Abstract
Aim
Our aim was to evaluate overall survival and parameters prognosticating longer survival in a large and homogeneous group of patients treated with 177Lu-PSMA-617 radioligand therapy with heavily pretreated advanced metastatic castration resistant prostate cancer.
Methods
A total of 104 patients were treated with 351 cycles of 177Lu-PSMA-617. Prostate specific antigen (PSA) changes after the first cycle of therapy were documented prior to a second cycle. Patients were followed-up for overall survival (OS). Any PSA decline, PSA decline ≥50%, initial PSA, alkaline phosphatase (ALP), lactate dehydrogenase (LDH), visceral metastases and cumulative injected activity were analyzed and evaluated according to OS. Multivariable analysis with parameters with a p-value ≤0.05 in univariate analysis was performed, additionally adjusting for age and presence of visceral metastases.
Results
A total of 51 patients (49%) died during the observation period. The majority of patients (97%) presented with bone metastases, 77% with lymph node metastases and 32% with visceral metastases. All patients were treated with at least one line of chemotherapy. Either abiraterone or enzalutamide had been given in 100% of the patients. Any PSA decline occurred in 70 (67%) and a PSA decline ≥50% in 34 (33%) of patients after the first cycle. The median OS was 56.0 weeks (95%CI: 50.5–61.5). Initial PSA decline ≥50%, initial LDH, visceral metastases, second line chemotherapy or prior radium-223 did not have an effect on survival, whereas any initial PSA decline, initial ALP <220 U/L and cumulative injected activity ≥18.8 GBq were associated with a longer survival. A step-by-step analysis revealed a PSA decline ≥20.87% as the most noticeable cut-off prognosticating longer survival, which remained an independent prognosticator of improved OS in the multivariate analysis.
Conclusion
177Lu-PSMA-617 RLT is a new effective therapeutic and seems to prolong survival in patients with advanced mCRPC pretreated with chemotherapy, abiraterone and/or enzalutamide.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Radioligand therapy (RLT) using 177Lu labeled prostate specific membrane antigen (PSMA) ligands has been the subject of several studies within the last few years [1,2,3,4,5,6,7,8,9]. 177Lu-PSMA-617 is the most commonly used PSMA-targeted radiopharmaceutical for patients with advanced metastasized castration resistant prostate cancer (mCRPC) patients. These mainly retrospective studies have reported promising response rates and progression-free survival. The compound shows a favorable toxicity profile and often times improves quality of life [3, 9, 10]. In a preliminary analysis, this new therapeutic could show a prolongation of overall survival (OS) compared to a historical nearly matched group of patients [4].
The aim of this retrospective study was to evaluate OS and parameters prognosticating longer survival in a large and homogeneous group of patients treated with 177Lu-PSMA-617 with advanced mCRPC heavily pretreated with at least one line of chemotherapy and abiraterone and/or Enzalutamide.
Methods
Patient population
A total of 104 patients with mCRPC, who have been treated with 177Lu-PSMA-617 RLT between December 2014 and December 2016, were included in this retrospective analysis. Patients had received at least one line of chemotherapy (docetaxel and/or cabazitaxel) and at least one of the next generation antihormonal therapies (enzalutamide and /or abiraterone) to be included in the present analysis. Data concerning response and safety have in part been reported before by KR and HA [1, 3,4,5,6].
The decision to perform 177Lu-PSMA-617 RLT was made individually in each case by an interdisciplinary tumor board. 177Lu-PSMA-617 RLT was offered in case of lacking other therapeutic options and disease progression despite established therapies according to guidelines for mCRPC management [11]. All patients received detailed information on the possible adverse events and risks which may be associated with 177Lu-PSMA-617 therapy. All patients gave their written informed consent prior to treatment. This study was approved by the local ethics committee.
Patient preparation
PSMA imaging was performed in all patients using 68Ga-PSMA-11 PET-CT or PET-MRI, according to institutional guidelines, to confirm PSMA expression in tumor lesions.
