Abstract
Purpose
To assess the additional value of secretin-enhanced MRCP (SMRCP) over conventional MRCP in diagnosing divisum.
Methods
Retrospective HIPAA-compliant and IRB-approved review found 140 patients with SMRCP and ERCP correlation within 6 months of each other. All studies were anonymized and the SMRCP images (SMRCP image set) were separated from 2D and 3D MRCP and axial and coronal T2-weighted images (conventional MRI image set). Each image set on each patient was assigned different and randomized case numbers. Two reviewers (R1 and R2) independently reviewed the image sets for divisum vs. no divisum, complete divisum vs. incomplete divisum, and the certainty of diagnosis (1 = definitely certain, 2 = moderately certain, and 3 = unsure). ERCP findings were taken as gold standard.
Results
There was no difference in age and gender between the divisum (n = 97, with 13 incomplete divisum) and no divisum (n = 43) groups. In diagnosing divisum anatomy, the sensitivity was higher for SMRCP compared to conventional MRI for R1 (84.5 vs. 72.2, p = 0.02) but not R2 (89.7 vs. 84.4, p = 0.25). The specificity was higher in SMRCP image set compared to conventional MRI (R1: 88.1 vs. 76.2, p = 0.01; R2: 81.4 vs. 65.1, p < 0.001). The mean area under ROC curve was higher for SMRCP image set (R1: 0.86 vs. 0.74, p = 0.01; R2: 0.87 vs. 0.74, p = 0.01). The certainty of diagnosis was higher in SMRCP image set compared to conventional MRI (p = 0.02 for both reviewers). SMRCP was not found to be superior in distinguishing incomplete from complete divisum. The main reasons for erroneous SMRCP diagnosis were the presence of an ansa loop in the main duct and ductal strictures due to chronic pancreatitis.
Conclusion
Even though the reviewers had more sequences (axial and coronal) to evaluate in the non-secretin image set, there was some improvement in diagnosing divisum with SMRCP.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Diagnosis of pancreatic ductal anomalies is important since patients with these anomalies have an increased risk of recurrent acute and chronic pancreatitis [1–3]. Traditionally, it has been thought that this association is due to functional or anatomic stenosis at the papilla, resulting in pancreatic ductal hypertension [4–6]. More recent reports suggest that the increased risk of inflammation may be due to the association of ductal anomalies with genetic anomalies that predispose to pancreatitis [7–10].
The most common congenital pancreas anomaly is divisum, in which there is lack of fusion between ventral and dorsal anlages of pancreas. Incomplete divisum signifies a divisum anatomy where there is a narrow ductal communication between the Santorini and Wirsung ducts (Fig. 1). This connecting duct may act as a potential valve to reduce pressure within the main pancreatic duct. The prevalence of incomplete divisum is thought to be lower than that of complete divisum in western populations [11, 12]. Some studies show that incomplete divisum has a similar risk for recurrent acute pancreatitis as complete divisum [11, 13, 14]
Diagram of normal anatomy (A), ansa loop (B), complete divisum (C), and incomplete divisum (D). Note that the inset (D) shows a minor connection between ventral and dorsal ducts incomplete divisum. In ansa loop (B) the unexpected curving of the main pancreatic duct in the head may in some case cause confusion with divisum.
Secretin is an endogenous gastrointestinal polypeptide hormone, released by the duodenal mucosa in response to luminal acidity, which stimulates the water and bicarbonate secretion of exocrine pancreas and contracts the sphincter of Oddi temporarily [15]. These effects help to improve visualization of pancreatic ducts in MRCP and facilitate cannulation of the minor papilla during endoscopic retrograde cholangiopancreatography (ERCP) [16, 17]. The use of secretin may also reduce the risk of post-ERCP pancreatitis [18]. A few previous papers have reported the usefulness of secretin-enhanced MRCP (SMRCP) in congenital ductal anomalies [19–23]. Nevertheless, the use of secretin for improving MRCP has remained untested in large, blinded trials. Studies of conventional MRCP and SMRCP, using prospectively issued radiology reports, suggest a sensitivity of only 65%–73% for diagnosing divisum [24, 25]. We wanted to assess, in a large series of patients using blinded reviewers, the sensitivity of conventional MRCP and SMRCP for diagnosing complete and incomplete pancreas divisum. As a secondary objective, we wanted to assess in which situations SMRCP was likely to result in false positive or false negative diagnosis.
