Abstract
Objectives
To determine the efficacy and safety of percutaneous calcitonin and steroid injection in the treatment of aneurysmal bone cysts (ABCs).
Materials and methods
Our study was IRB-approved and HIPAA-compliant. We reviewed pre- and post-procedural imaging studies and medical records of all CT-guided percutaneous injections of ABCs with calcitonin and steroid performed at our institution between 2003 and 2015.
Results
Treatment success based on imaging was categorized as substantial (51–100 %), partial (1–50 %), or none (0 %) by comparing radiographs of the lesion before and after treatment. Our study group comprised 9 patients (7 female, 2 male; mean age 19 ± 5 (range 12–25) years). ABCs were located in the pubis (n = 3), femur (n = 2), and humerus/scapula/ilium/sacrum (n = 1 for each). One patient did not have any clinical or imaging follow-up. For the other 8 patients, clinical and imaging follow-up ranged from 1 to 93 months (mean 16 ± 29 months). One patient had two injections, and 1 patient had three injections. Six out of eight patients (75 %) had complete symptomatic relief and 2 patients (25 %) had partial symptomatic relief after initial injection. Imaging follow-up revealed substantial imaging response in 4 out of 8 patients (50 %). There was a partial imaging response in 2 patients (25 %) and no imaging response in 2 out of 8 patients (25 %), and all 4 of these patients had local recurrence. There were no complications.
Conclusion
Percutaneous CT-guided injection of ABCs with calcitonin and steroid is a safe and effective treatment. Lack of imaging response may necessitate more aggressive treatment to minimize local recurrence.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Aneurysmal bone cysts (ABCs) are benign neoplasms predominantly occurring during adolescence, and typically involve the metaphysis near the sites of greatest growth (e.g., around the knee, proximal humerus) [1–3]. Primary ABCs are true neoplasms, many of which are associated with a translocation of the CDH11 and USP6 genes [3–7]. On radiographs, ABCs appear as “ballooning” lesions, thinning and expanding the cortex. The expansile nature of the lesion can lead to pain and swelling. Pain can be constant even in the absence of a pathological fracture [2, 3, 8]. As ABCs are symptomatic even without fracture and represent true neoplasms that will not resolve spontaneously, therapy is required. ABCs have been conventionally treated with curettage and packing, especially if they involve weight-bearing bones [1]. Previously, sclerotherapy has been attempted, with different sclerosing agents and with variable success [3, 9–23].
We report here what is to our knowledge the largest case series to date of percutaneous CT-guided sclerotherapy of ABCs using calcitonin and steroid.
Materials and methods
Our study was IRB-approved and HIPAA-compliant. A retrospective review was performed to identify CT-guided percutaneous injections of ABCs with calcitonin and steroid performed at our institution between 2003 and 2015. Medical records and procedure reports were reviewed for pathology results, type and volume of injectate, aspirated cyst volume, complications, duration of follow-up, and clinical outcome including symptomatic relief, need for repeat procedures or surgery were recorded. Symptomatic relief was self-reported per the patient, documented in the clinical notes as decreased pain and/or improved functional status (e.g., return to sports).
Injection technique
All injections were performed by musculoskeletal radiologists as an outpatient procedure under conscious sedation. After standard skin antisepsis, sterile draping, and local skin anesthesia with approximately 1 mL of lidocaine HCl 1 % (Xylocaine; APP Pharmaceuticals, Schaumburg, IL, USA), a Bonopty 14-gauge penetration cannula with stylet and Bonopty drill (AprioMed, Londonderry, NH, USA) were advanced under CT guidance into the lesion. Aspiration was attempted before injection of contrast medium and medication to create space for the injectate and to decrease dilution of the injectate. Subsequently, 3–5 mL of iodinated contrast medium (iopamidol; Isovue M-300, Bracco Diagnostics, Monroe Township, NJ, USA) were injected to confirm the location of the Bonopty device within the lesion. An additional 3–5 mL of contrast medium was injected to determine if there was communication between the loculations. Sclerotherapy was performed by injecting 100–225 U of calcitonin (Miacalcin; Novartis Pharmaceuticals, Basel, Switzerland) mixed with 40–160 mg of triamcinolone acetonide 40 mg/mL (Kenalog; Bristol-Myers Squibb, New York, NY, USA) or methylprednisolone acetate (Depo-Medrol; Pfizer, New York, NY, USA). For the first two cases, 100 U of calcitonin was injected. As there were no complications, the dose was increased to 200 U or more if the lesion could accommodate the volume of injectate.
