Abstract
Both aerobic methane-oxidizing bacteria (MOB) and nitrite-dependent anaerobic methane oxidation (n-damo) organisms can be important methane sinks in a wetland. However, the influences of the vegetation type on aerobic MOB and n-damo communities in wetland, especially in constructed wetland, remain poorly understood. The present study investigated the influences of the vegetation type on both aerobic MOB and n-damo organisms in a constructed urban landscape wetland. Sediments were collected from eight sites vegetated with different plant species. The abundance (1.19–3.27 × 107 pmoA gene copies per gram dry sediment), richness (Chao1 estimator = 16.3–81.5), diversity (Shannon index = 2.10–3.15), and structure of the sediment aerobic MOB community were found to vary considerably with sampling site. In contrast, n-damo community abundance (8.74 × 105–4.80 × 106 NC10 16S rRNA gene copies per gram dry sediment) changed slightly with the sampling site. The richness (Chao1 estimator = 1–11), diversity (Shannon index = 0–0.78), and structure of the NC10 16S rRNA gene-based n-damo community illustrated slight site-related changes, while the spatial changes of the pmoA gene-based n-damo community richness (Chao1 estimator = 1–8), diversity (Shannon index = 0–0.99), and structure were considerable. The vegetation type could have a profound impact on the wetland aerobic MOB community and had a stronger influence on the pmoA-based n-damo community than on the NC10 16S-based one in urban wetland. Moreover, the aerobic MOB community had greater abundance and higher richness and diversity than the n-damo community. Methylocystis (type II MOB) predominated in urban wetland, while no known type I MOB species was detected. In addition, the ratio of total organic carbon to total nitrogen (C/N) might be a determinant of sediment n-damo community diversity and aerobic MOB richness.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Wetlands are the most important natural source of greenhouse gas methane (CH4), with annual average emission of 145 Tg (Chowdhury and Dick 2013). Aerobic methane-oxidizing bacteria (MOB) are considered as an efficient biofilter to mitigate the CH4 produced in deeper anoxic layers before the release into the atmosphere (Chowdhury and Dick 2013; Conrad 2007; Yun et al. 2015). So far, the known aerobic methanotrophs mainly belong to the phylum Proteobacteria and can be further classified into two groups (type I (γ-Proteobacteria) and type II (α-Proteobacteria)) (Dai et al. 2015; Yang et al. 2016; Yun et al. 2015). It has been well-documented that wetland microbial communities are plant-specific in both natural wetland and constructed wetland (Lee and Kang 2016; Long et al. 2016; Rietl et al. 2016), yet little is known about the links of the aerobic MOB community with the wetland plant type. The pmoA genes that encode the subunits of particulate methane monooxygenases (pMMOs) have been a well-established biomarker to study the distribution of the aerobic methanotrophic communities. Several previous studies suggested that the vegetation type could greatly affect the abundance of the aerobic MOB community in natural wetland (Chen et al. 2017; Yun et al. 2012, 2013, 2014, 2015), and two other studies documented the profound influence of the vegetation type on the structure of the aerobic MOB community in natural wetland (Danilova et al. 2016; Narihiro et al. 2011). The influence of the vegetation type on the wetland aerobic MOB community diversity remains in debate. Very little or considerable influence of the vegetation type on the aerobic MOB diversity was found in different natural wetlands (Yun et al. 2013, 2015). However, direct information on the influence of the vegetation type on the aerobic MOB community in constructed wetland is still lacking.
Nitrite-dependent anaerobic methane oxidation (n-damo), putatively performed by Candidatus Methylomirabilis oxyfera (within bacterial phylum NC10) (Ettwig et al. 2010), can be an important microbial methane sink in a wetland (Hu et al. 2014). Based on NC10 16S rRNA genes and n-damo pmoA genes, presence of n-damo organisms has been found in a number of natural and constructed wetlands (Chen et al. 2015; Shen et al. 2015; Wang et al. 2016; Yang et al. 2017; Zhu et al. 2015). However, there is still a paucity of knowledge on the influence of the vegetation type on wetland n-damo organisms. Chen et al. (2015) suggested considerable influences of the plant type on the abundance, diversity, and structure of the sediment n-damo bacterial community in coastal intertidal wetland. Moreover, the vegetation type could also remarkably shape the n-damo community in vertical flow constructed wetland treating heavily polluted river water (Yang et al. 2017). To date, the coexistence of aerobic MOB and n-damo organisms in a wetland system has not been addressed. The difference of abundance and diversity between these two methane-oxidizing microbial groups in a wetland system remains unclear. Therefore, the main objective of this current study was to investigate the influences of the vegetation type on aerobic MOB and n-damo organisms in a constructed urban landscape wetland. Their differences in abundance and diversity were also studied. In addition, the links of aerobic and anaerobic methanotrophic communities with wetland environmental factors were further explored.