Renal scintigraphy using 99mTc-MAG3 was performed in all patients to rule out any upper urinary tract obstruction. Additional renal laboratory parameters, liver parameters and blood counts were performed to rule out clinically relevant renal or liver function impairment and bone marrow depression.
Preparation and administration of 177Lu-PSMA-617
The commercially available GMP precursor PSMA-617 (ABX advanced biochemical compounds, Radeberg, Germany) was labelled with 177Lutetium (ITG Isotopes Technologies, Garching, Germany) in-house according to radiosynthesis procedures described in detail previously [4].
RLT was injected intravenously as a slow bolus in 30 to 60 s. Patient had to be hydrated, additional to drinking water, thus receiving at least 1000 mL of Ringer’s Solution intravenously following therapy administration to accelerate renal clearance. To reduce uptake in the salivary glands, cooling, using standard cooling packs, was started 30 min prior to therapy and maintained for up to 4 h after injection. To confirm 177Lu-PSMA-617 uptake and retention in tumor lesions at least one whole-body planar scintigraphic scan and a SPECT/CT scan of the thorax and abdomen was performed 48 h after injection. 177Lu-PSMA-617 RLT was repeated every 8 weeks until progression, death or withdrawal of consent by the patient.
Evaluation of response and survival
Patients were followed-up for OS as the primary clinical endpoint. The response rate was also evaluated using the Prostate Cancer Working Group 3 (PCWG3) criteria considering a PSA decline ≥50% as biochemical response [12]. Additionally, any change in PSA values was documented and analyzed. Patients received a median of three therapy cycles (Range: 1–8). Because of an inhomogeneous number of cycles, the focus of this study was on the best PSA changes occurring after the first cycle before receiving the second cycle and these parameters were analyzed according to effect on survival.
OS was defined as the time from the first therapy cycle to death from any cause. Multiple blood parameters were measured at baseline and were correlated with OS. A median baseline level for PSA of 360 ng/mL, the established useful cut-off value for ALP of 220 U/L and normal upper value for LDH, 225 U/L, were chosen as cut-off values for further differentiation of subgroups of patients.
Statistics
SPSS Statistics 24 (IBM Corporation, Somers, NY, USA) and R version 3.2.5 were used for statistical analysis. Descriptive statistics are reported as median and IQR for continuous variables and frequencies for categorical variables. To evaluate the impact on OS, multiple baseline and follow-up parameters were analyzed using Kaplan-Meier estimators, log-rank tests, and Cox proportional hazards regression models. The results of the regression analyses are presented as hazard ratios (HR) with corresponding 95% confidence intervals (95%CI). The optimal cutpoint for PSA decline was determined using maximally selected rank statistics (maxstat package for R using pmethod = “Lau94” for determination of the upper bound of the p-value) [13]. All inferential statistics are intended to be exploratory and are interpreted accordingly. P-values p ≤ 0.05 are considered statistically noticeable.
Results
Baseline demographics and clinical characteristics of all patients are given in Table 1. A total of 351 cycles of 177Lu-PSMA-617 were applied in 104 patients. The median administered dose was 6.1 GBq (IQR 5.9–6.3) and median cumulative injected activity in each patient was 18.8 GBq (IQR 12.9–24.75). A median of three cycles of 177Lu-PSMA-617 RLT were administered (range one to eight cycles). Three patients (3%) received only one cycle of the therapy and died within 16 weeks after therapy application (8, 9 and 16 weeks) because of disease progression or not willing to receive further therapy.
A total of 51 patients (49%) died during the observation period. The majority of patients (97%) presented with bone metastases, 77% with lymph node metastases and 32% with visceral metastases. All patients were treated with at least one line of chemotherapy prior to 177Lu-PSMA-617 RLT. Either abiraterone or enzalutamide had been given in 100% of the patients. Abiraterone was given in 80% and enzalutamide in 82% of patients. Both next generation antihormonal therapies had been given in 61% of the patients (Table 1).
