Abstract
Purpose
The purpose of this study was to determine the incidence of incidental pituitary uptake on whole-body 18F-fluorodeoxyglucose (FDG) positron emission tomography/computed tomography (PET/CT) and to investigate its clinical significance.
Methods
The files of 40,967 patients who underwent whole-body FDG PET/CT were retrospectively reviewed. Quantification of pituitary metabolic activity was obtained by using the maximum standardized uptake value (SUVmax). Hormone assays and pituitary MRIs were performed to assess pituitary lesions.
Results
Focally increased pituitary FDG uptake on PET/CT was found in 30 of 40,967 patients, accounting for an incidence of 0.073%. The mean SUVmax of 30 patients was 8.9 ± 6.6 (range: 3.2–32.6). Histological diagnosis was obtained in three patients and included two growth hormone-secreting adenomas and one non-functioning adenoma. Hormone assays were performed on serum samples from 11 patients, 2 of whom were shown to have hypersecretion of pituitary hormone. MRI was performed on 19 patients. Abnormal MRI findings suggesting a pituitary mass were found in 18 of 19 cases (94.7%). The mean SUVmax calculated without correction for partial volume effect for macroadenomas was significantly higher than the SUVmax for microadenomas (11.5 ± 8.4 vs 4.8 ± 1.3; p < 0.05). There were no cases diagnosed with metastasis to the pituitary gland during clinical follow-up.
Conclusion
Incidental pituitary FDG uptake was a very rare finding. Cases with incidental pituitary FDG uptake were diagnosed primarily with clinically non-functioning adenomas, and there were also a few functioning adenomas. Further evaluations, including hormone assays and pituitary MRI, are warranted when pituitary uptake is found on FDG PET/CT.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Pituitary incidentalomas are defined as asymptomatic lesions of the pituitary gland found on magnetic resonance imaging (MRI) or computed tomography (CT) scans of the head performed for various purposes [1]. They can be classified by size and hormone secretion status. Pituitary incidentalomas <10 mm are classified as microadenomas and those >10 mm are macroadenomas. Independent of size, hormone hypersecretion can occur with any incidentaloma, but hyposecretion is usually the result of a macroadenoma [1]. Overall, the majority of pituitary adenomas are benign masses which remain clinically silent and are associated with minimal morbidity and mortality. The prevalence of pituitary incidentalomas discovered on autopsy is approximately 10%, with the majority of these lesions being microadenomas [1–3]. The prevalence of pituitary incidentalomas discovered on neuroimaging such as MRI or CT is approximately 3.7–20% [2, 4].
Positron emission tomography (PET)/CT using 18F-fluorodeoxyglucose (FDG) is increasingly performed for staging or localization of metastatic disease in patients with various kinds of malignancies. Pituitary glands do not normally accumulate FDG and are not visualized on FDG PET imaging [5]. Bergström et al. [6] reported that PET using 11C-methionine was a useful tool for diagnosis of pituitary adenoma and assessment of treatment response in patients with pituitary adenoma. Some studies have reported that functioning pituitary macro- and microadenomas can show increased FDG uptake on PET [7, 8]. However, a survey of FDG-avid pituitary incidentalomas in a large population has not yet been undertaken, although there have been a few case reports [5, 9, 10].
We performed a multicentre (Keimyung University Dongsan Medical Center, DSMC; Kyungpook National University Hospital, KNUH; and Yeungnam University Medical Center, YUMC) retrospective observational study to determine the incidence of incidental pituitary uptake on whole-body FDG PET/CT scans of suspected or known cancer patients, in addition to patients undergoing health screening. We also investigated the clinical significance of unexpected hypermetabolic foci in the pituitary gland using MRI and pituitary hormone assays.
Materials and methods
Patient population
The files of 40,967 patients (20,220 men and 20,747 women) who underwent whole-body FDG PET/CT for assessment of known or suspected malignancy (n = 35,147), benign disease or cancer screening (n = 5,820) from August 2004 to June 2009 were retrospectively reviewed. All patients in whom the written report mentioned an FDG-avid focus localized by PET/CT to the pituitary region, regardless of intensity of uptake and findings on the CT component, were identified and their histories reviewed. Patients with a prior history of pituitary tumour or prior imaging abnormalities in the pituitary gland were excluded. On the basis of an imaging study, a patient who had a possible osseous metastasis in the sella turcica indistinguishable from pituitary metastasis was also excluded. All patients provided informed consent for the PET/CT study, for assessment of their clinical records and for follow-up. Follow-up included clinical review, evaluations of any pituitary symptom and/or follow-up MRI or PET/CT. Regular oncological follow-ups in cancer patients were also performed according to each patient’s clinical situation and cancer type.
