Abstract
Today, methane (CH4), nitrous oxide (N2O), and carbon dioxide (CO2) emissions represent approximately 98 % of the total greenhouse gas (GHG) inventory worldwide, and their share is expected to increase significantly in this twenty-first century. CO2 represents the most important GHG with approximately 77 % of the total GHG emissions (considering its global warming potential) worldwide, while CH4 and N2O are emitted to a lesser extent (14 and 8 %, respectively) but exhibit global warming potentials 23 and 298 times higher than that of CO2, respectively. Most members of the United Nations, based on the urgent need to maintain the global average temperature 2 °C above preindustrial levels, have committed themselves to significantly reduce their GHG emissions. In this context, an active abatement of these emissions will help to achieve these target emission cuts without compromising industrial growth. Nowadays, there are sufficient empirical evidence to support that biological technologies can become, if properly tailored, a low-cost and environmentally friendly alternative to physical/chemical methods for the abatement of GHGs. This study constitutes a state-of-the-art review of the microbiology (biochemistry, kinetics, and waste-to-value processes) and bioreactor technology of CH4, N2O, and CO2 abatement. The potential and limitations of biological GHG degradation processes are critically discussed, and the current knowledge gaps and technology niches in the field are identified.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Today, CH4, N2O, and CO2 emissions represent approximately 98 % of the total greenhouse gas (GHG) inventory worldwide, and their share is expected to increase in this twenty-first century based on their industrial and organic-based nature and the forthcoming scenario of increasing world population (European Environment Agency 2011; Environmental Protection Agency 2011). CH4, with a global warming potential 23 times higher than that of CO2 and an atmospheric concentration increasing at 0.2–1 % year−1, is mainly emitted from organic waste treatment activities such as landfilling, composting, and wastewater treatment (95 million tons CO2 equivalent in the European Union [EU]) and livestock farming (166 million tons CO2 equivalent in the EU) (European Environment Agency 2011). CH4 represents approximately 14 % of the total GHG emissions worldwide (Intergovernmental Panel on Climate Change 2007). Emissions from compost piles or animal houses typically contain 0–200 mg CH4 m−3, while these concentrations can increase up to 20–100 g CH4 m−3 in old landfills (Nikiema et al. 2007). On the other hand, N2O is not only a major GHG with a global warming potential 298 times higher than that of CO2 but it is also the most important O3-depleting substance emitted in the twenty-first century, with yearly atmospheric concentration increases of 0.3 % (Ravishankara et al. 2009). In the EU-27, N2O is mainly emitted in waste treatment activities (10 million tons CO2 equivalent), nitric and adipic acid production (27 million tons CO2 equivalent), and livestock farming (21 million tons CO2 equivalent) (European Environment Agency 2011). N2O contributes to 8 % of the GHG emissions worldwide (Intergovernmental Panel on Climate Change 2007). Concentrations ranging from 10 to 2,000 mg N2O m−3 are common in the emissions of these activities (Xu et al. 2004; Kampschreur et al. 2008). Even in tanks with novel microbial nitrogen removal processes such as nitritation/anaerobic ammonium oxidation (anammox), significant amounts of N2O have been recorded during wastewater treatment (Kampschreur et al. 2008). Finally, CO2 represents the most important GHG with approximately 77 % of the total GHG emissions worldwide and an annual atmospheric concentration increase of 0.5 % over the last decade (Intergovernmental Panel on Climate Change 2007). CO2 is mainly produced in electricity and heat production processes (1,400 million tons), transport (960 million tons), industrial and fuel manufacturing (700 million tons), and other sectors (e.g., commercial, residential, or agricultural; with 670 million tons). Typical CO2 concentrations in combustion gases range from approximately 90 to 270 g m−3 (European Environment Agency 2011).
Most members of the United Nations, based on the urgent need to maintain the global average temperature 2 °C above preindustrial levels, have committed themselves to significantly reduce their GHG emissions (Intergovernmental Panel on Climate Change 2007). In this context, an active abatement of these emissions will help to achieve these target emission cuts without compromising industrial growth. Besides, the development of cost-efficient GHG abatement methods might be of paramount importance in the near future since recent monitoring campaigns in waste treatment facilities, for instance, have shown that the CH4 and N2O emissions reported might be underestimated by one order of magnitude (Foley et al. 2010; Ahn et al. 2010). As a matter of fact, most international GHG inventories have based their estimations on Intergovernmental Panel on Climate Change emission factors, and there is increasing evidence that some of these factors, for instance, those applied to waste treatment activities, might lack a scientific basis or be based on studies under very specific and non-extrapolable conditions (Foley et al. 2010; Ahn et al. 2010). In addition, the gradual application of the EU Landfill Directive 1999/31 (enforcing a reduction in the organic matter content of the solid waste to be disposed) will result in emissions with lower CH4 concentrations, which will significantly limit the implementation of conventional energy recovery-based treatment technologies and will require the application of cost-efficient abatement methods for diluted CH4 streams.
However, despite the environmental relevance of CH4, N2O, and CO2 emissions, the development of cost-efficient and environmentally friendly GHG treatment technologies (especially of those intended for the treatment of emissions containing low GHG concentrations) has been scarce. Today, physical/chemical treatment methods for CH4 abatement such as activated carbon adsorption or incineration are either inefficient or costly at the low concentrations typically found in emissions from waste treatment and animal farming and possess a high CO2 footprint as a result of their intensive energy usage (Melse and Van der Werf 2005). On the other hand, conventional NO x treatment technologies such as selective catalytic reduction or selective noncatalytic reduction present prohibitive operating costs (and large environmental impacts) when treating large air flow rates containing low concentrations of NO x as a result of their intensive energy use (Skalska et al. 2010). Similarly, CO2 sequestration through physical/chemical methods, such as gas scrubbing with alkaline or amine solutions or direct injection into subsurface natural reservoirs (storage), entails prohibitive costs and secondary environmental pollution (Herzog 2001; Kumar et al. 2011). In this regard, biotechnologies can become, if properly tailored, a low-cost and environmentally friendly alternative to physical/chemical methods for the abatement of CH4, N2O, and CO2. Biotechnologies, which are based on the biocatalytic action of specialized bacteria, microalgae, or fungi, have been consistently proven as robust and efficient abatement methods for the treatment of industrial volatile organic compounds and malodors, exhibiting lower operating costs and environmental impacts than their physical/chemical counterparts (Estrada et al. 2011, 2012b). This study constitutes a state-of-the-art review of the microbiology (biochemistry, kinetics, and waste-to-value processes) and bioreactor technology of CH4, N2O, and CO2 abatement. The potential and limitations of biological GHG degradation processes are critically discussed, and the current knowledge gaps and technological research niches in the field are identified.