Imaging evaluation
Two musculoskeletal radiologists with 5 and 11 years of experience reviewed all the available imaging studies. The location, size, and number of loculations of the ABC (unilocular vs multilocular) were evaluated on CT and if available, MRI. Duration of follow-up and type of follow-up (clinical, imaging) was recorded. Treatment success based on imaging was performed by comparing pre-procedural and post-procedural radiographs and was categorized as follows: substantial (51–100 % of the lesion filled-in; Figs. 1, 2 and 3), partial (1–50 % of the lesion filled-in; Figs. 4), and none (0 % of the lesion filled-in). Post-procedure clinical symptoms were recorded from the clinical notes. If additional treatment (e.g., repeat injection, surgical intervention) was performed, this information was also recorded.
A 13-year-old boy with right shoulder pain. a Scapular Y-view radiograph of the scapula demonstrates multilobulated, expansile lytic lesion in the scapular body (arrows). MRI and CT appearance were consistent with an aneurysmal bone cyst (ABC; not shown). CT-guided biopsy established a pathological diagnosis of an ABC. The lesion was injected with calcitonin and methylprednisolone. b Scapular Y-view radiograph 1.5 months after calcitonin and steroid sclerotherapy demonstrates new bone formation (arrows) in the area of the previously seen ABC, consistent with substantial imaging response. The patient’s pain had resolved
A 14-year-old girl with a lesion in the right pubis, incidentally discovered when being evaluated for coccyx pain. The patient was asymptomatic in this region, but was treated because she was active and was considered a fracture risk. a Frontal radiograph of the pelvis demonstrates an expansile, multiseptated, lytic lesion in the right pubis (arrows). b Frontal radiograph of the pelvis 4 months after calcitonin and steroid sclerotherapy demonstrates increased overall density of the lesion (arrows), with some small remaining pockets of lucency, consistent with substantial imaging response
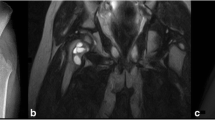
A 12 year-old-girl with right pubis lesion. a Frontal radiograph of the right pubis demonstrates an expansile lytic lesion in the right superior pubic ramus. b Frontal radiograph of the right pubis 1 month after calcitonin and steroid sclerotherapy demonstrates increased density throughout the lesion, consistent with a substantial imaging response. A few lucent areas remain (circle). The patient’s pain was nearly completely resolved. c Frontal radiograph of the right pubis 5 months after calcitonin and steroid sclerotherapy demonstrates progressively increased density throughout the lesion. The previously seen residual lucencies have filled in. The patient’s pain was completely resolved
A 21-year-old female rower with a left pubis lesion. a Frontal radiograph of the pelvis demonstrates an expansile lytic lesion in the left superior pubic ramus (arrow). b Frontal radiograph of the pelvis 2 months after calcitonin and steroid sclerotherapy demonstrates a small amount of increased density along the inferior aspect of the lesion, consistent with partial imaging response (arrow). The patient’s pain was nearly completely resolved
Results
Our study group comprised 9 patients (7 female, 2 male; mean age 19 ± 5 years, range 12–25 years). ABCs were located in the pubis (n = 3), femur (n = 2), humerus (n = 1), scapula (n = 1), ilium (n = 1), sacrum (n = 1). One patient had 2 injections, and 1 patient had 3 injections, for a total of 12 procedures. Patients had additional injections for persistent pain. Aspiration of bloody fluid was performed before injection in 9 out of 12 of procedures (75 %). No follow up or imaging was available for one of the patients. For the other 8 patients, average imaging and clinical follow-up were 12 ± 23 (range 1–69) months and 14 ± 23 (range 1–69) months respectively. The follow-up duration for each case individually is detailed in Table 1. Pathology confirmed the diagnosis of ABC in 5 out of 9 (56 %) patients. In 2 out of 9 patients (22 %) the pathology was nondiagnostic. In 2 out of 9 patients (22 %), there was only cytology, which did not show any malignant cells.
The patient results are detailed in Table 1. If more than one injection was performed, only the response to the first injection was evaluated. Six out of eight patients (75 %) had complete symptomatic relief and 2 patients (25 %) had partial symptomatic relief after initial injection. Imaging follow-up revealed substantial imaging response in 4 patients (50 %). There was a partial response in 2 patients (25 %) and little to no response in 2 patients (25 %), and all 4 of these patients required additional treatment. There were no complications.
