prognosis, and the liver is a primary site of metastasis. Almost 15% of patients have simultaneous liver metastases from colorectal cancer, and 60% of the metachronous metastases are in the liver [2].
Radical resection of liver metastases is recommended as the curative therapy to improve the prognosis. However, less than 20% of patients are candidates for the resection of liver lesions; for the majority, palliative chemotherapy is the preferred treatment strategy [3]. Therefore, the early prediction of the therapeutic response is important for selecting the appropriate chemotherapy approach.
Texture analysis, a new imaging biomarker, is a non-invasive method to assess the heterogeneity within a tumor. Tumors heterogeneity occurs due to variations in genomic subtypes, cell proliferation or apoptosis, metabolic activity, vascular structure, and other factors [4]. By analyzing the distribution and relationship of pixel or voxel-gray levels in the image, texture analysis provides a more detailed and quantitative evaluation of the lesion characteristics than morphological analysis. A series of studies on different tumors provided evidence that texture analysis could be used as a very promising imaging biomarker of tumor treatment response [5,6,7,8,9]. A few recent studies have proven that texture analysis of computed tomography (CT) images is feasible and has certain practical value in assessing the response of colorectal liver metastases (CRLMs) to chemotherapy [10,11,12,13]. In addition, MR images seem to display more accurate texture features than CT images [14].
However, few studies have evaluated the role of MR texture features in CRLMs. Therefore, the purpose of this study was to determine whether the texture features of pre-treated MR images of CRLMs are predictive of therapeutic response after cytotoxic chemotherapy.
Materials and methods
Patient population
This retrospective study was approved by our institutional review board, and the written informed consent was waived. Twenty-six patients with unrespectable CRLMs who underwent the standard first-line chemotherapy regimen using FOLFOX (oxaliplatin, leucovorin plus fluorouracil, repeated every 2 weeks, twice for one cycle), FOLFIRI (irinotecan, leucovorin plus fluorouracil, repeated every 2 weeks, twice for one cycle), or XELOX (oxaliplatin plus capecitabine, every 3 weeks for one cycle) at our institution between August 2014 and February 2016 were retrospectively reviewed. All patients underwent baseline MRI within 3 weeks before chemotherapy. Inclusion criteria consisted of: (a) histopathologically confirmed colorectal adenocarcinoma without the presence of any other malignant tumors; (b) the presence of liver metastases measuring > 1 cm in longest diameter with at least one lesion; (c) no previous treatment, including chemotherapy, radiotherapy, interventional treatment, or liver metastases resection, prior to baseline MRI; and (d) underwent the secondary imaging examination and response evaluation after four cycles of chemotherapy.
MR examinations
All MR images were performed before treatment on a 3.0 T MR magnet (Signa Horizon, GE Medical Systems, Milwaukee, WI) with a phased-array body coil. The MR imaging protocol and the parameters of sequences are detailed in Table 1. Enhanced images were acquired after the intravenous administration of gadopentetate dimeglumine using an axial and coronal LAVA sequence.
Texture analysis
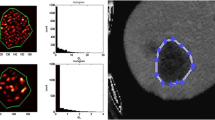
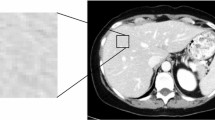
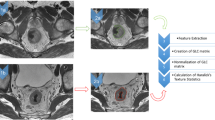
Texture analyses were performed using a dedicated script written in MATLAB (MATLAB R2011b, MathWorks, Inc., Natick, MA, USA). Following review of the MR images on PACS system, the largest cross-sectional slice of the hepatic lesions greater than 1 cm in longest diameter were selected and transferred to the texture analysis program [15]. The region of interest (ROI) was manually drawn along the tumor outer edge on axial T2-weighted images and then further refined by the exclusion of areas of great vessels with an agreement of two radiologists (with 3 and 13 years of experience in abdominal MRI, respectively). The reviewers were blinded to clinical information.
For each ROI, five histogram features (mean, variance, skewness, kurtosis, and entropy1) and five gray level co-occurrence matrix features (GLCM; angular second moment (ASM), entropy2, contrast, correlation, and inverse difference moment (IDM)) were extracted using this texture analysis software. Histogram statistics are calculated from the original image values and known as “first order” texture measures. The GLCM described here are used as a series of “second order” texture calculations, meaning that they consider the relationship between groups of two pixels in the original image. All the texture parameters are defined mathematically in Table 2. As the definitions have showed, higher variance, skewness, kurtosis, entropy1, contrast, entropy2 and lower ASM, correlation, IDM suggest increased heterogeneity within a ROI.
