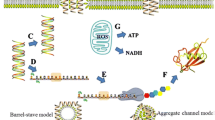
Abstract
Antibiotic growth promoters (AGPs) have been administered in livestock for decades to improve food digestion in growing animals, while also contributing to the control of microbial pathogens. The long-term and indiscrimate use of AGPs has generated genetic modifications in bacteria, leading to antimicrobial resistance (AMR), which can be disseminated to commensal and pathogenic bacteria. Thus, antimicrobial peptides (AMPs) are used to replaced AGPs. AMPs are found in all domains of life, and their cationic characteristics can establish electrostatic interactions with the bacterial membrane. These molecules used as growth promoters can present benefits for nutrient digestibility, intestinal microbiota, intestinal morphology, and immune function activities. Therefore, this review focuses on the application of AMPs with growth promoting potential in livestock, as an alternative to conventional antibiotic growth promoters, in an attempt to control AMR.
Key points
• The long-term and indiscriminate use of AGPs in animal food can cause AMR.
• AMPs can be used as substitute of antibiotics in animal food suplementation.
• Animal food suplementated with AMPs can provied economic efficiency and sustainable livestock production.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Antibiotics have been of vast importance to human health and are also employed in farm animal health to control disease and as growth promoters. Antibiotic growth promoters (AGPs) have been administered in sub-therapeutic doses with the role of eradicating or inhibiting pathogenic bacteria (Hugues and Heritage 2004). AGPs are administered in livestock to improve the animals’ digestion, so that they get the highest benefit from foodstuffs and grow into strong and healthy individuals (NOAH 2001; U.S. Food and Drug Administration 2015). Although the AGP mechanism of action is unclear, it is supposed that AGPs inhibit the sensitive populations of bacteria in the intestines, and decrease energy loss with fermentative processes (Jensen 1998). AGPs also act in reducing the frequency and severity of subclinical infections (George et al. 1982; Brennan et al. 2003); they decrease microbial use of nutrients, and boost nutrient absorption, due to intestinal wall thinning (Snyder and Wostmann 1987; Feighner and Dashkevicz 1987; Knarreborg et al. 2004; Huyghebaert et al. 2011). Thus, by regulating the microbial population and controlling microbial nutrients, energy is transformed into animal growth (Hugues and Heritage, 2004).
AGPs have been employed to improve the development of farm animals since the 1950s (Jones and Ricke 2003; Brown et al. 2017; Ronquillo and Hernandez 2017). Over the years, several antibiotics have been administered in livestock as GPs (Table 1). This use helped to produce meat on an industrial scale (Van Boeckel et al. 2015). However, the indiscriminate use of AGPs for decades caused genetic modifications in bacteria and has led to antimicrobial resistance (AMR), which can be disseminated to commensal and pathogenic bacteria (Aslam et al. 2018; Founou et al. 2016; Innes et al. 2020; Li et al. 2018). AGPs are therefore the subject of controversy associated with their risks and advantages.
Thus, antimicrobial peptides (AMPs) are being used to replace AGPs (Jenssen et al. 2006; Cheng et al. 2014; Zhao et al. 2016). AMPs are found in all domains of life, present chemical diversity and structure, and usually present cationic and amphipathic properties (Cardoso et al. 2020; Gomes et al. 2018; Spohn et al. 2019; Brogden 2005; Jenssen et al. 2006). The cationic characteristics of AMPs can establish electrostatic interactions with the bacterial membrane, which is commonly composed of negatively charged phospholipids (Hancock and Chapple 1999; Shai 2002). AMPs can interact with the outer membrane, disturbing its physical integrity, and may also be translocated across the membrane and act on internal targets (Hancock and Sahl 2006). AMPs exhibit activity against bacteria, fungi, viruses, and cancer (Cardoso et al. 2020; Hwang et al. 2011; Oshiro et al. 2019; Rodrigues et al. 2019; Saido-Sakanaka et al. 2004). In addition, these peptides can act indirectly by stimulating the host’s immune system (Ageitos et al. 2017; Hancock 2001; Ward et al. 2013; Wang et al. 2016; WHO 2014). Therefore, this review summarized the application of AMPs as growth promoters with potential for livestock, as an alternative to traditional antibiotics, in an effort to control AMR.