Discussion
Aneurysmal bone cysts are benign neoplasms that occur in young patients. Approximately 75 % of patients with ABC are less than 20 years old. ABCs can involve any part of the skeleton, but are most common in the long bones, spine, and pelvis. Within the long bones, ABCs are most frequently found in the metaphysis, but can extend into the epiphysis after physeal closure [2, 3, 5]. ABCs demonstrate a genetic translocation of the USP6 oncogene on chromosome 17, and therefore, they are true neoplasms, which will grow if they are not treated [3]. ABC-like areas have also been seen with other benign lesions, such as fibrous dyplasia, giant cell tumor, chondroblastoma, and chondromyxoid fibroma; these lesions are often called “secondary ABCs” and represent 30 % of ABCs [1]. However, these do not demonstrate the typical genetic translocation seen in de novo ABCs and may merely represent intra-lesional bleeding [8]. Both primary and secondary ABCs can be highly vascular, and when they present in locations such as the spine and pelvis, surgical treatment can be challenging in terms of complete resection without affecting vital structures, and also controlling bleeding [1–3, 8].
Besides surgical treatment, ABCs have been treated with embolization of feeding vessels, but this procedure can only performed if the afferent vessel can be identified, and carries a risk of accidental injury to other structures, such as the spinal cord in the case of vertebral lesions [3, 5, 24]. Embolization can also help to reduce hemorrhage before surgical treatment [3, 25]. Previously, sclerotherapy has been attempted by using different sclerosing agents with variable success. In the early 2000s, sclerotherapy was performed with Ethibloc, an alcoholic solution of corn protein. Garg et al. described the first series of 10 patients, and demonstrated complete symptom resolution. However, the series also described local transitory inflammatory reaction in 2 patients and an inflammatory, non-infectious collection in 1 patient [9]. Further case series confirmed the high efficacy of Ethibloc sclerotherapy, but also described a high rate of complications and ultimately a universal halt was placed on using this agent to treat ABCs [10–13]. A case report also described ABC regression after excisional biopsy [26].
Sclerotherapy using steroids alone has been used, with variable results. Scaglietti et al. first described the successful treatment of an ABC in 1974 and later published a series of 163 cases with complete ossification of the cyst in 55 % and at least partial ossification of the cyst in 45 %. Only 24 % of patients were treated with a single injection, and the remaining required additional injections. Complications were described as “rare,” but the rate was not reported [14, 15]. Cottalorda et al. described 2 patients with ABCs who were injected with methylprednisolone. The ABCs recurred and required re-treatment by other methods and this suggested that steroids might exacerbate ABCs [16]. Another report also suggests that steroids might worsen the lesion [3].
In a preliminary report, Shiels and Mayerson demonstrated the 100 % efficacy of doxycycline treatment of ABCs in 20 patients, although up to 14 treatments were required for each patient. Skin necrosis due to lesion overpressurization and extra-osseous leakage of the doxycycline occurred in 1 patient [17]. Lambot-Juhan et al. reported at least a partial symptomatic response in 90 % of patients in a series of 29 patients treated with 96 % ethanol injection; 79 % were treated with one or two injections. The only complication was a temporary nerve palsy due to extra-osseous leakage of the ethanol [18]. A recent surgical review cites 96 % ethanol injection as the preferred choice of sclerotherapy agent [3].
Sclerotherapy using calcitonin was first proposed in a paper by Szendröi et al. [19]. Calcitonin promotes new bone formation, but also suppresses osteoclast activity. Therefore, intralesional injection of calcitonin was thought to promote healing [19]. There have been multiple case reports and case series describing the efficacy of calcitonin sclerotherapy in treating ABCs, mostly in the upper cervical spine (C1 and C2) [19, 20, 22, 23]. In all of these reports, there have been no complications, suggesting that calcitonin might be a safe medication for injection. Calcitonin is a naturally occurring hormone that directly acts on bone, brain, kidneys, and possibly gonads. There are no known effects on muscle, fat, or other connective tissues. Therefore, there should theoretically be no risk for calcitonin leakage into the surrounding soft tissues. Calcitonin is also safely used as a systemic treatment agent for osteoporosis, hypercalcemia, Paget’s disease, bone pain related to inflammatory and degenerative diseases, complex regional pain syndrome, and osseous metastases, among other conditions [27–31]. However, until further investigative studies have been performed, calcitonin should still be injected with caution.