Biological CH4 removal
Microbiology of CH4 removal
Methanotrophs are methylotrophic bacteria able to utilize CH4 as the sole carbon and energy source, although several yeast genera such as Sporobolomyces and Rhodotorula or even the green microalgae Chlorella have been reported as methane oxidizers (Enebo 1967; Wolf and Hanson 1979). Most methanotrophs oxidize CH4 in the presence of O2, although some strains can couple the oxidation of CH4 to the reduction of sulfate, nitrate, nitrite, manganese, or iron (Beal et al. 2009; Knittel and Boetius 2009; Ettwig et al. 2010). Methanotrophs are ubiquitous in the environment and commonly found in tundra and wetlands, freshwater and eutrophic lakes, marine sediments and water columns, sewage sludge, rice paddies, agricultural soils and peat bogs, preferentially in the interface between aerobic and anaerobic areas with substantial fluxes of CH4 (Bowman 2006; Hanson and Hanson 1996). Three types of methanotrophs are distinguished: type I methanotrophs are characterized by uniform intracytoplasmic membranes throughout the cell as bundles of vesicular disks, membrane fatty acids with 14–16 carbons, and the use of the ribulose monophosphate pathway (RuMP) for formaldehyde assimilation. These methanotrophs belong to the Gammaproteobacteria class and include genera such as Methylomonas, Methylobacter, Methylomicrobium, and Methylococcus (Bratina et al. 1992). Encapsulated bacteria of the Clonothrix and Crenothrix genera within the Methylococcaceae family have been also recently identified as type I methanotrophs (Op den Camp et al. 2009). Type II methanotrophs are found within the Alphaproteobacteria class (Methylosinus and Methylocystis genera) and are characterized by intracytoplasmic membranes along the peripheral part of the cell, fatty acids with 18 carbons, and the use of the serine pathway for formaldehyde assimilation (Patt and Hanson 1978; Scott et al. 1981; Hanson and Hanson 1996). Type X methanotrophs contain membrane fatty acids with 16 carbons, use the RuMP pathway, posses a ribulose-1,5-bisphosphate carboxylase, grow at higher temperatures than types I and II, and belong mainly to the Methylococcus genera (Davies and Whittenbury 1970; Hanson and Hanson 1996). Despite the fact that most methanotrophs are included in the Gammaproteobacteria and Alphaproteobacteria classes, some of them belong to the NC10 phylum (nitrite-respiring methanotrophs), Verrucomicrobia, and Euryarchaeota (Hanson and Hanson 1996; Hou et al. 2008; Op den Camp et al. 2009; Ettwig et al. 2010; Semrau 2011).
All methanotrophs use methane monooxygenases (MMOs) during the aerobic oxidation of CH4 to methanol, utilizing reducing equivalents for the split of the O2 bonds. Two forms of MMOs have been identified in methanotrophs: soluble and particulate. The soluble MMO form (sMMO) was first detected in the cytosol of methanotrophs II and X and recently identified in type I (Koh et al. 1993; Hanson and Hanson 1996). This monooxygenase has a broader range of substrates than other monooxygenases and it is composed of a hydroxylase, a reductase, and a regulatory protein (Lipscomb 1994; Hanson and Hanson 1996; Wallar and Lipscomb 2001). sMMOs are synthesized when levels of Cu2+ are below 0.86 μmol g−1 dry weight (dw) and contain iron as an important cofactor in the reaction center (Hanson and Hanson 1996; Nielsen et al. 1997; Choi et al. 2003). On the other hand, particulate or membrane-bound MMOs (pMMO) are constitutive in all aerobic methanotrophs at Cu2+ concentrations above 0.86 μmol g−1 dw, except in the genera Methyloferula and Methylocella (Dalton 1992; Semrau et al. 2010). pMMO is also composed of three polypeptides, which exhibit a lower specific activity compared to sMMO, but nowadays, little is known about its molecular properties (Basu et al. 2003; Choi et al. 2005). Soluble MMOs employ NADH + H+ as reducing agent to oxidize the CH4 present in the intracellular medium (which previously diffused from the extracellular aqueous medium) to CH3OH, while pMMO requires a cytochrome complex to oxidize the CH4 present in the cytoplasm. Then, the periplasmic enzyme methanol dehydrogenase catalyzes the conversion of methanol to formaldehyde (Fig. 1) (O’Connor 1981; Anthony 1982; Dalton 1992; Hanson and Hanson 1996; Glass and Orphan 2012). Formaldehyde is then either converted to formic acid by formaldehyde dehydrogenase or to biomass through the RuMP pathway in type I and X methanotrophs or through the serine pathway in type II methanotrophs (Hanson and Hanson 1996).
Methane oxidation pathways in type I and II methanotrophs. Continuous lines represent a unidirectional flow in the pathway, while dashed lines represent alternative pathways. sMMO soluble methane monooxygenase, pMMO particulate methane monooxygenase, MDH methanol dehydrogenase, FDH formate dehydrogenase, FalDH formaldehyde dehydrogenase, cyt red/ox cytochrome reduced/oxidized, STHM serine hydroxymethyltransferase, H6PS hexulose-6-phosphate synthase. Adapted from Hanson and Hanson (1996) and Semrau et al. (2010)
Finally, carbon dioxide is produced from formate in an oxidation step catalyzed by formate dehydrogenase with the production of the reducing equivalents required at the initial steps of CH4 oxidation (Hanson and Hanson 1996; Chistoserdova 2011; Glass and Orphan 2012). Moreover, type X methanotrophs can assimilate formaldehyde through the RuMP pathway while fixing CO2 using a ribulose-1,5-bisphosphate carboxylase (Whittenbury 1981). For a more extensive discussion of the biochemistry of aerobic CH4 oxidation, readers are referred to specialized reviews (Higgins et al. 1981; Hanson and Hanson 1996; Semrau et al. 2010).
In contrast to aerobic CH4 oxidation, the information available in the literature about anaerobic CH4 oxidation is scarce. Moreover, anaerobic methane-oxidizing archaea, which are responsible for 7–25 % of the total CH4 oxidation worldwide, can undertake this process in symbiosis with sulfate-reducing bacteria, using CH4 as an electron donor to convert sulfate into sulfite (Hanson 1980; Hoehler et al. 1995, Reeburgh 2007). On the other hand, aerobic CH4-oxidizing bacteria can grow concomitantly with denitrifying bacteria, consuming oxygen and producing electron donors for denitrification (Bédard and Knowles 1989; Thalasso et al. 1997). In addition, a recently discovered denitrifying methanotroph Methylomirabilis oxyfera, from the phylum NC10, is able to perform intra-aerobic CH4 oxidation utilizing the oxygen contained in nitric oxide (NO) (Wu et al. 2011).
Microbial CH4 oxidation is often described by both Monod and Michaelis–Menten models. However, the different experimental setups and models used for the estimation of the kinetic parameters often hinder the direct comparison of the data available in literature (Table 1). The Michaelis–Menten constant (K M) determines the rate of the overall enzymatic activity and ranges from 31.7 × 10−9 to 2.8 × 10−5 M, the lowest value highlighting the high affinity of some bacterial enzymes for CH4 oxidation (Scheutz et al. 2009). Similarly, the Monod constant (K S) defines the affinity of microorganisms for the substrates and, in the particular case of CH4, ranges typically from 1 × 10−6 to 4.74 × 10−4 M. pMMO-bearing bacteria have been reported to exhibit a higher affinity for CH4 (lower K S values) than sMMO-bearing microorganisms (Bédard and Knowles 1989). Moreover, the maximum CH4 oxidation rates (V max) are often reported in a wide variety of units, with values ranging from 0.02 to 0.6 g m−3 h−1, from 1.48 × 10−4 to 25.7 g m−2 h−1, from 2.88 × 10−6 to 0.50 g g−1 h−1, and from 3.2 × 10−15 to 1.44 × 10−12 g cell−1 h−1. Varied biomass yields ranging from 0.02 to 0.8 g biomass g CH4 −1 are also typically found in the literature (Arcangeli and Arvin 1999).