There were 30 patients, who had no previous known pituitary tumour and no prior clinical or imaging suspicion of abnormalities in the pituitary gland, who met the inclusion criteria and comprised the study group for calculating the incidence of unexpected focal FDG uptake in the pituitary gland on whole-body PET/CT. The mean age of the study group of 30 patients (male: 13 patients) was 62.3 ± 14.1 years (range: 29–84 years). Primary malignant tumours included thyroid cancer (n = 4), colorectal cancer (n = 3), breast cancer (n = 3), prostate cancer (n = 3), lung cancer (n = 2) and cervical cancer (n = 2). There were eight patients who each had ovary, bladder, kidney, periampullary, gastric, or endometrial cancer, malignant melanoma or cholangiocarcinoma. Five patients underwent PET/CT for assessment of large vessel inflammation (n = 2), neurofibromatosis (n = 1) and cancer screening (n = 2).
Imaging acquisition and analysis of FDG PET/CT
Patients were instructed to fast except for glucose-free oral hydration for 4–6 h before injection of 370–555 MBq FDG. PET/CT was performed at 60–90 min following FDG injection. Blood glucose levels were measured before the injection of the tracer to ensure levels below 11 mmol/l. Unenhanced CT and whole-body PET were acquired sequentially using three BGO PET/CT scanners (one Discovery ST and two Discovery STE 16 systems, General Electric Healthcare, Milwaukee, WI, USA) and an LSO PET/CT scanner (Reveal RT-HiREZ, CTI Molecular Imaging Inc., Knoxville, TN, USA). The imaging field for whole-body PET/CT included the pituitary region.
Data obtained from CT acquisition were used for attenuation correction and fusion with PET images. The PET data were reconstructed iteratively using ordered subset expectation maximization software provided by the manufacturers. PET, CT and PET/CT images of the whole body were displayed in the axial, coronal and sagittal planes and were reviewed on a dedicated workstation (AW, General Electric Healthcare, Milwaukee, WI, USA). PET data were also displayed in a rotating maximum intensity projection.
PET studies showing single, well-circumscribed foci of increased uptake localized to the pituitary fossa on PET/CT images were reviewed. The PET component was assessed for intensity of FDG uptake. A nuclear medicine physician measured the maximum standardized uptake value (SUVmax) using a manually drawn region of interest in the transaxial slice showing the most intense uptake in the pituitary lesion.
Assessment of pituitary function and pituitary MRI
For assessment of pituitary function, hormone assays were performed to determine the serum levels of growth hormone (GH), prolactin (PRL), thyrotropin, follicle-stimulating hormone, luteinizing hormone and adrenocorticotropin (ACTH). For assessment of pituitary mass effect, clinical reviews for symptoms such as headache and visual disturbances and MRIs of the pituitary region were undertaken. MRI was performed using four different MR machines, two 1.5-T MRI units (Intera, Philips Medical Systems, Best, The Netherlands; and Symphony, Siemens, Erlangen, Germany) and two 3-T MRI units (Signa Excite, General Electric Medical Systems, Milwaukee, WI, USA), at three different institutions according to different protocols. Copies of MR studies were sent to the study reference centre (KNUH) for a second opinion and were centrally reviewed by a neuroradiologist with 10 years of experience. As minimal requirements, studies with sagittal and coronal T1-weighted images (T1WIs) obtained with <3 mm slice thickness, and contrast-enhanced T1-weighted images (CE MRIs) after administration of gadolinium chelates, were considered adequate. On MRI, a pituitary lesion with low signal intensity on T1WIs and high signal intensity on T2-weighted images (T2WIs), or a nodule with different contrast enhancement patterns from adjacent normal glandular structure on CE MRIs, was considered to be a pituitary adenoma. In cases without definite visible mass on MRI, abnormal findings such as a laterally displaced pituitary stalk (stalk deviation), suprasellar extension of pituitary gland (upward bulging), high signal intensity focus on T2WIs (cystic change), polycyclic shape of pituitary gland (figure-8 configuration) and invasion of the cavernous sinus (cavernous invasion) were considered to be presumptive pituitary microadenomas.