Methods
Patients
This retrospective Health Insurance Portability and Accountability ACT (HIPAA)-compliant study reviewed the MRI database between January 2002 and December 2009 for patients who underwent secretin-enhanced MRCP. Of 3764 patients, 878 had endoscopic retrograde pancreatography (ERCP) without intervening surgical therapy. Review of ERCP findings revealed 104 patients with divisum. Seven of these patients (all with complete divisum anatomy) were excluded since their MRCP images were not retrievable on PACS. The remaining 97 patients formed the subject group with divisum. Of the patients without divisum, the first consecutive 43 patients were included as the control group. These 140 patients formed the study cohort. Institutional review board permission for retrospective analysis of radiology and clinical databases, with waiver of informed consent, had been obtained.
MRI examinations
The MRI examinations were performed on a 1.5 T MRI scanner (Magnetom Avanto or Harmony, Siemens Medical Solutions, Erlangen, Germany). The T2-weighted, two-dimensional and three-dimensional MRCP and SMRCP protocols have been previously published [26, 27]. SMRCP was obtained after the injection of 16 micrograms of intravenous synthetic human secretin (ChiRhoStim, ChiRhoClin, Inc., Burtonsville, MD). For children, the dose was 0.2 μg/kg body weight. Coronal 4-cm-slab half-Fourier acquisition single-shot turbo spin-echo (HASTE) images obtained through the pancreas were obtained every 30 seconds for 10 or 15 min. Together with the presecretin coronal slab MRCP, there were 21 or 31 images in the secretin image set. Post-contrast images were not analyzed for this study.
Image review
All images were downloaded to an Apple Macintosh computer (iMac, running Mac OS X, Apple Inc., Cupertino, CA). The SMRCP images were separated from the rest of the image series and saved as the “secretin” image set. The T2-weighted axial and coronal images and two- and three-dimensional MRCP images were grouped together and saved as the “nonsecretin” image set. Post-gadolinium images were not included in this set. All images were anonymized using a DICOM viewer (OsiriX3.91, Pixmeo, Geneva, Switzerland). The two image sets per patient were labeled with different random case numbers.
Two abdominal radiologists with 15 (KS)- and 2 (BT)-year post-fellowship experience independently reviewed the randomized image sets in the order of the assigned case numbers. The reviewers were blinded to patient information, and were blinded as to which “secretin” and “nonsecretin” image sets belonged to the same patient. However, the reviewers knew whether an image set contained “secretin” or “nonsecretin” images.
The reviewers noted the presence or absence of divisum, whether the divisum was complete or not, and whether there was evidence of chronic pancreatitis. They also rated the confidence of diagnosing of divisum on a 1–3 scale: 1 = definite diagnosis, 2 = probable diagnosis, and 3 = unsure.
Following the initial review by the two reviewers, the reasons for the errors in identification of divisum anatomy (false positive and negative) were assessed by a radiologist (FA—17-year experience) who was unblinded and did not take part in the initial review.
Reference standard
For the diagnosis of pancreas divisum, ERCP represents the gold standard. Typically, a duodenoscope (JF-140, JF-130, TJF-160VR, Olympus America, Center Valley, PA) was advanced to the second portion of the duodenum. Pancreas divisum was confirmed via cannulation and injection of the dorsal pancreatic duct through the minor papilla. Pancreas divisum was suspected when cannulation of the major papilla demonstrated a small ventral duct system that did not communicate with the dorsal duct. The minor orifice was cannulated using a highly tapered cannula and less commonly a sphincterotome. Complete pancreas divisum was confirmed when contrast entering the minor papilla traversed the pancreatic body with no evidence of communication with the ventral pancreatic duct. Incomplete divisum was confirmed when contrast entering the minor papilla traversed the pancreatic body and there was a vestigial communication with the ventral system (Fig. 1). ERCP examinations were reported prospectively by one of six expert endoscopists. In addition, they were reported contemporaneously by one of two expert gastrointestinal radiologists with over 15-year experience in viewing endoscopic images. The ERCP images were not reviewed for this study, since they are much less diagnostic than the original endoscopists’ reports. For instance, divisum is difficult to diagnose on retrospective review of ERCP images since the pancreatic and bile ducts are injected separately and contrast may not persist in both ducts on the same image.