Materials and methods
Site description and sampling
The studied urban landscape wetland (with the total surface area of about 0.179 km2), located in Guangzhou City (southwest China), was constructed in 2012. The local region has a humid subtropical monsoon climate, with the annual mean air temperature of 21–23 °C and precipitation of 1500–2000 mm (Yi et al. 2016). In this study, triplicate sediments (0–10 cm) at eight sites (A–H) were collected in June in 2016 using a Kajak tube core sampler (KC Denmark A/S, Holmbladsvej, Silkeborg, Denmark) (Supplementary Fig. S1). Sites A–H were vegetated with Alisma plantago-aquatica Linn., Typha angustifolia, Cyperus papyrus, Nelumbo nucifera Gaertn., Canna indica L., Thalia dealbata Fraser, Acorus calamus L., and Hydrocleys nymphoides, respectively. Wetland sediment physicochemical properties (including pH, total nitrogen (TN), total phosphorus (TP), total organic carbon (TOC), and the ratio of TOC to TN (C/N) were listed in Supplementary Table S1.
Quantitative PCR assay
Genomic DNA from each wetland sediment sample was extracted using a Powersoil DNA extraction kit (MO BIO Laboratories, Carlsbad, CA, USA), and the DNA quality and quantity were assessed using a NanoDrop 2000 spectrophotometer (Thermo Fisher Scientific, Waltham, MA, USA). Aerobic MOB was quantified using the primers A189F/Mb661R targeting the pmoA genes (Kolb et al. 2003). The abundance of wetland sediment n-damo organisms was assessed with the NC10-specific primer sets qP1F/qP1R (Ettwig et al. 2009; Wang et al. 2012). The amplification program was as follows: 95 °C for 10 min, 40 cycles of 95 °C for 45 s, 55 °C for 30 s, followed by 72 °C for 30 s, and finally 72 °C for 5 min. Standard curves were generated with a series of tenfold dilutions of plasmid DNA harboring the target genes. Negative controls containing no template DNA were performed following the same amplification protocol. The efficiency and coefficient (R 2) of qPCR assays for aerobic MOB and n-damo organisms were 97% and 0.992, and 95% and 0.995, respectively.
Clone library analysis
The pmoA genes of wetland sediment aerobic MOB were amplified using the primer pair A189F/Mb661R according to the literature (Yang et al. 2014). The pmoA genes of sediment n-damo bacteria were amplified with a nested approach (first-step primers A189_b/cmo682, followed by primers cmo182/cmo568) following the same condition reported in our previous study (Long et al. 2017a, b). The NC10 16S rRNA genes were also amplified using a nested approach (first-step primers 202F/1545R, followed by primers p1F/p2R), as previously described (Wang et al. 2012). The resulting PCR products were purified with the TIANquick Mini Purification Kit (Tiangen Biotech (Beijing) Co., LTD, Beijing, China). The purified amplicons from triplicate wetland sediment samples were pooled in equal amounts and then ligated into the pEASY®-T1 Cloning Vector and then transformed into Trans1-T1 Phage Resistant Chemically Competent Cell (TransGen Biotech Co., Ltd., Beijing, China). Positive clones containing an insert of the correct size were randomly selected and then sequenced at SinoGenoMax Co., Ltd. (Beijing, China). The obtained sequences were deposited in the GenBank database (KY995713–KY995870 for n-damo pmoA genes, KY995871–KY996307 for aerobic MOB pmoA genes, and KY313904–KY314112 for NC10 16S rRNA genes). These sequences were divided into operational taxonomic units (OTUs) with ≥ 97% similarity. Community α-diversity estimates (Chao1 richness estimator and Shannon diversity) were obtained with the MOTHUR program (Schloss et al. 2009). The number of NC10 16S rRNA gene sequences from each wetland sample was rarefied at 20 (the lowest NC10 16S rRNA gene sequence number within a single sample). Moreover, phylogenetic analyses of the representative pmoA or NC10 16S rRNA gene sequences were conducted with MEGA 6.0 (Tamura et al. 2013) using the neighbor-joining method. The confidence levels were tested using bootstrap analysis with 1000 replicates. In addition, weighted Unifrac distance of wetland sediment samples was generated with R library GUniFrac, and environment clustering analysis was further conducted using R (version i386, 3.3.2) (https://www.r-project.org/).
Statistical analysis
The difference in the abundance of aerobic MOB or n-damo organisms among wetland sediment samples was checked at the significance level of 5% with one-way analysis of variance (ANOVA). The links of wetland sediment physicochemical parameters with the abundance, richness, and diversity of aerobic MOB or n-damo organisms were discriminated based on Spearman’s rank correlation analysis (using the software SPSS 20.0) (IBM, Armonk, NY, USA). Moreover, the correlations between methanotrophic microbial community composition and wetland environmental parameters were established with redundancy analysis (RDA) using Monte Carlo tests (with the software CANOCO 4.5 (Microcomputer Power, Ithaca, NY, USA)). The relative abundance of sequences in each microbial OTU and sediment physicochemical parameters were assigned as species input and environmental input, respectively.