Response and survival
Follow-up data after the first cycle of 177Lu-PSMA-617 RLT concerning PSA response was available for all patients. Any PSA decline occurred in 70 (67%) and a PSA decline ≥50% in 34 (33%) of patients after the first cycle prior to receiving further therapy cycles.
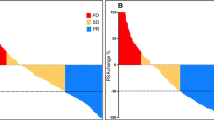
For the entire cohort, the Kaplan-Meier analysis showed a median OS of 56.0 weeks (95%CI: 50.5–61.5; Fig. 1). Univariate analysis of OS showed a noticeable longer median OS of 62.9 weeks (95%CI: 51.5–74.3) in patients with any PSA decline than in patients with PSA progression (47 weeks; 95%CI: 39.5–54.6; log-rank P = 0.004; Fig. 2a) with a hazard ratio of 0.38 (95%CI: 0.19–0.67, P = 0.005). A comparison between patients with a PSA decline ≥50% and <50% did not show a significant difference concerning OS (log rank P = 0.1; Fig. 2b). However, stepwise analyzing the percentage of PSA decline after the first cycle as a prognosticator of OS, a PSA decline ≥20.87% was determined as the best cutpoint with a median OS of 68 weeks (95%CI: 57.9–78.1) vs. 44 weeks (95%CI: 38.5–49.5; Fig. 3) and a hazard ratio of 0.28 (95%CI: 0.15–0.51). Adjusting for the fact that several cutpoints were tested, the upper bound of the p-value for the comparison of the two groups is 0.015. Baseline ALP levels <220 U/L and the cumulative injected activity ≥18.8 GBq were also associated with a longer OS (Table 2). For the analysis of the cumulative injected activity the inhomogeneous number of cycles must be kept in mind, i.e., patients with longer survival had a higher chance to receive a higher cumulative activity. Initial PSA and LDH levels, presence of visceral metastases, prior secondline chemotherapy and prior therapy with Radium-223 did not show any noticeable association with OS in the univariate analyses (Table 2).
A multivariable analysis with parameters with a p-value ≤0.05 in the univariate analyses was performed, additionally adjusting for age and presence of visceral metastases. A PSA decline ≥20.87% remained an independently strong prognosticator of OS. Absence of visceral metastases may also prolong survival, but did not reach statistical significance in the present population (Fig. 4).
Discussion
In the present study data from 104 mCRPC patients treated with a total of 351 cycles of 177,Lu-PSMA-617 RLT were retrospectively analyzed with focus on OS and parameters being prognostic for longer OS. Patients were already pretreated with at least one line of chemotherapy and one of the next generation anti-hormonal therapies. In this group of heavily pretreated mCRPC patients the established biochemical response defined as a PSA decline ≥50% [12] was not prognostic for a longer OS, whereas any PSA decline after the first cycle was noticeably associated with a longer OS (62.9 vs 47.0 weeks). These results are in line with prior works in rather more inhomogeneous and smaller groups of patients: 56 vs. 29 weeks by Bräuer et al. and 68 vs. 33 weeks by Ahmadzadehfar et al. showing the better outcome in patients with any PSA decline after the first therapy cycle [2, 6]. Furthermore, a step-by-step analysis of percentage of PSA decline revealed that a PSA decline of ≥20.87% was as a strong prognosticator of longer OS in the present study. These results underline that classically defined biochemical response may not be the most relevant prognosticator for survival. The presence of visceral metastases was previously reported to be predictive for response to 177Lu-PSMA-617 RLT [3]. In the present study visceral metastases did not have any effect on outcome in the univariate analysis. In the multivariate analysis a PSA decline ≥20.87% was a strong prognosticator of OS (Fig. 4). Prior second line chemotherapy and Radium-223, initial ALP, PSA and LDH levels did not have a strong effect on survival, whereas the cumulative injected activity ≥18.8 GBq was associated with a longer OS in the univariate analysis. In this case there might be a bias due to patients with a longer survival receiving a higher number of therapy cycles resulting in higher cumulative activity.
