Abstract
Objective
To intra-individually assess the association of inflammation severity and cartilage composition measured by RAMRIS synovitis sub-score and delayed gadolinium-enhanced magnetic resonance imaging of the cartilage (dGEMRIC) of metacarpophalangeal (MCP) joints in patients with rheumatoid arthritis (RA).
Methods
Forty-three patients with RA according to ACR/EULAR classification criteria (age 52.9 ± 14.5 years, range, 18–77 years) were included in this study. All study participants received 3-T MRI scans of the metacarpophalangeal joints of the second and third finger (MCP 2 and 3). The severity of synovitis was scored according to the RAMRIS synovitis sub-score by two readers in consensus. In the cases with identical synovitis sub-scores, two radiologists decided in consensus on the joint with more severe synovitis. Cartilage composition was assessed with dGEMRIC. To test the association of inflammation severity and cartilage damage and in order to eliminate inter-patient confounders, each patient’s MCP 2 and 3 were dichotomized into the joint with more severe synovitis versus the joint with less severe synovitis for a paired Wilcoxon test of dGEMRIC value.
Results
There was a significant difference of dGEMRIC value (median of difference: 47.12, CI [16.6; 62.76]) between the dichotomized MCPs (p = 0.0001). There was a significant correlation between dGEMRIC value and RAMRIS synovitis grading of the joint with more severe synovitis (r = 0.5; p < 0.05) and the joint with less severe synovitis (r = 0.33; p < 0.05).
Conclusions
Our data concur with the concept that synovitis severity is associated with cartilage damage. The local inflammatory status on a joint level correlated significantly with the extent of cartilage degradation in biochemical MRI.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Biochemical MRI of cartilage is a validated tool for assessing cartilage degradation in clinical trials of RA and osteoarthritis [1–3]. Delayed gadolinium-enhanced MRI of the cartilage (dGEMRIC) is a magnetic resonance imaging (MRI) feature to visualize proteoglycan loss in cartilage composition [4–6]. It has been demonstrated that cartilage changes measured by dGEMRIC correlate with histological analysis [7]. Based on improvements in MRI techniques, it is possible to assess cartilage composition of small joints that are frequently affected in RA [4]. A loss of proteoglycans has been demonstrated in early RA and seems to precede morphological changes in cartilage of small finger joints [2].
Uncontrolled RA is characterized by progressive joint destruction and long-term functional disability [8]. Inflammation of the synovial membrane is associated with destruction of bone and cartilage [9]. The degree of inflammation highly correlates with functional impairment and the development of joint destruction over time leads to disability [10–12].
The therapy with disease-modifying antirheumatic drugs (DMARD) and biologicals aims at disease control and can halt the progression of joint destruction [13, 14].
This has put monitoring of joint damage in the focus of radiologic attention in the follow-up of RA. In 2003, the Outcome Measures in RA Clinical Trials (OMERACT) group with the RA MRI Score (RAMRIS) established a highly reliable sum-score based on the semi-quantitative rating of the severity of synovitis, bone marrow edema and erosions in hand and wrist joints [15]. The RAMRIS scoring system has been applied in therapy-response trials in RA [16, 17]. However, the system does not consider cartilage destruction in RA. In 33 patients with RA, Herz et al. investigated the relation between inflammation of synovia and cartilage degradation measured with biochemical MRI in an inter-individual study design. Synovitis was determined with the RAMRIS synovitis sub-score and cartilage degradation was assessed with dGEMRIC. They found a correlation between high synovitis sub-score and low dGEMRIC values, suggestive of cartilage damage [1].
Our hypothesis was that cartilage damage measured by dGEMRIC of MCP joints in patients with rheumatoid arthritis (RA) is associated with the severity of joint inflammation on a patient level.
Materials and methods
Patients
This study was approved by the institutional review board, and informed consent was obtained from all patients. Forty-three patients with rheumatoid arthritis, including (35 female; eight male, age 52.9 ± 14.5 years, range, 18–77 years, disease duration 2.9 ± 4.9 years, range, <0.5–19 years, DAS28 3.7 ± 1.5) were enrolled in this retrospective study. All patients fulfilled the 2010 American College of Rheumatology/European League Against Rheumatism Rheumatoid arthritis classification criteria [18, 19]. All 43 patients received 3-T MRI (Magnetom Trio; Siemens Healthcare) scans of the clinically dominant hand.