Results
Abundance of aerobic methane-oxidizing bacteria and nitrite-dependent anaerobic methane oxidation organisms
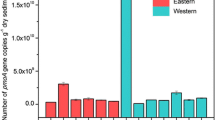
Since there was no suitable qPCR primer set for the quantification of n-damo pmoA genes so far, the abundance of wetland n-damo organisms was estimated by targeting NC10 16S rRNA genes (Zhu et al. 2015). In the studied urban landscape wetland, the number of aerobic MOB pmoA genes was 1.19–3.27 × 107 copies per gram dry sediment, while sediment NC10 16S rRNA genes ranged between 8.74 × 105 and 4.80 × 106 copies per gram dry sediment (Fig. 1). The calculated ratio of NC10 16S rRNA genes to aerobic MOB pmoA genes was 5.7–25.8%. Significant difference in the aerobic MOB pmoA gene copy number was observed among different sampling sites (P < 0.05). Sediments at sites C and F had the largest aerobic MOB community size. Moreover, sediment at site D displayed less aerobic MOB pmoA gene copies than sediments at sites A, E, and H (P < 0.05) but more than sediments at sites B and G (P < 0.05). In addition, sediments at sites C and E displayed a relatively higher NC10 16S rRNA gene copy; however, no significant difference in sediment n-damo community abundance was found among other sites (P > 0.05).
Abundance of aerobic MOB and n-damo communities in urban wetland sediments. Sites A–H were vegetated with Alisma plantago-aquatica Linn., Typha angustifolia, Cyperus papyrus, Nelumbo nucifera Gaertn., Canna indica L., Thalia dealbata Fraser, Acorus calamus L., and Hydrocleys nymphoides, respectively. Values are the average of triplicate samples. Vertical bars indicate standard deviations. Different letters above the columns indicate a significant difference (P < 0.05)
Diversity and richness of aerobic methane-oxidizing bacteria and nitrite-dependent anaerobic methane oxidation organisms
In this study, each wetland sediment aerobic MOB pmoA gene library included 53–55 sequences and 13–29 OTUs (Table 1). Based on aerobic MOB pmoA genes, the Chao1 richness estimators and Shannon diversity indices of wetland sediment aerobic MOB communities were 16.3–81.5 and 2.10–3.15, respectively. Each wetland sediment sample included 20 NC10 16S rRNA gene sequences and was composed of 1–5 NC10 16S OTUs. Moreover, wetland sediments contained 19 or 20 n-damo pmoA gene sequences that could be furthered grouped into 1–5 OTUs. Based on NC10 16S rRNA and n-damo pmoA genes, the Chao1 richness estimators and Shannon diversity indices of wetland sediment n-damo organisms were 1–11 and 1–8, and 0–0.78 and 0–0.99, respectively. There results illustrated spatial variations of the diversity and richness of both aerobic MOB and n-damo communities in the studied urban landscape wetland. In addition, aerobic MOB community had a much higher diversity and richness than n-damo community.
Clustering analysis of aerobic methane-oxidizing bacteria and nitrite-dependent anaerobic methane oxidation community compositions
Based on aerobic MOB pmoA genes, wetland sediment samples displayed a considerable difference in aerobic MOB community compositions (Fig. 2a). Sediments at sites E and G formed a clade, while other sediments formed another clade. Moreover, based on NC10 16S rRNA genes, wetland sediment samples at sites C, D, E, F and H displayed the identical n-damo community compositions, and their n-damo community compositions were only slightly different from those of other wetland sediment samples (at sites A, B and G) (Fig. 2b). However, based on n-damo pmoA genes, wetland sediment samples illustrated a considerable difference in n-damo community compositions (Fig. 2c). Sediment at site C was distantly separated from other sediments. Sediments at sites D, E, G and H could be grouped together, while wetland sediments at sites A, B and F could form another group.
Clustering analysis of aerobic MOB communities based on pmoA genes (a) and n-damo communities based on NC10 16S rRNA genes (b) and n-damo pmoA genes (c). AM, BM, CM, DM, EM, FM, GM, and HM represent the aerobic MOB pmoA gene libraries constructed with sediments from sites A–H, respectively. AS, BS, CS, DS, ES, FS, GS, and HS represent the NC10 16S rRNA gene libraries constructed with sediments from sites A–H, respectively. AD, BD, CD, DD, ED, FD, GD, and HD represent the n-damo pmoA gene libraries constructed with sediments from sites A–H, respectively
Phylogeny of aerobic methane-oxidizing bacteria and nitrite-dependent anaerobic methane oxidation communities
The sequences from major aerobic MOB pmoA OTUs (including at least three sequences in all sediment MOB pmoA clone libraries) could be also divided into three clusters (Fig. 3a). The proportion of each MOB pmoA cluster displayed a considerable variation with sampling site (Fig. 4a). Cluster I included a total of 270 MOB pmoA sequences and was the largest aerobic MOB pmoA group. The sequences in this cluster could be related to those from several cultivated Methylocystis species (type II MOB). Cluster I-like MOB were dominant in each wetland sediment. Moreover, cluster II included only 12 MOB pmoA sequences that were detected in sediments from sites A, C, D, E and G. The sequences in cluster II could be grouped with those from rice soil, lake sediment, and disused tin mining pond (Ho et al. 2011; Liu et al. 2015; Sow et al. 2014). In addition, cluster III contained 33 MOB pmoA sequences. These MOB pmoA sequences were mainly distributed in sediments from sites D, E, G and H, and they could be related to the MOB pmoA sequences from various soil and sediment ecosystems.