Concern about growth promoters in livestock
As mentioned, the most common growth promoters applied in livestock are antibiotics (AGPs). However, the excessive use of antimicrobials has already contributed to the emergence of global public health problems, such as antimicrobial resistance, hypersensitivity responses, and damage to normal bacterial biota (Ronquillo and Hernandez 2017; Bacanlı and Başaran 2019). Thus, concern about the risks associated with AGPs in livestock and the consequences for human health has been increasing (Hughes and Heritage 2004; Marquardt and Li 2018; Tona 2018; Ma et al. 2020).
AMR development generally occurs through mutations (vertical AMR acquisition) and gene horizontal transfer (horizontal AMR acquisition) (Nadeem et al. 2020; Vidovic and Vidovic 2020). Occurrence of vertical AMR acquisition may be related with the exposure of bacterial populations to antibiotics, even in low concentrations. In this situation, any mutation that confers partial or full resistance against these antibiotics can be positively selected and transferred to subsequent generations, and this resistance may be against a specific antibiotic or a whole class (Tenover 2006; Davies and Davies 2010; Vidovic and Vidovic 2020). Horizontal AMR acquisition may occur by transference of resistant genetic elements such as plasmids or transposons, by horizontal transfer such as conjugation, transduction, or other mechanisms. Such processes can provide resistance against several antibiotic classes (Fig. 1) (Tenover 2006; Medina et al. 2020; Nadeem et al. 2020; Vidovic and Vidovic 2020).
Some studies estimated that approximately 90% of antibiotics applied in farm animals can be excreted by urine and/or feces. Furthermore, a number of antibiotics applied in animals can be stored in tissues such as muscle, milk, eggs, and fat. These residues can be dispersed by water and waste-water systems, or by fertilizers that employ manure, and cause the contamination of soil and consequently affect the soil microbiota, water, and plants (Ronquillo and Hernandez 2017; Iwu et al. 2020). Humans can be contaminated by AGPs in different ways, by ingestion in food, including milk, eggs, and meat, or by consuming water with residues of AGPs (Ben et al. 2019; Kraemer et al. 2019).
In this context, regulatory agencies, such as the European Commission and the US Food and Drug Administration (FDA), have established limits for antibiotic residues in animal foods (European Commission 1998; FDA 2016; FAO 2018). In 2006, the use of AGPs was completely banned in the European Union countries, and the use of antibiotics is permitted only for veterinary purposes (ten Doeschate and Raine 2006). In recent years, countries such as Canada, India, China, and Malaysia have restricted the use of AGPs in livestock, but many countries do not present any formal restrictions on AGPs (Brown et al. 2017; Ronquillo and Hernandez 2017; Salim et al. 2018; Bacanlı and Başaran 2019; Ben et al. 2019).
Thus, in recent years, the use of AGPs has been reduced and gradually replaced by effective dietary supplements, such as probiotics and/or prebiotics, enzymes, and novel antimicrobial peptides. In addition, the application of antibiotics in livestock requires control and prudence (Ma et al. 2020; Magnusson 2020; Patel et al. 2020).
Antimicrobial peptide growth promoters
Antimicrobial peptides (AMPs) display a broad spectrum of activity against bacteria, fungi, viruses, and cancer, and these characteristics have already been widely discussed (Cardoso et al. 2020; Hwang et al. 2011; Rodrigues et al. 2019; Saido-Sakanaka et al. 2004). Normally, the activity of AMPs can be related to bacterial membrane interaction. This interaction can occur associated with ion channel/pore formation and/or detergent-like effect, indicating the molecular basis of their attraction to membranes (Brogden 2005; Nguyen et al. 2011). Furthermore, AMPs display different modes of action, like membrane disruption, increased membrane permeability, and/or disturbances in key cellular processes by interacting with intracellular targets (Yeaman and Yount 2003; Nguyen et al. 2011; Wimley and Hristova 2011; Sani and Separovic 2016). Besides, the use of AMPs as a growth promoter has demonstrated beneficial effects on nutrient digestibility, the intestinal microbiota, intestinal morphology, and immune function activities (Fig. 1) (Liu et al. 2008; Xiao et al. 2015; Gadde et al. 2017).