Statistical analysis
Numeric data were expressed as mean ± standard deviation. The chi-square test was used to analyse the differences in incidence among subgroups by gender, age and reason for study. Student’s t test was used to analyse the difference in SUVmax between microadenomas and macroadenomas. The Mann-Whitney test was used to analyse SUVmax differences between patients with normal pituitary function and patients with hypersecretion of pituitary hormone. Correlation analyses between the SUVmax and pituitary gland size or volume were performed using Spearman’s rank correlation coefficient. A p value less than 0.05 was considered statistically significant.
Results
Focally increased FDG uptake localized to the pituitary gland by PET/CT was found in 30 of 40,967 patients, accounting for an incidence of 0.073% [95% confidence interval (CI): 0.047–0.099]. The clinical characteristics, hormone assay results, intensity of FDG uptake and MRI findings for the 30 patients are summarized in Table 1. The incidences of incidental pituitary uptake on FDG PET/CT were not significantly different for men versus women (men: 0.064%, 95% CI: 0.029–0.099; women: 0.082%, 95% CI: 0.043–0.121; p = 0.509) or among age subgroups (<50 years: 0.048%, 95% CI: 0.001–0.094; 50–69 years: 0.090%, 95% CI: 0.043–0.138; ≥70 years: 0.138%, 95% CI: 0.036–0.241; p = 0.216). The incidence of incidentally found pituitary lesions on FDG PET/CT in patients with known or suspected malignancy (0.071%, 95% CI: 0.043–0.099) was similar to that in patients with benign disease or undergoing cancer screening (0.086%, 95% CI: 0.011–0.161; p = 0.699). The overall intensity of FDG uptake in the final study group of 30 patients was a mean SUVmax of 8.9 ± 6.6 (range: 3.2–32.6).
Of the 30 patients, 4 complained of nonspecific headache, but none of the 30 patients (13 men and 17 women) complained of visual field defects or cranial nerve palsies. On physical examination, one patient had enlarged hands and feet, prognathism and frontal bossing, suggesting hypersecretion of GH. Assessment of pituitary function using hormone assays was performed in 11 patients, 2 of whom were shown to have hypersecretion of pituitary hormones. Elevated GH and PRL concentrations were seen in one patient, and elevated GH was noted in one patient. There was no significant difference in SUVmax between patients with normal hormone levels and patients with hormone hypersecretion (8.9 ± 5.2 vs 6.8 ± 1.2, respectively; p = 0.18). Histological diagnosis after surgical resection was obtained in three cases, including two GH-secreting adenomas and one non-functioning adenoma.
MRI of the pituitary gland was performed in 19 patients. Abnormal pituitary findings suggesting metastasis from a primary malignancy were not found in any patient. Gadolinium-enhanced pituitary masses on T1WIs were found in 11 of 19 patients (57.9%) with a mean diameter of 18.2 mm (range: 5.6–30.1 mm). Only 1 of 11 lesions (9.0%) detected on MRI was less than 10 mm in diameter. Of the 19 patients, 18 (94.7%) had abnormal MRI findings, suggesting a pituitary mass. Stalk deviation was found in 15 patients, upward bulging in 12 patients, visible mass in 11 patients, cystic change in 1 patient, figure-8 configuration in 2 patients and cavernous invasion in 5 patients. One case did not demonstrate any pituitary abnormality on MRI. Seven patients with abnormal MRI findings but no definite mass lesion were diagnosed with presumptive pituitary microadenoma. The mean SUVmax of ten macroadenomas was significantly higher than the mean SUVmax of eight microadenomas (11.5 ± 8.4 vs 4.8 ± 1.3, respectively; p < 0.05; Figs. 1 and 2).