Statistical analysis
The presence of divisum (complete or incomplete) and the lack of divisum were noted. Categorical variables from the qualitative assessment and frequency of occurrence in different groups were analyzed with descriptive statistics and Fisher’s exact test. The correlations between the measurements of the two reviewers were performed using Shrout–Fleiss random effect ICC coefficient [28]. Receiver operating characteristic (ROC) curve analysis was used to evaluate the usefulness of secretin in diagnosing divisum, and in differentiating complete divisum from incomplete divisum. A two-tailed p value less than 0.05 was used to indicate statistical significance. Statistical analysis was performed using MedCalc 11.1 (MedCalc Software, Mariakerke, Belgium) (for ROC curves) and SPSS 17.0. (SPSS Inc, Chicago, Ill).
Results
Patients
There were 107 females and 33 males, making up the cohort of 140 patients. Mean age was 45.4 years (range 5–85 years). The divisum group had 72 females and 25 males group with mean age of 46.3 years (range 9–85 years). The control group had 35 females and 8 males with mean age of 43.2 years (range 5–84 years).The indications for the MRI examinations were unexplained abdominal pain (n = 63), assessment of chronic pancreatitis (n = 38), assessment of recurrent acute pancreatitis (n = 23), sphincter of Oddi dysfunction (n = 11), and miscellaneous (n = 5). There was no significant difference in age (p = 0.17) or gender (p = 0.78) between the divisum and control groups. There were more patients with ERCP diagnosis of chronic pancreatitis in the divisum group (32/97 = 33%) that in the control group (12/43 = 28%, p = 0.02). The mean duration between MRI and ERCP was 34 days (range 0–148 days).
Divisum vs. no divisum
Based on ERCP data, there were 97 patients in the divisum group and 43 patients in the control group. The sensitivity and specificity of the two reviewers for the secretin and non-secretin image sets are given in Table 1. The sensitivity of reviewer 1 was significantly higher in the secretin set (p = 0.02) but no difference in sensitivity was seen for reviewer 2 (p = 0.25). The specificity of both reviewers, particularly reviewer 2, was higher for the secretin group (p = 0.01 and p < 0.001, respectively). The area under receiver operating characteristic curves (AUC) is also given in Table 1 (Fig. 2). There was a significantly higher AUC for the secretin group compared to the non-secretin group (p = 0.01 for both reviewers).
Receiver operating characteristics (ROC) curves of secretin-enhanced MRCP (SMRCP) and conventional MRI image sets in diagnosing divisum anatomy. Solid curve represents conventional MRI results of reviewer 1 and dashed curve that of reviewer 2. Dotted curve represents SMRCP results of reviewer 1 and dot-dashed curve those of reviewer 2. Area under curve (AUC) values are given in Table 1. There was a significant increase in AUC for conventional MRI compared to ADC measurements for both reviewers (p = 0.01). Diagonal line represents AUC of 0.50.
Table 2 gives the degree of certainty recorded by the two reviewers in diagnosing the presence or absence of divisum. In general, the confidence of diagnosing divisum was significantly improved with MRCP (p = 0.02, both reviewers) (Figs. 3, 4). The frequency of certain diagnosis was increased with SMRCP for reviewer 1 (p = 0.01) but not for reviewer 2 (p = 0.21). The frequency of unsure diagnosis with SMRCP was significantly lower for reviewer 2 (p = 0.003).
59-Year-old female with unexplained abdominal pain. A Presecretin MRCP showed distal common bile duct (CBD) (dashed arrow) but there was poor visualization of pancreatic duct. It was not possible to determine anatomy with certainty. B SMRCP clearly showed main duct (white arrow) draining via dorsal (Santorini) duct (black arrowhead), and crossing distal CBD (dashed arrow), indicating divisum. Distal end of dorsal duct was distended consistent with a Santorinicele (white arrowhead). This may be due to increased pressure of duct at minor ampulla, and occasionally cause pain. Increased fluid is seen in the duodenum (black arrows). Prior to MRI, patient ingested 300 mL of iron colloid suspension (ferumsoxysl, GastroMark, Mallinckrodt, Raleigh, NC) which made gastric and duodenal lumen dark on presecretin MRCP. Exocrine secretions reaching duodenum after secretin stimulation increased duodenal luminal signal.