In this regard, studies using lactoferricin-lactoferrin (LF-chimera) were used to supplement piglet feed. The results demonstrated an increase in body weight and in the average daily gain (ADG) of 13.3 and 29.3%, respectively, compared with pigs fed control diets (Tang et al. 2012). Other studies tested the growth and digestive capacity after administering AMPs in poultry and pigs (Wang et al. 2016). The use of synthetic AMPs, such as AMP-A3 and AMP-P5 (both derived from the amino acid substitution of Helicobacter pylori-HP and cecropin-magainin2 fusion, respectively), increased the efficiency of gain of weanling pigs and broilers, with additional benefits concerning nutrient uptake and intestinal morphology. The maximal AMP concentrations tested were 90 and 60 mg kg−1 for AMP-A3 and AMP-P5, respectively. The results showed the effect for both body weight increases and intestinal injury reduction (Choi et al. 2013; Yoon et al. 2012, 2013, 2014). Another study evaluated the response of pig antibacterial peptides (PABP) in growth performance and small intestine mucosal immune responses in broilers. The authors reported that this PABP added to drinking water (20 and 30 mg/L) or supplemented in feed (150 and 200 mg/kg) can enhance growth performance, raise the intestinal ability to absorb nutrients, and improve the mucosal immunity of the intestine (Bao et al. 2009).
A different group used the AMP Epinephelus lanceolatus piscidin (EP), heterologously expressed and used as a dietary supplement for Gallus gallus domesticus. Treatment groups included control, and EP supplemented the diet at different doses (0.75, 1.5, 3.0, 6.0, and 12%). The results indicated that EP supplementation increased G. domesticus weight gain, feed efficiency, IL-10, and IFN-γ production, when compared to control (Tai et al. 2020). The pediocin A from Pediococcus pentosaceus FBB61 was tested by Grilli et al. (2009) as feed supplementation, and also tested against the Clostridium perfringens proliferation in broilers. The authors used a control group and another group where 80 AU.g−1 of pediocin A was added to the feed. The broilers were fed for 21 days, and they were challenged with culture of C. perfringens type A, which was administered by mouth on days 14, 15, and 16, twice daily (106 cfu/broiler). According to the authors, supplementation with pediocin A increased broiler growth performance during the challenge with C. perfringens (Grilli et al. 2009).
AMPs can also be used as AGPs in aquaculture, as described by Gyan et al. (2020). The application of AMPs can enhance the innate immune system, and boosts growth performance and disease resistance in Pacific white leg shrimp. In this study, different concentrations of AMP were tested in feed supplementation (0% until 1%). The results demonstrated the optimum concentration of AMP is 0.4% (400 g/kg). Researchers also observed that excess AMP in supplementation negatively affected the growth performance and immune system of the shrimp. Other studies which demonstrate efficient results using AMPs as growth promoters in livestock are shown in Table 2.
Concluding remarks and prospects
In the last 70 years, AGPs have been synonymous with productivity in livestock farming. However, the extensive use of these growth promoters has contributed to the development of bacterial strains with antimicrobial resistance. Antimicrobial resistance represents a worldwide problem and is treated with concern by the WHO. Hence, the European Union and the USA have limited the use of antibiotics in animal production. In an attempt to maintain livestock production, studies using AMPs as growth promoters have taken place, showing effective results in animal weight gains, and in some cases improving host immunity.
AMPs can therefore be an excellent way to substitute antibiotics due to characteristics such as a lower risk of inducing antimicrobial resistance, good inhibitory effects, and ease of degradation. Further studies using AMPs will allow a better understanding of the effects on the gastrointestinal ecosystem, and this will enable the best use of antimicrobial peptides for economic efficiency and sustainable livestock production.