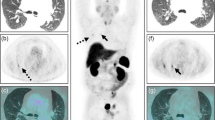
Focal FDG uptake in the pituitary region of a 62-year-old man, who underwent PET/CT for cancer screening. Transaxial and sagittal PET images (left) show focally increased FDG uptake (arrow) in the pituitary region (with a measured SUVmax of 17.8). Transaxial and sagittal PET/CT fusion images (centre) localize this focal uptake to the pituitary gland. Coronal and sagittal MRI images (right) demonstrate an 18.6-mm gadolinium-enhanced pituitary mass, stalk deviation and upward bulging. He was asymptomatic, and his serum pituitary hormone levels were within normal ranges. In clinical follow-ups, there were no developments of symptoms associated with either pituitary mass or hormonal abnormalities
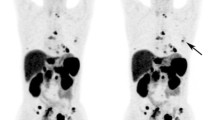
Except for three patients undergoing surgical resection, regular follow-ups (duration: 19.4 ± 3.5 months) were performed, including clinical examinations focused on relevant neurological and endocrinological symptoms. During the follow-up period, none of the patients complained of any symptoms associated with pituitary mass effect. Follow-up MRIs were performed in three patients 6–12 months after baseline studies. There were no significant changes in pituitary mass sizes found in any of the patients. Follow-up PET/CT scans were performed during or after completion of chemotherapy in six patients, and persistent pituitary FDG uptake was demonstrated, regardless of FDG uptake changes in the primary malignant lesion (Fig. 3). No case was diagnosed with metastasis to the pituitary gland or sella turcica during clinical oncological follow-ups of the cancer patients.
Pretreatment and post-chemotherapy 18F-FDG PET/CT scans in a 66-year-old man with small cell lung cancer. a Maximum intensity projection shows foci of increased FDG uptake in the left upper lung (arrow, with a measured SUVmax of 7.7) and left adrenal gland (arrowhead, with a measured SUVmax of 8.1). The hypermetabolic left lung mass was diagnosed as small cell lung cancer by transthoracic needle biopsy. b Sagittal PET/CT fusion image shows focal increased FDG uptake in the pituitary gland (with a measured SUVmax of 12.7). c Maximum intensity projection after six courses of chemotherapy shows almost complete disappearance of focal FDG uptake in the left upper lung (arrow) and decreased FDG uptake in the left adrenal gland (arrowhead, with a measured SUVmax of 5.3). d Sagittal PET/CT fusion image shows persistent FDG uptake in the pituitary gland (with a measured SUVmax of 12.1) regardless of FDG uptake changes in the left upper lung and adrenal gland
Discussion
FDG PET/CT is used in the evaluation of most types of malignancy, which has resulted in significant clinical impact on patient management [11, 12]. FDG PET/CT is more accurate than CT or other conventional imaging modalities for the diagnosis of previously unknown, recurrent or metastatic cancer foci, based in part on its whole-body imaging capabilities [12–14]. Incidental foci of abnormal FDG uptake detected during whole-body PET/CT examinations may represent previously unknown malignant sites, physiological variants or benign lesions unrelated to cancer [12–14]. There have been only a few cases with unexpected pituitary FDG uptake previously reported, primarily as case reports [5, 9, 10]. A large-scale study has not been reported. Our study is the first large survey of incidental pituitary FDG uptake on PET/CT, and it demonstrates that incidental pituitary FDG uptake on FDG PET/CT is a very rare finding. The incidence of unexpected FDG-avid pituitary lesions was 0.073%, which is much lower than the incidence of FDG-avid incidentalomas in other endocrine organs such as the thyroid and adrenal glands [15–18]. In addition, the incidence of unexpected FDG-avid pituitary lesions in our present study is much lower than that of incidentalomas found on autopsy or MRI [1–4]. We think that several factors may contribute to the different results. First, the majority of pituitary incidentalomas are microadenoma. In post-mortem studies, only a few cases of macroadenoma have been found [3, 19–21]. Buurman and Saeger [21] reported 334 microadenoma cases and only 3 macroadenoma cases from autopsies of 3,048 patients. Second, compared to MRI, whole-body PET/CT has lower spatial resolution for pituitary imaging. Whole-body PET/CT scans were performed on all patients. A majority of small pituitary masses cannot be found by whole-body PET/CT. Finally, not all pituitary adenoma are FDG-avid tumours. De Souza et al. [8] reported 12 positive findings of 17 functioning pituitary microadenomas. The incidence calculations in our present study only include FDG-avid pituitary tumours.