The rates of CH4 oxidation depend on parameters such as dissolved oxygen and CH4 concentrations, moisture content in the media, temperature, pH, type of nitrogen source, or copper levels. Hence, both types I and II methanotrophs exhibit their maximum oxidation rate at gas oxygen concentrations ranging from 1.5 to 10.5 % v/v (Whittenbury and Dalton 1981; Wilshusen et al. 2004). Furthermore, high CH4/O2 ratios stimulated the growth of type II methanotrophs (high K M), while low CH4/O2 ratios stimulated the growth of type I methanotrophs (low K M) (Bender and Conrad 1995; Amaral and Knowles 1995). These findings support the hypothesis that sMMOs are usually expressed at high CH4 concentrations and pMMO at low CH4 concentrations (Lontoh and Semrau 1998; Henckel et al. 2000). The optimum moisture content for methanotroph growth was shown to be packing media specific, which will be discussed later on in the reactor section. Methanotrophs are mesophilic microorganisms, although thermotolerant and thermophilic Methylothermus, Methylocaldum, Methylococcus, or Verrucomicrobia strains have been isolated from hot springs, with an optimal growth in the range of 42–65 °C (Bodrossy et al. 1999; Tsubota et al. 2005). Psychrophilic strains of Methylobacter, Methylosphaera, and Methylomonas have been isolated from tundra soils, Antarctic meromictic lakes, and deep igneous groundwater, exhibiting an optimal growth range of 3.5–15 °C (Omelchenko et al. 1993; Bowman et al. 1997; Kalyuzhnaya et al. 1999). Hence, maximum oxidation rates in composite soils occurred at 31 °C, while in most peat soils, the optimal temperature was 25 °C (Bédard and Knowles 1989; Whalen et al. 1990; Dunfield et al. 1993; Bender and Conrad 1995). On the other hand, low CH4 oxidation rates were recorded in samples from forest and landfill cover soils at 2–5 °C (Whalen and Reeburgh 1996; Christophersen et al. 2000). Species from the Methylocella and Methylocapsa genera or the Verrucomicrobia phylum exhibiting an optimum growth at pH 2–2.5 have been isolated from acidic environments, but most methanotrophs preferably live and oxidize CH4 at pH 7–7.65 (Bender and Conrad 1995; Dedysh et al. 1998, 2002; Pol et al. 2007; Dunfield et al. 2007). Oxidation rates by acidophilic methanotrophs have been found to be higher in samples from acidic peat soils (pH values of 4–5) compared to those from neutral soils (pH values of 6–8) (Born et al. 1990; Dunfield et al. 1993). Despite type II methanotrophs being able to express the enzyme nitrogenase at low O2 levels and perform N2 fixation, nitrate and ammonia are the preferred N sources (Quayle 1972). Type I methanotrophs are dominant in environments with low methane concentrations and high inorganic nitrogen levels, while type II methanotrophs are often predominant in scenarios with high CH4/N ratios (Amaral and Knowles 1995). Microbial stimulation or inhibition mediated by inorganic nitrogen sources such as ammonia or nitrate depend on the nitrogen and CH4 concentration, the pH, and the type of methanotroph. Hence, optimal growth and CH4 oxidation rates were recorded at 12–61 mM ammonia for some methanotrophic communities, while some studies revealed that concentrations of 4–10 mM could reduce CH4 oxidation by 30 % (Bender and Conrad 1995). Some in situ investigations suggest that high ammonia concentrations could inhibit CH4 oxidation either by toxicity or enzymatic competition, although such detrimental effects could be also due to a nitrite accumulation from ammonia oxidation (Bédard and Knowles 1989). In this context, ammonia fertilizers seem to be more hazardous to type II than to type I methanotrophs (Mohanty et al. 2006). Copper positively regulates the activity of pMMO/sMMO and controls the expression of their genes (Stanley et al. 1983; Dalton et al. 1984). However, Cu2+ concentrations have to be controlled in order to maintain copper homeostasis and prevent metal toxicity. The optimal growth of most methanotrophs is achieved at cooper concentrations lower than 4.3 mM (Bender and Conrad 1995), although a combination of high Cu levels and organic material can favor the rapid growth of type II methanotrophs (Graham et al. 1993). Besides, in Cu-limiting scenarios, some aerobic methanotrophs excrete a molecule called methanobactin able to bind cooper in the extracellular medium and actively transport it into the cell at concentrations as low as 0.7–1 μM (Kim et al. 2004; Balasubramanian et al. 2011).
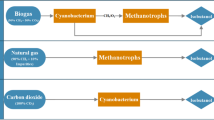
Microbial CH4 oxidation can be coupled with the production of high added-value biotechnological products. Thus, some methanotrophs can utilize acetyl-CoA from the serine or RuMP pathways to form polyhydroxyalkanoates such as polyhydroxybutyrate (PHB), an important commodity in the bioplastic industry (Higgins et al. 1981; Lidstrom and Stirling 1990). PHB generation can be stimulated under N-, S-, or Mg-limiting conditions. For instance, N limitations supported the production of PHB at 39 % (w/w) in Methylobacterium organophilum in a bioreactor and even at 57 % (w/w) in serological glass bottles (Zúñiga et al. 2011). Exopolysaccharides at 62 % (w/w) can be also produced from CH4 oxidation using Methylocystis parvus (Hou et al. 1978; Chida et al. 1983). CH4-oxidizing bacteria, as well as algae, fungi, or yeasts, can be also used in single-cell protein production for human and animal consumption. For instance, Norferm Danmark A/S in Norway produces 8,000 tons protein year−1 (BioProtein) from Methylococcus capsulatus Bath (Winder 2004).
Reactors for biological CH4 oxidation
Several bioreactor configurations such as biofilters, biotrickling filters, airlifts, and stirred tanks have been used for CH4 abatement from air emissions (Fig. 2). Biotechnologies for CH4 abatement have been implemented in enclosed bioreactors where the polluted air (containing O2) is supplied by forced ventilation in either upflow or downflow mode and in open bioreactors where the CH4-bearing emission (e.g., landfill gas) is supplied upwards at the bottom of the system, while O2 diffuses from the air at the top of the bioreactor (Gebert et al. 2001; Gebert and Gröngröft 2006)
Enclosed laboratory-scale bioreactors are the most popular CH4 abatement systems reported in the literature likely due to the fact that operational parameters such as temperature and moisture content can be controlled. On the other hand, open bioreactors constitute the most implemented technology to treat real CH4 emissions from landfills at full scale. Unfortunately, forced air ventilation to provide the O2 needed by aerobic methanotrophs is not recommended when treating real landfill CH4 emissions to avoid explosion risks (CH4 concentration in landfill gas in the range of 30–70 % v/v) (Kallistova et al. 2005; Zamorano et al. 2007). Therefore, O2 supply in open passively vented biofilters strongly depends on climate conditions, and O2-limiting conditions are often encountered in these bioreactors (Humer and Lechner 1999; Gebert et al. 2001; Berger et al. 2005). In this regard, a three-dimensional numerical model has been recently developed, incorporating the effect of advection–diffusion gas flows, heat, and moisture on CH4 oxidation in open biofilters (Hettiarachchi et al. 2011). This innovative model allowed for the estimation of CH4 removal under several environmental conditions confirming that CH4 oxidation in open biofilters strongly depends on environmental factors.