References
Ageitos JM, Sánchez-Pérez A, Calo-Mata P, Villa TG (2017) Antimicrobial peptides (AMPs): ancient compounds that represent novel weapons in the fight against bacteria. Biochem Pharmacol 133:117–138. https://doi.org/10.1016/j.bcp.2016.09.018
Aguirre ATA, Acda SP, Angeles AA, Oliveros MCR, Merca FE, Cruz FA (2015) Effect of bovine lactoferrin on growth performance and intestinal histologic features of broiler. Philipp J Vet Anim Sci 41(1)
Aslam B, Wang W, Arshad MI, Khurshid M, Muzammil S, Rasool MH, Nisar MA, Alvi RF, Aslam MA, Qamar MU, Salamat MKF, Baloch Z (2018) Antibiotic resistance: a rundown of a global crisis. Infect Drug Resist 11:1645. https://doi.org/10.2147/IDR.S173867
Bacanlı M, Başaran N (2019) Importance of antibiotic residues in animal food. Food Chem Toxicol 125:462–466. https://doi.org/10.1016/j.fct.2019.01.033
Bao H, She R, Liu T, Zhang Y, Peng KS, Luo D, Yue Z, Ding Y, Hu Y, Liu W, Zhai L (2009) Effects of pig antibacterial peptides on growth performance and intestine mucosal immune of broiler chickens. Poult Sci J 88(2):291–297. https://doi.org/10.3382/ps.2008-00330
Ben Y, Fu C, Hu M, Liu L, Wong MH, Zheng C (2019) Human health risk assessment of antibiotic resistance associated with antibiotic residues in the environment: a review. Environ Res 169:483–493. https://doi.org/10.1016/j.envres.2018.11.040
Brennan J, Skinner J, Barnum DA, Wilson J (2003) The efficacy of bacitracin methylene disalicylate when fed in combination with narasin in the management of necrotic enteritis in broiler chickens. Poult Sci J 82(3):360–363. https://doi.org/10.1093/ps/82.3.360
Brogden KA (2005) Antimicrobial peptides: pore formers or metabolic inhibitors in bacteria? Nat Rev Microbiol 3:238–250. https://doi.org/10.1038/nrmicro1098
Brown K, Uwiera RR, Kalmokoff ML, Brooks SP, Inglis GD (2017) Antimicrobial growth promoter use in livestock: a requirement to understand their modes of action to develop effective alternatives. Int J Antimicrob Agents 49(1):12–24. https://doi.org/10.1016/j.ijantimicag.2016.08.006
Cao LT, Wu JQ, Xie F, Hu SH, Mo Y (2007) Efficacy of nisin in treatment of clinical mastitis in lactating dairy cows. J Dairy Sci 90(8):3980–3985. https://doi.org/10.3168/jds.2007-0153
Cardoso MH, Orozco RQ, Rezende SB, Rodrigues G, Oshiro KG, Cândido ES, Franco OL (2020) Computer-aided design of antimicrobial peptides: are we generating effective drug candidates? Front Microbiol 10:3097. https://doi.org/10.3389/fmicb.2019.03097
Ceotto-Vigoder H, Marques SLS, Santos INS, Alves MDB, Barrias ES, Potter A, Alviano DS, Bastos MCF (2016) Nisin and lysostaphin activity against preformed biofilm of Staphylococcus aureus involved in bovine mastitis. J Appl Microbiol 121(1):101–114. https://doi.org/10.1111/jam.13136
Cheng G, Hao H, Xie S, Wang X, Dai M, Huang L, Yuan Z (2014) Antibiotic alternatives: the substitution of antibiotics in animal husbandry. Front Microbiol 5:69–83. https://doi.org/10.3389/fmicb.2014.00217
Choi SC, Ingale SL, Kim JS, Park YK, Kwon IK, Chae BJ (2013) Effects of dietary supplementation with an antimicrobial peptide-P5 on growth performance, nutrient retention, excreta and intestinal microflora and intestinal morphology of broilers. Anim Feed Sci Technol 185(1–2):78–84. https://doi.org/10.1016/j.anifeedsci.2013.07.005
Davies J, Davies D (2010) Origins and evolution of antibiotic resistance. Microbiol Mol Biol Rev 74(3):417–433. https://doi.org/10.1128/MMBR.00016-10
