Abstract
Purpose
Adiposis dolorosa (Dercum’s disease) is a condition of benign, painful subcutaneous lipomatous lesions associated with weakness, endocrine and lipid abnormalities, and mental disturbances. There is little information documenting the cross-sectional imaging findings that differentiate it from lipomatous and neoplastic soft tissue masses, or massive localized lymphedema. The purpose of this study was to provide a radiological case series of adiposis dolorosa.
Methods
A 10-year retrospective review of the picture archiving and communications system was performed. Two musculoskeletal radiologists reviewed images to confirm and document imaging features, location, size, and patient demographics. Medical records were reviewed to characterize patients into three groups: one group met at least three of the four criteria of Dercum’s syndrome, the second group met less than three criteria, and the third group had clinical diagnosis of cellulitis of the lower extremity.
Results
Seventeen cases (25 masses) of adiposis dolorosa were found, nine cases of which met at least three criteria of Dercum’s syndrome. All cases in the first two groups demonstrated skin thickening and lymphedema of subcutaneous fat, which was fluid attenuation on CT and low or intermediate T1-weighted and high STIR/T2-weighted MR signal. Two cases with pathology showed mild fatty infiltration with fibrous septa, and the third case showed massive localized lymphedema. The third group of ten cellulitis patients demonstrated non-mass-like subcutaneous edema with similar CT attenuation and MR signal characteristics to the first two groups, but differed by the presence of post-contrast enhancement and non-mass-like appearance in 90 %.
Conclusion
Imaging findings of adiposis dolorosa and massive localized lymphedema overlap, as do the symptoms and pathological features. Due to the mass-like engorgement of the soft tissues and pain, patients will often undergo imaging to exclude neoplasm or infection. Knowledge of these conditions and the characteristic imaging findings is important to prevent unnecessary biopsy and misdiagnosis.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Adiposis dolorosa, also known as Dercum’s and Ander’s disease, was first described in 1888 as an entity of multiple painful lipomas in overweight, postmenopausal females with mental or emotional disturbances [1]. Although adiposis dolorosa occurs predominantly in females, cases in men have been documented [2]. The masses of adiposis dolorosa are a benign entity, classified as lipomatous lesions by the World Health Organization (WHO) [3]. The etiology of Dercum’s disease is unknown, although there is some evidence for an autosomal dominant inheritance pattern with incomplete penetrance [4, 5].
Due to a lack of unique histopathological findings, the term “Dercum’s syndrome” was proposed to describe the process. Dercum’s syndrome includes (1) painful fatty masses; (2) generalized obesity, usually in females of menopausal age; (3) asthenia, weakness, and fatigability; (4) mental disturbances, including emotional instability, depression, epilepsy, mental confusion, and true dementia [6]. The exact etiology of the pain is unknown. Several theories have been suggested, including capillary microthrombi, endocrine abnormalities, microtrauma, stretching of nerves, and defective fatty acid synthesis [1, 7–10].
The painful masses tend to occur in the extremities near the hip and knee joints as well as on the trunk [11]. They rarely involve the hands or feet. Masses are often multiple, freely mobile on physical examination, and painful to the touch [11]. There is a paucity of literature with radiological images specifically defined as Dercum’s syndrome. A case report by Amine et al. demonstrated lumps of adipose tissue in an arc-shaped configuration around the ankle joints on both ultrasound (US) and magnetic resonance imaging (MRI) [12]. Another case report by Kawale et al. showed thickening of the scalp tissue on computed tomography (CT) and MRI [13]. A recent paper by Tins et al. discussed MRI findings of fatty lesions of the superficial fat of the extremities with nodular increased fluid signal and no contrast enhancement [14]. However, not all of these lesions in this case series were painful, which is typically a required component of Dercum’s syndrome, nor where they as pendulous appearing as the clinical images provided by Dercum and Brodovsky [1, 2].
The purpose of our study was to provide a radiological case series of patients meeting the criteria of Dercum’s syndrome, describing the salient imaging features that may allow differentiation from cellulitis or other soft tissue masses, such as atypical lipoma, and defining the role of imaging in these patients.
