Abstract
Purpose
The aim of the study was to compare sequential 177Lu-DOTA-TATE planar scans (177Lu-DOTA-TATE) in patients with metastasized neuroendocrine tumours (NET) acquired during peptide receptor radionuclide therapy (PRRT) for dosimetry purposes with the pre-therapeutic 68Ga-DOTA-TATE positron emission tomography (PET)/CT (68Ga-DOTA-TATE) maximum intensity projection (MIP) images obtained in the same patients concerning the sensitivity of the different methods.
Methods
A total of 44 patients (59 ± 11 years old) with biopsy-proven NET underwent 68Ga-DOTA-TATE and 177Lu-DOTA-TATE imaging within 7.9 ± 7.5 days between the two examinations. 177Lu-DOTA-TATE planar images were acquired at 0.5, 2, 24, 48 and 72 h post-injection; lesions were given a score from 0 to 4 depending on the uptake of the radiopharmaceutical (0 being lowest and 4 highest). The number of tumour lesions which were identified on 177Lu-DOTA-TATE scans (in relation to the acquisition time after injection of the therapeutic dose as well as with regard to the body region) was compared to those detected on 68Ga-DOTA-TATE studies obtained before PRRT.
Results
A total of 318 lesions were detected; 280 (88%) lesions were concordant. Among the discordant lesions, 29 were 68Ga-DOTA-TATE positive and 177Lu-DOTA-TATE negative, whereas 9 were 68Ga-DOTA-TATE negative and 177Lu-DOTA-TATE positive. The sensitivity, positive predictive value and accuracy for 177Lu-DOTA-TATE as compared to 68Ga-DOTA-TATE were 91, 97 and 88%, respectively. Significantly more lesions were seen on the delayed (72 h) 177Lu-DOTA-TATE images (91%) as compared to the immediate (30 min) images (68%). The highest concordance was observed for bone metastases (97%) and the lowest for head/neck lesions (75%). Concordant lesions (n = 77; mean size 3.8 cm) were significantly larger than discordant lesions (n = 38; mean size 1.6 cm) (p < 0.05). No such significance was found for differences in maximum standardized uptake value (SUVmax). However, concordant liver lesions with a score from 1 to 3 in the 72-h 177Lu-DOTA-TATE scan had a lower SUVmax (n = 23; mean 10.9) than those metastases with a score of 4 (n = 97; mean SUVmax 18) (p < 0.05).
Conclusion
Although 177Lu-DOTA-TATE planar dosimetry scans exhibited a very good sensitivity for the detection of metastases, they failed to pick up 9% of lesions seen on the 68Ga-DOTA-TATE PET/CT. Three-dimensional dosimetry using single photon emission computed tomography/CT could be applied to investigate this issue further. Delayed (72 h) images are most suitable for drawing regions of interest for dosimetric calculations.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Neuroendocrine tumours (NET) are rare neoplasms that secrete peptides and neuroamines. They can cause distinct clinical syndromes, including carcinoid syndrome, but many are clinically silent until late presentation with mass effect [1]. Most NET express a high density of somatostatin receptors, mainly subtype 2, which eventually get internalized after ligand binding [2]. These characteristics allow imaging by somatostatin receptor scintigraphy (typically with 111In-labelled pentetreotide or 99mTc-labelled somatostatin analogues) as well as peptide receptor radionuclide therapy (PRRT).
68Ga-DOTA-TATE, a selective somatostatin receptor subtype 2 positron emission tomography (PET) radiotracer [3], is an emerging diagnostic tool for effectively identifying primary tumours and for assessing the extent of the disease with high spatial resolution [4–7]. The maximum standardized uptake value (SUVmax) of tumour lesions on 68Ga-DOTA-TATE PET/CT has recently been shown to correlate with the receptor density [8, 9].
Treatment should be highly individualized based on NET characteristics, previous therapies and general health status of the patient. PRRT with 177Lu-DOTA-TATE has proven to be an effective and safe therapy option [10–15]. 177Lu has a half-life of 6.7 days and emits both beta radiation of 497 keV (78%) and gamma radiation of 208.3 keV (11%), allowing imaging and dosimetry after therapy. Dosimetry calculations for PRRT are usually performed using 177Lu-DOTA-TATE whole-body planar scans (177Lu-DOTA-TATE) acquired during PRRT.
The purpose of this study was to compare 177Lu-DOTA-TATE dosimetry (planar) scans acquired during PRRT in metastasized NET with 68Ga-DOTA-TATE PET/CT maximum intensity projection (MIP) images to find the sensitivity of the two different methods for the detection of metastases, which could have consequences for answering the question of whether all the lesions seen on PET/CT are taken into consideration for tumour dosimetry calculations.