European Commission (1998) Agricultural situation and prospects in the central and eastern countries – Poland, directorate general for agriculture brussels
FAO (2018) World Livestock: Transforming the livestock sector through the sustainable development goals. Rome. 222 pp. Licence: CC BY-NC-SA 3.0 IGO
FDA U (2016) NARMS Integrated Report: 2014-The National Antimicrobial Resistance Monitoring System: Enteric Bacteria
Feighner SD, Dashkevicz MP (1987) Subtherapeutic levels of antibiotics in poultry feeds and their effects on weight gain, feed efficiency, and bacterial cholyltaurine hydrolase activity. Appl Environ Microbiol 53(2):331–336
Founou LL, Founou RC, Essack SY (2016) Antibiotic resistance in the food chain: a developing country-perspective. Front Microbiol 7:1881. https://doi.org/10.3389/fmicb.2016.01881
Gadde U, Kim WH, Oh ST, Lillehoj HS (2017) Alternatives to antibiotics for maximizing growth performance and feed efficiency in poultry: a review. Anim Health Res Rev 18(1):26–45. https://doi.org/10.1017/S1466252316000207
Gyan WR, Yang Q, Tan B, Jan SS, Jiang L, Chi S, Dong X, Liu H, Shuang Z (2020) Effects of antimicrobial peptides on growth, feed utilization, serum biochemical indices and disease resistance of juvenile shrimp, Litopenaeus vannamei. Aquac Res 51(3):1222–1231. https://doi.org/10.1111/are.14473
Gomes B, Augusto MT, Felício MR, Hollmann A, Franco OL, Gonçalves S, Santos NC (2018) Designing improved active peptides for therapeutic approaches against infectious diseases. Biotechnol Adv 36(2):415–429. https://doi.org/10.1016/j.biotechadv.2018.01.004
George BA, Quarle CL, Fagerberg DJ (1982) Virginiamycin effects on controlling necrotic enteritis infections in chickens. Poult Sci J 61:447–450. https://doi.org/10.3382/ps.0610447
Grilli E, Messina MR, Catelli E, Morlacchini M, Piva A (2009) Pediocin A improves growth performance of broilers challenged with Clostridium perfringens. Poult Sci J 88(10):2152–2158. https://doi.org/10.3382/ps.2009-00160
Hancock RE, Sahl HG (2006) Antimicrobial and host-defense peptides as new anti-infective therapeutic strategies. Nat Biotechnol 24(12):1551–1557. https://doi.org/10.1038/nbt1267
Hancock RE (2001) Cationic peptides: effectors in innate immunity and novel antimicrobials. The Lancet Infect Dis 1(3):156–164. https://doi.org/10.1016/S1473-3099(01)00092-5
Hancock R, Chapple D (1999) MINIREVIEW Peptide antibiotics. Antimicrob Agents Chemother 1317–1323. https://doi.org/10.1128/AAC.43.6.1317
Hugues P, Heritage J (2004) Antibiotic growth-promoters in food animals. FAO Animal Production and Health Paper 129–152
Huyghebaert G, Ducatelle R, Van Immerseel F (2011) An update on alternatives to antimicrobial growth promoters for broilers. Vet J 187(2):182–188. https://doi.org/10.1016/j.tvjl.2010.03.003
Hwang B, Hwang JS, Lee J, Lee DG (2011) The antimicrobial peptide, psacotheasin induces reactive oxygen species and triggers apoptosis in Candida albicans. Biochem Biophys Res Commun 405:267–271. https://doi.org/10.1016/j.bbrc.2011.01.026
Innes GK, Randad R, Korinek A, Davis MF, Price LB, So AD, Heaney CD (2020) External societal costs of antimicrobial resistance in humans attributable to antimicrobial use in livestock. Annu Rev Public Health 41. https://doi.org/10.1146/annurev-publhealth-040218-043954. Accessed in 10 January 2021