Phylogenetic tree of representative aerobic MOB pmoA gene sequences (a), NC10 16S rRNA gene sequences (b) and n-damo pmoA gene sequences (c) and the reference sequences from Genbank. The obtained sequences beginning with “AM” –“HM”, “AS” –“HS” and “AD” –“HD” are referred to aerobic MOB pmoA gene sequences, NC10 16S rRNA gene sequences and n-damo pmoA gene sequences retrieved from sites A–H, respectively. The number in parentheses represents the total sequences from all wetland sediment samples in the same OTU. Numbers at the nodes indicate the levels of bootstrap support based on neighbor-joining analysis of 1000 resampled datasets. The values less than 50 are not listed. The bar respectively represents 5%, 2% and 1% sequence divergence in aerobic MOB pmoA, NC10 16S and n-damo pmoA trees
Compositions of aerobic MOB pmoA clusters (a), NC10 16S rRNA clusters (b), and n-damo pmoA clusters (c) in urban wetland sediments. AM, BM, CM, DM, EM, FM, GM, and HM represent the aerobic MOB pmoA gene libraries constructed with sediments from sites A–H, respectively. AS, BS, CS, DS, ES, FS, GS, and HS represent the NC10 16S rRNA gene libraries constructed with sediments from sites A–H, respectively. AD, BD, CD, DD, ED, FD, GD, and HD represent the n-damo pmoA gene libraries constructed with sediments from sites A–H, respectively
The sequences from all NC10 16S OTUs were assigned into three clusters (Fig. 3b). Cluster 1 comprised of a total of 158 16S rRNA gene sequences that were closely related to the 16S rRNA gene sequence from M. oxyfera (Ettwig et al. 2010). Sediments from sites A, C, D, E, F and H solely contained cluster 1-like organisms, and cluster 1-like organisms also predominated in sediments from sites G and B (Fig. 4b). These results further confirmed the slight difference in NC10 16S rRNA gene-based n-damo community compositions. Clusters 2 and 3 only included one sequence from sites G and B, respectively. The two wetland sediment sequences were closely related to those from soil or sediment ecosystems.
The sequences from major n-damo pmoA OTUs (including at least two sequences in all sediment n-damo pmoA clone libraries) could be also separated into three clusters (Fig. 3c). The proportion of each n-damo pmoA cluster varied considerably with the sampling site (Fig. 4c). Cluster a included a total of 58 n-damo pmoA sequences that could be closely related to the pmoA sequence from M. oxyfera (Ettwig et al. 2010). Cluster a-like sequences were mainly detected in sediments from sites D, F, and F. Cluster b comprised of 34 n-damo pmoA sequences that were mainly retrieved from sediments from sites A and B. The sequences in cluster b were related to those from diverse soil and sediment ecosystems. Moreover, cluster c was the largest n-damo pmoA group containing 62 sequences, and they were mainly distributed in sediments from sites C, E and H. The sequences in cluster c could be grouped together with those from leachate cover soil, waste water treatment plant, constructed wetland sediment, and reservoir sediment.
Relationship between environmental factors and aerobic methane-oxidizing bacteria and nitrite-dependent anaerobic methane oxidation communities
Spearman’s rank correlation analysis illustrated that sediment C/N was negatively correlated to n-damo pmoA Shannon diversity (P < 0.05) but positively to MOB pmoA Chao1 richness (P < 0.05) (Table 2). The relative abundance of cluster a-like n-damo pmoA sequences exhibited a negative correlation with the concentrations of sediment TOC and TP (P < 0.05 or P < 0.01). The proportion of cluster b-like n-damo pmoA sequences was positively correlated to pH (P < 0.05), while the proportion of cluster c-like n-damo pmoA sequences illustrated a positive correlation with the level of sediment TN (P < 0.01). Moreover, the proportion of cluster I-like MOB pmoA sequences was positively correlated to pH (P < 0.05). However, no significant correlation was found between the determined wetland sediment environmental parameters and the abundance of aerobic MOB and n-damo organisms (P > 0.05).
Wetland sediment environmental factors in the first two RDA dimensions respectively accounted for 67.79% and 13.11% of the total variance for the NC10 16S OTU composition (Fig. 5b). TP (P = 0.017, F = 2.7, 999 permutations) had a significant contribution to the NC10 16S-based n-damo organisms–environment relationship. Moreover, wetland environmental factors in the first two RDA dimensions totally accounted 39.8 and 35.14% for the variance of MOB pmoA and n-damo pmoA OTU compositions, respectively (Fig. 5a, c). However, no environmental factor was found to significantly contribute to the pmoA-based n-damo organisms (or aerobic MOB)–environment relationship.