Materials and methods
Patients
We retrospectively reviewed 44 consecutive patients (30 male, 14 female aged 23–83 years, mean 59 ± 11 years) with histologically proven NET (31 gastroenteropancreatic NET, 6 pulmonary carcinoids, 2 cases of medullary thyroid cancer, 1 renal NET and 4 cases of metastatic NET of unknown primary) (Table 1). All of the patients were enrolled at the Department of Nuclear Medicine/PET Centre, Bad Berka, Germany, where the whole study was performed. They were all referred for PRRT with 177Lu-DOTA-TATE due to progressive disease after failure of other treatment modalities. All patients underwent examinations with 68Ga-DOTA-TATE and 177Lu-DOTA-TATE within 7.9 ± 7.5 days between the studies. The patients did not receive any additional therapy in between the two scans.
Radiochemistry
The radiopharmaceuticals were prepared in the radiopharmacy of the Zentralklinik Bad Berka (Bad Berka, Germany).
Preparation of 68Ga-DOTA-TATE
The elution of 68Ga from the 68Ge/68Ga generator (obtained from Eckert and Ziegler, Berlin, Germany) as well as its labelling and associated quality control were performed as described in a previous publication [16]. 68Ga labelling of DOTA-TATE was performed following the procedure described by Meyer et al. [17]. The processed and eluted 68Ga fraction was directly transferred to solutions containing 30–50 μg of DOTA-TATE. Labelling yields of >95% were achieved within 10 min. Overall yields reached 70% at 20 min after generator elution relative to the eluted 68Ga activity, not corrected for decay.
Preparation of 177Lu-DOTA-TATE
The 177Lu labelling of the DOTA-peptides was performed as previously described [18]. Briefly, a solution of 500 μg of 2.5-dihydroxybenzoic acid and 20 μg of the corresponding DOTA-peptide in 50 μl of 0.4-M sodium acetate buffer (pH, 5.5) was added to a solution of 1 GBq of 177Lu in 30 μl of 0.05-M HCl. The mixture was heated to 90°C for 30 min and then diluted with 0.9% saline solution, followed by a sterile filtration. Quality control was performed by RP-18 HPLC, solvent A: water, solvent B: acetonitrile (both with 0.1% trifluoroacetic acid), gradient: 0–2 min 95% A, 20 min 95% B, flow rate: 1.2 ml/min, column: LiChrospher 100 RP-18 EC-5 μm 250×4 mm. The radiochemical purity was always greater than 99%. Samples were taken for sterility and pyrogenicity testing.
Imaging
68Ga-DOTA-TATE PET/CT
All 68Ga-DOTA-TATE PET/CT scans were accomplished using a dedicated hybrid PET/CT, a camera combining a PET unit and a two-slice CT unit (Biograph Duo, Siemens Medical Solutions). The patients were advised to stop the long-acting formulation octreotide therapy 4–6 weeks before the PET/CT examination. The patients fasted for at least 6 h before the image acquisition and were asked to drink 1.5 l of water-equivalent oral contrast dispersion (Gastrografin). This dispersion is used routinely in PET/CT without known adverse side effects to the accumulation of 68Ga-DOTA-TATE. Whole-body examinations (brain to mid-thigh) were performed with the patient in a supine position with arms raised.
Starting 60 min after an intravenous injection of approximately 100 MBq of 68Ga-DOTA-TATE (containing 5–10 μg of DOTA-TATE peptide for 100 MBq 68Ga), a contrast-enhanced CT was acquired in the craniocaudal direction with a 30-s delay (administration of 100 ml of intravenous contrast by an automated injection pump). CT was performed in spiral mode using a continuous acquisition at 130 kVp, 115 mAs, 4-mm collimation, 5-mm slice width, a table feed of 8 mm per rotation at 0.8-s rotation time and 2.4-mm slice spacing. During the CT, our protocol uses a limited breath-hold technique: patients are asked to hold their breath in normal expiration only for the time that the CT takes to cover the lower lung and liver, which is typically less than 15 s.
Immediately after acquisition of the CT, a PET emission scan was performed without changing the patient’s position, starting in the caudocranial direction with the bladder/pelvis region being scanned first. Emission data were acquired at 7–8 bed positions for 1–3 min at each position (depending upon the patient’s weight and height) resulting in a total emission scan of no more than 24 min and a total PET/CT examination time of about 30 min (including patient positioning, CT and PET imaging).
177Lu-DOTA-TATE dosimetry whole-body scans
177Lu-DOTA-TATE dosimetry whole-body scans were obtained after intravenous injection of a therapeutic dose with a mean injected activity of 6,734 ± 936 MBq of 177Lu-DOTA-TATE (containing ~150 mg of DOTA-TATE peptide for 7.5 GBq 177Lu) under a nephroprotection regime [19]. For kidney protection, an infusion of 1,500 ml of a combination of lysine and arginine (Lys-Arg) was used; 500 ml of L-lysine HCl 5% plus 250 ml of L-arginine HCl 10% plus 250 ml of saline were mixed and brought to a pH of 7.4. The osmolarity of this solution was 400 mosmol/l, and it contained 25 g of lysine and 25 g of arginine. The infusion was started 30 min before the administration of the therapeutic dose and was continued for 4 h. The radiopharmaceutical was coadministered over 10–15 min by using a second infusion pump system. As reported, the coinfusion of amino acids reduces renal exposure and allows for higher absorbed doses to reach tumours [19]. Imaging was performed at 30 min (immediate) to 2 (early), 24, 48 and 72 h (delayed) post-injection (MedisoMedical Imaging Systems, Budapest, Hungary, X-RING and SPIRIT DH-V gamma camera; MeGP collimator; peak at 208 keV; 15% energy window; 15 cm/min).