Methods and materials
Institutional review board (IRB) approval was obtained for a 10-year retrospective review of the Picture Archiving and Communications System (PACS). Using the Primordial© search engine embedded within the GE ISite PACS, a keyword search was performed to find cases interpreted by radiologists as Dercum, Dercum’s, Anders, adiposa dolorosa, or adiposis dolorosa. Additionally, cases that were previously saved in teaching files were reviewed. Exclusion criteria included patients with biopsy-proven malignancy in the extremity, biopsy-proven infection in the area, or deep venous thrombosis in the extremity.
Two fellowship trained musculoskeletal radiologists (4 and 7 years of experience) reviewed the reports and images to confirm findings and further document specific features. The radiology information system (GE-RIS) database and the electronic medical record (Cerner Powerchart) were reviewed to collect demographic data on age, sex, height, weight, symptoms of pain or infection, and indication for imaging. Indication for CT, MR, and US was divided into two groups, mass related and non-mass related. "Mass related" was defined as patients imaged to evaluate a soft-tissue mass with or without pain. "Non-mass related" was an examination performed not to evaluate the mass. The mass was an incidental finding. All patient notes were reviewed to assess for the criteria of Dercum’s syndrome, including asthenia, weakness, fatigability, emotional instability, depression, epilepsy, mental confusion, and true dementia. The combination of these symptoms, weight, and imaging characteristics was used to determine how many of the four criteria of Dercum’s syndrome were met per patient. Overweight was defined as a body mass index (BMI) of 30 or more. Clinical notes were reviewed to determine if the physicians recorded these patients as having Dercum’s syndrome.
A total of 17 patients were found. Mean patient age was 55 years (range 18 to 79), and 70.6 % were female (12 of 17). The data set was divided into two groups. The first consisted of patients meeting at least three of the four criteria for Dercum’s syndrome, and the second group consisted of those only meeting one or two of the criteria. For group 1, mean patient age was 61.3 years (range 43–78) and 55.6 % (5/9) were females, while for group 2, mean patient age was 55.2 years (range 18–79) and 87.5 % were female. Finally, histopathology results and cultures were reviewed if available.
A third group of age-, sex-, and body mass index (BMI)-matched patients with clinical diagnosis of lower extremity cellulitis was also reviewed to provide a comparison of imaging features to the Dercum’s groups. This group consisted of 10 patients with mean age of 52.3 years (range 34–72), and 60 % were female.
Recorded imaging features included the total number of lesions, location of the lesion, side of the body, three plane diameter, depth of soft tissue compartmental involvement (skin, skin and subcutaneous fat, skin and subcutaneous fat and muscle), and neurovascular features (involvement, sparing, or displacement) on the best available imaging modality.
Specific CT features recorded were the predominant lesion attenuation (fat, fluid, soft tissue, bone), pattern of the attenuation component (rounded/mass like, radial pattern, mixed), lesion definition (well defined/ill defined), presence of calcification, and enhancement pattern (none, non-mass like, mass like).
MR imaging features documented included both signal intensity on T1- and T2-weighted or short tau inversion recovery (STIR) sequences (low, intermediate, high in respect to skeletal muscle) as well as heterogeneous vs. homogeneous appearance on T1- and T2-weighted images.
For CT and MR, enhancement was determined to be non-mass like if the area enhancement was poorly circumscribed and faded at the periphery. Enhancement was determined to be mass like if the area of enhancement was well defined. The presence of skin thickening was defined as greater than two times the normal thickness.
Ultrasound imaging features recorded included homogeneous or heterogeneous ultrasound appearance, echotexture (hypoechoic, hyperechoic, or isoechoic to the subcutaneous fat), and presence of increased power and/or color Doppler flow. The presence of skin thickening was defined as greater than two times the normal thickness.
Results
Total number of masses in the Dercum’s/MLL groups was 25 with 8 patients having bilateral masses and 9 with a single lesion.