Biofilters are by far the most common bioreactors used for CH4 removal although innovative multiphase systems operated in different bioreactor configurations have emerged in the last 5 years. Despite being relatively simple systems, biofilters constitute the most studied and implemented technology to date and can support high CH4 removal rates when parameters such as the O2 concentration or the moisture content are optimized. For instance, biofilter operation in landfills at O2 concentrations of 18–28 μM (1.7–2.6 % in the gas phase) resulted in maximum oxidation rates of 1.78 × 10−6 mol CH4 g−1 dw h−1 (Gebert et al. 2003). The optimum moisture content was shown to depend on water activity, which itself is often packing media specific, with maximum CH4 oxidation rates at a moisture content of 11 % (w/w) in composite soils, 10–20 % in landfill cover soils, and 20–35 % in other types of soils, while moisture contents of 5, 56, or 71 % in composite soils supported low oxidations rates (Whalen et al. 1990; Bender and Conrad 1995). The intensive research conducted on biofiltration over the last 20 years has resulted in important advances in packing material technology, evolving from a bed of simple materials such as compost, pine bark, or perlite to more sophisticated structured packings such as metallic or polymeric ring and foams or custom-made nutrients containing polymeric pellets. Besides, optimum environmental conditions, nutrient requirements, and potential inhibitors of CH4 oxidation in biofilters have been recently identified (Veillette et al. 2012). These breakthroughs in biofiltration technology have resulted in significant reductions in the EBRTs required to efficiently abate CH4 emissions. For instance, ECs of 1.2–25 g m−3 h−1 have been recorded in conventional biofilters operated at EBRTs of 10–360 h (Table 2) (du Plessis et al. 2003; Berger et al. 2005; Melse and Van der Werf 2005), while third generation enclosed biofilters operated at EBRTs of 0.3–20 h were able to reach ECs of 65–280 g m−3 h−1 (Gebert and Gröngröft 2006; Nikiema and Heitz 2009; Park et al. 2009). To the best of our knowledge, Park et al. (2009) reported the highest abatement performance in a biofilter treating CH4 with a maximum EC of 280 g m−3 h−1 (corresponding to an RE of 50 %) at an EBRT of 1.2 h. Girard et al. (2011) achieved an EC of 14.5 g m−3 h−1 in a biofilter operated at an EBRT of 0.07 h. Interestingly, a similar EC to that obtained by Park et al. (2009) would be expected if the ECs obtained by Girard et al. (2011) were multiplied by 17, which is the EBRT ratio between these reports.
Several bioreactor configurations and operation modes have been recently investigated in order to overcome the typical mass transfer limitations encountered in conventional biofilters as a result of the low water solubility of this GHG (Fig. 2). Rocha-Rios et al. (2009, 2011) studied the performance of the so-called two-phase partitioning bioreactors (TPPBs) for CH4 abatement, which are based on the addition of a nonaqueous phase (e.g., a liquid solvent or a solid polymer) with a high affinity for CH4, resulting in higher CH4 absorptions and driving forces for mass transfer (Quijano et al. 2009). Silicone oil, exhibiting 15 times higher affinity for CH4 than water is one of the most commonly used nonaqueous phases in TPPBs. In this context, a silicone oil-based TPPB implemented in a stirred tank reached a maximum EC of 106 g m−3 h−1 at an EBRT as short as 0.08 h (corresponding to 4.8 min) (Rocha-Rios et al. 2009). This important reduction in the EBRT dramatically decreased the size of the abatement unit and consequently the overall technology cost. In the quest for less energy-demanding systems than stirred tank reactors, TPPBs have been also implemented in airlift systems with a maximum EC of 22 g m−3 h−1 at an EBRT of 0.12 h. Nevertheless, a better performance was reported in a TPPB operated as a biotrickling filter, exhibiting a maximum EC of 51 g m−3 h−1 at an EBRT of 0.08 h (Rocha-Rios et al. 2009). Likewise, Avalos et al. (2012) recently reported that a biotrickling filter (without the nonaqueous phase) using stones as packing material and provided with a nonionic surfactant was able to reach a maximum EC of 21 g m−3 h−1 at EBRTs as short as 0.07 h (corresponding to 4.2 min). Although promising results have been obtained in TPPBs and single liquid-phase biotrickling filters, more research is still necessary to boost the overwhelming CH4 abatement potential of biotechnologies.
At this point, it is important to stress that little attention has been given to the microbiological aspects in bioreactors devoted to CH4 abatement (Scheutz et al. 2009). In some studies, the bioreactors were operated for months to stimulate the growth of the indigenous methanotrophic microorganisms present in the packing materials, which resulted in either very long start-up periods (du Plessis et al. 2003; Einola et al. 2008) or in a poor CH4 abatement performance (Berger et al. 2005). A more efficient performance and shorter start-up periods were recorded when the bioreactor was inoculated with methanotrophic biomass from the leachate (or directly with a portion of the packing material) of a CH4-treating biofilter (Nikiema and Heitz 2009; Avalos et al. 2012). The enrichment of methanotrophs from activated sludge of wastewater treatment plants is also another common strategy to produce acclimated inocula and to reduce process start-up periods (Rocha-Rios et al. 2009). On the other hand, Avalos et al. (2012) found that clogging issues were very different in a biotrickling filter packed with clay spheres, polypropylene spheres, and stones, despite using the same biomass and inoculation protocol, the reactor packed with stones being more susceptible to clogging and presenting stability problems. These recent studies, therefore, confirm that microbiology and reactor/packing material design are important research areas to be pursued due to their key role on bioreactor start-up and abatement performance.
Biological N2O removal
Microbiology of N2O removal
To date, research on N2O emission control has been mainly focused on minimizing and/or preventing N2O generation and its release to the atmosphere. Once N2O is formed, removal by in situ consumption might be also promoted before implementing end-of-pipe technologies (Desloover et al. 2012). The mitigation strategies developed to date were based on microbiological studies investigating the influence of process operational conditions on N2O production rates, especially in wastewater treatment plants and composting facilities (Kampschreur et al. 2009; Rassamee et al. 2011). In this context, the elucidation of the complex mechanisms governing the microbial production and consumption of N2O is crucial to develop cost-effective end-of-pipe biotechnologies for N2O abatement.