Image interpretation
Images were independently visually interpreted on a HERMES workstation by two experienced nuclear medicine physicians. Images acquired at different time points on 177Lu-DOTA-TATE and 68Ga-DOTA-TATE MIP images were visually inspected in varying scales. In 177Lu-DOTA-TATE images, each focal, abnormal tracer uptake was recorded by anatomical localization and by an intensity uptake score following a 5-point scale adapted from the Krenning score for Octreoscan [12]: absence of uptake (grade 0), lower than (grade 1), equal to (grade 2) or greater than (grade 3) normal liver tissue or higher than normal spleen or kidney uptake (grade 4). In 68Ga-DOTA-TATE MIP images, each focal, abnormal tracer uptake was recorded by anatomical localization and SUVmax. Then lesions seen at different time points on 177Lu-DOTA-TATE were compared to the 68Ga-DOTA-TATE MIP images.
For regional analysis, the body was divided into five sections: head/neck, thorax, liver, abdomen/pelvis and bones. In patients with generalized metastases (more than 15 lesions), only 1 target lesion in each anatomical region was selected for the analysis.
In 115 of 318 lesions, the size of the metastasis (on CT) and the SUVmax values were also measured.
Histopathological confirmation was not possible in all of the lesions. Hence, the lesions detected on 68Ga-DOTA-TATE PET/CT were considered to be positive by follow-up for a minimum duration of 6 months with 68Ga-DOTA-TATE PET/CT, CT, MRI, 18F-fluorodeoxyglucose (FDG) PET/CT, ultrasound or biochemical markers.
Statistical analysis
Data were defined as mean ± SD or total number (%). Sensitivity, positive predictive value and accuracy were calculated for detection of lesions on 68Ga-DOTA-TATE and 177Lu-DOTA-TATE studies. Group comparisons between 68Ga-DOTA-TATE and 177Lu-DOTA-TATE were performed using an independent sample t test. Spearman’s correlation was applied for correlations between the different data. Separate analysis was made for concordant (177Lu-DOTA-TATE and 68Ga-DOTA-TATE positive) and discordant lesions (177Lu-DOTA-TATE negative 68Ga-DOTA-TATE positive or 177Lu-DOTA-TATE positive 68Ga-DOTA-TATE negative). The Statistical Package for the Social Sciences 15 (SPSS 15) statistical software system (SPSS® Inc., Chicago, IL, USA) was used for the calculations. A p value <0.05 was considered as statistically significant.
Results
Interlesional analysis
A total of 318 lesions were evaluated in this study; 280 (88%) lesions were concordant. Among the discordant lesions (12%), 29 (9%) were 68Ga-DOTA-TATE positive and 177Lu-DOTA-TATE negative and 9 (3%) were 68Ga-DOTA-TATE negative and 177Lu-DOTA-TATE positive.
The sensitivity, positive predictive value and accuracy for the detection of NET metastases for 177Lu-DOTA-TATE as compared to 68Ga-DOTA-TATE were 91, 97 and 88%, respectively.
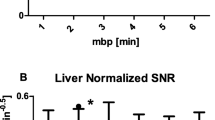
Significantly more lesions (Fig. 1) were seen on the delayed (72 h) 177Lu-DOTA-TATE images (91%) as compared to the immediate (30 min) (68%) or early (2 h) scans (82%) [p = 0.000 and odds ratio 1.4 (95% confidence interval 1.2–1.6)].
Among the concordant lesions, the highest agreement was observed for bone metastases (97%) followed by liver (88%), abdomen (83%) and lesions in the thorax (80%). The lowest concordance was found for metastases in the head/neck region (75%) (Fig. 2).
The analysis of tumour size showed that concordant lesions (n = 77; mean size 3.8 cm) were significantly larger than discordant lesions (n = 38; mean size 1.6 cm) (p < 0.05)
The analysis of SUVmax showed no significant difference between concordant and discordant lesions.
There was a significant correlation between the 72-h 177Lu-DOTA-TATE visual intensity scores and the 68Ga-DOTA-TATE SUVmax for lesions in the thorax, abdomen/pelvis and skeletal system (p < 0.05). This correlation was not shown for liver lesions. However, concordant liver lesions with a 177Lu-DOTA-TATE score from 1 to 3 in the 72-h 177Lu-DOTA-TATE scan had a lower SUVmax (n = 23; mean 10.9) than those metastases with a 177Lu-DOTA-TATE score of 4 (n = 97; mean SUVmax 18) (p < 0.05) (Tables 2 and 3).