Response evaluation
Chemotherapy response was determined by evaluating the changes in tumor size on a lesion-by-lesion basis. The maximum diameter of tumor was measured to the nearest millimeter on axial T2-weighed scans. We divided the response into two groups [16]: the responding group (≥ 30% reduction in the maximum transverse diameter) and non-responding group (< 30% reduction in the maximum transverse diameter).
Statistical analysis
The texture parameters of tumors were statistically analyzed to find the differences in baseline MR histogram parameters between the two groups. Mean values of the ten parameters were compared between the groups of responding and non-responding using Student’s t test or Mann–Whitney U test when not normally distributed. The potential predictive parameters to differentiate the responding group from the non-responding group were subsequently tested using multivariable logistic regression analysis. A P value of less than 0.05 was considered significant. The diagnostic ability of the texture parameters to predict the treatment outcome was assessed by receiver-operating characteristic (ROC) curve analysis. All data were analyzed using SPSS (version 21.0; Chicago, IL, USA) and MedCalc (version 12.7.2; Ostend, Belgium).
Results
Patients and lesions
A total of 26 consecutive patients (12 F/14 M, mean age 58.5 ± 9.7 years) who met the criteria were recruited from our hospital between August 2014 and February 2016. All patients underwent baseline standard MRI followed by the first-line chemotherapy using FOLFOX (n = 9), FOLFIRI (n = 12), or XELOX (n = 5). The number of liver metastatic lesions was stratified into: 1–4 (10 patients), 5–10 (7 patients), or > 10 (9 patients). In all patients, 107 responding (Fig. 1) and 86 non-responding (Fig. 2) lesions were evaluated.
Texture statistics
The mean values of the five histogram parameters and five GLCM parameters are provided in Table 3. Variance, entropy1, contrast, and entropy2 were statistically higher in the responding group than in the non-responding group (P < 0.05). ASM, correlation, and IDM of the responding group were independently lower than those of the non-responding group (P < 0.05). However, the mean (P = 0.186), skewness (P = 0.311), and kurtosis (P = 0.763) did not show significant differences between the two groups.
Variance (P < 0.001) and ASM (P = 0.001) remained potential predictive values to discriminate responding from non-responding lesions when tested using multivariable logistic regression analysis. ROC curve analyses were performed to evaluate the performance to predict the response to chemotherapy. Corresponding areas under the ROC curves (AUCs) and the associated criteria, sensitivities, specificities, positive predictive values, and negative predictive values, are shown in Table 4 and Fig. 3. Among them, the highest AUC of the predictor (PRE1) from the association of variance and ASM was 0.814, with good sensitivity (71.0%) and specificity (84.9%).
Discussion
In this study, we found that MR texture features derived from histogram and GLCM quantified on T2-weighted images correlated with the chemotherapeutic response of patients with CRLMs. The data showed that responding lesions had higher baseline variance and lower ASM than non-responding lesions. The highest AUC of the predictor from the association of variance and ASM reached 0.814, with good sensitivity (71.0%) and specificity (84.9%).
As higher variance and lower ASM correlate with the complexity and non-uniformity of image texture, which in turn reflect tumor heterogeneity. The results of our study suggest that heterogeneous tumors seem to have a more favorable response to therapy, which may be related to the hypoxic micro-environment, irregular angiogenesis, and extracellular vascular permeability that are characteristic of these tumors. Thus, to some extent, heterogeneous tumors have a higher metabolic burden and greater distribution of tumor blood vessels [17, 18].
The effect of chemotherapy relies on the delivery of chemotherapeutic agents, which is associated with vascular supply to and the metabolism of metastatic liver cells. Theoretically, tumors with greater heterogeneity can provide a wealth of information on therapeutic response, which can be used to make efficacy predictions, to more effectively deliver chemotherapy drugs to the lesions and to improve drug absorption and bioavailability.
The association between the texture features derived from medical imaging and clinical outcomes such as therapeutic response and survival has already been proven in a variety of tumor types [7,8,9, 19, 20]. There are also some reports showing that texture parameters derived from CT images are conducive to predicting the chemotherapy response of patients with CRLMs [11,12,13]. Some previously published data lend support to the results of our study. For example, in a study evaluating 77 patients with CRLMs and assessing texture features derived from CT [11], texture parameters were correlated with tumor grade, baseline serum CEA, KRAS mutation status and overall survival, their result demonstrated that more homogeneous tumors with less entropy and smaller standard deviation were more aggressive in their biology (higher tumor grade and poorer overall survival). To the best of our knowledge, until now, only one published study has evaluated CRLMs by MR texture analyses [21]. In contrast to our results, they found that the mean of the responding group were significantly lower than that of the non-responding group (P = 0.001), but no significant differences in variance, skewness ,and kurtosis were found between the two groups [21]. A possible contributing factor might be that, in their study histogram parameters were quantized on apparent diffusion coefficient (ADC) maps performed on a 1.5T MR system. Different MR scanners with different acquisition parameters may also affect the texture measurements.