RDA ordination plot for the first two principal dimensions of the links of wetland sediment physicochemical parameters with the OTU compositions of aerobic MOB pmoA gene sequences (a), NC10 16S rRNA gene sequences (b), and n-damo pmoA gene sequences (c). AM, BM, CM, DM, EM, FM, GM, and HM represent the aerobic MOB pmoA gene libraries constructed with sediments from sites A–H, respectively. AS, BS, CS, DS, ES, FS, GS, and HS represent the NC10 16S rRNA gene libraries constructed with sediments from sites A–H, respectively. AD, BD, CD, DD, ED, FD, GD, and HD represent the n-damo pmoA gene libraries constructed with sediments from sites A–H, respectively
Discussion
Abundance of wetland aerobic methane-oxidizing bacteria and nitrite-dependent anaerobic methane oxidation organisms
It has been well-documented that the vegetation type greatly affected the aerobic MOB community size in natural wetland (Chen et al. 2017; Yun et al. 2012, 2013, 2014, 2015), yet the relationship between vegetation type and aerobic MOB abundance in constructed wetland remains unknown. In the present study, the abundance of the aerobic MOB community in the constructed urban wetland illustrated a considerable difference at sites with different vegetation types. This suggested that the vegetation type might be a key determinant of the aerobic MOB abundance in urban wetland. The vegetation type can affect methanogenic archaeal community and methanogenesis pathways (Jiang et al. 2010; Tian et al. 2012). Hence, it could be assumed that the vegetation type had a considerable influence on methane production in the anoxic environments of the constructed urban wetland (Yang et al. 2017). In addition, the MOB community far outnumbered the n-damo community, suggesting that aerobic MOB might play a more important role than n-damo organisms in methane oxidization in constructed urban wetland.
The influence of the plant type on the sediment n-damo community abundance has been found in both natural wetland (Chen et al. 2015) and vertical flow constructed wetland (Yang et al. 2017). In this study, although sediments vegetated with C. papyrus and C. indica L. had a relatively higher n-damo community abundance, sediments vegetated with other plant species differed slightly in the n-damo community abundance. This suggested that the vegetation type might have a slight impact on the sediment n-damo community abundance in urban wetland.
Richness and diversity of wetland aerobic methane-oxidizing bacteria and nitrite-dependent anaerobic methane oxidation organisms
The influence of the vegetation type on the wetland aerobic MOB diversity remains unclear. The vegetation type illustrated a considerable influence on the aerobic MOB diversity in Xianghai wetland (a natural wetland in northeast China) (Yun et al. 2013), but a very slight (or no) influence in Sanjiang wetland (another natural wetland in northeast China) (Yun et al. 2015). In this study, in the constructed urban wetland, the diversity and richness of the aerobic MOB community differed considerably at sites with different plant species. Hence, the present study further provided the first evidence that the vegetation type could have a profound influence on the aerobic MOB diversity and richness in constructed urban wetland. Moreover, so far, the links between environmental factors and the aerobic MOB diversity and richness remain elusive. In this study, Spearman’s rank correlation analysis indicated that the aerobic MOB richness in urban wetland was positively correlated to sediment C/N. However, there was no clear link of the aerobic MOB diversity with the determined wetland environmental factors. In addition, Good’s coverage estimators of aerobic MOB pmoA libraries were relatively low (usually less than 80%) at the current sequencing depth. This implied that deeper sequencing might result in more OTUs. The application of high-throughput sequencing analysis to study wetland aerobic MOB communities might yield more solid data.
The plant type could considerably affect the richness and diversity of n-damo organisms (based on either NC10 16S rRNA genes or n-damo pmoA genes) in coastal intertidal wetland (Chen et al. 2015). The plant type could also regulate the richness and diversity of n-damo organisms (based on n-damo pmoA genes) in a vertical flow constructed wetland treating heavily polluted river water (Yang et al. 2017). In this study, the richness and diversity of the n-damo community in the constructed urban wetland were investigated based on the two genes. Good’s coverage estimators of NC10 16S rRNA libraries and n-damo pmoA libraries were usually ≥ 90%, indicating that NC10 16S rRNA and n-damo pmoA OTUs had been ideally captured at the current sequencing depth. At a given sampling site, the value of either the Chao1 richness estimator or the Shannon diversity index usually differed based on the two genes. This was consistent with the results found in natural wetland (Chen et al. 2015; Wang et al. 2016). Moreover, except at site B, the NC10 16S-based n-damo community richness and diversity illustrated a slight site-related change. In contrast, the site-related change of the pmoA-based n-damo community richness and diversity was more remarkable. These results suggested that the vegetation type might have a stronger influence on the pmoA-based n-damo community richness and diversity than on the NC10 16S-based ones in urban wetland. To date, little is known about the links of the n-damo community diversity with wetland environmental factors. A recent study suggested that the wetland n-damo community diversity could be regulated by pH (Zhu et al. 2015). In the present study, the result of Spearman’s rank correlation analysis further suggested that the pmoA-based n-damo community diversity in urban wetland could decrease with increasing sediment C/N. This was in agreement with the result found in the Dongjiang River (Long et al. 2017a) but was contrary to the result reported in the freshwater lakes on the Yunnan Plateau (Liu et al. 2015). Therefore, the influence of C/N on the sediment n-damo community diversity might depend on habitat type.