N2O may be biologically produced by both nitrification and denitrification processes (Fig. 3). Nitrification is a sequential and predominantly autotrophic process where NH3 is first oxidized to NO2 − by ammonia-oxidizing bacteria and then further oxidized to NO3 − by nitrite-oxidizing bacteria. Conventional denitrification involves NO3 − reduction by heterotrophic denitrifiers to N2 through a stepwise reduction sequence involving NO2 −, NO, and N2O. Thus, the main routes for N2O production are hydroxylamine oxidation, nitrifier denitrification, and heterotrophic denitrification (Kampschreur et al. 2009; Desloover et al. 2012; Wunderlin et al. 2012) (Fig. 3).
Overview of the main metabolic routes involved in N2O production and biodegradation in bacterial cultures. The enzymes responsible of each individual reaction are ammonia monooxygenase (AMO), hydroxylamine oxidoreductase (HAO), nitrite oxidoreductase (NXR), nitrate reductase (NAR), nitrite reductase (NIR), nitric oxide reductase (NOR), and nitrous oxide reductase (NOS). Adapted from Wrage et al. (2001), Desloover et al. (2012), and Glass and Orphan (2012)
Hydroxylamine (NH2OH) is an intermediate in the nitritation step (oxidation of NH3 to NO2 −). The oxidation of NH3 to NH2OH is catalyzed by an ammonia monooxygenase (Wood 1986; Wrage et al. 2001), while the oxidation of NH2OH to NO2 − is catalyzed by a hydroxylamine oxidoreductase (Hooper and Terry 1979). N2O production occurs via chemical decomposition of NH2OH in a process called chemodenitrification or through NO2 − reduction with electron donors such as organic (e.g., amines) or inorganic (e.g., Fe2+ and Cu2+) compounds (Wunderlin et al. 2012). During nitrifier denitrification, NH3 is partially oxidized to NO2 − and then further reduced to NO, N2O, and N2 in a process carried out by a very specific group of microorganisms (Wrage et al. 2001). Since nitrifier denitrification involves both nitritation and denitrification steps, N2O may thus be produced in both processes (Colliver and Stephenson 2000). Finally, N2O production during heterotrophic denitrification is caused by an interruption in the last reduction step before reaching the most reduced compound (N2). Thus, imbalances in the production and consumption of the enzyme nitrous oxide reductase (NOS), which catalyzes N2O reduction to N2, have been pointed out as the main responsible of N2O accumulation (Wunderlin et al. 2012; Glass and Orphan 2012). In brief, suboptimal conditions for nitrification and denitrification are intrinsically related to N2O emissions. For instance, when nitrification occurs under oxygen-limiting conditions, ammonia-oxidizing bacteria use NO2 − as the terminal electron acceptor instead of O2, leading to higher N2O emissions (Colliver and Stephenson 2000; Wunderlin et al. 2012). Indeed, emissions of N2O have been also recorded in anammox tanks, although these emissions could not be directly attributed to the anaerobic oxidation of ammonium (Kampschreur et al. 2008). Likewise, the enzyme NOS catalyzing N2O reduction during heterotrophic denitrification is the most sensitive to O2, causing incomplete denitrification and N2O accumulation at high O2 concentrations (Tallec et al. 2008; Uggetti et al. 2012). High NO2 − concentrations can also induce an enhanced reduction of this compound to N2O during nitrification and lower denitrification rates, resulting in an accumulation of NO and N2O (Kampschreur et al. 2009).
Based on the fact that the aerobic oxidation of N2O, to the best of our knowledge, has not been reported yet, any biotechnology devoted to N2O removal must focus on reducing N2O to N2 by favoring the activity of the enzyme (NOS). In this context, the enzyme NOS is known to require Cu in larger quantities than other enzymes, and the reduction of N2O is thus feasible when Cu is present above a certain threshold concentration (Glass and Orphan 2012). Granger and Ward (2003) observed a low NOS activity in denitrifiers grown at 0.3 nM of total dissolved Cu and a complete consumption of N2O when Cu was increased up to 10 nM. Environments rich in sulfides might support low N2O reduction rates since sulfide scavenging and metal precipitation may reduce Cu bioavailability. Very few studies have investigated the potential of microorganisms for N2O reduction, and the few studies available focused on O2-free emissions, conditions which are far from those present in most real N2O emissions. For instance, Apel and Turick (1992) assessed the ability of nine bacterial species to reduce N2O to N2 under anaerobic conditions (headspace filled with N2O and He) in organic synthetic media. Seven species were able to successfully consume N2O, with Pseudomonas denitrificans and Paracoccus denitrificans exhibiting the best N2O biodegradation performance. The optimum temperature for those two species was in the range of 30–35 °C, and maximum N2O removal rates of 0.017 and 0.015 mM mg−1 dw h−1 were obtained for Pseudomonas denitrificans (initial N2O concentration of 0.9 mM) and Paracoccus denitrificans (initial N2O concentration of 1.6 mM), respectively. Miyahara et al. (2010) reported that Pseudomonas stutzeri TR2 was able to aerobically denitrify with a low production of N2O, suggesting that bioaugmentation with this strain could reduce N2O emissions from aerobic wastewater treatment systems due to its improved denitrification ability in the presence of oxygen. P. stutzeri TR2 grew at a rate of 0.32 h−1, comparable to the specific growth rates of anaerobic denitrifiers, and was classified by the authors as an expert anaerobic denitrifier.
Bioreactors for N2O abatement
The implementation of N2O control strategies is likely to become mandatory in the coming years due to the elevated contribution of this GHG to global warming and ozone depletion. To date, only physical/chemical technologies such as thermal decomposition (Löffler et al. 2002), selective catalytic reduction (Muramatsu et al. 1997; Satsuma et al. 2000), or adsorption (Golden et al. 2004) have been used for the removal of N2O from off-gases. The application of these abatement methods usually involves the consumption of expensive and/or hazardous chemicals, generation of secondary pollution, and high operating costs when treating high flow rates at the typical low N2O concentrations. Conversely, biotechnologies offer a more environmentally friendly and cost-effective platform for the removal of N2O based on their lower energy requirement, absence of hazardous chemicals or catalyst, and innocuous nature of their final end products (N2 and biomass) (Devinny et al. 1999; Boswell 2001).