Group 1
A total of nine patients met at least three of the four criteria for Dercum’s syndrome (see Table 2). Three of the nine (33.3 %) met all four criteria. Physicians called two Dercum’s syndrome. Seven of nine patients underwent imaging for a mass-related indication. A total of 12 masses (3 bilateral) were found, 9 in the thigh (8 medial, 1 lateral), 1 in the posterior ankle, 1 in the anterior abdominal wall, and 1 in the posterior neck at the C6-7 level. Mean mass size was 18.6 cm AP (range 9.1 to 29.2 cm) × 14.1 cm CC (range 7.8 to 23 cm) × 9.68 cm transverse diameter (range 4.9 to 16.6 cm). All lesions were confined to the skin and subcutaneous tissue without involvement of the fascia or deep muscle. No cases demonstrated adjacent muscle edema. Neurovascular structures coursed normally through the involved areas without morphological change or displacement.
There were five CT (group 1) examinations. All lesions revealed sessile or pedunculated mass-like areas in the subcutaneous fat with a radial pattern of fluid attenuation with ill-defined borders (Fig. 1). No calcifications were noted. There was no contrast enhancement. All lesions demonstrated skin thickening.
CT appearance of group 1 masses. (a) Axial and (b) coronal CT images in a 56-year-old male show a focal pedunculated mass-like area in the medial proximal left thigh. There is skin thickening and fluid attenuation stranding of the subcutaneous fat. There is no fascial or muscular involvement. This patient had three of the four criteria of Dercum’s syndrome
There were six MRI (group 1) examinations. All cases demonstrate heterogeneous low-to-intermediate T1-weighted and high T2-weighted signal on MRI (Fig. 2). There was no post-contrast enhancement. All lesions demonstrated skin thickening.
MRI appearance of group 1 masses. (a) Axial T1-weighted (TR 425, TE 7), (b) axial T2-weighted (TR 7,049, TE 80) fat suppressed, and (c) axial T1-weighted (TR 425, TE 7) fat-suppressed post-gadolinium images in a 57-year-old female with a left thigh mass demonstrate a pedunculated mass-like area along the medial left thigh that has skin thickening, low signal intensity areas of subcutaneous edema on T1, and high signal intensity on fluid-sensitive images. There is a separation of the mass from the fascial planes of the left thigh. There is no contrast enhancement
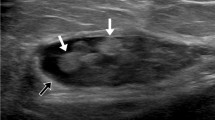
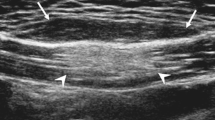
There were two ultrasound (group 1) examinations. Ultrasound images revealed heterogeneous areas of hyperechoic tissue in the subcutaneous fat layer with ill-defined margins. No color or power Doppler flow was present. All lesions demonstrated skin thickening (Fig. 3).
Group 2
A total of eight patients only met one or two criteria of Dercum’s syndrome. Five of eight patients underwent imaging for a mass-related indication. Thirteen total masses were analyzed (5 bilateral masses), 11 of which were in the medial thigh and 2 in the lateral thigh. Five CT, three MR, and two US examinations were available for review. Mean diameter of the masses was 15.02 cm AP (range 7.5 to 29.9 cm) × 18.4 cm CC (range 10.6 to 35.7 cm) × 10.1 cm transverse (range 3.1 to 32.6 cm). All lesions were confined to the skin and subcutaneous tissue without involvement of the fascia or deep muscle. No cases demonstrated adjacent muscle edema. Neurovascular structures coursed normally through the involved areas without morphological change or displacement.
Similar to group 1, CT and MR images demonstrated sessile or pedunculated mass-like areas in the subcutaneous fat with a radial pattern of attenuation on CT (Fig. 4) and low T1- and high T2-weighted signal on MRI (Fig. 5). No enhancement was present on post-gadolinium images. There was associated skin thickening in all cases with no involvement of the fascia or deeper muscle. There was no calcification. Ultrasound images revealed areas of hyperechoic tissue in the subcutaneous fat layer with ill-defined margins and associated skin thickening. No central color or power Doppler flow was present (Fig. 6). No cystic areas were present. Table 1 provides a comparison of the two groups, and Table 2 identifies the criteria met in each group.