MR imaging
MRI was performed of the dominantly affected hand on a 3-T MRI system (Magnetom Trio; Siemens Healthcare) to assess synovitis T1-weighted images pre–and post-contrast were performed of the MCP joints with a maximum slice thickness of 3 mm in at least two coronal planes and a transversal fat-suppressed T1-weighted sequence [field of view (FOV) 13 × 18 cm, matrix size 256 × 182 pixels]. Gadolinium-MRI contrast agent was applied intravenously (0.4 ml/kg body weight of Gd-DTPA2-, Magnevist; Schering).
Biochemical MRI with dGEMRIC of the MCP joints of the index and middle fingers was performed with two 4-cm loop surface coils placed above and beneath the MCP joint. The size of the coils and the FOV limited the examination to two adjacent joints: MCP 2 and 3. Subjects were imaged in a prone position with the hand extended over the head. dGEMRIC was acquired 40 min after contrast agent administration.
Variable flip-angle three-dimensional gradient-echo imaging (with two flip angles) was used for T1 calculation [4]. Flip angles were set to 5° and 26°. Twenty-two sagittal slices with a thickness of 2 mm were positioned perpendicular to the joint spaces. The FOV was 73 × 90 mm. The matrix of 312 × 384 provided an in-plane resolution of 233 μm. Total acquisition time was 2.25 min.
To reduce movement artefacts, motion correction was performed on each patient’s MCP joint before image analysis. For motion correction we used the software STROKETOOL (http://www.digitalimagesolutions.de, Frechen, Germany) as described elsewhere in detail [20].
Image analysis
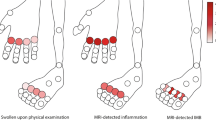
Standard MR images were read in consensus by two radiologists. Images were evaluated for synovitis (range, 0–3) according to the Outcome Measures in RA Clinical Trials (OMERACT) group RAMRIS guidelines established in 2003 [15]. Synovitis scoring was performed of second and third MCP. In 20 cases of identical RAMRIS synovitis sub-scores in MCP 2 and 3, a subjective gradation into the joint with more severe synovitis and the joint with less severe synovitis was undertaken by two radiologists in consensus with 2 and 8 years of experience in musculoskeletal radiology (Fig. 1). The two radiologists were blinded to the dGEMRIC values. Based on this data, the RAMRIS synovitis sub-score of second and third MCP, each patient’s pair of MCP2 and MCP3 was dichotomized into the joint with more severe synovitis vs. the joint with less severe synovitis (Fig. 2). The synovitis sub-score grading in a joint with more vs. a joint with less severe synovitis refers to dichotomization. The intraindividual analyses were chosen to eliminate interindividual cofactors, such as the different inflammatory status on a joint level between different patients.
Transversal fat-suppressed T1-weighted sequence [field of view (FOV) 13 × 18 cm, matrix size 256 × 182 pixels] of a patient with RA showed identical RAMRIS synovitis sub-scores of MCP 2 and 3 (white arrows). In this condition, two radiologists decided in consensus on the joint with more severe synovitis and the joint with less severe synovitis. In this patient, MCP 3 was defined as the joint with more severe synovitis. Additionally, there is a tenosynovitis of the flexor tendon of the third finger
Representative dGEMRIC image of the second (a) and third (b) metacarpophalangeal joint of a patient with RA. Sagittal contrast-enhanced T1-weighted images (repetition time 15 ms, time to echo 3.34 ms, flip angle 5°) with a dGEMRIC color map overlay. Color-coding indicates high glycosaminoglycan (GAG) content (green-blue) to low GAG content (red-orange). Transversal fat-suppressed T1-weighted sequence [field of view (FOV) 13 × 18 cm, matrix size 256 × 182 pixels] of the same patient with MCP synovitis (c). In this patient, MCP 3 is the joint with the higher RAMRIS synovitis sub-score. dGEMRIC values demonstrate low GAG content in the joint with more severe synovitis
Molecular imaging with dGEMRIC was performed of second and third MCP. To determine cartilage quality, T1 maps were analyzed using region of interest (ROI) measurements. T1 values were calculated pixelwise using the formula
where
and S1(x,y,z), S2(x,y,z) are the pixel intensities corresponding to the different flip angles. Gradient-echo images with a flip angle of 5° were used as an anatomic reference for cartilage identification, and ROIs were set in the phalangeal and metacarpal cartilage of the MCP joints of the index and middle fingers. The ROIs were transferred to the co-registered T1 map. The dGEMRIC value, T1 [ms] and ROI size (number of pixels) were recorded.
Statistical analysis
Paired Wilcoxon signed-rank test for dichotomous analyses and Spearman rho correlations between RAMRIS scores and dGEMRIC values of MCP 2 and 3 were performed using SPSS software, version 22. p values less than 0.05 were considered significant. The values for dGEMRIC are presented as the mean ± SD.