Although the number of studies on the application of biological reactors for the removal of N2O is scarce, their potential has been demonstrated. For instance, Desloover et al. (2011) recently assessed the performance of a bioelectrochemical system with a cation exchange membrane separating the biological anodic and cathodic compartments. In this system, the electrons originated in the anode from sodium acetate biodegradation were supplied to the denitrifying bacteria present in the biocathode and promoted the reduction of N2O to N2 according to the equation: N2O + 2e − + 2H+ → N2 + H2O (E 0 = +1.36 V). N2O removal rates up to 1 kgN2O Nm−3 net cathodic compartment day−1 were achieved, with a cathodic coulombic efficiency of ≈100 %. However, although this technology constitutes a promising alternative due to its cost efficiency, challenges regarding its scale-up and sensitivity to aerobic environments still need to be solved (Desloover et al. 2011). Conventional biotechnologies for off-gas treatment such as biofiltration, biotrickling filtration, or bioscrubbing have been rarely applied for N2O abatement, with most of the studies reported for NO removal based on autotrophic nitrification and heterotrophic denitrification (Shanmugasundram et al. 1993; Nascimento et al. 2000; Chou and Lin 2000; Woertz et al. 2001). For instance, Utami et al. (2012) evaluated the performance of a biofilter packed with a cow manure-based compost batchwise for 9 h at different operating conditions, with the highest N2O REs (61 %) obtained at an EBRT of 58 min and a moisture content of 50 %. Likewise, Hood (2011) recorded N2O REs ranging from 14 to 17 % in a biofilter packed with compost and woodchips (30/70 %) at an EBRT of 7.6 s and N2O inlet concentrations of 0.68–1.24 mg m−3. Lower N2O removal efficiencies (0.7 %) were supported by a biofilter packed with pine nuggets and lava rock operated at an EBRT of 5 s, a relative humidity of 90 %, and inlet N2O concentrations of 428 ± 22.2 ppbv (Akdeniz et al. 2011). The low performance recorded in these biofilters was probably due to the lack of a specific microbial community acclimated to N2O biodegradation under aerobic conditions, since these systems were not specifically designed for the abatement of N2O. Besides, the presence of hydrogen sulfide in some of these air emissions might had promoted the precipitation of metals such as copper, whose presence is mandatory for the synthesis of the enzyme NOS and the correct functioning of N2O-degrading communities. In brief, the high sensitivity of the biological N2O reduction step towards the presence of O2, which is inherent in most N2O emissions, and the lack of economic or legislative incentive for its removal to date have hindered the development of high performance biological N2O abatement technologies (Desloover et al. 2012). Nevertheless, the few studies addressing the biological removal of N2O have shown promising results but require further process optimization.
Biological CO2 removal
Microbiology of CO2 removal
Biological CO2 fixation is carried out via photosynthesis by all terrestrial plants, although microorganisms such as microalgae and cyanobacteria can do it at higher rates and with 10–50 times better efficiency (Costa et al. 2000). Among microalgae, Chlorophyta (including genera such as Chlamydomonas, Chlorella, Dunaliella, Haematococcus, Botryococcus, or Scenedesmus), Euglenophyta (Euglena), Bacillariophyta (Nitzschia), Rhodophyta (Porphyridium), and Eustigmatophyta (Nannochloropsis) rank among the most investigated for biological CO2 fixation (Richmond 2004; Madigan et al. 2006; Barsanti and Gualtieri 2006). Despite constituting a phylum of bacteria rather distant taxonomically, cyanobacteria or blue–green algae are frequently considered as microalgae because of their ability to carry out photosynthesis. Cyanobacteria group includes genera such as Nostoc, Oscillatoria, Synechococcus, Synechocystis, Spirulina, Arthrospira, Anabaena, or Pediastrum. Both types of photosynthetic microorganisms, from now on referred to as microalgae, are ubiquitous in terrestrial and aquatic environments and can survive in extreme environments such as geysers and hot water springs, desert soils, and Antarctic locations (Madigan et al. 2006; Kumar et al. 2011).
Microalgae use light as the sole source of energy and CO2 as the sole carbon source during photosynthesis. Nevertheless, some species such as Chlorella vulgaris, Dunaliella salina, Euglena gracilis, and Tetraselmis tetrathele also exhibit a concomitant heterotrophic metabolism capable of utilizing from simple organic substrates like acetate, glucose, glutamate, lactate, or amino acids (Gladue and Maxey 1994, Ogbonna and Tanaka 1998) to complex aromatics such as cresols or naphthalene (Semple et al. 1999). Microalgae photosynthesis involves a redox process considered as oxygenic when H2O acts as the electron donor:
At least 8 mol of photons is required to obtain a mole of CH2O, with 218 KJ energy mol−1 photons (Ho et al. 2011). CO2 assimilation during microalgal photosynthesis involves two stages: light reactions where H2O oxidation to O2 results in the production of ATP and NADPH and dark reactions where CO2 is reduced to organic compounds utilizing NADP+/NAD+ as the electron acceptor.
Light reactions are initiated with light harvesting by the antenna complexes of the thylakoid membranes (Staehelin 1986; Taiz and Zeiger 2002). The excitation energy is then conveyed to the reaction center, where pigments such as chlorophyll a absorb a few photons per second with a conversion efficiency of 25–27 % (Taiz and Zeiger 2002). These photons mediate the photolysis of H2O with the subsequent reduction of chlorophyll P680 from the photosystem II (PSII) (Iverson 2006), the electron transport continuing through quinones, cytochromes, and plastocyanin, finally reaching photosystem I (PSI) and reducing the P700 pigment (Hill and Bendall 1960; Cerveny et al. 2009). Simultaneously, protons are pumped from the thylakoid lumen in order to obtain ATP (Fig. 4).
Carbon and electron fluxes during photosynthesis in green microalgae (upper figure) and in cyanobacteria (lower figure). The ATP and NADPH produced during the electron transport in the light reactions are used later on for CO2 reduction in the Calvin cycle. The term “electron transporters” include several components such as quinones, the cytochrome system, or plastocyanin. R RuBisCo, P phosphoribulokinase, CA carbonic anhydrase, PSII photosystem II, PSI photosystem I
Dark reactions, also known as Calvin cycle, imply CO2 fixation and, consequently, the use of the NAD(P)H and ATP obtained in the light reactions (Calvin 1989; Taiz and Zeiger 2002; Nelson and Cox 2005). The Calvin cycle includes three basic stages: carboxylation of the CO2 acceptor ribulose-1,5-bisphosphate to 3-phosphoglycerate by the enzyme ribulose bisphosphate carboxylase (RuBisCo), reduction of 3-phosphoglycerate to glyceraldehydes-3-phosphate utilizing the obtained ATP and NADPH, and regeneration of the CO2 acceptor through a phosphorylation mediated by the enzyme phosphoribulokinase (Fig. 4). Twelve NAD(P)H molecules and 18 ATP molecules are needed to produce a molecule of fructose-6-phosphate from 6 molecules of CO2 using the RuBisCo (Hall and Rao 1999; Taiz and Zeiger 2002, Madigan et al. 2006):
RuBisCo also exhibits an oxygenase activity and O2 can compete with CO2 as substrate, resulting in a process named photorespiration (Miziorko and Lorimer 1983; Edwards and Walker 2004). Photorespiration is favored at high O2/CO2 ratios, high temperatures, or high irradiations and can cause a decrease of up to 30–50 % in the photosynthetic efficiency (PE) of microalgae, with the subsequent reduction in CO2 assimilation (Zhu et al. 2008; Gioardano et al. 2005). The enzyme RuBisCo in microalgae exhibits a low affinity for CO2, since typical C3 plants present K M values in the range of 15–25 μM, green algae over 30 μM, and cyanobacteria up to 200 μM (Moroney and Somanchi 1999; Savir et al. 2010). Due to the low affinity of some microalgal species for CO2, most microalgae and cyanobacteria present specific organelles hosting key enzymes such as RuBisCo and inorganic carbon-concentrating mechanisms (CCMs) (Matsuda et al. 1998; Raven et al. 2008). CCMs consist of ATP-driven plasma membrane pumps induced by low levels of dissolved CO2. Inorganic carbon-accumulating pumps (CO2 and HCO3 −) increase the CO2 levels in the vicinity of RuBisCo and consequently reduce the extent of photorespiration, even at atmospheric CO2 levels (0.035 %) (Fig. 4) (Moroney and Somanchi 1999). By using these CCMs, microalgae can concentrate HCO3 − more than 20-folds over ambient CO2 levels and cyanobacteria more than 100-folds (Miller et al. 1990). The incorporation of CO2 into the Calvin cycle requires the conversion of HCO3 − to CO2 by a carbonic anhydrase, whose level of expression is also governed by CO2 concentrations (Price and Badger 1989; Price et al. 1992; Karlsson et al. 1998).