Among the discordant lesions, 29 of 318 were 68Ga-DOTA-TATE positive and 177Lu-DOTA-TATE negative. Among these lesions, ten (34%) were located in the liver, eight (28%) in the abdomen/pelvis region, six (21%) in the thorax, three (10%) in the bone and two (7%) in the head/neck region. Fifteen (52%) of the 68Ga-DOTA-TATE positive and 177Lu-DOTA-TATE negative lesions were lymph nodes, ten (34%) liver metastases, three (10%) bone metastases and one pleural metastasis.
There were 9 of 318 lesions which were 68Ga-DOTA-TATE negative and 177Lu-DOTA-TATE positive (this pattern was observed in 5 patients overall). Seven lesions (in three patients) were found in the liver (Fig. 3), one was located in the pelvis and one in the neck.
Example of discordant lesions. a Liver metastases were only seen on 177Lu-DOTA-TATE planar scintigraphy performed 2 h after injection in a patient with NET. b 68Ga-DOTA-TATE PET/CT shows a hypodense lesion on the CT scan which reveals a donut pattern (MIP images, transverse PET, transverse CT and transverse fused PET/CT)
In this study, all lesions which were detected by either imaging modality were located in parts of the body covered by 68Ga-DOTA-TATE PET/CT (brain to mid-thigh). The 177Lu-DOTA-TATE scan did not reveal any additional lesion in the distal extremities.
Patient-based analysis
A total of 14 patients had 5 or less metastases, 20 had 6–8 and 10 had more than 10 metastases. In all patients, both imaging techniques were positive for at least one lesion; however, in six patients, 68Ga-DOTA-TATE showed malignant involvement in regions that were negative for 177Lu-DOTA-TATE (in two patients 68Ga-DOTA-TATE revealed liver metastases, in another two patients thoracic lesions and in two patients abdominal spread). In one patient, 177Lu-DOTA-TATE showed liver involvement that was not detected on 68Ga-DOTA-TATE.
Discussion
68Ga-DOTA-TATE is a somatostatin receptor subtype 2 ligand which is being increasingly used for somatostatin receptor PET/CT imaging of NET. It offers very good target to non-target imaging properties that make it an ideal PET radiotracer [7, 20]. 68Ga-DOTA-TATE PET/CT has proven to be an accurate method for detecting primary tumours and metastases, in particular for well-differentiated NET [21, 22]. The higher resolution of PET/CT systems with the ability of acquiring tomographic images, directly correlated to anatomy, results in significantly higher sensitivity for lesion detection when compared to 111In- or 99mTc-labelled somatostatin analogues [23].
In patients with inoperable or metastatic well-differentiated NET, PRRT with 177Lu-DOTA-TATE has shown very encouraging results [14, 18] with only minor side effects and good overall median duration of survival [13]. 177Lu, apart from intermediate beta energy (suitable for PRRT), also emits gammas suitable for scintigraphy and subsequent dosimetry. As part of the routine practise, in the Department of Nuclear Medicine/PET Centre, Bad Berka, Germany, all of the patients undergoing PRRT with 177Lu-DOTA-TATE are scanned in order to perform dosimetric calculations. Whole-body planar images are acquired and a standard with a known aliquot of the injected dose is also counted. In our study, we have retrospectively compared the dosimetric 177Lu-DOTA-TATE whole-body planar scans with the pre-therapeutic 68Ga-DOTA-TATE scans performed for diagnostic purposes. This is why the 177Lu-DOTA-TATE images included in this study are performed with a therapeutic dose of radioligand. As the 177Lu-DOTA-TATE images were performed for dosimetric purposes, no complementary projections or tomographic images were acquired.
As the aim of the study is to report to what extent lesions in NET detected on 68Ga-DOTA-TATE can also be identified on dosimetric 177Lu-DOTA-TATE scans under therapeutic conditions, data related to adverse reactions after the injection of the 177Lu-DOTA-TATE, side effects or therapeutic outcome of the PRRT are beyond the purpose of the study and are not reported.
In the present study, the 177Lu-DOTA-TATE planar dosimetry scan proved to be an accurate method for the detection of NET metastases with a sensitivity and positive predictive value of 91 and 97% as compared to 68Ga-DOTA-TATE and a lesion concordance rate between the two modalities of 88%. However, it failed to pick up 9% of lesions seen on 68Ga-DOTA-TATE PET/CT.
The highest concordance rate was found for the delayed 177Lu-DOTA-TATE scans. This observation may be related to the kinetics of 177Lu-DOTA-TATE with progressive continuous accumulation in NET metastases and decreasing background activity, which results in a better lesion to background ratio (Figs. 4 and Fig. 5). This was especially evident for bone metastases where physiological uptake in normal bone tissue is very low, providing a very good target to non-target ratio (Fig. 6).
