Abstract
Objective
To determine the efficacy of percutaneous sclerotherapy in the treatment of aneurysmal bone cysts.
Materials and methods
Seventeen patients (7 girls, 10 boys) with aneurysmal bone cysts were treated by the percutaneous approach with Ethibloc (n=14) and histoacryl glue (n=3) in our institution between January 1994 and June 2000. The cysts were located in the extremities (n=6), pelvis (n=2), spine (n=2), mandible (n=5), rib (n=1) and sphenoid bone (n=1). Percutaneous sclerotherapy was performed with fluoroscopic and/or computed tomographic guidance under general anesthesia. Clinical and imaging follow-up lasted from 24 months to 9 years and 6 months (mean: 57.3 months). The results were quantified as: excellent (residual cyst less than 20% of the initial involvement), satisfactory (residual cyst 30–50%), unsatisfactory (residual cyst more than 50%).
Results
The age of the patients ranged from 4 years and 6 months to 15 years and 8 months (mean: 11 years and 2 months). In nine patients, the therapeutic procedure was repeated 2–5 times. Excellent regression was observed in 16 (94%), satisfactory results in 1 (6%). There was no failure (unsatisfactory result or no response to treatment) in this reported series. The complications were minor and included: local inflammatory reaction (n=2), small blister (n=1), and leakage (n=1). Relief of symptoms was achieved in all patients. No recurrence was noted during follow-up.
Conclusion
Percutaneous sclerotherapy of aneurysmal bone cysts with Ethibloc is safe and effective. It is an important alternative to surgery, especially when surgery is technically impossible or not recommended in high-risk patients.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
In 1970, in a classic review article, Biesecker et al. [1] wrote "the lack of understanding about the origin and growth of aneurysmal bone cysts has led to an empirical approach to their treatment". Thirty years later, despite the persistent uncertainty of their origin, treatments have improved, mostly owing to sclerotherapy and arterial embolization. We report a retrospective study of 17 aneurysmal bone cysts (ABC) treated with sclerotherapy, combined or not with surgery and arterial embolization. The aim of our research was to provide guidelines for the management of ABC in children (Fig. 1).
Materials and methods
The records of 17 patients with ABC who underwent sclerotherapy at the authors' institution between January 1994 and June 2000 were reviewed.
Patients were referred for sclerotherapy by orthopedic surgeons. Circumstances of diagnosis, topography, and size of the ABC are summarized in Table 1. Multiple bone involvement was found in a single case (patient 1, thoracic spine ABC). The diameter of the lesion before sclerotherapy ranged from 1.6 to 12 cm (average 4.6 cm).
Pathological examination was available in nine cases. Pathological material was obtained at the time of attempted surgical excision in six cases (patients 1, 3, 5, 7, 12, 13), and after surgical biopsy because of atypical radiological presentation in three cases (patients 8, 11, 14). According to pathology, one ABC was associated with ossifying fibromas (patient 7) and one with a unicameral bone cyst (patient 12). Pathologic examination of the mandibular lesion of patient 8 was compatible with a giant-cell granuloma or an ABC. The others were considered as isolated ABC (Fig. 2a, b, 3a, b, 4a–d).