PET scanning is based on the principle that metabolically active cells such as those in tumours are more likely than normal cells to take up the labelled glucose analogue, FDG, to become detectable. It has been primarily used for patients with malignant tumours, where it is now a commonly used imaging technique for the staging and follow-up of those patients [22]. For benign tumours, PET scans are usually negative, because the cells of these tumours are metabolically less active than malignant cells. However, the cells of functioning tumours that are hormone-secreting tumours may be more active and therefore more likely to take up FDG. For Cushing’s syndrome, FDG PET has been reported to be beneficial in discovering the sites of ectopic ACTH secretion, since these ACTH-secreting tumours are usually small and difficult to localize [23, 24]. FDG PET for pituitary adenomas has been anecdotally reported [25, 26]. In the early 1990s, a study of 20 cases of pituitary microadenomas (17 cases with Cushing’s disease and 3 cases with acromegaly) reported that FDG PET was positive in 12 cases and MRI was positive in 13 cases. The study concluded that FDG PET was complementary to MRI, because FDG PET was positive in five cases with negative or questionable results on MRI [8]. Another study evaluated PET in 24 cases of pituitary macroadenomas, none of which were ACTH-secreting. The scans were positive in all cases, but there was no uptake that could characterize a specific type of hormone and there was no correlation between the amount of hormone secreted and PET activity [27]. Campeau et al. [5] also reported on a patient with mucosa-associated lymphoid tissue lymphoma who had a non-functioning pituitary adenoma with FDG avidity. Our present study also found no statistically significant difference in the SUVmax of pituitary lesions with or without pituitary hormone hypersecretion. The SUVmax of pituitary lesions were also not statistically different with regard to sex or underlying disease. Macroadenomas had higher FDG uptakes than microadenomas. There have been no reports on the mechanism of FDG uptake in functioning and non-functioning pituitary adenomas. However, the size of a pituitary mass would be considered one of the important factors for the degree of pituitary FDG uptake.
Pituitary tumours are difficult to detect radiologically. In our present study, 57.9% of patients who had a pituitary FDG focus were found to have a detectable pituitary mass on MRI. This result is believed to be due to the small sizes of the masses and possibly to inadequate MR protocol. The majority (65.4%) of microadenomas were reported to show peak enhancement at a point in time later than peak enhancement of the adjacent anterior pituitary lobe, and the remaining 34.6% of microadenomas were reported to be enhanced before enhancement of the anterior pituitary [28]. Although dynamic spin-echo imaging of the pituitary gland provides improved visibility and clarity of lesions compared with routine scanning after contrast administration, the detection rate has varied because of lesion sizes and enhancement patterns. In cases with enlarged pituitary glands seen on MRI, it was possible that there were invisible microadenomas on routine MRI scanning after contrast administration [29].
In our present study, there was no case diagnosed with metastasis to the pituitary gland or sella turcica during clinical follow-up of cancer patients. None of the patients complained of any symptoms associated with pituitary mass effect during the follow-up period, and significant changes in size suggesting pituitary metastasis were not found in follow-up MRI. Follow-up PET/CT demonstrated persistent pituitary FDG uptake regardless of FDG uptake changes in the primary malignant lesion. By contrast, Soussan et al. [10] reported that pituitary gland involvement of non-Hodgkin’s lymphoma was detected on FDG PET/CT, and follow-up PET/CT after chemotherapy demonstrated total disappearance of focal pituitary FDG uptake. Therefore, we can postulate that the persistent pituitary FDG uptake seen in our patients could reflect benign aetiology rather than metastasis. In our study, patients with a prior history of pituitary tumour or prior imaging abnormalities in the pituitary gland were excluded. We excluded a patient with pituitary FDG uptake that could not be distinguished from an osseous metastasis to the sella turcica. These factors may have affected the results of our present study.
There are several limitations to the current study. First, histological diagnoses were only made in three patients, and hormonal assays and MRI evaluation were not performed in all patients with incidental pituitary uptake on FDG PET/CT, because of the advanced status of the primary malignancy or clinician indifference. Second, our present study was conducted by retrospective review of the PET/CT reports written by ten readers over more than 4 years. Inter-reader variations of criteria used for measurement of pituitary uptake may exist. In each institute, however, evaluations of PET/CT images were based on the consensus of at least two nuclear medicine physicians, and there was no significant difference in the incidence of incidentalomas among the institutions (data not shown). Some readers could have seen a focus of pituitary FDG uptake but neglected to mention it, since this finding was not related to the pathology of interest and it would not have had an impact on management. However, interpretations of whole-body PET/CT scans have been performed according to systematic reviews, and significant incidental findings such as unexpected thyroid uptake or unexpected breast uptake were also recorded. Thus, there would be low probability that there was underestimation of the actual incidence. Third, there may be a question regarding the reliability of uptake measurements using SUVmax. SUV depends on acquisition, reconstruction and region of interest parameters. In our present study, four different PET/CT scanners were used. Thus, there may have been some differences between institutions regarding image acquisition and reconstruction. To minimize these differences, reconstruction processing and imaging analyses were performed at a single institution using the same software for all calculations. Similarly, we established minimum requirements for MRI images, because of the differences in MRI protocols. Finally, quantitative accuracy of PET is affected by partial volume effects that result in underestimation of the SUV, especially in small-sized lesions. In the present study, correction for partial volume effect was not performed, because some microadenomas were not identified on MRI as definite pituitary masses. In such cases, we cannot accurately measure the size of a microadenoma to perform partial volume correction.