Example of concordant lesions. 177Lu-DOTA-TATE planar dosimetry scan anterior projection, performed 24 h after injection (left) in a patient with NET, demonstrates multiple metastases in the mediastinum. The tumour uptake in the lesions is higher than normal liver (grade 4). 68Ga-DOTA-TATE (right) (a MIP images, b coronal fused PET/CT, c transverse fused PET/CT and d CT) shows the same lesions (arrow) but with less intense uptake than on the 177Lu-DOTA-TATE scan. Activity standard for dosimetric purposes (star)
Sequential 177Lu-DOTA-TATE anterior planar scans for the same patient as in Fig. 4. The abnormal mediastinal uptake is higher in the delayed scan. Observe the decreasing background activity over time. Notice the activity in the gall bladder in the 24-h scan (arrow) and the subsequent increasing bowel activity due to the physiological elimination in the following images. Activity standard for dosimetric purposes (star)
The remaining “discordant group” (12%) could be classified into two categories:
-
First, 68Ga-DOTA-TATE positive/177Lu-DOTA-TATE negative: Most of the lesions missed by 177Lu-DOTA-TATE in this group were found in the liver and in the abdomen/pelvis region and were lymph nodes. The most probable explanation for these findings is that, in these regions, there is a high background activity due to physiological elimination of the radiotracer through the bowel, as well as organ superposition, which result in a lower sensitivity in planar images, especially in small structures like lymph nodes. This assumption is supported by the fact that concordant lesions were significantly larger than discordant lesions (Table 4). In addition, the 68Ga-DOTA-TATE studies provide a higher spatial resolution than the 177Lu-DOTA-TATE planar images [21–23]. Therefore, dosimetry calculations based on planar 177Lu-DOTA-TATE scans will miss some of the lesions. This could possibly be avoided by three-dimensional dosimetry using single photon emission computed tomography (SPECT)/CT. This hybrid modality has proven to increase the diagnostic accuracy [24, 25] and its use for dosimetry has already been suggested [26, 27]; however, SPECT/CT needs to prove its superiority for dosimetric purposes.
Table 4 Concordant and discordant lesions according to SUVmax and size
Another possible explanation for this discrepancy could be that the tumour cell accumulation capacity under therapeutic conditions may be altered due to the biological effects of the radiation from the 177Lu-DOTA-TATE. In our opinion, this explanation is not very probable because the radiobiological effects are more likely to be expected in the delayed scans and especially in the lesions with a very high uptake. However, the delayed scans showed more lesions when compared to the immediate or early scans and the lesions with very high uptake are more likely to be visible in the scans.
-
Second, 68Ga-DOTA-TATE negative/177Lu-DOTA-TATE positive: This pattern was present for nine lesions in five patients and mainly observed in liver lesions in a few patients (seven lesions in three patients). Two of these 177Lu-DOTA-TATE positive liver lesions (in one patient) were detected 6 months later, also by 68Ga-DOTA-TATE, performed after 177Lu-DOTA-TATE therapy. The explanation for the disparity between the imaging procedures in these lesions may concern the somatostatin analogue compound labelled to the radioisotope (in our study the same compound with different concentrations); the physical and chemical characteristics of the two different radioisotopes (68Ga and 177Lu) as well as the dose injected; and the histological characteristics of the lesions (in our study a wide and heterogeneous group) as well as the intra-individual variations.
Although 177Lu-DOTA-TATE and 68Ga-DOTA-TATE had been performed with the same somatostatin analogue (DOTA-TATE), a possible explanation for the difference in radiotracer uptake could be the higher amount of peptide used for 177Lu-DOTA-TATE therapy. Velikyan et al. studied nine patients with gastroenteropancreatic NET. Six of them underwent three sequential PET/CT examinations with intravenous injections of 68Ga-DOTATOC (specific radioactivity: 25–50 MBq/nmol) preceded by 0.50 and 250 or 500 μg (0, 49, 245 and 491 nmol, respectively) octreotide, which was administered 10 min before the tracer injection. The tracer accumulation in the tumours varied and depended on the total amount of the peptide administered. Compared to the baseline study (0 μg of octreotide), there was first an increase (50 μg of octreotide) and then a decrease (250 or 500 μg of octreotide) in the tumour uptake. An exception was one patient showing a continuously increasing tumour uptake. Interestingly, a significant mass effect was also found on the tracer binding in non–tumoural tissues [28].
Discordances in the biodistribution of any molecule when labelled with different radionuclides for diagnostic or therapeutic purposes are known. Small structural modifications, chelator substitution or metal replacement were shown to considerably affect the binding affinity [3, 18, 29–31]. A possible explanation for the image discrepancies could be related to the different characteristics of the two chemical elements (ionic radii, radiometal effects).