Community structure of wetland aerobic methane-oxidizing bacteria and nitrite-dependent anaerobic methane oxidation organisms
Two previous studies have revealed a profound influence of the vegetation type on the aerobic MOB community structure in natural wetlands (Danilova et al. 2016; Narihiro et al. 2011). In this study, the results of both clustering and phylogenetic analyses displayed a considerable site-related shift in the aerobic MOB community structure in the constructed urban wetland, suggesting a profound influence of the vegetation type on the aerobic MOB community structure. Moreover, to date, there has been no consensus on the dominant MOB species in wetland ecosystems. A dominance of Methylocystis (type II MOB) was found in Riganqiao peatlands on the Qinghai–Tibetan Plateau (Deng et al. 2013), while Methylobacter (type I MOB) predominated in littoral wetland of Lake Namco on the Tibetan Plateau (Yun et al. 2014) and in Xianghai wetland (Yun et al. 2013). A dominance of Methylobacter and Methylocystis organisms was found in Sanjiang wetland (Yun et al. 2015) and Luoshijiang wetland (Yang et al. 2014). In addition, Methylomonas (type I MOB) and Methylocystis predominated in the Sphagnum-covered and Sasa-invaded wetlands (Narihiro et al. 2011). In this study, no known type I MOB species was detected, whereas Methylocystis species (type II MOB) predominated in the constructed urban wetland. This suggested that Methylocystis species might play a crucial role in methane oxidization in constructed urban wetland. The environmental factors driving the distribution of Methylocystis species in wetland ecosystems remain unknown. In this study, although no clear link was found between wetland environmental factors and the overall aerobic MOB community structure, the pH was found to be a possibly key factor regulating the distribution of Methylocystis species in this constructed urban wetland. Since the wetland sediment pH ranged between 5.27 and 5.94 and a higher pH value favored the relative abundance of Methylocystis species, it might be assumed that a moderate acidic condition was more suitable for the growth of Methylocystis species.
The plant type could greatly affect the structure of the n-damo community (based on either NC10 16S rRNA genes or n-damo pmoA genes) in coastal intertidal wetland (Chen et al. 2015). Based on n-damo pmoA genes, Yang et al. (2017) reported a considerable influence of the vegetation type on the n-damo community structure in a vertical flow constructed wetland treating heavily polluted river water. In this study, the results of both clustering and phylogenetic analyses revealed that, in the urban landscape wetland, the n-damo community composition had a slight site-related change based on NC10 16S rRNA genes, but a considerable variation based on n-damo pmoA genes. These results indicated that the vegetation type might have a stronger influence on the pmoA-based n-damo community structure than on the NC10 16S-based one in the urban wetland. Information on the factors structuring the overall wetland n-damo community is still very limited. Chen et al. (2015) suggested that the wetland n-damo community structure could be shaped by ammonium and nitrite. In this study, TP was found to be a possible determinant of the NC10 16S-based n-damo community structure in urban wetland. Moreover, a previous study suggested that, in freshwater wetland, only a very small proportion of NC10 16S rRNA gene sequences were closely related to the 16S rRNA gene sequence of M. oxyfera, while most of the n-damo pmoA gene sequences could be closely related to the pmoA gene sequence of M. oxyfera (Wang et al. 2016), which was consistent with the result observed in coastal intertidal wetland (Chen et al. 2015). In contrast, in the here studied urban wetland, most of the retrieved NC10 16S rRNA gene sequences (158 out of 160) had close relationships with the 16S rRNA gene sequence of M. oxyfera, while only a small proportion of 58 out of 154 were closely related to the pmoA gene sequence of M. oxyfera. Although no determined environmental factor had a clear link with the overall community composition of pmoA-based n-damo organisms, TOC and TP were correlated with the proportion of cluster a-like n-damo organisms, and pH and TN had a correlation with the proportion of cluster b- and c-like n-damo organisms, respectively. Therefore, it might be the collective effect of several factors rather than a single one manipulating the overall community composition of pmoA-based n-damo organisms in urban wetland ecosystems.
In conclusion, in the urban landscape wetland, the richness, diversity, and structure of both the MOB community and the pmoA-based n-damo community varied considerably with the sampling site, while the NC10 16S-based n-damo community illustrated a slight site-related change. A considerable site-related abundance variation was detected in the aerobic MOB community but only a smaller one in the NC10 16S-based n-damo community. The vegetation type could have a profound impact on the wetland aerobic MOB community and the pmoA-based n-damo community, instead of on the NC10 16S-based n-damo community. Moreover, Methylocystis (type II MOB) predominated in the urban wetland, and it might be mainly responsible for methane oxidization.