The vast majority of patients with CRLMs are ineligible for surgical resection and are instead recommended the first-line chemotherapy, including FOLFOX, FOLFIRI or XELOX. However, almost half of the patients with CRLMs exhibit no therapeutic response even after administration of the first-line chemotherapy [22, 23]. For these individuals, the addition of targeted agents such as bevacizumab or cetuximab may prolong the progression-free survival [24]. Hence, the prediction of chemotherapy response as early as possible is important for clinical treatment decisions and to screen suitable patients. Size-based measurements are restrictive in therapeutic response predictions [25]. Additional criteria such as density and enhancement pattern, may provide more information but fail to provide a quantifiable measure of response [26,27,28]. Texture analyses may enable the extraction of more useful quantization parameters from the image. The results of this study showed that the baseline MR texture features of histogram and GLCM were associated with the response to chemotherapy.
There are several limitations in our study. First, the study is limited by its small sample size and retrospective background. Second, only a single slice of the hepatic metastatic lesion was assessed, which may not adequately represent the heterogeneous characteristics of the whole tumor. Although previous studies comparing 2D vs. 3D measurements of single lesions reported quite comparable results [13, 15]. Furthermore, therapeutic response was determined on a lesion-by-lesion basis by evaluating changes in tumor size after chemotherapy, which does not accurately reflect the pathological chemotherapeutic response. Ideally, the response of individual lesions should be correlated to surgical findings and pathological results. However, the vast majority of patients in the study presented with multiple metastases and were thus excluded from surgery. Further studies are needed that include more cases with corresponding pathological information and clinical outcomes after chemotherapy.
Conclusion
Pretreatment response evaluations using imaging studies may benefit therapeutic decision making. MR texture analysis on T2-weighted images is a non-invasive technique to extract tumor heterogeneity information from standard MR images without the need for contrast agent injection. Our results suggest that pre-therapeutic MR texture features have the potential to predict the therapeutic response of colorectal liver metastases. However, larger-scale prospective studies are needed to establish its clinical application.
References
Torre LA, Bray F, Siegel RL, et al. (2015) Global cancer statistics, 2012. CA 65:87–108
Kemeny N (2006) Management of liver metastases from colorectal cancer. Oncology (Williston Park) 20:1185–1186
Leporrier J, Maurel J, Chiche L, et al. (2006) A population-based study of the incidence, management and prognosis of hepatic metastases from colorectal cancer. Br J Surg 93:465–474
Asselin M, O’Connor JPB, Boellaard R, Thacker NA, Jackson A (2012) Quantifying heterogeneity in human tumours using MRI and PET. Eur J Cancer 48:447–455
Cook GJR, Yip C, Siddique M, et al. (2013) Are pretreatment 18F-FDG PET tumor textural features in non-small cell lung cancer associated with response and survival after chemoradiotherapy? J Nucl Med 54:19–26
Liu J, Mao Y, Li Z, et al. (2016) Use of texture analysis based on contrast-enhanced MRI to predict treatment response to chemoradiotherapy in nasopharyngeal carcinoma. J Magn Reson Imaging 44:445–455
Michoux N, Van den Broeck S, Lacoste L, et al. (2015) Texture analysis on MR images helps predicting non-response to NAC in breast cancer. BMC Cancer 15:574
De Cecco CN, Ganeshan B, Ciolina M, et al. (2015) Texture analysis as imaging biomarker of tumoral response to neoadjuvant chemoradiotherapy in rectal cancer patients studied with 3-T magnetic resonance. Investig Radiol 50:239–245
De Cecco CN, Ciolina M, Caruso D, et al. (2016) Performance of diffusion-weighted imaging, perfusion imaging, and texture analysis in predicting tumoral response to neoadjuvant chemoradiotherapy in rectal cancer patients studied with 3T MR: initial experience. Abdom Radiol 41:1728–1735
Miles KA, Ganeshan B, Griffiths MR, Young RCD, Chatwin CR (2009) Colorectal cancer: texture analysis of portal phase hepatic CT images as a potential marker of survival. Radiology 250:444–452