38-Year-old female with two prior episodes of acute pancreatitis. A Presecretin MRCP clearly showed CBD (dashed arrow). Main pancreatic duct (white arrowhead) and dorsal duct (black arrow) were barely visible. Confidence of diagnosing divisum was low. B SMRCP at 7 minutes post-injection clearly showed main pancreatic duct (white arrowhead) draining via dorsal duct (black arrow) which crossed over CBD (dashed arrow), indicating divisum. Use of secretin increased confidence of diagnosing divisum. Exocrine fluid output barely filled duodenal (D2) lumen (black arrowhead) and was considered suboptimal. This may suggest impaired exocrine function of pancreas, even though there were no ductal changes to suggest chronic pancreatitis.
Incomplete divisum vs. complete divisum
Of the 97 patients with divisum anatomy, 13 had incomplete divisum (Figs. 5, 6) and the rest had complete divisum (Figs. 3, 4). The sensitivity and specificity as well as the area under ROC curve (AUC) for the two reviewers are given in Table 3. The only significant finding was the increased sensitivity for incomplete divisum on secretin image set for reviewer 1. There was no difference in the AUC for secretin and non-secretin image sets for both reviewers (p = 0.09 and 0.66, respectively). SMRCP did not substantially contribute to the differentiating incomplete from complete divisum.
42-Year-old female with suspected chronic pancreatitis. A Presecretin MRCP showed main pancreatic duct (white arrow) but it was not possible to determine whether there was divisum anatomy. B SMRCP image showed that main pancreatic duct (white arrow) drained via dorsal duct (black arrow) indicating divisum anatomy. Ventral duct (black arrowhead) was seen with terminal arborization (white arrowhead). Connection between ventral (Wirsung) and dorsal (Santorini) duct systems was not clearly seen, and this case was incorrectly called as complete divisum by one of the reviewers on SMRCP image set. C. Initial ERCP injection of ventral duct (black arrow) showed arborization (white arrowheads). There was flash filling of main duct (white arrows) with ventral duct injection, indicating incomplete divisum.
34-Year-old male with suspected sphincter of Oddi dysfunction. Presecretin MRCP (A) and SMRCP (B) showed main pancreatic duct (white arrow) draining via dorsal duct (black arrow) which crossed over CBD (dashed arrow), in keeping with divisum. On image A, an indeterminate wispy duct (black arrowhead) was seen in the pancreatic head. On SMRCP (B), the ventral duct (white arrowhead) was seen to fill the main duct (white arrow) via small duct (black arrowhead). Both reviewers called non-secretin image set as complete divisum and SMRCP image set as incomplete divisum. C. ERCP confirmed incomplete divisum. Injection of ventral duct (white arrowhead) filled the main duct (arrow) via connecting duct (black arrowhead).
Analysis of erroneous MRCP findings
The most common reason for missing divisum on non-secretin image set was the inability to adequately visualize the main pancreatic duct due to technical factors. Technically poor quality images were responsible for false negative calls on the non-secretin image set for both reviewers (Figs. 3, 4). In the secretin image set, the technical quality appeared generally better, but we did not formally assess technical quality since this evaluation is subjective and prone to bias. The most common reason for not identifying divisum anatomy on the secretin image set was the presence of chronic pancreatitis with ductal strictures in the head. The presence of chronic pancreatitis was responsible for all cases where both reviewers had false negative calls on the secretin image sets image. The presence of chronic pancreatitis was also responsible for 5 of 6 unsure diagnosis of reviewer 1 and 5 of 7 unsure diagnosis of reviewer 2 on the secretin image set. The presence of an ansa loop in the pancreatic head makes it difficult to determine whether the main duct joins the ventral or dorsal system and leads to both false positive and false negative diagnosis of divisum (Fig. 7).