Another probable explanation could be the diverse radiotracer kinetics that depend on the lesion type (cystic or solid) and the differences in the biological half-life of the two radiotracers [3]. It is known that 177Lu-DOTA-TATE has a higher residence time in tumours when compared with other 177Lu-labelled peptides [32]. Possibly, in some type of (cystic) lesions, the uptake is very slow and the target to non-target ratio is only high enough to detect such a lesion long after the intravenous injection (“filling in effect”). It can be specifically observed in the liver, where the physiological uptake is relatively high (as compared for example to bone). This might explain why a lesion is only detected by a radiotracer with a long half-life like 177Lu-DOTA-TATE (160 h) and not by 68Ga-DOTA-TATE (68 min). Mirzaei et al. had reported additional lesions detected in therapeutic scans with 177Lu-DOTA-TATE when compared to the diagnostic scan performed with 99mTc-EDDA/HYNIC-TOC in the same patients. In their study, a diagnostic scan was performed 2–3 h after the radiotracer injection while the post-therapeutic scan was performed 24 and 72 h post-injection [26].
It is, therefore, of great interest to perform PET/CT studies in these patients with long-lived PET isotopes like 44Sc or 86Y, especially in pre-therapeutic use. Jamar et al. [19] and Helisch et al. [29] described the use of 86Y-DOTATOC before the therapeutic use of 90Y. However, the limited availability of these isotopes and their decay mode with additional high-energy gamma rays, requiring elaborate correction algorithms, preclude their routine clinical use. Nevertheless, in this case, the use of long-lived PET radiotracers enables a direct comparison in the same modality, avoiding the comparison between planar and tomographic images.
Of the other two lesions, one was found in the pelvis (possibly corresponding to an intrauterine lesion DD myoma) and one was found in the head/neck region. Unfortunately, there is no available histological confirmation for this latter lesion, but it was confirmed as an enlarging metastasis in follow-up studies.
Limitations of the study
The study included a rather heterogeneous population of different NET subtypes. This has to be balanced against a reasonably sized patient population, especially with regard to the fact that these tumours are relatively rare. The histopathological confirmation was not ethically possible in patients with discordant lesions. However, each patient was widely studied with close clinical monitoring and follow-up studies for a minimum duration of 6 months with 68Ga-DOTA-TATE PET/CT, CT, MRI, 18F-FDG PET/CT, ultrasound or biochemical markers.
Conclusion
177Lu-DOTA-TATE planar dosimetry scans exhibit a very good sensitivity for the detection of metastases in NET, showing good concordance with 68Ga-DOTA-TATE. The highest concordance rate was found for the delayed 177Lu-DOTA-TATE (72 h) images, these being the most suitable images for dosimetric calculations. However, 177Lu-DOTA-TATE failed to pick up 9% of lesions seen on 68Ga-DOTA-TATE PET/CT. Most of the missed lesions were small lesions located in the liver and in the abdominal/pelvis region. The most probable explanation for this discrepancy is the better tomographic resolution of 68Ga-DOTA-TATE. Dosimetry calculations based only on planar 177Lu-DOTA-TATE scans will certainly miss some of the lesions, giving an erroneous result. This could possibly be avoided by three-dimensional dosimetry using SPECT/CT. A few lesions were only detected on 177Lu-DOTA-TATE scans. Higher amounts of peptides used in 177Lu-DOTA-TATE scans, different radiotracer kinetics, depending on the lesion type, or differences in the chemical or physical radionuclide characteristics might explain this discrepancy.
References
Modlin IM, Oberg K, Chung DC, Jensen RT, de Herder WW, Thakker RV, et al. Gastroenteropancreatic neuroendocrine tumours. Lancet Oncol 2008;9:61–72.
Reubi JC. Peptide receptors as molecular targets for cancer diagnosis and therapy. Endocr Rev 2003;24:389–427.
Reubi JC, Schär JC, Waser B, Wenger S, Heppeler A, Schmitt JS, et al. Affinity profiles for human somatostatin receptor subtypes SST1-SST5 of somatostatin radiotracers selected for scintigraphic and radiotherapeutic use. Eur J Nucl Med 2000;27:273–82.
Baum RP, Prasad V, Hommann M, Hörsch D. Receptor PET/CT imaging of neuroendocrine tumors. Recent Results Cancer Res 2008;170:225–42.
Prasad V, Ambrosini V, Hommann M, Hoersch D, Fanti S, Baum RP. Detection of unknown primary neuroendocrine tumours (CUP-NET) using (68)Ga-DOTA-NOC receptor PET/CT. Eur J Nucl Med Mol Imaging 2010;37:67–77.
Sainz-Esteban A, Prasad V, Carril JM, Baum RP. Pancreatic neuroendocrine tumor with involvement of the inferior mesenteric vein diagnosed by Ga-68 DOTA-TATE PET/CT. Clin Nucl Med 2010;35:40–1.