Results
dGEMRIC value of the joint with more severe synovitis was 369 ± 137 ms, dGEMRIC value of the joint with less severe synovitis was 421 ± 129 ms. RAMRIS synovitis sub-score of the joint with more severe synovitis was 2.51 (range, 1–3), synovitis sub-score of the joint with less severe synovitis MCP was 1.86 (range, 0–3). There was a significant difference between the dGEMRIC value of dichotomized MCPs (p = 0.0001; Fig. 3). The median of difference was 47.12, CI [16.6; 62.76].
dGEMRIC values of the joint with more severe synovitis (red) compared to the dGEMRIC values of the joint with less severe synovitis (blue). There was a significant difference between both groups, indicating an association between MRI synovitis sub-score on a joint level and biochemical cartilage composition (p = 0.0001)
There was a significant negative correlation between dGEMRIC value and RAMRIS synovitis sub-scores of the joint with more severe synovitis in dichotomous analysis (r = 0.5; p < 0.05; Fig. 4) as well as between dGEMRIC value and RAMRIS synovitis sub-scores of the joint with less severe synovitis (r = 0.33; p < 0.05; Fig. 5). In our patient population, only four patients showed a higher dGEMRIC value in the joint with more severe synovitis compared to the joint with less severe synovitis (Fig. 6).
Dichotomous dGEMRIC values of MCP 2 and 3 in percent, the joint with less severe synovitis was represented as 100%. The left side represents the dGEMRIC value of the joint with less severe synovitis (100%). The right side shows the dGEMRIC value of the joint with more severe synovitis (in relation to 100% of the joint with less severe synovitis). Out of 43 examined hands, 39 MCP pairs showed lower dGEMRIC values of the joints with more severe synovitis (black lines)
Discussion
dGEMRIC, as one method of biochemical MRI detecting cartilage degradation, is increasingly commonly used in clinical trials on cartilage changes in RA [2, 21, 22]. With dGEMRIC, it is possible to detect proteoglycan loss after the intravenous application of negatively charged contrast agent (gadolinium diethylenetriamine pentaacetate anion—Gd-DTPA). The negatively charged Gd-DTPA penetrates cartilage in an inverse relationship to the concentration of negatively charged glycosaminoglycan side chains of proteoglycan. A depletion of proteoglycan content in degenerated cartilage results in an accumulation of the paramagnetic gadolinium ions. This consecutively accelerates T1 relaxation time [23].
Our data show that high inflammatory MRI scores were associated with cartilage proteoglycan loss on a patient level. The joint with a higher RAMRIS synovitis sub-score demonstrated a significantly lower dGEMRIC value in the intra-individual analysis representing a higher degree of cartilage destruction. In our patient population, only four patients showed a higher dGEMRIC value of the joint with more severe RAMRIS synovitis sub-score compared to the joint with a lower RAMRIS synovitis sub-score.
Clinical remission with cessation of inflammatory activity is a major aim in the treatment of RA [24]. A possible dissociation of systemic inflammatory activity from joint destruction was reported and puts preservation of joint integrity into focus of therapy [8]. MRI is a validated tool in monitoring progression of joint destruction in RA. Gandjbakhch and colleagues reported on a subclinical inflammation in RA patents in remission, which may be an explanation for structural progression despite effective treatment [24]. Tiderius et al. demonstrated that cartilage damage in biochemical MRI continues irrespective of disease activity following therapy escalation with TNF-alpha-blockers [21, 25].
Several other studies suggested a relationship between synovitis severity and joint damage [1, 24]. We found that the degree of cartilage proteoglycan loss was significantly associated with MRI sub-score of synovitis severity in a particular pair of adjacent joints.
Herz et al. showed a significant inter-individual correlation between MRI synovitis sub-score and cartilage proteoglycan loss in a cohort study design [1]. In our study we intra-individually examined MCP 2 and 3 with regard to cartilage proteoglycan loss and RAMRIS synovitis sub-score on a patient level to diminish confounders between subjects such as disease duration, age, gender, or therapy effects.
Our results support the concept that inflammatory severity is associated with cartilage damage on a single joint level.
Our study has limitations. No synovial and cartilage biopsies for histological analysis as a gold standard in evaluation of joint inflammation were available. Only few studies prepared synovial biopsies as gold standard [26]. However, RAMRIS synovitis sub-score and dGEMRIC are well-established methods to assess synovial inflammation [27] and cartilage damage [7]. Additionally, the absolute values of dGEMRIC vary among different studies and protocols [1]. The lack of a standard protocol for biochemical cartilage imaging limits the comparability of dGEMRIC between individual studies.