Iwu CD, Korsten L, Okoh AI (2020) The incidence of antibiotic resistance within and beyond the agricultural ecosystem: a concern for public health. Microbiol Open 9(9):e1035. https://doi.org/10.1002/mbo3.1035
Jensen BB (1998) The impact of feed additives on the microbial ecology of the gut in young pigs. J Anim Feed Sci 7:45–64. https://doi.org/10.22358/jafs/69955/1998
Jenssen H, Hamill P, Hancock RE (2006) Peptide antimicrobial agents. Clin Microbiol Rev 19(3):491–511. https://doi.org/10.1128/CMR.00056-05
Jones FT, Ricke SC (2003) Observations on the history of the development of antimicrobials and their use in poultry feeds. Poult Sci J 82(4):613–617. https://doi.org/10.1093/ps/82.4.613
Knarreborg A, Lauridsen C, Engberg RM, Jensen SK (2004) Dietary antibiotic growth promoters enhance the bioavailability of alpha-tocopheryl acetate in broilers by altering lipid absorption. J Nutr 134:1487–1492. https://doi.org/10.1093/jn/134.6.1487
Kogut MH, Genovese KJ, He H, Swaggerty CL, Jiang YW (2012) BT cationic peptides: small peptides that modulate innate immune responses of chicken heterophils and monocytes. Vet Immunol Immunopathol 145(1–2):151–158. https://doi.org/10.1016/j.vetimm.2011.10.023
Kogut MH, Genovese KJ, He H, Swaggerty CL, Jiang Y (2013) Modulation of chicken intestinal immune gene expression by small cationic peptides as feed additives during the first week posthatch. Clin Vaccine Immunol 20(9):1440–1448. https://doi.org/10.1128/CVI.00322-13
Kraemer SA, Ramachandran A, Perron GG (2019) Antibiotic pollution in the environment: from microbial ecology to public policy. Microorganisms 7(6):180. https://doi.org/10.3390/microorganisms7060180
Li Z, Hu Y, Yang Y, Lu Z, Wang Y (2018) Antimicrobial resistance in livestock: antimicrobial peptides provide a new solution for a growing challenge. Anim Front 8(2):21–29. https://doi.org/10.1093/af/vfy005
Liu T, She R, Wang K, Bao H, Zhang Y, Luo D, Hu Y, Ding Y, Wang D, Peng K (2008) Effects of rabbit sacculus rotundus antimicrobial peptides on the intestinal mucosal immunity in chickens. Poult Sci J 87(2):250–254. https://doi.org/10.3382/ps.2007-00353
Ma JL, Zhao LH, Sun DD, Zhang J, Guo YP, Zhang ZQ, Ma QG, Ji C, Zhao LH (2020) Effects of dietary supplementation of recombinant plectasin on growth performance, intestinal health and innate immunity response in broilers. Probiotics Antimicro Proteins 1–10. https://doi.org/10.1007/s12602-019-9515-2
Magnusson U (2020) Prudent and effective antimicrobial use in a diverse livestock and consumer’s world. J Anim Sci 98:S4–S8. https://doi.org/10.1093/jas/skaa148
Marquardt RR, Li S (2018) Antimicrobial resistance in livestock: advances and alternatives to antibiotics. v 8(2):30–37. https://doi.org/10.1093/af/vfy001
Medina MJ, Legido-Quigley H, Hsu LY (2020) Antimicrobial resistance in one health. In: Global health security. Springer, pp 209–229. https://doi.org/10.1007/978-3-030-23491-1_10
Messaoudi S, Madi A, Prévost H, Feuilloley M, Manai M, Dousset X, Connil N (2012) In vitro evaluation of the probiotic potential of Lactobacillus salivarius SMXD51. Anaerobe 18(6):584–589. https://doi.org/10.1016/j.anaerobe.2012.10.004
Nadeem SF, Gohar UF, Tahir SF, Mukhtar H, Pornpukdeewattana S, Nukthamna P, Moula Ali AM, Bavisetty SCB, Massa S (2020) Antimicrobial resistance: more than 70 years of war between humans and bacteria. Crit Rev Microbiol 46(5):578–599. https://doi.org/10.1080/1040841X.2020.1813687
National Office of Animal Health (NOAH) (2001) Antibiotics for animals. http://www.noah.co.uk/issues/antibiotics.htm. Accessed 18 Jan 2021