CT appearance of a group 2 mass. (a) Axial CT image in a 67-year-old female with a painful thigh mass demonstrates a pendulous mass of 32 cm AP diameter × 28 cm transverse diameter between the thighs. It appears to originate from the right thigh. There is diffuse skin thickening and ill-defined increased attenuation of the subcutaneous fat. (b) Scout radiograph from the CT demonstrates the pendulous nature of the mass
MR appearance of a group 2 mass. (a) Axial T1-weighted (TR 674, TE 10), (b) axial STIR (TR 5,500, TE 37) fat-suppressed, and (c) axial T1-weighted (TR 542, TE 9.7) post-gadolinium fat suppressed images in a 52-year-old female show a mass along the posteromedial soft tissues of the right thigh with skin thickening, low T1- and high T2-weighted signal in the subcutaneous fat, and no significant post-contrast enhancement
No patients had records of rheumatic disease, cellulitis, or malignancy. Three of the 16 patients had lesion removal because of the size or discomfort with pathology results. Two cases in group 1 showed no signs of malignancy or infection. These two patients demonstrated adipose tissue traversed by large fibrous septa with no atypical cells. Patient 2 also had vascular aggregates at the intersection of adipose and fibrous tissues. The third patient, part of group 2, demonstrated pathology findings diagnosed as massive localized lymphedema, described as acute and chronic inflammatory infiltrate associated with hemorrhage and edema.
Group 3
Ten patients who were imaged for the indication of cellulitis comprised group 3. Five cases involved the medial thigh, one the posterior thigh, and four the thigh diffusely. Six CT, three MR, and one US examination were available for review. All lesions were confined to the skin and subcutaneous tissue without involvement of the fascia or deep muscle. No cases demonstrated adjacent muscle edema. Neurovascular structures coursed normally through the involved areas without morphological change or displacement. In nine of ten patients, CT and MR images demonstrated non-mass-like areas of high attenuation on CT (Fig. 7a and b) and low T1- and high T2-weighted signal on MRI. There was associated skin thickening in all but one case. The imaging appearance differed from the first two groups in that the attenuation was not in a radial pattern, but instead was more linear, diffuse, and non-mass like. There was also post-contrast enhancement in all three cases of contrast administration. One patient with a CT of the left thigh had a more mass-like appearance (12.3 cm transverse × 15.6 cm AP × 19.3 cm CC diameter) of high attenuation in a radial pattern in the subcutaneous tissues with skin thickening (Fig. 7c and d). This patient underwent biopsy because of the eventual development of an abscess, which confirmed clinical symptoms of cellulitis. This case had been given a differential diagnosis of adiposis dolorosa in the radiology report.
CT appearance of cellulits. (a) Axial and (b) coronal images of a 44-year-old male with a clinical diagnosis of cellulitis shows the non-mass-like appearance of fluid attenuation in the subcutaneous tissues of the right medial thigh with associated skin thickening. (c) Axial and (b) coronal CT images of the only patient in group 3 who had a mass-like appearance of cellulitis. Images show a pedunculated area of skin thickening with a radial pattern of edema in the subcutaneous fat of the medial left thigh. Clinical history and physical examination findings in this patient pointed to cellulitis, and biopsy confirmed clinical suspicion
The ultrasound case of cellulitis showed skin thickening and a cobblestone appearance of edema in the subcutaneous fat that was non-mass like. However, this case did have central power Doppler flow.
Discussion
The earliest descriptions of Dercum’s disease and syndrome included clinical pictures without radiological examples. The cases in the literature illustrating Dercum’s lesions show nonspecific areas of soft tissue prominence with increased fat and skin thickening [12, 13]. Our series is the largest to date of what was interpreted by musculoskeletal radiologists at our institution as lesions typical of Dercum’s disease and (in group 1) meeting at least three of the four clinical criteria for Dercum’s syndrome. The masses tended to be prominent pedunculated or sessile areas of subcutaneous fat edema in a radial pattern, with skin thickening and non-mass-like enhancement. These findings are consistent with the benign behavior of Dercum’s lesions. The thigh was the most frequent location in both groups. The medial thigh was involved five to eight times more frequently than the lateral thigh, suggesting friction may be a contributing factor in the development of this entity.