Patient 14. Aneurysmal bone cyst of the left acetabulum and ischium revealed by a fracture in a 12-year-old girl. a Plain radiograph shows an expansile lesion with septa that involves the left acetabulum and ischion. b Follow-up plain radiograph obtained 48 months later after three sessions of sclerotherapy shows excellent bone mineralization
Patient 2. An 11-year-old boy with an aneurysmal bone cyst of the second metatarsus. a Plain film shows an expansile lesion of the second metatarsus. b Follow-up plain radiograph obtained 21 months later after two sessions of sclerosing treatment shows complete reconstruction with normal bone volume and mineralization
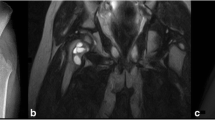
Patient 5. A 15-year-old girl with right root compression operated on for resection of the expansile lytic bone lesion. The surgery was limited because of important bleeding. a Computed tomography before sclerosing treatment shows an expansile lesion of the right sacrum (patient in prone position). b MR imaging (FSE T2:3600/90) reveals a high signal intensity lesion in the right sacrum. c Two years 10 months later, follow-up CT-scan performed after four sessions of sclerosing treatment reveals an excellent reossification of the sacral bone. d MR imaging (IRFSE: 3600 /172/126 confirms the result
The data collected for each case included: age, sex, circumstances of diagnosis (incidental finding or clinical complaints), location, and size of the ABC. Treatments were classified into three types: surgery, arterial embolization, and sclerotherapy. Surgery was intralesional (curettage) or extralesional (segmental or marginal "en bloc" resection). Embolization was performed with polyvinyl alcohol particles under general anesthesia. Sclerotherapy was performed in the angiographic suite or in the operating room using general anesthesia. Direct puncture of the aneurysmal bone cyst with or without CT-scan guidance was performed with a 20 or 18 G Disp pencil-point needle (Cook, Bloomington, Ind.). When good venous reflux was obtained, the ABC was opacified with contrast medium under fluoroscopy. Digitalized subtraction angiograms were obtained to evaluate both the draining venous system and the ABC volume.
Ethibloc (Ethnor Laboratories/Ethicon, Norderstedt, Germany) is a sclerosing agent not approved by the FDA. Ethibloc contains an alcoholic (60%) solution of 210 mg zein (corn protein)/ml alcohol, 162 mg sodium diatrizoate/ml alcohol (as a radiopaque marker), 145 mg oleum papaveris/ml alcohol (which is added to keep the substance sterile). Ethibloc is available in sterilized ready-to-inject syringes (7.5 ml).
We diluted Ethibloc with 5 cm3 100% ethanol. A volume equivalent to the estimated size of the ABC, as determined by the volume of contrast material needed to fully opacify the lesion, was injected under fluoroscopic control. If the venous drainage was accessible, sclerotherapy was performed under manual compression of the draining veins.
When the ABC was located in the spine or in the skull, we used n-Butyl Cyanoacrylate (Histoacryl blue. Indermil; Loctite, Dublin, Republic of Ireland). This embolic agent is diluted with lipiodol. It is also not FDA approved.
When available, results of pathological examination were recorded.
Radiographs, CT scans, and MRIs were reviewed from the time the patients were referred to our institution to the end of follow-up.
Results of therapy were classified as unsatisfactory, satisfactory, or excellent. Unsatisfactory results were progression of the lesion, recurrence, or more than 50% of residual cyst. Satisfactory results were partial healing (stabilization with less than 50% of residual cyst without clinical complaints). Excellent results were noted when 80% or more of bone sclerosis was seen at follow-up.
Results
The average age at the time of diagnosis was 11 years and 2 months (range: 4 years 6 months to 15 years 8 months). There were ten males and seven females.
Follow-up ranged from 2 years to 9 years and 6 months (mean: 57.3 months, median: 54.5 months, SD: 28.7 months). There were 16 (94%) excellent results and 1 (6%) satisfactory result.
Six patients had previously undergone unsuccessful surgery. In one patient (patient 5), surgery was interrupted before excision because of massive hemorrhage. The four remaining patients underwent a total of six curettages with subsequent recurrence (patients 3, 7, 12, 13). Four curettages among the six required bone grafts. No cryosurgery was performed. The average interval between curettage and subsequent sclerotherapy was 9.4 months (range: 4–20 months). Patient 12 had four intralesional injections of steroids prior to sclerotherapy with Ethibloc.
Two patients underwent intraoperative sclerotherapy. In patient 1 (spinal lesion) sclerotherapy was performed in the operative room to reduce intraoperative bleeding. In patient 17 (sphenoid lesion) surgical exposure was mandatory because of the proximity of cerebral structures, and sclerotherapy was not followed by surgical excision.
If total opacification was not possible through a single puncture, sclerotherapy was completed through additional punctures. An alcoholic solution of zein (Ethibloc; Ethnor Laboratories/Ethicon, Norderstedt, Germany) was used in 14 patients. The 3 patients with sphenoid and spinal ABC (patients 1, 11, 17) were treated with cyanoacrylate (Indermil; Loctite, Dublin, Republic of Ireland).