Lubner MG, Stabo N, Lubner SJ, et al. (2015) CT textural analysis of hepatic metastatic colorectal cancer: pre-treatment tumor heterogeneity correlates with pathology and clinical outcomes. Abdom Imaging 40:2331–2337
Rao S-X, Lambregts DM, Schnerr RS, et al. (2016) CT texture analysis in colorectal liver metastases: a better way than size and volume measurements to assess response to chemotherapy? United Eur Gastroenterol J 4:257–263
Ahn SJ, Kim JH, Park SJ, Han JK (2016) Prediction of the therapeutic response after FOLFOX and FOLFIRI treatment for patients with liver metastasis from colorectal cancer using computerized CT texture analysis. Eur J Radiol 85:1867–1874
Zhang X, Gao X, Liu BJ, et al. (2015) Effective staging of fibrosis by the selected texture features of liver: which one is better, CT or MR imaging? Comput Med Imaging Graph 46:227–236
Ng F, Kozarski R, Ganeshan B, Goh V (2013) Assessment of tumor heterogeneity by CT texture analysis: can the largest cross-sectional area be used as an alternative to whole tumor analysis? Eur J Radiol 82:342–348
Cui Y, Zhang X-P, Sun Y-S, Tang L, Shen L (2008) Apparent diffusion coefficient: potential imaging biomarker for prediction and early detection of response to chemotherapy in hepatic metastases. Radiology 248:894–900
Ganeshan B, Goh V, Mandeville HC, et al. (2013) Non-small cell lung cancer: histopathologic correlates for texture parameters at CT. Radiology 266:326–336
Ng F, Ganeshan B, Kozarski R, Miles KA, Goh V (2013) Assessment of primary colorectal cancer heterogeneity by using whole-tumor texture analysis: contrast-enhanced CT texture as a biomarker of 5-year survival. Radiology 266:177–184
Ganeshan B, Panayiotou E, Burnand K, Dizdarevic S, Miles K (2012) Tumour heterogeneity in non-small cell lung carcinoma assessed by CT texture analysis: a potential marker of survival. Eur Radiol 22:796–802
Oh JS, Kang BC, Roh J-L, et al. (2015) Intratumor textural heterogeneity on pretreatment (18)F-FDG PET images predicts response and survival after chemoradiotherapy for hypopharyngeal cancer. Ann Surg Oncol 22:2746–2754
Liang HY, Huang YQ, Yang ZX, Ying-Ding Zeng MS, Rao SX (2016) Potential of MR histogram analyses for prediction of response to chemotherapy in patients with colorectal hepatic metastases. Eur Radiol 26:2009–2018
Tournigand C, André T, Achille E, et al. (2004) FOLFIRI followed by FOLFOX6 or the reverse sequence in advanced colorectal cancer: a randomized GERCOR study. J Clin Oncol 22:229–237
Colucci G, Gebbia V, Paoletti G, et al. (2005) Phase III randomized trial of FOLFIRI versus FOLFOX4 in the treatment of advanced colorectal cancer: a multicenter study of the Gruppo Oncologico Dell’Italia Meridionale. J Clin Oncol 23:4866–4875
Kabbinavar FF, Schulz J, McCleod M, et al. (2005) Addition of bevacizumab to bolus fluorouracil and leucovorin in first-line metastatic colorectal cancer: results of a randomized phase II trial. J Clin Oncol 23:3697–3705
Egger ME, Cannon RM, Metzger TL, et al. (2013) Assessment of chemotherapy response in colorectal liver metastases in patients undergoing hepatic resection and the correlation to pathologic residual viable tumor. J Am Coll Surg 216:845–857
Chun YS, Vauthey J-N, Boonsirikamchai P, et al. (2009) Association of computed tomography morphologic criteria with pathologic response and survival in patients treated with bevacizumab for colorectal liver metastases. JAMA 302:2338–2344
Shindoh J, Loyer EM, Kopetz S, et al. (2012) Optimal morphologic response to preoperative chemotherapy: an alternate outcome end point before resection of hepatic colorectal metastases. J Clin Oncol 30:4566–4572
Chung WS, Park MS, Shin SJ, et al. (2012) Response evaluation in patients with colorectal liver metastases: RECIST version 1.1 versus modified CT criteria. Am J Roentgenol 199:809–815
Acknowledgments
The authors declare that there is no conflict of interest regarding the publication of this paper. This study was supported by the National Natural Science Foundation of China (Grant No. 81501437).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, H., Li, W., Hu, F. et al. MR texture analysis: potential imaging biomarker for predicting the chemotherapeutic response of patients with colorectal liver metastases. Abdom Radiol 44, 65–71 (2019). https://doi.org/10.1007/s00261-018-1682-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-018-1682-1