From our survey of incidental pituitary uptake on FDG PET/CT, our study demonstrates that (1) the incidence of focal pituitary FDG uptake was 0.073% (95% CI: 0.047–0.099); (2) there were no cases of metastatic pituitary lesions, and the majority of patients with focal pituitary FDG uptake were considered to have clinically and radiologically non-functioning adenomas; (3) 2 of 3 patients undergoing surgical resection were diagnosed with GH-secreting adenomas; (4) 2 of 11 patients in whom hormonal assays were performed were shown to have pituitary hormone hypersecretion; (5) 18 of 19 patients undergoing MRI had abnormal findings suggesting pituitary adenoma; and (6) there was a significant difference in the mean SUVmax calculated without correction for partial volume effect between microadenomas and macroadenomas.
Conclusion
Incidental pituitary uptake on FDG PET was a very rare finding. Most cases of incidental pituitary FDG uptake were diagnosed as clinically non-functioning adenomas, and there were a few functioning adenomas. Further evaluations, including hormone assays and pituitary MRI, are warranted when pituitary uptake is found on FDG PET/CT.
References
Shirodkar M, Jabbour SA. Endocrine incidentalomas. Int J Clin Pract 2008;62:1423–31.
Molitch ME, Russell EJ. The pituitary “incidentaloma”. Ann Intern Med 1990;112:925–31.
Molitch ME. Pituitary tumours: pituitary incidentalomas. Best Pract Res Clin Endocrinol Metab 2009;23:667–75.
Chidiac RM, Aron DC. Incidentalomas. A disease of modern technology. Endocrinol Metab Clin North Am 1997;26:233–53.
Campeau RJ, David O, Dowling AM. Pituitary adenoma detected on FDG positron emission tomography in a patient with mucosa-associated lymphoid tissue lymphoma. Clin Nucl Med 2003;28:296–8.
Bergström M, Muhr C, Lundberg PO, Långström B. PET as a tool in the clinical evaluation of pituitary adenomas. J Nucl Med 1991;32:610–5.
Alzahrani AS, Farhat R, Al-Arifi A, Al-Kahtani N, Kanaan I, Abouzied M. The diagnostic value of fused positron emission tomography/computed tomography in the localization of adrenocorticotropin-secreting pituitary adenoma in Cushing’s disease. Pituitary 2009;12(4):309–14.
De Souza B, Brunetti A, Fulham MJ, Brooks RA, DeMichele D, Cook P, et al. Pituitary microadenomas: a PET study. Radiology 1990;177:39–44.
Koo CW, Bhargava P, Rajagopalan V, Ghesani M, Sims-Childs H, Kagetsu NJ. Incidental detection of clinically occult pituitary adenoma on whole-body FDG PET imaging. Clin Nucl Med 2006;31:42–3.
Soussan M, Wartski M, Ezra J, Glaisner S, Pecking AP, Alberini JL. Non-Hodgkin lymphoma localization in the pituitary gland: diagnosis by FDG-PET/CT. Clin Nucl Med 2008;33:111–2.
Kluetz PG, Meltzer CC, Villemagne VL, Kinahan PE, Chander S, Martinelli MA, et al. Combined PET/CT imaging in oncology. Impact on patient management. Clin Positron Imaging 2000;3:223–30.
Israel O, Mor M, Guralnik L, Hermoni N, Gaitini D, Bar-Shalom R, et al. Is 18F-FDG PET/CT useful for imaging and management of patients with suspected occult recurrence of cancer? J Nucl Med 2004;45:2045–51.
Bohuslavizki KH, Klutmann S, Kröger S, Sonnemann U, Buchert R, Werner JA, et al. FDG PET detection of unknown primary tumors. J Nucl Med 2000;41:816–22.