Eight patients had a course of 2–4 sequential sclerotherapies and nine patients had a single session of sclerotherapy (average: 1.7 procedures per patient). The only adverse reaction was a transient leakage of alcoholic zein in a metatarsal ABC.
Arteriography was performed in five patients (patients 1, 5, 11, 14, 17): three before surgery to reduce peroperative bleeding (patients 1, 11, 17) and two before sclerotherapy because of the large size of the ABC (patients 5, 14). In two cases without large feeding arteries (patients 1, 14), embolization was not performed. Arterial embolization was performed in the remaining three patients. A branch of the internal iliac artery was embolized in patient 5 (large ABC of the sacrum), a lumbar artery embolized in patient 11 (spinal lumbar lesion), and a maxillary artery embolized in patient 17 (sphenoid ABC).
Two patients with spinal lesion underwent surgery after sclerotherapy: patient 1 (en bloc excision) and patient 11 (curettage).
Discussion
The main controversy about ABC management is based on whether pathological examination is mandatory or not. Some authors do not believe that ABC are secondary to other lesions [2, 3, 4]. Even if ABC are considered secondary lesions, biopsy specimens from curettage material could overlook the primary lesion [4, 5]; then the only way to assess the lack of associated lesion would be the pathological examination obtained from a total excision. Such surgery implies massive bone resection and bone reconstruction. According to a number of authors [5, 6, 7, 8, 9, 10, 11, 12, 13] and in our own experience, we believe that pathological examination is not mandatory when the clinical presentation and radiological appearance are typical: secondary ABC most often have radiological features that are characteristic of their primary lesion [6, 14]. Furthermore, we consider a copious blood return through the first puncture a necessary requisite for diagnosis.
We do not intend here to review the radiological criteria of ABC. Briefly, we consider a lesion typical of ABC when the lesion is osteolytic eccentric or central with circumferential expansion, expansile with cortical thinning [15], and metaphyseal in location in long bones [4, 15, 16]. Fluid-fluid levels seen with a CT scan, MRI or US are not considered pathognomonic [17].
The need for therapy and its efficiency are difficult to assess since some ABC heal spontaneously [11] or following a trauma (biopsy or pathologic fracture) [4]. The ideal treatment should be safe and permanent without recurrences.
Extralesional excision is the most definite treatment of ABC because it prevents recurrence [2, 10], but its disadvantages are numerous: growth-plate injury, prolonged immobilization and hospitalization, and important loss of bone, necessitating subsequent bone grafts [4]. Therefore, intralesional excision (curettage) is the most frequently performed surgery, but it is often incomplete in large lesions because of massive intraoperative bleeding [10]. It leads to a 18 to 59% recurrence rate [1, 3, 4, 5, 15, 16], depending on whether additional cryosurgery is performed. Curettage is also associated with a risk of epiphysiodesis [16]. Finally, surgical approach may be difficult at certain sites.
Radiotherapy was recommended in inoperable cases prior to the availability of sclerotherapy and arterial embolization. The numerous and severe complications of radiation therapy are well known (secondary osteosarcomas, osteonecrosis, fusion of the growth plate, gonadal damage, radiation myelopathy) [1, 2, 4, 15, 16, 17, 18].
Embolization of the feeding vessels was initially a preoperative procedure performed to reduce intraoperative bleeding [19, 20]. Thereafter, embolization was shown to be effective after surgical failure [21] or as a single therapy [10, 12, 22, 23]. However, ABC often lack large feeding arteries to be embolized, and repeated procedures are frequently required when arterial embolization is selected as the single therapy [10]. When the ABC is cranial or spinal in location, the vicinity of arteries supplying the brain or the spinal cord makes the procedure hazardous because of the risk of aberrant emboli. Also, we recommend that only histoacryl be used for cranial or spinal ABC considering the important inflammatory reaction induced by Ethibloc.
Sclerotherapy is nowadays the primary treatment of ABC for several authors because it is safe, easy to perform and cost effective, and far less aggressive than surgery. Patients are discharged 6 h postprocedure, and they do not have to be immobilized. Potential complications are pulmonary embolism, osteomyelitis, and complications related to general anesthesia. Guilbaud et al. [8] reported a case of aseptic osteitis that required prolonged immobilization. None was observed in our series. Leakage of alcoholic zein is a well-known occurrence that has no clinical significance following soft-tissue vascular malformation sclerotherapy [8].