References
Chen J, Zhou ZC, JD G (2015) Complex community of nitrite dependent anaerobic methane oxidation bacteria in coastal sediments of the Mai Po wetland by PCR amplification of both 16S rRNA and pmoA genes. Appl Microbiol Biotechnol 99:1463–1473
Chen XP, Ma H, Zheng Y, Liu JM, Liang X, He CQ (2017) Changes in methane emission and methanogenic and methanotrophic communities in restored wetland with introduction of Alnus trabeculosa. J Soils Sediments 17:181–189
Chowdhury TR, Dick RP (2013) Ecology of aerobic methanotrophs in controlling methane fluxes from wetlands. Appl Soil Ecol 65:8–22
Conrad R (2007) Microbial ecology of methanogens and methanotrophs. Adv Agron 96:1–63
Dai Y, Wu Z, Xie SG, Liu Y (2015) Methanotrophic community abundance and composition in plateau soils with different plant species and plantation ways. Appl Microbiol Biotechnol 99:9237–9244
Danilova OV, Belova SE, Gagarinova IV, Dedysh SN (2016) Microbial community composition and methanotroph diversity of a subarctic wetland in Russia. Microbiology 85:583–591
Deng YC, Cui XY, Luke C, Dumont MG (2013) Aerobic methanotroph diversity in Riganqiao peatlands on the Qinghai–Tibetan Plateau. Environ Microbiol Rep 5:566–574
Ettwig KF, van Alen T, van de Pas-Schoonen KT, Jetten MS, Strous M (2009) Enrichment and molecular detection of denitrifying methanotrophic bacteria of the NC10 phylum. Appl Environ Microbiol 75:3656–3662
Ettwig KF, Butler MK, Le Paslier D, Pelletier E, Mangenot S, Kuypers MMM, Schreiber F, Dutilh BE, Zedelius J, de Beer D, Gloerich J, Wessels HJCT, van Alen T, Luesken F, Wu ML, van de Pas-Schoonen KT, Op den Camp HJM, Janssen-Megens EM, Francoijs KJ, Stunnenberg H, Weissenbach J, Jetten MSM, Strous M (2010) Nitrite-driven anaerobic methane oxidation by oxygenic bacteria. Nature 464:543–548
Ho A, Luke C, Cao ZH, Frenzel P (2011) Ageing well: methane oxidation and methane oxidizing bacteria along a chronosequence of 2000 years. Environ Microbiol Rep 3:738–743
Hu BL, Shen LD, Lian X, Zhu Q, Liu S, Huang Q, He ZF, Geng S, Cheng DQ, Lou LP, XY X, Zheng P, He YF (2014) Evidence for nitrite-dependent anaerobic methane oxidation as a previously overlooked microbial methane sink in wetlands. Proc Natl Acad Sci U S A 111:4495–4500
Jiang N, Wang Y, Dong X (2010) Methanol as the primary methanogenic and acetogenic precursor in the cold Zoige wetland at Tibetan Plateau. Microb Ecol 60:206–213
Kolb S, Knief C, Stübner S, Conrad R (2003) Quantitative detection of methanotrophs in soil by novel pmoA-targeted real-time PCR assays. Appl Environ Microbiol 69:2423–2429
Lee SH, Kang H (2016) The activity and community structure of total bacteria and denitrifying bacteria across soil depths and biological gradients in estuary ecosystem. Appl Microbiol Biotechnol 100:1999–2010
Liu Y, Zhang JX, Zhao L, Li YZ, Yang YY, Xie SG (2015) Aerobic and nitrite-dependent methane-oxidizing microorganisms in sediments of freshwater lakes on the Yunnan Plateau. Appl Microbiol Biotechnol 99:2371–2381
Long Y, Yi H, Chen SL, Zhang ZK, Cui K, Bing YX, Zhuo QF, Li BX, Xie SG, Guo QW (2016) Influences of plant type on bacterial and archaeal communities in constructed wetland treating polluted river water. Environ Sci Pollut Res 23:19570–19579
Long Y, Jiang XJ, Guo QW, Li BX, Xie SG (2017a) Sediment nitrite dependent methane-oxidizing microorganisms temporally and spatially shift in the Dongjiang River. Appl Microbiol Biotechnol 101:401–410
Long Y, Guo QW, Li NN, Li BX, Tong TL, Xie SG (2017b) Spatial change of reservoir nitrite-dependent methane-oxidizing microorganisms. Ann Microbiol 67:165–174
Narihiro T, Hori T, Nagata O, Hoshino T, Yumoto I, Kamagata Y (2011) The impact of aridification and vegetation type on changes in the community structure of methane-cycling microorganisms in Japanese wetland soils. Biosci Biotechnol Biochem 75:1727–1734
Rietl AJ, Overlander ME, Nyman AJ, Jackson CR (2016) Microbial community composition and extracellular enzyme activities associated with Juncus roemerianus and Spartina alterniflora vegetated sediments in Louisiana saltmarshes. Microb Ecol 71:290–303