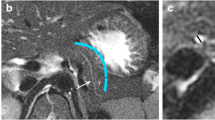
21-Year-old female with history of recurrent acute pancreatitis. Presecretin (A) and post-secretin (B) MRCP images showed the main pancreatic duct (white arrows) apparently coursing toward major papilla to join CBD (dashed arrows). Ansa loop (white arrowheads) and arborization of ventral duct (black arrowheads) were noted in pancreatic head. Post-secretin image (B) did not only dispute findings of presecretin MRCP, but also showed prominent dorsal duct (black arrow) ending in minor papilla. Both reviewers read this case as “no divisum” on presecretin and post-secretin image sets. C. At ERCP, dorsal duct was cannulated (black arrow) and injection directly filled main pancreatic duct (white arrow) indicating divisum anatomy. Stent was placed in ventral duct (black arrowhead). There was thin communication (white arrowhead) indicating incomplete divisum. Diagnosis of incomplete divisum may be difficult on SMRCP and conventional MRI. In addition, the presence of ansa loop in pancreatic head makes diagnosis of divisum harder.
Discussion
Pancreas divisum occurs in 5%–10% of the population in European and North American series [11, 29]. There is a significantly higher risk of chronic or recurrent acute pancreatitis in patients with divisum [2–6, 12, 29]. ERCP has been the traditional method of diagnosing divisum. However, dorsal duct cannulation is associated with a high incidence of acute pancreatitis (8%–10%), even if minor papilla sphincterotomy is performed [30]. Incomplete divisum is a relatively new entity for radiologists but is well known in the gastrointestinal endoscopy literature. There is some debate as to its prevalence and significance. Large ERCP series from Japan and Korea give the prevalence of incomplete divisum as 0.6%–1.3% [13, 31]. Divisum is relatively uncommon in this population [32], and incomplete and complete divisum have comparable frequencies. Studies from USA report a prevalence of 0.6% for incomplete divisum [11, 12] which is much lower than that of complete divisum. In our study, 878 ERCP patients were initially assessed and 91 (10.4%) had complete divisum and 13 (1.5%) had incomplete divisum. The risk of recurrent acute or chronic pancreatitis is thought to be similar in incomplete and complete divisum [11, 13, 14].
Initial reports suggested a high accuracy of MRCP in diagnosing divisum [33]. More recent studies indicated that MRCP had a sensitivity of only 32%–73% in diagnosing divisum [24, 25, 34]. In one study, one-fifth (21.2%) of MRCPs were non-diagnostic since key segments of the pancreatic duct were not seen [24]. Conventional MRCP and SMRCP were compared in one paper which found that seven of 30 (23.3%) cases of divisum were better seen after secretin injection [20]. This study did not use ERCP as the gold standard, so true sensitivity or specificity could not be determined. Another publication compared secretin and non-secretin MRCP images side-by-side [23]. ERCP was used as the gold standard, and MRCP diagnoses were made in consensus. Conventional MRCP and SMRCP detected divisum in 23 and 30 patients, respectively, indicating a 30% increase in sensitivity for SMRCP. No false positive SMRCP diagnosis was found. To our knowledge, no prior study has rigorously compared SMRCP with conventional MRCP in a large cohort of patients with divisum and control subjects.
Our study found that SMRCP demonstrated a significantly higher sensitivity and specificity for differentiating the presence and absence of divisum, for at least one of the reviewers. SMRCP resulted in increased sensitivities of 5%–12% and increased specificities of 12%–16%. The differing sensitivity and specificity among the two reviewers may be related to their experience in assessing SMRCP. The area under ROC curve was higher for SMRCP compared to conventional MRCP (p = 0.01 for both reviewers). Both reviewers were more confident in the diagnosis of divisum when reviewing SMRCP compared to conventional MRCP (p = 0.02, both reviewers).
No prior study has assessed the value of MRCP in differentiating incomplete and complete divisum. ERCP may be an imperfect gold standard in this respect. It is usual practice to inject contrast with the minimal necessary force, to reduce the risk of ERCP-induced pancreatitis. As a result, a tenuous connection between the dorsal and ventral systems may not be adequately opacified. Our results indicate that the sensitivity for diagnosing incomplete divisum increased significantly for one reviewer (31%–69%, p < 0.01), but not for the second reviewer (23%–32%, p = 0.39). Neither reviewer showed a difference in specificity with SMRCP. The mean area under ROC curve for differentiating complete and incomplete divisum was not increased by the use of secretin. SMRCP was not able to easily differentiate complete and incomplete divisum due to the inability of detecting the small duct that connects the ventral and dorsal systems (Figs. 5, 7).