Several sclerosing agents have been proposed for the treatment of benign bone cysts. Acetate of methyl prednisolone did not demonstrate any efficiency in a series of 12 ABC reported by Scaglietti et al. [24]. We have no experience with calcitonin and radionuclide, which seemed to be efficient as the sole therapy in small series [25, 26]. Alcoholic solution of zein was first used with success in a series of four benign bone cysts (two unicameral and two aneurysmal) by Adamsbaum et al. in 1993 [27]. Because we already had the expertise in using Ethibloc in various vascular malformations, we decided to use it in all but three patients who were treated with cyanoacrylate because of the site of involvement (skull, spine).
Indeed, there are special considerations in the management of ABC located near the brain or the spinal cord. Sclerotherapy must be achieved with much care because of risk of leakage or an erroneous course of the sclerosing agent in the surrounding veins with subsequent central nervous injury. To diminish the risk of regional inflammation and erroneous emboli in spinal and juxta cerebral ABC, we have chosen to use cyanoacrylate rather than Ethibloc. If there are severe neurological signs at presentation, early decompression is mandatory and preoperative arterial embolization and/or simultaneous sclerotherapy significantly reduce peroperative bleeding. When surgical decompression is hazardous (possible damage to neural structures, massive hemorrhage) sclerotherapy alone can be sufficient, as demonstrated in patient 17 (sphenoid ABC), whose neurological deficits progressively subsided following sclerotherapy.
In our initial experience, several sessions of sclerotherapy were performed within a year. With further expertise we have reached the conclusion that one should wait 9–12 months to evaluate the induced sclerosis and perform additional sclerotherapy only if some cysts remain.
In patient 13, the third sclerotherapy session was cancelled because there was no blood return after puncture of the sole remaining cyst; the latter had probably become a "solid variant of ABC," which represents a lesion at a reparative stage that rarely recurs after curettage [26, 27, 28, 29].
Since the aim of the treatment is to avoid pain, pathological fractures and the progression of the lesion, partial sclerosis (more than 50%) without growing of the ABC is considered a satisfactory result.
Post-treatment follow-up should be at least 24 months, as most recurrences take place within 2 years [3, 10, 15, 16]. It could be shorter if radiographic evidence of complete sclerosis and absence of clinical symptoms are recorded. CT scan and radiographs are the best modalities to assess the reossification.
Our algorithm for the management of ABC is summarized in Fig. 1.
We believe that sclerotherapy is the treatment of choice for aneurysmal bone cyst because it is far less aggressive than surgery and more effective. Early surgery remains mandatory if there are neurological symptoms in spinal and juxta cerebral locations, but preoperative sclerotherapy dramatically reduces intraoperative blood loss.
References
Biesecker JL, Marcove RC, Huvos AG, et al (1970) Aneurysmal bone cysts. A clinicopathologic study of 66 cases. Cancer 26:615–625
Tillman BP, Dahlin DC, Lipscomb PR, et al (1968) Aneurysmal bone cyst: an analysis of ninety-five cases. Mayo Clin Proc 43:478–495
Ruiter DJ, van Rijssel TG, van der Velde EA (1977) Aneurysmal bone cysts: a clinicopathological study of 105 cases. Cancer 39:2231–2239
Campanacci M, Capanna R, Picci P (1986) Unicameral and aneurysmal bone cysts. Clin Orthop 204:25–36
Szendroi M, Cser I, Konya A, et al (1992) Aneurysmal bone cyst. A review of 52 primary and 16 secondary cases. Arch Orthop Trauma Surg 111:318–322