Agress H, Cooper BZ. Detection of clinically unexpected malignant and premalignant tumors with whole-body FDG PET: histopathologic comparison. Radiology 2004;230:417–22.
Jana S, Zhang T, Milstein DM, Isasi CR, Blaufox MD. FDG-PET and CT characterization of adrenal lesions in cancer patients. Eur J Nucl Med Mol Imaging 2006;33:29–35.
Karantanis D, Bogsrud TV, Wiseman GA, Mullan BP, Subramaniam RM, Nathan MA, et al. Clinical significance of diffusely increased 18F-FDG uptake in the thyroid gland. J Nucl Med 2007;48:896–901.
Chen W, Parsons M, Torigian DA, Zhuang H, Alavi A. Evaluation of thyroid FDG uptake incidentally identified on FDG-PET/CT imaging. Nucl Med Commun 2009;30:240–4.
Bae JS, Chae BJ, Park WC, Kim JS, Kim SH, Jung SS, et al. Incidental thyroid lesions detected by FDG-PET/CT: prevalence and risk of thyroid cancer. World J Surg Oncol 2009;7:63.
Parent AD, Bebin J, Smith RR. Incidental pituitary adenomas. J Neurosurg 1981;54:228–31.
Kurosaki M, Saeger W, Lüdecke DK. Pituitary tumors in the elderly. Pathol Res Pract 2001;197:493–7.
Buurman H, Saeger W. Subclinical adenomas in postmortem pituitaries: classification and correlations to clinical data. Eur J Endocrinol 2006;154:753–8.
Gambhir SS, Czernin J, Schwimmer J, Silverman DH, Coleman RE, Phelps ME. A tabulated summary of the FDG PET literature. J Nucl Med 2001;42:1S–93.
Pacak K, Ilias I, Chen CC, Carrasquillo JA, Whatley M, Nieman LK. The role of [(18)F]fluorodeoxyglucose positron emission tomography and [(111)In]-diethylenetriaminepentaacetate-D-Phe-pentetreotide scintigraphy in the localization of ectopic adrenocorticotropin-secreting tumors causing Cushing’s syndrome. J Clin Endocrinol Metab 2004;89:2214–21.
Kumar J, Spring M, Carroll PV, Barrington SF, Powrie JK. 18Flurodeoxyglucose positron emission tomography in the localization of ectopic ACTH-secreting neuroendocrine tumours. Clin Endocrinol (Oxf) 2006;64:371–4.
Sasajima T, Mineura K, Kowada M, Shishido F, Uemura K. Positron emission tomography (PET) of an invasive ACTH-producing pituitary adenoma. Rinsho Hoshasen 1990;35:407–10. Japanese.
Komori T, Martin WH, Graber AL, Delbeke D. Serendipitous detection of Cushing’s disease by FDG positron emission tomography and a review of the literature. Clin Nucl Med 2002;27:176–8.
Francavilla TL, Miletich RS, DeMichele D, Patronas NJ, Oldfield EH, Weintraub BD, et al. Positron emission tomography of pituitary macroadenomas: hormone production and effects of therapies. Neurosurgery 1991;28:826–33.
Bonneville JF, Cattin F, Gorczyca W, Hardy J. Pituitary microadenomas: early enhancement with dynamic CT—implications of arterial blood supply and potential importance. Radiology 1993;187:857–61.
Bonneville JF, Bonneville F, Cattin F. Magnetic resonance imaging of pituitary adenomas. Eur Radiol 2005;15:543–8.
Acknowledgments
This work was supported by Nuclear Research & Development Program of National Research Foundation of Korea (NRF) funded by Ministry of Education, Science & Technology (MEST) (grant code: 2010-0017515) and this work was supported by the Grant of the Korean Ministry of Education, Science and Technology (The Regional Core Research Program/Antiaging and Well-being Research Center).
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary materials
Below is the link to the electronic supplementary material.
ESM 1
(PDF 67 kb)
Rights and permissions
About this article
Cite this article
Jeong, S.Y., Lee, SW., Lee, H.J. et al. Incidental pituitary uptake on whole-body 18F-FDG PET/CT: a multicentre study. Eur J Nucl Med Mol Imaging 37, 2334–2343 (2010). https://doi.org/10.1007/s00259-010-1571-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-010-1571-5