Srirajaskanthan R, Kayani I, Quigley AM, Soh J, Caplin ME, Bomanji J. The role of 68Ga-DOTATATE PET in patients with neuroendocrine tumors and negative or equivocal findings on 111In-DTPA-octreotide scintigraphy. J Nucl Med 2010;51:875–82.
Miederer M, Seidl S, Buck A, Scheidhauer K, Wester HJ, Schwaiger M, et al. Correlation of immunohistopathological expression of somatostatin receptor 2 with standardised uptake values in 68Ga-DOTATOC PET/CT. Eur J Nucl Med Mol Imaging 2009;36:48–52.
Kaemmerer D, Peter L, Lupp A, Schulz S, Sänger J, Prasad V, et al. Molecular imaging with (68)Ga-SSTR PET/CT and correlation to immunohistochemistry of somatostatin receptors in neuroendocrine tumours. Eur J Nucl Med Mol Imaging 2011;38:1659–68.
Forrer F, Valkema R, Kwekkeboom DJ, de Jong M, Krenning EP. Neuroendocrine tumors. Peptide receptor radionuclide therapy. Best Pract Res Clin Endocrinol Metab 2007;21:111–29.
van Essen M, Krenning EP, Bakker WH, de Herder WW, van Aken MO, Kwekkeboom DJ. Peptide receptor radionuclide therapy with 177Lu-octreotate in patients with foregut carcinoid tumours of bronchial, gastric and thymic origin. Eur J Nucl Med Mol Imaging 2007;34:1219–27.
Kwekkeboom DJ, Bakker WH, Kam BL, Teunissen JJ, Kooij PP, de Herder WW, et al. Treatment of patients with gastro-entero-pancreatic (GEP) tumours with the novel radiolabelled somatostatin analogue [177Lu-DOTA(0),Tyr3]octreotate. Eur J Nucl Med Mol Imaging 2003;30:417–22.
Kwekkeboom DJ, de Herder WW, Kam BL, van Eijck CH, van Essen M, Kooij PP, et al. Treatment with the radiolabeled somatostatin analog [177Lu-DOTA 0,Tyr3]octreotate: toxicity, efficacy, and survival. J Clin Oncol 2008;26:2124–30.
Kwekkeboom DJ, de Herder WW, van Eijck CH, Kam BL, van Essen M, Teunissen JJ, et al. Peptide receptor radionuclide therapy in patients with gastroenteropancreatic neuroendocrine tumors. Semin Nucl Med 2010;40:78–88.
Sowa-Staszczak A, Pach D, Chrzan R, Trofimiuk M, Stefańska A, Tomaszuk M, et al. Peptide receptor radionuclide therapy as a potential tool for neoadjuvant therapy in patients with inoperable neuroendocrine tumours (NETs). Eur J Nucl Med Mol Imaging 2011;38:1669–74.
Zhernosekov KP, Filosofov DV, Baum RP, Aschoff P, Bihl H, Razbash AA, et al. Processing of generator-produced 68Ga for medical application. J Nucl Med 2007;48:1741–8.
Meyer GJ, Mäcke H, Schuhmacher J, Knapp WH, Hofmann M. 68 Ga-labelled DOTA-derivatised peptide ligands. Eur J Nucl Med Mol Imaging 2004;31:1097–104.
Kwekkeboom DJ, Bakker WH, Kooij PP, Konijnenberg MW, Srinivasan A, Erion JL, et al. [177Lu-DOTA0Tyr3]octreotate: comparison with [111In-DTPA0]octreotide in patients. Eur J Nucl Med 2001;28:1319–25.
Jamar F, Barone R, Mathieu I, Walrand S, Labar D, Carlier P, et al. 86Y-DOTA0-D-Phe1-Tyr3-octreotide (SMT487)—a phase 1 clinical study: pharmacokinetics, biodistribution and renal protective effect of different regimens of amino acid co-infusion. Eur J Nucl Med Mol Imaging 2003;30:510–8.
Rufini V, Calcagni ML, Baum RP. Imaging of neuroendocrine tumors. Semin Nucl Med 2006;36:228–47.
Kayani I, Bomanji JB, Groves A, Conway G, Gacinovic S, Win T, et al. Functional imaging of neuroendocrine tumors with combined PET/CT using 68Ga-DOTATATE (DOTA-DPhe1,Tyr3-octreotate) and 18F-FDG. Cancer 2008;112:2447–55.
Kayani I, Conry BG, Groves AM, Win T, Dickson J, Caplin M, et al. A comparison of 68Ga-DOTATATE and 18F-FDG PET/CT in pulmonary neuroendocrine tumors. J Nucl Med 2009;50:1927–32.
Gabriel M, Decristoforo C, Kendler D, Dobrozemsky G, Heute D, Uprimny C, et al. 68Ga-DOTA-Tyr3-octreotide PET in neuroendocrine tumors: comparison with somatostatin receptor scintigraphy and CT. J Nucl Med 2007;48:508–18.