Nguyen LT, Haney EF, Vogel HJ (2011) The expanding scope of antimicrobial peptide structures and their modes of action. Trend Biotechnol 29(9):464–472. https://doi.org/10.1016/j.tibtech.2011.05.001
Oshiro KG, Rodrigues G, Monges BED, Cardoso MH, Franco OL (2019) Bioactive peptides against fungal biofilms. Front Microbiol. https://doi.org/10.3389/fmicb.2019.02169
Patel SJ, Wellington M, Shah RM, Ferreira MJ (2020) Antibiotic stewardship in food-producing animals: challenges, progress, and opportunities. Clin Ther. https://doi.org/10.1016/j.clinthera.2020.07.004
Ronquillo MG, Hernandez JCA (2017) Antibiotic and synthetic growth promoters in animal diets: review of impact and analytical methods. Food Control 72:255–267. https://doi.org/10.1016/j.foodcont.2016.03.001
Rodrigues G, Silva GGO, Buccini DF, Duque HM, Dias SC, Franco OL (2019) Bacterial proteinaceous compounds with multiple activities toward cancers and microbial infection. Front Microbiol 10. https://doi.org/10.3389/fmicb.2019.01690
Saido-Sakanaka H, Ishibashi J, Momotani E, Amano F, Yamakawa M (2004) In vitro and in vivo activity of antimicrobial peptides synthesized based on the insect defensin. Peptides 25:19–27. https://doi.org/10.1016/j.peptides.2003.12.009
Salim HM, Huque KS, Kamaruddin KM, Haque Beg A (2018) Global restriction of using antibiotic growth promoters and alternative strategies in poultry production. Sci Prog 101(1):52–75. https://doi.org/10.3184/003685018X15173975498947
Sani MA, Separovic F (2016) How membrane-active peptides get into lipid membranes. Acc Chem Res 49(6):1130–1138. https://doi.org/10.1021/acs.accounts.6b00074
Shai Y (2002) Mode of action of membrane active antimicrobial peptides. Pep Sci 66:236–248. https://doi.org/10.1002/bip.10260
Snyder DL, Wostmann BS (1987) Growth rate of male germfree Wistar rats fed ad libitum or restricted natural ingredient diet. Lab Anim Sci 37:320–325
Spohn R, Daruka L, Lázár V, Martins A, Vidovics F, Grézal G, Méhi O, Kintses B, Számel M, Jangir PK, Csörgő B, Györkei Á, Bódi Z, Faragó A, Bodai L, Földesi I, Kata D, Maróti G, Pap B, Wirth R, Papp B, Pál C (2019) Integrated evolutionary analysis reveals antimicrobial peptides with limited resistance. Nat Commun 10(1):1–13. https://doi.org/10.1038/s41467-019-12364-6
Tai HM, Huang HN, Tsai TY, You MF, Wu HY, Rajanbabu V, Chang HY, Pan CY, Chen JY (2020) Dietary supplementation of recombinant antimicrobial peptide Epinephelus lanceolatus piscidin improves growth performance and immune response in Gallus gallus domesticus. PLoS One Mar 11:15(3):e0230021. https://doi.org/10.1371/journal.pone.0230021
Tang X, Fatufe AA, Yin Y, Tang Z, Wang S, Liu Z, Li TJ (2012) Dietary supplementation with recombinant lactoferrampin-lactoferricin improves growth performance and affects serum parameters in piglets. J Anim Vet Adv 11:2548–2555
ten Doeschate RAHM, Raine H (2006) History and current use of feed additives in the European Union: legislative and practical aspects. Avian Gut Funct Health Dis 28:1
Tenover FC (2006) Mechanisms of antimicrobial resistance in bacteria. Am J Med 119(6):S3–S10
Tona GO (2018) Current and future improvements in livestock nutrition and feed resources. Anim Husbandry Nutr 147–169. https://doi.org/10.5772/intechopen.73088
U.S. Food and Drug Administration. 2015. Veterinary feed directive. Available at: https://www.fda.gov/AnimalVeterinary/ucm071807.htm. Accessed 15 Jan 2021.