To our knowledge, our cohort is the largest to date, examining the efficacy and safety of percutaneous calcitonin and steroid injection in the treatment of ABCs and the first lesions treated other than the spine. All of our treated patients had at least partial symptomatic relief, and 50 % had a substantial imaging response, which did not require further treatment. The apparent discordance between the clinical and imaging response may be in part due to the choice of radiographs for evaluating the imaging response. Radiographs were selected because they involve a low level of radiation, the patients are young, and they may require serial examinations. CT would be more accurate in evaluating a response, but would expose patients to a much higher radiation dose. Low-dose CT, if comparable in dose with a radiographic examination, may be considered as an alternative for follow-up. Orthopedic surgeons at our institution typically see the patient for a follow-up visit in their clinic 1 month after the procedure. Radiographs obtained at 1 month typically demonstrate radiographic changes, which continue to evolve over time. A reasonable follow-up plan would be at 1 month, 6 months, and 12 months, with additional imaging if the patient has new or persistent symptoms.
One of the cases that had no imaging response consisted of a recurrence around a previously curettaged lesion, with graft material in the lesion. The graft was a physical barrier to accessing some portions of the lesion in this case. The material of the graft was unlikely to respond to the calcitonin as the native bone would, and it could also inhibit the medication in some way. Finally, only the medication that contacts the cyst wall has the potential to act on the bone to produce healing of the lesion. Therefore, methods of increasing the volume of the injectate without increasing the dose may help to improve the efficacy of this treatment. Interestingly, among the group of patients who had a substantial imaging and clinical response, 50 % had a uniloculated lesion and 50 % had a multiloculated lesion. This initial series supports the notion that a multiloculated lesion can be successfully treated by percutaneous calcitonin and steroid sclerotherapy. Other factors that may influence the efficacy of the medication include the age of the patient and the location of the lesion. Cyst aspiration did not appear to correlate with the efficacy of the injection in our series, possibly because the cyst fills with blood soon after aspiration. These areas are all potential areas for investigation in the future.
In conclusion, percutaneous CT-guided injection of ABCs with calcitonin and steroid is an effective treatment, and no complications occurred in our series. A lack of imaging response may necessitate more aggressive treatment to minimize local recurrence. Future prospective studies are needed to confirm our findings.
References
Campanacci M, Capanna R, Picci P. Unicameral and aneurysmal bone cysts. Clin. Orthop. 1986;204:25–36
Bonakdarpour A, Levy WM, Aegerter E. Primary and secondary aneurysmal bone cyst: a radiological study of 75 cases. Radiology. 1978;126:75–83.
Mascard E, Gomez-Brouchet A, Lambot K. Bone cysts: unicameral and aneurysmal bone cyst. Orthop Traumatol Surg Res. 2015;101:S119–127.
Fletcher CDM. WHO classification of tumours of soft tissue and bone. Lyon: IARC Press; 2013.
Cottalorda J, Bourelle S. Modern concepts of primary aneurysmal bone cyst. Arch Orthop Trauma Surg. 2007;127:105–14.
Althof PA, Ohmori K, Zhou M, Bailey JM, Bridge RS, Nelson M, et al. Cytogenetic and molecular cytogenetic findings in 43 aneurysmal bone cysts: aberrations of 17p mapped to 17p13.2 by fluorescence in situ hybridization. Mod Pathol. 2004;17:518–25.
Oliveira AM, Perez-Atayde AR, Inwards CY, Medeiros F, Derr V, Hsi B-L, et al. USP6 and CDH11 oncogenes identify the neoplastic cell in primary aneurysmal bone cysts and are absent in so-called secondary aneurysmal bone cysts. Am J Pathol. 2004;165:1773–80.
Dahlin DC, McLeod RA. Aneurysmal bone cyst and other nonneoplastic conditions. Skeletal Radiol. 1982;8:243–50.
Garg NK, Carty H, Walsh HP, Dorgan JC, Bruce CE. Percutaneous Ethibloc injection in aneurysmal bone cysts. Skeletal Radiol. 2000;29:211–6.
Falappa P, Fassari FM, Fanelli A, Genovese E, Ascani E, Crostelli M, et al. Aneurysmal bone cysts: treatment with direct percutaneous Ethibloc injection: long-term results. Cardiovasc Intervent Radiol. 2002;25:282–90.
Dubois J, Chigot V, Grimard G, Isler M, Garel L. Sclerotherapy in aneurysmal bone cysts in children: a review of 17 cases. Pediatr Radiol. 2003;33:365–72.
de Gauzy JS, Abid A, Accadbled F, Knorr G, Darodes P, Cahuzac JP. Percutaneous Ethibloc injection in the treatment of primary aneurysmal bone cysts. J Pediatr Orthop Part B. 2005;14:367–70.
George HL, Unnikrishnan PN, Garg NK, Sampath JS, Bass A, Bruce CE. Long-term follow-up of Ethibloc injection in aneurysmal bone cysts. J Pediatr Orthop Part B. 2009;18:375–80.