In chondromalacia and osteoarthritis, increased cartilage perfusion in dynamic MRI has been published, suggestive of increased extracellular spaces in these conditions [28]. This finding yet awaits confirmation by other study groups. In RA, no data on cartilage perfusion are available yet. Possibly hyperperfusion leads to a bias in dGEMRIC values in RA. We found a direct correlation between high inflammatory MRI scores and low dGEMRIC values on a paired joint level, but we also detected low dGEMRIC values in patients with low or moderate synovitis.
Conclusions
The significant association of cartilage composition and RAMRIS synovitis sub-score supports the concept that inflammation and cartilage damage are coupled on a local joint level.
References
Herz B, Albrecht A, Englbrecht M, Welsch GH, Uder M, Renner N, Schlechtweg P, Paul D, Lauer L, Engelke K, Janka R, Rech J, Schett G, Finzel S: Osteitis and synovitis, but not bone erosion, is associated with proteoglycan loss and microstructure damage in the cartilage of patients with rheumatoid arthritis. Ann Rheum Dis 2013.
Miese F, Buchbender C, Scherer A, Wittsack HJ, Specker C, Schneider M, et al. Molecular imaging of cartilage damage of finger joints in early rheumatoid arthritis with delayed gadolinium-enhanced magnetic resonance imaging. Arthritis Rheum. 2012;64:394–9.
Liebl H, Joseph G, Nevitt MC, Singh N, Heilmeier U, Karupppasamy S, Jungmann PM, McCulloch CE, Lynch JA, Lane NE, Link TM. Early T2 changes predict onset of radiographic knee osteoarthritis: data from the osteoarthritis initiative. Ann Rheum Dis 2014.
Miese FR, Ostendorf B, Wittsack HJ, Reichelt DC, Mamisch TC, Zilkens C, et al. Metacarpophalangeal joints in rheumatoid arthritis: delayed gadolinium-enhanced MR imaging of cartilage–a feasibility study. Radiology. 2010;257:441–7.
Bashir A, Gray ML, Burstein D. Gd-DTPA2- as a measure of cartilage degradation. Magn Reson Med. 1996;36:665–73.
Burstein D, Velyvis J, Scott KT, Stock KW, Kim YJ, Jaramillo D, et al. Protocol issues for delayed Gd(DTPA)(2-)-enhanced MRI (dGEMRIC) for clinical evaluation of articular cartilage. Magn Reson Med. 2001;45:36–41.
Zilkens C, Miese FR, Crumbiegel C, Kim YJ, Herten M, Antoch G, et al. Magnetic resonance imaging and histology of ovine hip joint cartilage in two age populations: a sheep model with assumed healthy cartilage. Skeletal Radiol. 2013;42:699–705.
Lafeber FP, Van der Laan WH. Progression of joint damage despite control of inflammation in rheumatoid arthritis: a role for cartilage damage driven synovial fibroblast activity. Ann Rheum Dis. 2012;71:793–5.
Smolen JS, Aletaha D, Steiner G. Does damage cause inflammation? revisiting the link between joint damage and inflammation. Ann Rheum Dis. 2009;68:159–62.
Welsing PM, Landewé RB, van Riel PL, Boers M, van Gestel AM, van der Linden S, et al. The relationship between disease activity and radiologic progression in patients with rheumatoid arthritis: a longitudinal analysis. Arthritis Rheum. 2004;50:2082–93.
Aletaha D, Smolen J, Ward MM. Measuring function in rheumatoid arthritis: identifying reversible and irreversible components. Arthritis Rheum. 2006;54:2784–92.
Jansen LM, van der Horst-Bruinsma IE, van Schaardenburg D, Bezemer PD, Dijkmans BA. Predictors of radiographic joint damage in patients with early rheumatoid arthritis. Ann Rheum Dis. 2001;60:924–7.
Smolen JS, Aletaha D. Monitoring rheumatoid arthritis. Curr Opin Rheumatol. 2011;23:252–8.
Landewé R, van der Heijde D, Klareskog L, van Vollenhoven R, Fatenejad S. Disconnect between inflammation and joint destruction after treatment with etanercept plus methotrexate: results from the trial of etanercept and methotrexate with radiographic and patient outcomes. Arthritis Rheum. 2006;54:3119–25.
Ostergaard M, Peterfy C, Conaghan P, McQueen F, Bird P, Ejbjerg B, et al. OMERACT rheumatoid arthritis magnetic resonance imaging studies. Core set of MRI acquisitions, joint pathology definitions, and the OMERACT RA-MRI scoring system. J Rheumatol. 2003;30:1385–6.