Wong KK, Cahill JM, Frey KA, Avram AM. Incremental value of 111-In pentetreotide SPECT/CT fusion imaging of neuroendocrine tumors. Acad Radiol 2010;17:291–7.
Even-Sapir E, Keidar Z, Bar-Shalom R. Hybrid imaging (SPECT/CT and PET/CT)–improving the diagnostic accuracy of functional/metabolic and anatomic imaging. Semin Nucl Med 2009;39:264–75.
Mirzaei S, Bastati B, Lipp RW, Knoll P, Zojer N, Ludwig H. Additional lesions detected in therapeutic scans with 177Lu-DOTATATE reflect higher affinity of 177Lu-DOTATATE for somatostatin receptors. Oncology 2011;80:326–9.
Sandström M, Garske U, Granberg D, Sundin A, Lundqvist H. Individualized dosimetry in patients undergoing therapy with (177)Lu-DOTA-D-Phe (1)-Tyr (3)-octreotate. Eur J Nucl Med Mol Imaging 2010;37:212–25.
Velikyan I, Sundin A, Eriksson B, Lundqvist H, Sörensen J, Bergström M, et al. In vivo binding of [68Ga]-DOTATOC to somatostatin receptors in neuroendocrine tumours—impact of peptide mass. Nucl Med Biol 2010;37:265–75.
Helisch A, Förster GJ, Reber H, Buchholz HG, Arnold R, Göke B, et al. Pre-therapeutic dosimetry and biodistribution of 86Y-DOTA-Phe1-Tyr3-octreotide versus 111In-pentetreotide in patients with advanced neuroendocrine tumours. Eur J Nucl Med Mol Imaging 2004;31:1386–92.
de Araújo EB, Caldeira Filho JS, Nagamati LT, Muramoto E, Colturato MT, Couto RM, et al. A comparative study of 131I and 177Lu labeled somatostatin analogues for therapy of neuroendocrine tumours. Appl Radiat Isot 2009;67:227–33.
Antunes P, Ginj M, Zhang H, Waser B, Baum RP, Reubi JC, et al. Are radiogallium-labelled DOTA-conjugated somatostatin analogues superior to those labelled with other radiometals? Eur J Nucl Med Mol Imaging 2007;34:982–9.
Esser JP, Krenning EP, Teunissen JJ, Kooij PP, van Gameren AL, Bakker WH, et al. Comparison of [(177)Lu-DOTA(0), Tyr(3)]octreotate and [(177)Lu-DOTA(0), Tyr(3)]octreotide: which peptide is preferable for PRRT? Eur J Nucl Med Mol Imaging 2006;33:1346–51.
Acknowledgments
The authors would like to thank the nursing and technician staff in the Bad Berka Zentralklinik for their technical assistance. We would also like to thank all supporting personnel of the radiopharmacy department for their expert help and effort. We are grateful for the support from the coworkers of the Department of Nuclear Medicine/PET Centre, Bad Berka, Germany. All of the patients included in this study were enrolled in the Department of Nuclear Medicine/PET Centre, Bad Berka, Germany. Image acquisition, patients’ therapeutic management and follow-up were also performed in the Department of Nuclear Medicine/PET Centre, Bad Berka, Germany. Aurora Sainz-Esteban performed this study during a European programme of internship abroad between the Zentralklinik Bad Berka (Bad Berka, Germany) and the Hospital Universitario Marqués de Valdecilla (Santander, Spain).
Authors’ contributions
Each author has contributed significantly to the submitted work. Aurora Sainz-Esteban contributed to the conception and design of the study, acquired, analysed and interpreted the data and wrote the manuscript. Vikas Prasad contributed to the conception and design of the study, performed the PET/CT and the dosimetry studies, analysed the data and critically revised and approved the final manuscript. Christiane Schuchardt and Carolin Zachert performed the dosimetry studies and critically read the manuscript. José Manuel Carril critically revised and approved the final manuscript. Richard Paul Baum conceived the study, performed the PET/CT and dosimetry studies and critically revised and approved the final manuscript.
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
The work was performed in the Department of Nuclear Medicine and Centre for PET/CT, Zentralklinik Bad Berka, Germany.
This work was presented in part at the EANM Congress 2009 in Barcelona.
The first two authors (Aurora Sainz-Esteban and Vikas Prasad) contributed equally to the work.
Rights and permissions
About this article
Cite this article
Sainz-Esteban, A., Prasad, V., Schuchardt, C. et al. Comparison of sequential planar 177Lu-DOTA-TATE dosimetry scans with 68Ga-DOTA-TATE PET/CT images in patients with metastasized neuroendocrine tumours undergoing peptide receptor radionuclide therapy. Eur J Nucl Med Mol Imaging 39, 501–511 (2012). https://doi.org/10.1007/s00259-011-2003-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-011-2003-x