Scaglietti O, Marchetti PG, Bartolozzi P. Final results obtained in the treatment of bone cysts with methylprednisolone acetate (depo-medrol) and a discussion of results achieved in other bone lesions. Clin Orthop Relat Res. 1982;165:33–42
Scaglietti O, Marchetti PG, Bartolozzi P. The effects of methylprednisolone acetate in the treatment of bone cysts. Results of three years follow-up. J Bone Joint Surg Br. 1979;61-B:200–4.
Cottalorda J, Kohler R, Chotel F, de Gauzy JS, Lefort G, Louahem D, et al. Recurrence of aneurysmal bone cysts in young children: a multicentre study. J Pediatr Orthop Part B. 2005;14:212–8.
Shiels WE, Mayerson JL. Percutaneous doxycycline treatment of aneurysmal bone cysts with low recurrence rate: a preliminary report. Clin Orthop Relat Res. 2013;471:2675–83.
Lambot-Juhan K, Pannier S, Grévent D, Péjin Z, Breton S, Berteloot L, et al. Primary aneurysmal bone cysts in children: percutaneous sclerotherapy with absolute alcohol and proposal of a vascular classification. Pediatr Radiol. 2012;42:599–605.
Szendröi M, Cser I, Kónya A, Rényi-Vámos A. Aneurysmal bone cyst. A review of 52 primary and 16 secondary cases. Arch Orthop Trauma Surg. 1992;111:318–22.
Gladden ML, Gillingham BL, Hennrikus W, Vaughan LM. Aneurysmal bone cyst of the first cervical vertebrae in a child treated with percutaneous intralesional injection of calcitonin and methylprednisolone. A case report. Spine. 2000;25:527–30; discussion 531.
Rai AT, Collins JJ. Percutaneous treatment of pediatric aneurysmal bone cyst at C1: a minimally invasive alternative: a case report. AJNR Am J Neuroradiol. 2005;26:30–3.
Tonomura ET, Ramos P, Hemais PM, Marchiori E, Gasparetto EL. Aneurysmal bone cyst at C2: imaging evaluation after intralesional injection of calcitonin and methylprednisolone. Arq Neuropsiquiatr. 2008;66:711–5.
Ohashi M, Ito T, Hirano T, Endo N. Percutaneous intralesional injection of calcitonin and methylprednisolone for treatment of an aneurysmal bone cyst at C-2. J Neurosurg Pediatr. 2008;2:365–9.
Boriani S, De Iure F, Campanacci L, Gasbarrini A, Bandiera S, Biagini R, et al. Aneurysmal bone cyst of the mobile spine: report on 41 cases. Spine. 2001;26:27–35.
Amendola L, Simonetti L, Simoes CE, Bandiera S, De Iure F, Boriani S. Aneurysmal bone cyst of the mobile spine: the therapeutic role of embolization. Eur Spine J. 2013;22:533–41.
Scott I, Connell DG, Duncan CP. Regression of aneurysmal bone cyst following open biopsy. Can Assoc Radiol J. 1986;37:198–200.
Wang J, Yao M, Xu J-H, Shu B, Wang Y-J, Cui X-J. Bisphosphonates for prevention of osteopenia in kidney-transplant recipients: a systematic review of randomized controlled trials. Osteoporos Int. 2016;27:1683–90.
Mirrakhimov AE. Hypercalcemia of malignancy: an update on pathogenesis and management. North Am J Med Sci. 2015;7:483–93.
Singer F. Paget’s disease of bone. In: De Groot LJ, Beck-Peccoz P, Chrousos G, Dungan K, Grossman A, Hershman JM, et al., editors. Endotext. South Dartmouth (MA): MDText.com; 2000. Available from: http://www.ncbi.nlm.nih.gov/books/NBK279033/. Accessed 27 July 2016
Eriksen EF. Treatment of bone marrow lesions (bone marrow edema). Bonekey Rep. 2015;4:755.
Palmer G. Complex regional pain syndrome. Aust Prescr. 2015;38:82–6.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
None.
Conflicts of interest
The authors declare that they have no conflicts of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
Informed consent
Informed consent was waived for individual participants included in the study. The study was approved by the local Institutional Review Board (IRB) and was HIPAA-compliant.
Rights and permissions
About this article
Cite this article
Chang, C.Y., Kattapuram, S.V., Huang, A.J. et al. Treatment of aneurysmal bone cysts by percutaneous CT-guided injection of calcitonin and steroid. Skeletal Radiol 46, 35–40 (2017). https://doi.org/10.1007/s00256-016-2503-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00256-016-2503-7