Dabska M, Buraczewski J (1969) Aneurysmal bone cyst. Pathology, clinical course and radiologic appearances. Cancer 23:371–389
Bollini G, Jouve JL, Cottalorda J, et al (1998) Aneurysmal bone cyst in children: analysis of twenty-seven patients. J Pediatr Orthop B 7:274–285
Guibaud L, Herbreteau D, Dubois J, et al (1998) Aneurysmal bone cysts: percutaneous embolization with an alcoholic solution of zein—series of 18 cases. Radiology 208:369–373
Garg NK, Carty H, Walsh HP, et al (2000) Percutaneous Ethibloc injection in aneurysmal bone cysts. Skeletal Radiol 29:211–216
Boriani S, De Iure F, Campanacci L, et al (2001) Aneurysmal bone cyst of the mobile spine: report on 41 cases. Spine 26:27–35
Malghem J, Maldague B, Esselinckx W, et al (1989) Spontaneous healing of aneurysmal bone cysts. A report of three cases. J Bone Joint Surg Br 71:645–650
De Cristofaro R, Biagini R, Boriani S, et al (1992) Selective arterial embolization in the treatment of aneurysmal bone cyst and angioma of bone. Skeletal Radiol 21:523–527
Chartrand-Lefebvre C, Dubois J, Roy D, et al (1996) Direct intraoperative sclerotherapy of an aneurysmal bone cyst of the sphenoid. AJNR 17:870–872
Martinez V, Sissons HA (1988) Aneurysmal bone cyst. A review of 123 cases including primary lesions and those secondary to other bone pathology. Cancer 61:2291–2304
Vergel De Dios AM, Bond JR, Shives TC, et al (1992) Aneurysmal bone cyst. A clinicopathologic study of 238 cases. Cancer 69:2921–2931
Marcove RC, Sheth DS, Takemoto S, et al (1995) The treatment of aneurysmal bone cyst. Clin Orthop 311:157–163
Tsai JC, Dalinka MK, Fallon MD, et al (1990) Fluid-fluid level: a nonspecific finding in tumors of bone and soft tissue. Radiology 175:779–782
Gladden ML Jr, Gillingham BL, Hennrikus W, et al (2000) Aneurysmal bone cyst of the first cervical vertebrae in a child treated with percutaneous intralesional injection of calcitonin and methylprednisolone. A case report. Spine 25:527–530; discussion 531
Dysart SH, Swengel RM, van Dam BE (1992) Aneurysmal bone cyst of a thoracic vertebra. Treatment by selective arterial embolization and excision. Spine 17:846–848
Papagelopoulos PJ, Currier BL, Shaughnessy WJ, et al (1998) Aneurysmal bone cyst of the spine. Management and outcome. Spine 23:621–628
Radanovic B, Simunic S, Stojanovic J, et al (1989-1990) Therapeutic embolization of aneurysmal bone cyst. Cardiovasc Intervent Radiol 12:313–316
Cigala F, Sadile F (1996) Arterial embolization of aneurysmal bone cysts in children. Bull Hosp Jt Dis 54:261–264
Cory DA, Fritsch SA, Cohen MD, et al (1989) Aneurysmal bone cysts: imaging findings and embolotherapy. AJR 153:369–373
Scaglietti O, Marchetti PG, Bartolozzi P (1979) The effects of methylprednisolone acetate in the treatment of bone cysts. Results of three years follow-up. J Bone Joint Surg Br 61:200–204
Szendroi M, Antal I, Liszka G, et al (1992) Calcitonin therapy of aneurysmal bone cysts. J Cancer Res Clin Oncol 119:61–65
Bush CH, Drane WE (2000) Treatment of an aneurysmal bone cyst of the spine by radionuclide ablation. AJNR 21:592–594
Adamsbaum C, Kalifa G, Seringe R, et al (1993) Direct Ethibloc injection in benign bone cysts: preliminary report on four patients. Skeletal Radiol 22:317–320
Sanerkin NG, Mott MG, Roylance J (1983) An unusual intraosseous lesion with fibroblastic, osteoclastic, osteoblastic, aneurysmal and fibromyxoid elements. "Solid" variant of aneurysmal bone cyst. Cancer 51:2278–2286
Gipple JR, Pritchard DJ, Unni KK (1992) Solid aneurysmal bone cyst. Orthopedics 15:1433–1436
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dubois, J., Chigot, V., Grimard, G. et al. Sclerotherapy in aneurysmal bone cysts in children: a review of 17 cases. Ped Radiol 33, 365–372 (2003). https://doi.org/10.1007/s00247-003-0899-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00247-003-0899-4