A reasonable differential diagnosis for Dercum’s disease would include cellulitis, an atypical lipomatous lesion, and massive localized lymphedema. Cellulitis is an infection of the skin and subcutaneous tissue. It is poorly defined similarly to Dercum’s disease and massive localized lymphedema. Cellulitis demonstrates high attenuation on CT with loss of the well-defined margin between the subcutaneous fat and underlying muscle [15]. On MRI, cellulitis demonstrates reticulated areas of intermediate T1-weighted signal and high signal on fluid-sensitive sequences within the subcutaneous fat [16]. As shown in group 3, cellulitis may occasionally have focal or circumferential soft tissue swelling, but typically does not have the sessile or pedunculated areas of soft tissue prominence mimicking a tumor on clinical inspection. Additionally, cellulitis has a more rapid onset of symptoms and appears different on clinical examination. An atypical lipomatous lesion is the proper term to describe subcutaneous lesions with histologic features identical to well-differentiated liposarcoma. This terminology is used because of the limited morbidity and lack of significant potential for dedifferentiation [17]. On CT, the lesion has predominantly fat attenuation with prominent thick septa and focal nodular regions usually less than 2 cm in size. The septations within an atypical lipomatous lesion are almost always thicker and more numerous than the septa within the normal adjacent subcutaneous tissue. The non-lipid components will demonstrate intermediate T1-weighted signal, high T2-weighted signal, and moderate to marked contrast enhancement [17] and be more sharply defined than the non-lipomatous components of DD or MLL. The atypical lipomatous lesion (subcutaneous well-differentiated liposarcoma) will have well-defined lesion borders, unlike cellulitis, Dercum’s disease, and massive localized lymphedema, which are all poorly defined.
Two of the three pathology cases in our series demonstrated findings of adipose tissue with fibrosepta without inflammation, which would fit with the imaging findings and exclude infection and malignancy. However, the third case (a group 2 lesion) demonstrated acute and chronic inflammation and was interpreted by the pathologist as massive localized lymphedema.
Massive localized lymphedema (MLL) was first described by Farshid and Weiss in 1988 [18]. A recent series of 22 patients by Maduch described large pendulous masses, usually located about the thighs, primarily in obese patients [19]. The description and appearance of imaging findings in their patients are similar to those found in our patient series. CT images of patients with MLL published by Wang [20] and Wu [21] are nearly identical to our CT images in both groups. MR images in a series of three patients [22, 23] also are similar to the images of both sets of our patients, with large pendulous masses of adipose tissue along the lower extremities with edema tracking along the fibrous septae in a lace-like fashion with associated dermal thickening. Thus, imaging findings of Dercum’s lesions and MLL appear quite similar in both the literature and our case series. Additionally, the patient populations are similar: obese, middle-aged individuals. Obese patients often have comorbid conditions, including diabetes, hypothyroidism, sleep apnea, and cardiac issues, which are associated with weakness and fatigue.
The physiology of MLL includes impedance of lymphatic flow due to obesity, leading to accumulation of protein-rich lymphatic fluid in the subcutaneous fatty tissues. This is often associated with reduced tissue oxygenation, resulting in fibrosis and a chronic inflammatory state. The inflammation and fibrosis often result in painful masses, further complicating differentiation between MLL and Dercum’s disease. Similar to a recent study by Tin, not all of the Dercum’s disease masses were painful (Table 2), which is the typical description. These particular patients were being imaged for another clinical indication, and the masses were incidental findings.
A potential difference between Dercum’s disease and MLL may be found on clinical examination, where in MLL the overlying skin displays a typical peau d’orange appearance of congested dermal lymphatics. Cracks, pits, fissures, or open wounds may develop, leading to leakage of lymph onto the skin, which can result in cellulitis [24]. However, theoretically in Dercum’s disease, the focal fatty masses could impede lymphatic flow, and the radial pattern of edema may represent lymphedema. Thus, imaging and clinical differentiation of these two entities is still not absolute. We are unsure whether these diagnoses can coexist.