The rates of photosynthetic CO2 assimilation depend on factors such as the temperature, pH, light intensity, O2 and CO2 levels, or presence of inhibitory compounds. Optimal temperatures for microalgae growth and photosynthesis vary from 15 to 25 °C (Tamiya 1957). Despite higher temperatures inducing a reduced photosynthesis due to a decrease in the solubility of CO2, some microalgae species such as Chlorella have been reported to grow optimally in the range of 30–35 °C, which are typically encountered in outdoor cultivations (Pulz 2001). In this context, the isolation of thermophilic species is mandatory in order to implement microalgae-based CO2 capture methods in power plants, whose exhaust gas emissions are discharged at high temperatures. For instance, Synechococcus elongatus was able to fix CO2 at 60 % v/v and 52 °C at rates comparable to those recorded at 20 % v/v and 25 °C (Miyairi 1995). Likewise, thermophilic species of Chlorogloeopsis were able to fix CO2 at 50 °C (Ono and Cuello 2007). Microalgae and cyanobacteria preferentially grow at a neutral pH, although species like Spirulina platensis exhibit an optimum pH of 9 and Chlorococcum littorale a pH of 4 (Kodama et al. 1993; Hu et al. 1998). The effect of pH on microalgae growth is complex since it is difficult to dissociate the direct effects on microalgae growth from collateral effects such as the modification in the CO2/HCO3 −/CO3 2− and NH3/NH4 + equilibria or in phosphorus or heavy metal availability (Muñoz and Guieysse 2006). The photosynthetic activity increases linearly with increasing light intensities up to 400 μmol photon m−2 s−1, although some species such as Scenedesmus or Chlorella exhibit saturation thresholds of 100–200 μmol photon m−2 s−1 (≈5–10 % sunlight) (Hanagata et al. 1992; Muñoz and Guieysse 2006; Tredici 2009). In this context, S. platensis was able to withstand light energy fluxes of up to 8,000 μmol photon m−2 s−1 at a culture density of 8.4 ± 1.6 g L−1 under optimum mixing (Hu et al. 1996). Despite the fact that high light intensities can cause photoinhibition over PSII, microalgae strategies such as the reduction in the pigment content, PSII inactivation, or growth under heterotrophic or mixotrophic conditions can minimize its pernicious effects (Behrenfeld et al. 1998; Melis 1999; Ogbonna and Tanaka 2000; Carlsson et al. 2007). Indeed, genetic modifications of microalgae have emerged as a promising tool to reduce the size of the antenna complexes to minimum values of 37 chlorophyll molecules for PSII and 95 for PSI, which can increase photosynthetic efficiencies by 50 % (Nakajima and Ueda 1997; Mitra and Melis 2008; Eriksen 2008). Despite the high solar energy fluxes impinging on the earth surface, microalgae cultivation is often limited by light supply. Hence, considering the sole absorption of the photosynthetically active radiation from the total impinging solar radiation and the energy losses due to reflection, respiration, photosaturation, and photoinhibition, the maximum PE in microalgal culture often decreases from the expected 12.4 % to values of ≈5 % or even lower (Tredici 2009). Microalgae and cyanobacteria are able to efficiently grow at CO2 atmospheric levels (≈0.0387 % v/v) but are frequently cultivated at CO2 levels in the range of 5–15 % v/v in order to increase microalgal productivities (Kumar et al. 2010a). Microalgae tolerance to CO2 concentrations is species-dependent with optimum ranges of 2–15 % v/v for S. platensis and 15 % for Nannochloropsis salina (Doucha et al. 2005; Kumar et al. 2010b). Several strains of the thermophilic red alga Cyanidium caldarium were even capable of growing at CO2 levels of 100 % (Seckbach and Ikan 1972). On the other hand, high dissolved O2 concentrations (>35 mg L−1) in the cultivation broth favor photorespiration and O2 radical formation, with the subsequent decrease in microalgal productivity (Pulz 2001; Carvalho et al. 2006; Ho et al. 2011). Finally, heavy metals, NH3, or industrial gases such as SO x and NO x can inhibit microalgae growth. In the context of CO2 capture from flue gases, NO x is not as toxic as SO x towards microalgae and can be even used as nitrogen source. Thus, several Nannochloropsis species can grow at 100 ppmv of NO, while Dunaliella tertiolecta can withstand NO x concentrations of up to 1,000 ppmv (Yoshihara et al. 1996; Nagase et al. 1998). In contrast, high levels of SO x (>400 ppmv) severely reduce the cultivation pH and the photosynthetic rate (Packer 2009; Kumar et al. 2010b). In this regard, Tetraselmis species can grow in flue gases containing 14 % v/v CO2, 185 ppmv of SO x , and 125 ppmv of NO x (Matsumoto et al. 1995).
The ability of C. vulgaris, Botryococcus braunii, C. littorale, Scenedesmus sp., Chlamydomonas reinhardtii, and Spirulina sp. for CO2 capture was recently assessed based on their high CO2 fixation rates and ability to yield high added-value products (de Morais and Costa 2007a, b, c; Packer 2009; Ota et al. 2009; Chen et al. 2010; Yoo et al. 2010). Moreover, typical maximum specific growth rates (μ max) of microalgae under photosynthetic, heterotrophic, and mixotrophic conditions are 0.110, 0.098, and 0.198 h−1 for C. vulgaris and 0.061, 0.040, and 0.048 h−1 for Scenedesmus acutus, respectively (Ogawa and Aiba 1981).
Microalgae CO2 capture can also be coupled with the production of high added-value biotechnological products, which would significantly improve the process economy. Thus, health-promoting molecules from Chlorella species (Richmond 1990; Gouveia et al. 1996), β-carotenes from D. salina (Metting 1996; Ben-Amotz 1999), pharmaceuticals and phycobiliproteins from S. platensis (Spolaore et al. 2006; Raja et al. 2008), ketocarotenoid astaxanthins from Haematococcus pluvialis (Lee and Ding 1994; Spolaore et al. 2006), carotenes or cosmetics from Arthrospira (Richmond 1986; Viskari and Colyer 2003; Spolaore et al. 2006), and eicosapentaenoic acid from Nannochloropsis species (Boussiba et al. 1987; Chisti 2007; Chen et al. 2010) rank among the most common high added-value products marketed nowadays and can significantly contribute to the economic viability of microalgae-based CO2 capture technologies.