Van Boeckel TP, Brower C, Gilbert M, Grenfell BT, Levin SA, Robinson TP, Teillant A, Laxminarayan R (2015) Global trends in antimicrobial use in food animals. Proc Natl Acad Sci USA 112(18):5649–5654. https://doi.org/10.1073/pnas.1503141112
Vidovic N, Vidovic S (2020) Antimicrobial resistance and food animals: Influence of livestock environment on the emergence and dissemination of antimicrobial resistance. Antibiotics 9(2):52. https://doi.org/10.3390/antibiotics9020052
Xiao H, Shao F, Wu M, Ren W, Xiong X, Tan B, Yin Y (2015) The application of antimicrobial peptides as growth and health promoters for swine. J Anim Sci Biotechnol 6(1):1–6. https://doi.org/10.1186/s40104-015-0018-z
Ward BP, Ottaway NL, Perez-Tilve D, Ma D, Gelfanov VM, Tschöp MH, DiMarchi RD (2013) Peptide lipidation stabilizes structure to enhance biological function. Mol Metabolism 2(4):468–479. https://doi.org/10.1016/j.molmet.2013.08.008
Wang G, Song Q, Huang S, Wang Y, Cai S, Yu H, Ding X, Zeng X, Zhang J (2020) Effect of antimicrobial peptide microcin J25 on growth performance, immune regulation, and intestinal microbiota in broiler chickens challenged with Escherichia coli and Salmonella sp. Animals (Basel) 10(2). https://doi.org/10.3390/ani10020345
Wang S, Zeng X, Yang Q, Qiao S (2016) Antimicrobial peptides as potential alternatives to antibiotics in food animal industry. Int J Mol Sci 17(5):603. https://doi.org/10.3390/ijms17050603
Wimley WC, Hristova K (2011) Antimicrobial peptides: successes, challenges and unanswered questions. J Membr Biol 239(1):27–34. https://doi.org/10.1007/s00232-011-9343-0
World Health Organization (2014) WHO’s first global report on antibiotic resistance Reveals serious, worldwide threat to public health. Available from: http://www.who.int/mediacentre/news/releases/2014/amr-report/en/. Accessed 10 Jan 2021
Zhao Y, Zhang M, Qiu S, Wang J, Peng J, Zhao P, Zhu R, Wang H, Li Y, Wang K, Yan W, Wang R (2016) Antimicrobial activity and stability of the D-amino acid substituted derivatives of antimicrobial peptide polybia-MPI. AMB Express 6:122. https://doi.org/10.1186/s13568-016-0295-8
Yeaman MR, Yount NY (2003) Mechanisms of antimicrobial peptide action and resistance. Pharmacol Rev 55(1):27–55. https://doi.org/10.1124/pr.55.1.2
Yoon JH, Ingale SL, Kim JS, Kim KH, Lee SH, Park YK, Lee SC, Kwona IK, Chae BJ (2014) Effects of dietary supplementation of synthetic antimicrobial peptide-A3 and P5 on growth performance, apparent total tract digestibility of nutrients, fecal and intestinal microflora and intestinal morphology in weanling pigs. Livest Sci 159:53–60. https://doi.org/10.1016/j.livsci.2013.10.025
Yoon JH, Ingale SL, Kim JS, Kim KH, Lee SH, Park YK, Kwona IK, Chae BJ (2012) Effects of dietary supplementation of antimicrobial peptide-A3 on growth performance, nutrient digestibility, intestinal and fecal microflora and intestinal morphology in weanling pigs. Anim Feed Sci Technol 177(1–2):98–107. https://doi.org/10.1016/j.anifeedsci.2012.06.009
Yoon JH, Ingale SL, Kim JS, Kim KH, Lohakare J, Park YK, Park JC, Kwon IK, Chae BJ (2013) Effects of dietary supplementation with antimicrobial peptide-P5 on growth performance, apparent total tract digestibility, faecal and intestinal microflora and intestinal morphology of weanling pigs. J Sci Food Agric 93(3):587–592. https://doi.org/10.1002/jsfa.5840
Funding
This work was supported by grants from Fundação de Apoio à Pesquisa do Distrito Federal (FAPDF), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) (MC 88887.351521/2019–00), Conselho Nacional de Desenvolvimento e Tecnológico (CNPq), and Fundação de Apoio ao Desenvolvimento do Ensino, Ciência e Tecnologia do Estado de Mato Grosso do Sul (FUNDECT), Brazil.
Author information
Authors and Affiliations
Contributions
GRR and MRM revised the literature and wrote, and OLF revised the manuscript. All authors have read and approved the manuscript.
Corresponding author
Ethics declarations
Ethical statement
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Rodrigues, G., Maximiano, M.R. & Franco, O.L. Antimicrobial peptides used as growth promoters in livestock production. Appl Microbiol Biotechnol 105, 7115–7121 (2021). https://doi.org/10.1007/s00253-021-11540-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-021-11540-3