Østergaard M, Emery P, Conaghan PG, Fleischmann R, Hsia EC, Xu W, et al. Significant improvement in synovitis, osteitis, and bone erosion following golimumab and methotrexate combination therapy as compared with methotrexate alone: a magnetic resonance imaging study of 318 methotrexate-naive rheumatoid arthritis patients. Arthritis Rheum. 2011;63:3712–22.
Hetland ML, Ejbjerg B, Horslev-Petersen K, Jacobsen S, Vestergaard A, Jurik AG, et al. group Cs: MRI bone oedema is the strongest predictor of subsequent radiographic progression in early rheumatoid arthritis. Results from a 2-year randomised controlled trial (CIMESTRA). Ann Rheum Dis. 2009;68:384–90.
van Gestel AM, Prevoo ML, Van’t Hof MA, van Rijswijk MH, van de Putte LB, van Riel PL. Development and validation of the european league against rheumatism response criteria for rheumatoid arthritis. Comparison with the preliminary american college of rheumatology and the world health organization/international league against rheumatism criteria. Arthritis Rheum. 1996;39:34–40.
Aletaha D, Neogi T, Silman AJ, Funovits J, Felson DT, Bingham CO, et al. 2010 rheumatoid arthritis classification criteria: An american college of rheumatology/european league against rheumatism collaborative initiative. Arthritis Rheum. 2010;62:2569–2581.
Miese F, Kröpil P, Ostendorf B, Scherer A, Buchbender C, Quentin M, et al. Motion correction improves image quality of dGEMRIC in finger joints. Eur J Radiol. 2011;80:e427–431.
Tiderius CJ, Sandin J, Svensson J, Dahlberg LE, Jacobsson L. Knee cartilage quality assessed with dGEMRIC in rheumatoid arthritis patients before and after treatment with a TNF inhibitor. Acta Radiol. 2010;51:1034–7.
Buchbender C, Scherer A, Kröpil P, Körbl B, Quentin M, Reichelt DC, et al. Cartilage quality in rheumatoid arthritis: comparison of T2* mapping, native T1 mapping, dGEMRIC, δr1 and value of pre-contrast imaging. Skeletal Radiol. 2012;41:685–92.
Crema MD, Roemer FW, Marra MD, Burstein D, Gold GE, Eckstein F, et al. Articular cartilage in the knee: current MR imaging techniques and applications in clinical practice and research. Radiographics. 2011;31:37–61.
Gandjbakhch F, Conaghan PG, Ejbjerg B, Haavardsholm EA, Foltz V, Brown AK, et al. Synovitis and osteitis are very frequent in rheumatoid arthritis clinical remission: results from an MRI study of 294 patients in clinical remission or low disease activity state. J Rheumatol. 2011;38:2039–44.
Christensen AF, Horslev-Petersen K, Christgau S, Lindegaard HM, Lottenburger T, Junker K, et al. Uncoupling of collagen ii metabolism in newly diagnosed, untreated rheumatoid arthritis is linked to inflammation and antibodies against cyclic citrullinated peptides. J Rheumatol. 2010;37:1113–20.
Vordenbäumen S, Sewerin P, Lögters T, Miese F, Schleich C, Bleck E, et al. Inflammation and vascularisation markers of arthroscopically-guided finger joint synovial biopsies reflect global disease activity in rheumatoid arthritis. Clin Exp Rheumatol. 2014;32:117–20.
Conaghan P, Lassere M, Østergaard M, Peterfy C, McQueen F, O’Connor P, et al. OMERACT rheumatoid arthritis magnetic resonance imaging studies. Exercise 4: an international multicenter longitudinal study using the RA-MRI score. J Rheumatol. 2003;30:1376–9.
Sanz R, Martí-Bonmatí L, Rodrigo JL, Moratal D. MR pharmacokinetic modeling of the patellar cartilage differentiates normal from pathological conditions. J Magn Reson Imaging. 2008;27:171–7.
Acknowledgments
The study was supported by a grant from the Deutsche Forschungsgemeinschaft (DFG - MI 1575/1-1).
Conflict of interest
There are no conflicts of interest to disclose.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Schleich, C., Müller-Lutz, A., Sewerin, P. et al. Intra-individual assessment of inflammatory severity and cartilage composition of finger joints in rheumatoid arthritis. Skeletal Radiol 44, 513–518 (2015). https://doi.org/10.1007/s00256-014-2045-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00256-014-2045-9