Review of false positive and negative results in our study indicated that radiologists must be cautious in pancreas divisum in cases of ansa loop (Figs. 1), a relatively common normal anatomic variant of pancreatic ductal fusion in the head [35]. Severe ductal stricture and reduced reserve of exocrine pancreas in chronic pancreatitis contributed to false negative results.
We are aware of some of the limitations of this study. The study was retrospective and used ERCP as a gold standard. Thus, the study population may be skewed from the general population. The number of incomplete pancreas divisum cases was small, since this condition is rare. We tried to make this study as unbiased as possible using a robust system of case anonymization. Nevertheless, the reviewers knew which anonymized image sets were secretin-enhanced and which were not. The use of secretin increases the fluid content in the duodenum and allows for this differentiation. Short of electronically removing the duodenum and small bowel on every image (not a trivial task), it would be impossible to overcome this bias.
In conclusion, SMRCP yields a higher overall sensitivity and specificity compared to conventional MRCP in diagnosing divisum. It may, in some circumstances, increase the certainty of diagnosing complete pancreas divisum. SMRCP was not found to be useful in differentiating complete and incomplete pancreas divisum. Nevertheless, SMRCP may obviate the need for a diagnostic ERCP in many cases of suspected divisum.
References
Fischer M, Hassan A, Sipe BW, et al. (2010) Endoscopic retrograde cholangiopancreatography and manometry findings in 1,241 idiopathic pancreatitis patients. Pancreatology 10(4):444–452
Takuma K, Kamisawa T, Tabata T, Egawa N, Igarashi Y (2010) Pancreatic diseases associated with pancreas divisum. Dig Surg 27(2):144–148
Gonoi W, Akai H, Hagiwara K, et al. (2011) Pancreas divisum as a predisposing factor for chronic and recurrent idiopathic pancreatitis: initial in vivo survey. Gut 60(8):1103–1108
Cotton PB (1980) Congenital anomaly of pancreas divisum as cause of obstructive pain and pancreatitis. Gut 21(2):105–114
Gregg JA (1977) Pancreas divisum: its association with pancreatitis. Am J Surg 134(5):539–543.
Satterfield ST, McCarthy JH, Geenen JE, et al. (1988) Clinical experience in 82 patients with pancreas divisum: preliminary results of manometry and endoscopic therapy. Pancreas 3(3):248–253.
Bertin C, Pelletier AL, Vullierme MP, et al. (2012) Pancreas divisum is not a cause of pancreatitis by itself but acts as a partner of genetic mutations. Am J Gastroenterol 107(2):311–317.
Cavestro GM, Zuppardo RA, Bertolini S, et al. (2010) Connections between genetics and clinical data: Role of MCP-1, CFTR, and SPINK-1 in the setting of acute, acute recurrent, and chronic pancreatitis. Am J Gastroenterol 105(1):199–206
Garg PK, Khajuria R, Kabra M, Shastri SS (2009) Association of SPINK1 gene mutation and CFTR gene polymorphisms in patients with pancreas divisum presenting with idiopathic pancreatitis. J Clin Gastroenterol 43(9):848–852.
Whitcomb DC (2012) Genetics of alcoholic and nonalcoholic pancreatitis. Curr Opin Gastroenterol. 28(5):501–506
Jacob L, Geenen JE, Catalano MF, et al. (1999) Clinical presentation and short-term outcome of endoscopic therapy of patients with symptomatic incomplete pancreas divisum. Gastrointest Endosc 49(1):53–57
Lehman GA, Sherman S (1998) Diagnosis and therapy of pancreas divisum. Gastrointest Endosc Clin N Am 8(1):55–77
Kim MH, Lee SS, Kim CD, et al. (2001) Incomplete pancreas divisum: is it merely a normal anatomic variant without clinical implications? Endoscopy 33(9):778–785
Kamisawa T, Tu Y, Egawa N, Tsuruta K, Okamoto A (2006) Clinical implications of incomplete pancreas divisum. JOP 7(6):625–630
Chey WY, Chang TM (2003) Secretin, 100 years later. J Gastroenterol 38(11):1025–1035.