Rahbar et al. previously reported a significantly longer OS in a preliminary analysis of patients treated with 177Lu-PSMA-617 RLT comparing to a historical population being treated with best supportive care prior to availability of 177Lu-PSMA-617 [4]. Kulkarni et al. reported of outcome in a heterogeneous group of patients at an earlier stage of disease being treated by different 177Lu labeled PSMA targeting ligands, but at the time of the analysis the median OS was not reached [14]. Baum et al. reported a median OS of 13.7 months also in a healthier group of patients at an earlier stage of disease with a history of chemotherapy in only 44% of the patients and abiraterone and enzalutamide was given in 37% and 20% of the patients prior to PSMA targeted therapy [15], therefore the results are not comparable with the presenting analysis of patients pretreated with chemotherapy in 100%.
The Comet-1 trial was a randomized controlled trial in a comparable group of equally heavily pretreated mCRPC patients with OS as the primary endpoint [16]. Patients were progressive and were treated with at least one line of chemotherapy and abiraterone and/or enzalutamide. OS was compared between those treated with cabozantinib plus placebo and those treated with prednisone plus placebo. OS in the cabozantinib group was 11.0 months vs. 9.8 month in the placebo group (log rank P = 0.213). Compared to this study the median OS in the present study was 13 months (56 weeks; Fig. 1) in patients treated with 177Lu-PSMA-617 RLT. Looking at the patients with any PSA decline and PSA decline ≥20.87% after the first cycle the median OS was even longer with 14.7 and 16.0 months, respectively, (62.9 and 68 weeks, Table 2). 177Lu-PSMA-617 RLT seems to be effective in prolonging OS, especially in those patients responding to the first cycle of therapy. These results have to be confirmed in prospective controlled studies, but they give us the direction for planning future randomized trials. 177Lu-PSMA-617 RLT has the potential to change the management of mCRPC patients in the future. Although currently 177Lu-PSMA-617 RLT is performed in patients with advanced disease and multiple prior treatments, the improvement of survival might be significant.
Conclusions
177Lu-PSMA-617 RLT is a new and effective therapeutic and seems to prolong survival in patients with advanced mCRPC pretreated with chemotherapy, abiraterone and/or enzalutamide. The effect on survival is even greater in patients already responding to the first cycle of the therapy, especially if a PSA decline ≥20.87% occurs. The time is ready for prospective clinical trials to bring this promising therapeutic to regulatory approval.
References
Ahmadzadehfar H, Rahbar K, Kurpig S, et al. Early side effects and first results of radioligand therapy with (177)Lu-DKFZ-617 PSMA of castrate-resistant metastatic prostate cancer: a two-centre study. EJNMMI Res. 2015;5:114.
Brauer A, Grubert LS, Roll W, et al. 177Lu-PSMA-617 radioligand therapy and outcome in patients with metastasized castration-resistant prostate cancer. Eur J Nucl Med Mol Imaging. 2017.
Rahbar K, Ahmadzadehfar H, Kratochwil C, et al. German Multicenter Study Investigating 177Lu-PSMA-617 Radioligand Therapy in Advanced Prostate Cancer Patients. J Nucl Med. 2017;58:85–90.
Rahbar K, Bode A, Weckesser M, et al. Radioligand Therapy With 177Lu-PSMA-617 as A Novel Therapeutic Option in Patients With Metastatic Castration Resistant Prostate Cancer. Clin Nucl Med. 2016;41:522–8.
Rahbar K, Schmidt M, Heinzel A, et al. Response and Tolerability of a Single Dose of 177Lu-PSMA-617 in Patients with Metastatic Castration-Resistant Prostate Cancer: A Multicenter Retrospective Analysis. J Nucl Med. 2016;57:1334–8.