Pathological differentiation seems the closest to a gold standard, although the literature is still not conclusive. When Dercum first described the disease in 1888, the most interesting histologic feature noted was inflammation of the nerves within the painful adipose tissue [25]. However, this feature has yet to be reliably reproduced in subsequent studies. Instead, reports of the histologic features vary from unremarkable adipose tissue to adipose tissue with mild pathological findings [26]. Pathological features identified include reactive changes, chronic inflammation (lymphocytes, plasma cells, and multinucleated giant cells), increased connective tissue, granulomas, microthrombi, and angiomas [27, 28]. In MLL, histology shows dermal fibrosis, expansion of fibrous septa between fat lobules with increased numbers of stromal fibroblasts, lymphatic proliferation and lymhangiectasia, multinucleated fibroblastic cells, and marked vascular proliferation. Thus, some of the features overlap with DD and some of our pathology results [29].
In our practice, we will describe these lesions as typical of Dercum’s disease and/or massive localized lymphedema. This interpretation will prevent unnecessary biopsy or characterization as a malignant entity since the imaging excludes neoplastic entities, particularly atypical lipoma.
Limitations of our study include that pathology results were only available in 3 of the 17 cases. Additionally, clinical histories were retrospective and dependent upon physician-reported symptoms and diagnoses. Our institution does not have a psychiatric unit; thus patients with psychiatric disturbances may have been missed, as those records are not accessible to us. Also in this era of transparency and the patient portal, non-psychiatrists may be reluctant to suggest psychiatric diagnoses in the medical record. Finally, our search only investigated cases diagnosed as Dercum’s disease by our radiologists rather than looking for Dercum’s syndrome in the electronic medical record. There may be bias in the interpretation of our radiologists.
In conclusion, this is the largest case series demonstrating the imaging findings of Dercum’s syndrome and/or massive localized lymphedema. These two entities appear identical by imaging and physical examination of skin findings (peau d’orange, cracks, pits, fissures, or open wounds). Cellulitis has similar imaging characteristics and ill-defined borders, but will not have the mass-like clinical presentation of sessile or pedunculated soft tissue prominence. Atypical lipoma (subcutaneous well-differentiated liposarcoma) will have sharply defined lesion margins and sharp distinction between lipomatous and non-lipomatous components. The typical imaging findings are important to understand and aid in the exclusion of malignancy.
References
Dercum FX. Three cases of a hitherto unclassified affection resembling in its grosser aspects obesity, but associated with special nervous symptoms: adiposis dolorosa. Am J Med Sci. 1892;104:521.
Brodovsky S, Westreich M, Leibowitz A, Schwartz Y. Adiposis dolorosa (Dercum’s disease): 10-year follow-up. Ann Plast Surg. 1994;33:664–8.
Christopher DM, Unni KK, Mertens F. WHO Classification of Tumors. Pathology and Genetics: Tumors of Soft Tissue and Bone. Lyon, France: IARC Press; 2002. p. 19–34.
Cantu JM, Ruiz-Barquin E, Jimenez M, et al. Autosomal dominant inheritant in adiposis dolorosa. Humangeretik. 1973;18:89.
Lynch HT, Harlan WL. Hereditary factors in adiposis dolorosa. Am J Hum Genet. 1936;15:184.
Jensen JJ, Kiilerich S. Adiposis dolorosa: Dercum’s disease. Ugeskr Leeger. 1991;153(50):3564.
Mella BA. Adiposis dolorosa. Univ Mich Med Cent J. 1967;33:79.
Eisman J, Swezey RL. Juxta-articular adiposis dolorosa: what is it? Report of 2 cases. Ann Rheum Dis. 1979;38:479.
Dolph JL, Demuth RJ, Miller SH. Familial multiple lipomatosis. Plast Reconstr Surg. 1980;66:620.