Devereaux BM, Fein S, Purich E, et al. (2003) A new synthetic porcine secretin for facilitation of cannulation of the dorsal pancreatic duct at ERCP in patients with pancreas divisum: a multicenter, randomized, double-blind comparative study. Gastrointest Endosc 57(6):643–647
Devereaux BM, Lehman GA, Fein S, et al. (2002) Facilitation of pancreatic duct cannulation using a new synthetic porcine secretin. Am J Gastroenterol 97(9):2279–2281
Jowell PS, Branch MS, Fein SH, et al. (2011) Intravenous synthetic secretin reduces the incidence of pancreatitis induced by endoscopic retrograde cholangiopancreatography. Pancreas 40(4):533–539
Nicaise N, Pellet O, Metens T, et al. (1998) Magnetic resonance cholangiopancreatography: interest of IV secretin administration in the evaluation of pancreatic ducts. Eur Radiol 8(1):16–22
Manfredi R, Costamagna G, Brizi MG, et al. (2000) Pancreas divisum and “santorinicele”: diagnosis with dynamic MR cholangiopancreatography with secretin stimulation. Radiology 217(2):403–408
Song MH, Kim MH, Lee SK, Seo DW (2002) The clinical usefulness of secretin-enhanced magnetic resonance pancreatography in patients with pancreas divisum and idiopathic acute pancreatitis. Gastrointest Endosc 55(3):454–455
Hellerhoff KJ, Helmberger H 3rd, Rosch T, et al. (2002) Dynamic MR pancreatography after secretin administration: image quality and diagnostic accuracy. Am J Roentgenol 179(1):121–129
Matos C, Metens T, Deviere J, et al. (2001) Pancreas divisum: evaluation with secretin-enhanced magnetic resonance cholangiopancreatography. GastrointestEndosc 53(7):728–733
Mosler P, Akisik F, Sandrasegaran K, et al. (2012) Accuracy of magnetic resonance cholangiopancreatography in the diagnosis of pancreas divisum. Dig Dis Sci 57(1):170–174
Carnes ML, Romagnuolo J, Cotton PB (2008) Miss rate of pancreas divisum by magnetic resonance cholangiopancreatography in clinical practice. Pancreas 37(2):151–153
Tirkes T, Akisik F, Tann M, Balci NC (2009) Imaging of the pancreas with secretin enhancement. Top Magn Reson Imaging 20(1):19–24
Akisik MF, Sandrasegaran K, Aisen AA, et al. (2006) Dynamic secretin-enhanced MR cholangiopancreatography. Radiographics 26(3):665–677
Shrout PE, Fleiss JL (1979) Intraclass correlations: uses in assessing rater reliability. Psychol Bull 86(2):420–428
Delhaye M, Cremer M (1992) Clinical significance of pancreas divisum. Acta Gastroenterol Belg 55(3):306–313
Moffatt DC, Cote GA, Avula H, et al. (2011) Risk factors for ERCP-related complications in patients with pancreas divisum: a retrospective study. Gastrointest Endosc 73(5):963–970.
Kamisawa T, Egawa N, Tu Y, Tsuruta K, Okamoto A (2007) Pancreatographic investigation of embryology of complete and incomplete pancreas divisum. Pancreas 34(1):96–102.
Liao Z, Gao R, Wang W, et al. (2009) A systematic review on endoscopic detection rate, endotherapy, and surgery for pancreas divisum. Endoscopy 41(5):439–444
Bret PM, Reinhold C, Taourel P, et al. (1996) Pancreas divisum: evaluation with MR cholangiopancreatography. Radiology 199(1):99–103
Kamisawa T, Tu Y, Egawa N, et al. (2007) MRCP of congenital pancreaticobiliary malformation. Abdom Imaging 32(1):129–133
Koshinaga T, Fukuzawa M (2000) Pancreatic ductal morphological pattern and dilatation in postoperative abdominal pain in patients with congenital choledochal cyst: an analysis of postoperative pancreatograms. Scand J Gastroenterol 35(12):1324–1329
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sandrasegaran, K., Cote, G.A., Tahir, B. et al. The utility of secretin-enhanced MRCP in diagnosing congenital anomalies. Abdom Imaging 39, 979–987 (2014). https://doi.org/10.1007/s00261-014-0131-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-014-0131-z