Schloss PD, Westcott SL, Ryabin T, Hall JR, Hartmann M, Hollister EB, Lesniewski RA, Oakley BB, Parks DH, Robinson CJ, Sahl JW, Stres B, Thallinger GG, Van Horn DJ, Weber CF (2009) Introducing MOTHUR: open-source, platform-independent, community supported software for describing and comparing microbial communities. Appl Environ Microbiol 75:7537–7541
Shen LD, Huang Q, He ZF, Lian X, Liu S, He YF, Lou LP, XY X, Zheng P, BL H (2015) Vertical distribution of nitrite-dependent anaerobic methane-oxidising bacteria in natural freshwater wetland soils. Appl Microbiol Biotechnol 99:349–357
Sow SLS, Khoo G, Chong LK, Smith TJ, Harrison PL, Ong HKA (2014) Molecular diversity of the ammonia-oxidizing bacteria community in disused tin-mining ponds located within Kampar, Perak, Malaysia. World J Microbiol Biotechnol 30:757–766
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol 30:2725–2729
Tian JQ, Chen H, Dong XZ, Wang YF (2012) Relationship between archaeal community structure and vegetation type in a fen on the Qinghai–Tibetan Plateau. Biol Fertil Soils 48:349–356
Wang Y, Zhu G, Harhangi HR, Zhu B, Jetten MSM, Yin C, Op den Camp HJM (2012) Co-occurrence and distribution of nitrite-dependent anaerobic ammonium and methane-oxidizing bacteria in a paddy soil. FEMS Microbiol Lett 336:79–88
Wang Y, Huang P, Ye F, Jiang Y, Song LY, Op den Camp HJM, Zhu GB, SJ W (2016) Nitrite-dependent anaerobic methane oxidizing bacteria along the water level fluctuation zone of the Three Gorges Reservoir. Appl Microbiol Biotechnol 100:1977–1986
Yang MX, Guo QW, Tong TL, Li NN, Xie SG, Long Y (2017) Vegetation type and layer depth influence nitrite-dependent methane-oxidizing bacteria in constructed wetland. Arch Microbiol 199:505–511
Yang YY, Shan JW, Zhang JX, Zhang XL, Xie SG, Liu Y (2014) Ammonia- and methane-oxidizing microorganisms in high-altitude wetland sediments and adjacent agricultural soils. Appl Microbiol Biotechnol 98:10197–10209
Yang YY, Zhao Q, Cui YH, Wang YL, Xie SG, Liu Y (2016) Spatio-temporal variation of sediment methanotrophic microorganisms in a large eutrophic lake. Microb Ecol 71:9–17
Yi XH, Jing DD, Wan JQ, Ma YW, Wang Y (2016) Temporal and spatial variations of contaminant removal, enzyme activities, and microbial community structure in a pilot horizontal subsurface flow constructed wetland purifying industrial runoff. Environ Sci Pollut Res 23:8565–8576
Yun JL, Zhuang GQ, Ma AZ, Guo HG, Wang YF, Zhang HX (2012) Community structure, abundance, and activity of methanotrophs in the Zoige wetland of the Tibetan Plateau. Microb Ecol 63:835–843
Yun J, Yu Z, Li K, Zhang H (2013) Diversity, abundance and vertical distribution of methane-oxidizing bacteria (methanotrophs) in the sediments of the Xianghai wetland, Songnen Plain, northeast China. J Soils Sediments 13:242–252
Yun JL, YW J, Deng YC, Zhang HX (2014) Bacterial community structure in two permafrost wetlands on the Tibetan Plateau and Sanjiang Plain, China. Microb Ecol 68:360–369
Yun JL, Zhang HX, Deng YC, Wang YF (2015) Aerobic methanotroph diversity in Sanjiang wetland, northeast China. Microb Ecol 69:567–576
Zhu GB, Zhou LL, Wang Y, Wang SY, Guo JH, Long XE, Sun XB, Jiang B, Hou QY, Jetten MSM, Yin CQ (2015) Biogeographical distribution of denitrifying anaerobic methane oxidizing bacteria in Chinese wetland ecosystems. Environ Microbiol Rep 7:128–138
Funding
This work was supported by Integration Research and Demonstration of Water Quality Conservation and Ecological Restoration Technology of Urban Lake Landscape Water: the Major Projects of Synergy Creation of Production, Learning and Research of Guangzhou (No. 201508020078) and Basic scientific research business of Central level public welfare scientific research institution-Key technology research and equipment development of sudden oil spill pollution control in River Basin.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
The authors declare that they have no competing interests.
Electronic supplementary material
ESM 1
(PDF 255 kb)
Rights and permissions
About this article
Cite this article
Chen, S., Chen, J., Chang, S. et al. Aerobic and anaerobic methanotrophic communities in urban landscape wetland. Appl Microbiol Biotechnol 102, 433–445 (2018). https://doi.org/10.1007/s00253-017-8592-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-017-8592-y