Bonatus TJ, Alexander AH. Dercum’s disease (adiposis dolorosa): a case report and review of the literature. Clin Orthop. 1986;205:251.
Kling DH. Juxta articular adiposis dolorosa. Arch Surg. 1937;34:599.
Amine B, Leguilchard F, Benhamou CL. Dercum's disease (adiposis dolorosa): a new case-report. Joint Bone Spine. 2004;71(2):147–9.
Kawale J et al. Adiposis dolorosa of scalp presenting with severe headache: an unusual case. J Headache Pain. 2010;11(6):539–41.
Tins BJ, Matthews C, Haddaway M, et al. Adiposis dolorosa (Dercum’s disease): MRI and ultrasound appearances. Clin Radiol. 2013;68:1047–53.
Beauchamp NJ. Jr1, Scott WW Jr, Gottlieb LM, Fishman EK. CT evaluation of soft tissue and muscle infection and inflammation: a systematic compartmental approach. Skelet Radiol. 1995;24(5):317–24.
Kransdorf MJ, Murphey MD. Imaging of Soft Tissue Tumors. Publication Date: May 2, 2006 | ISBN-10: 0781747716 | ISBN-13: 978-0781747714 | Edition: Second
Walker EA, Salesky JS, Fenton ME, Murphey MD. Magnetic resonance imaging of malignant soft tissue neoplasms in the adult. Radiol Clin N Am. 2011;49(6):1219–34.
Farshid G, Weiss SW. Massive localized lymphedema in the morbidly obese: a histologically distinct reactive lesion simulating liposarcoma. Am J Surg Pathol. 1998;22:1277–83.
Manduch M, Oliveira AM, Nascimento AG, et al. Massive localized lympoedemea: a clinicopathological study of 22 cases and review of the literature. J Clin Pathol. 2009;62:808–11.
Wang NS, Walters RF, Warren SJ. Massive localized lymphedema: a soft tissue process that may present to dermatologists. Am J Dermatopathol. 2010;32:380–3.
Wu D, Gibbs J, Corral D, Intengan M, Brooks JJ. Massive localized lymphedema: additional locations and association with hypothyroidism. Hum Pathol. 2000;31(9):1162–8.
Narayanarao T, Suvarchala A, Krishnababu G. Pseudosarcoma-massive localized lymphoedema in morbidly obese—a rare entity: case report. Int J Surg Case Rep. 2012;3(8):389–91.
Khanna M, Naraghi A, Salonen D, et al. Massive localized lymphoedema: clinical presentation and MR imaging characteristic. Skeletal Radiol. 2011;40:647–52.
Wang NS, Walters RF, Warren SJ. Massive localized lymphedema: a soft tissue process that may present to dermatologists. Am J Dermatopathol. 2010;32(4):380–3.
Dercum FX. A subcutaneous connective tissue dystrophy of the arms and back, associated with symptoms resembling myxoedema. Univ Med Mag (Philadelphia). 1888;1:1–11.
Campen RB, Sang CN, Duncan LM. Case records of the Massachusetts general hospital. Case 25-2006. A 41-year-old woman with painful subcutaneous nodules. N Engl J Med. 2006;355:714–22.
Herbst KL, Asare-Bediako S. Adiposis dolorosa is more than painful fat. Endocrinologist. 2007;17:326–34.
Hansson E, Svensson H, Stenram U, Brorson H. Histology of adipose tissue inflammation in Dercum's disease, obesity and normal weight controls: a case control study. J Inflamm (Lond). 2011;8:24.
Manduch M, Oliveira AM, Nascimento AG, Folpe AL. Massive localized lymphedema: a clinicopathological study of 22 cases and review of the literature. J Clin Pathol. 2009;62(9):80–11.
Conflicts of interest
No conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Petscavage-Thomas, J.M., Walker, E.A., Bernard, S.A. et al. Imaging findings of adiposis dolorosa vs. massive localized lymphedema. Skeletal Radiol 44, 839–847 (2015). https://doi.org/10.1007/s00256-015-2114-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00256-015-2114-8