Ahmadzadehfar H, Wegen S, Yordanova A, et al. Overall survival and response pattern of castration-resistant metastatic prostate cancer to multiple cycles of radioligand therapy using [177Lu]Lu-PSMA-617. Eur J Nucl Med Mol Imaging. 2017;44(9):1448–54.
Ahmadzadehfar H, Zimbelmann S, Yordanova A, et al. Radioligand therapy of metastatic prostate cancer using 177Lu-PSMA-617 after radiation exposure to 223Ra-dichloride. Oncotarget. 2017;8:55567–74.
Fendler WP, Kratochwil C, Ahmadzadehfar H, et al. 177Lu-PSMA-617 therapy, dosimetry and follow-up in patients with metastatic castration-resistant prostate cancer. Nuklearmedizin. 2016;55:123–8.
Yordanova A, Becker A, Eppard E, et al. The impact of repeated cycles of radioligand therapy using [177Lu]Lu-PSMA-617 on renal function in patients with hormone refractory metastatic prostate cancer. Eur J Nucl Med Mol Imaging. 2017.
Fendler WP, Reinhardt S, Ilhan H, et al. Preliminary experience with dosimetry, response and patient reported outcome after 177Lu-PSMA-617 therapy for metastatic castration-resistant prostate cancer. Oncotarget. 2017;8:3581–90.
Cornford P, Bellmunt J, Bolla M, et al. EAU-ESTRO-SIOG Guidelines on Prostate Cancer. Part II: Treatment of Relapsing, Metastatic, and Castration-Resistant Prostate Cancer. Eur Urol. 2017;71:630–42.
Scher HI, Morris MJ, Stadler WM, et al. Trial Design and Objectives for Castration-Resistant Prostate Cancer: Updated Recommendations From the Prostate Cancer Clinical Trials Working Group 3. J Clin Oncol. 2016;34:1402–18.
Hothorn TaL B. On the Exact Distribution of Maximally Selected Rank Statistics. Computational Statistics & Data Analysis. 2003;43:121–37.
Kulkarni HR, Singh A, Schuchardt C, et al. PSMA-Based Radioligand Therapy for Metastatic Castration-Resistant Prostate Cancer: The Bad Berka Experience since 2013. J Nucl Med. 2016;57:97S–104S.
Baum RP, Kulkarni HR, Schuchardt C, et al. Lutetium-177 PSMA radioligand therapy of metastatic castration-resistant prostate cancer: Safety and efficacy. J Nucl Med. 2016;57(7):1006–13.
Smith M, De Bono J, Sternberg C, et al. Phase III Study of Cabozantinib in Previously Treated Metastatic Castration-Resistant Prostate Cancer: COMET-1. J Clin Oncol. 2016;34:3005–13.
Acknowledgments
We thank the Radiochemistry Group at both Departments of Nuclear Medicine for their highly reliable production of 177Lu-PSMA-617, and the nursing staff and the nuclear medicine technologists for their support.
Disclosures
The University of Münster received consulting fees from ABX Advanced Biochemical Compounds, Radeberg, Germany for K.R., M.B. and M.E.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no financial or non-financial competing interests.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. This article does not contain any studies with animals performed by any of the authors. This study was approved by the local ethics committee (No. 2016–585-f-S, Ethikkommission der Ärztekammer Westfalen-Lippe und der Westfälischen Wilhelms-Universität Münster).
Rights and permissions
About this article
Cite this article
Rahbar, K., Boegemann, M., Yordanova, A. et al. PSMA targeted radioligandtherapy in metastatic castration resistant prostate cancer after chemotherapy, abiraterone and/or enzalutamide. A retrospective analysis of overall survival. Eur J Nucl Med Mol Imaging 45, 12–19 (2018). https://doi.org/10.1007/s00259-017-3848-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-017-3848-4