Photobioreactors for CO2 abatement
Photobioreactors (PBRs) are considered “direct” CO2 capture methods where a CO2-rich off-gas is directly pumped into the cultivation medium of the bioreactor in contrast to higher plant biomass systems based on CO2 diffusion (Benemann 1997). The first approach to an engineered microalgae cultivation system dates back to the World War II, when Germany looked for innovative protein sources to replace animal proteins during shortage periods (Carvalho et al. 2006). The concept was further developed during the 1950s and 1960s, but the focus shifted from protein production towards bioenergy production during the first energy crisis in the early 1970s (Chaumont 1993). Today, algal mass cultivation for bioenergy production is still perceived as economically nonsustainable by itself based on the high microalgae production costs (in the range of 4–70 € kg−1 microalgae depending on the PBR scale) and the relatively low cost of the energy obtained from them (Norsker et al. 2011; Acién et al. 2012a). Hence, the integration of microalgal CO2 capture with the production of high added-value products or wastewater treatment is nowadays regarded as the only alternative to make the process economically viable (Morweiser et al. 2010; Wang et al. 2008). In this context, recent studies have also shown the possibility of recycling the culture medium after biomass harvesting and a suitable sterilization in the mass production of microalgae to minimize nutrient losses (González López et al. 2013).
The most important parameters in PBR design and operation are light distribution and CO2 supply, the latter becoming even more relevant when the main goal of the system is CO2 capture (Morweiser et al. 2010). Scale-up of PBRs is particularly difficult due to the self-shading of cells, which causes the nonhomogeneous distribution of light, and increased mixing rates are sometimes not applicable due to the sensitivity of some photosynthetic strains to shear stress (García Camacho et al. 2011). Apart from the parameters mentioned in the “Microbiology of CO2 removal” section, the control of sterile conditions, mixing, and removal of O2 (by high turbulence or stripping with inert gases) is also of paramount relevance during PBR design and operation (Pulz 2001). Moreover, PBR design can be also tailored in order to increase the illuminated area-to-volume ratio, reduce the light path, and increase the frequency of the light–dark cycles, with the subsequent increase in microalgae productivity and CO2 removal rates (Pulz 2001; Richmond 2004). In this context, the shorter the light path is, the higher the frequency of the light–dark cycles and the photosynthetic rates. These frequencies also depend on the optimal cell density (OCD) in the cultivation broth due to the increased mutual shading at increasing microalgae concentrations (Richmond 2004; Tredici 2009). Since the OCD and light path are inversely correlated, PBRs with narrow light path (i.e., 1–2 cm) and high irradiances can support high-density cultures (Zou et al. 2000). The combination of both environmental and operational parameters, together with the selection of the optimum microbial strain, determines the CO2 capture potential, which can be estimated from microalgae productivities by applying the stoichiometric CO2 requirement factor for microalgae growth of 1.85 g CO2 g−1 biomass (Table 3) (Carvalho et al. 2006; Posten 2009). The main PBR configurations will be discussed following the traditional classification into open and enclosed systems.
Open photobioreactors
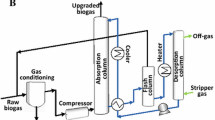
Open ponds are the simplest and cheapest technology for microalgae cultivation and can be classified into natural water bodies (lakes, lagoons, or ponds) or artificial ponds (Singh and Sharma 2012). Of them, raceway ponds, also named as high rate algal ponds (HRAP), are the most applied configuration due to their higher microalgal productivities (compared to other open alternatives) at a reasonable energy cost (200 MJ ha−1 day−1 or 0.03–0.2 W m−3 for mixing) (Tredici 2009). In these HRAPs, the microalgae broth is continuously recirculated around a closed loop channel using a rotating paddlewheel (the most cost-effective mixing mode), while CO2 (or air) can be injected at different locations of the PBR via CO2 diffusion pumps or external CO2 bubble columns (Fig. 5a) (Xu et al. 2009; Muñoz et al. 2012). The main advantages of HRAPs are their simplicity of construction and operation and their lower investment (in the range of 2.3–20 € m−2) and operating costs compared to enclosed PBRs (Lundquist et al. 2010; Craggs et al. 2012). However, open configurations present critical disadvantages inherent to their design such as a high vulnerability to external contamination with undesired wild microalgae species or predators, a poor light utilization as a result of the low light–dark frequencies supported by the hydrodynamics of the ponds, a high water footprint by evaporation (up to 40 L m−2 day−1 in desertic climates) (Massey University 2011, large land requirements, and low CO2 transfer efficiencies as a result of the technical limitations for CO2 supply (Xu et al. 2009). Overall, open ponds support lower biomass productivities than enclosed PBRs (Table 3) and, therefore, a lower potential for CO2 capture (Ugwu et al. 2008). Nevertheless, despite the previously mentioned limitations, open ponds still hold a promising potential for CO2 capture in particular applications such as biogas upgrading (the process of enriching biogas in CH4 by removing CO2 and H2S). For instance, Muñoz et al. (2012) achieved removal efficiencies of 90 % for CO2 and 100 % for H2S in a HRAP coupled with an independent bubble absorption column (60 cm depth) where the microalgae broth was continuously recycled. Likewise, Mandeno et al. (2005) achieved reductions in the CO2 content of biogas from 40 to 50 % by sparging the biogas into a HRAP in a countercurrent pit to improve CO2 mass transfer (Mandeno et al. 2005). Despite presenting a lower potential for CO2 fixation (up to 0.20 g CO2 L−1 day−1) than their enclosed counterparts, HRAPs are the PBRs with the highest ratio of CO2 fixed/CO2 emitted (≈90.6 kg CO2 fixed kg−1 CO2 emitted, considering an energy consumption of 0.02 kWh kg−1 CO2 assimilated and the average EU-27 specific CO2 emission factor of 0.46 tonnes CO2 emitted MWh−1) (Covenant of Mayors 2012).
Enclosed bioreactors
Enclosed PBRs are more efficient at maintaining axenic microalgae cultures, allow for a better control over the process variables (temperature, pH, and CO2 concentration), and minimize both CO2 and water losses (Singh and Sharma 2012). In order to improve light penetration and minimize photoinhibition effect, enclosed PBRs present high illuminated area-to-volume ratios (30–70 m−1 in enclosed systems vs 3–10 m−1 in HRAPs). In enclosed PBRs, CO2 is supplied at high concentrations (even pure CO2 is directly supplied) by means of high-performance diffusion systems at certain locations of the PBR or directly injected in the recirculating cultivation broth to avoid inorganic carbon limitations since most PBRs are always designed considering light supply as the limiting parameter for microalgae growth (Carvalho et al. 2006). Finally, the cultivation broth must be mixed with minimum energy requirements to prevent microalgae settling and facilitate the access of microalgae cells to the photic zone of the culture, while avoiding cell damage by excessive shear stress (Carvalho et al. 2006; Posten 2009). Enclosed PBRs are classified into three major categories, depending on their configuration:
-
1.
Vertical tubular reactors consists of a vertical pipe made of transparent materials such as polyethylene or glass and where a CO2-enriched gas is sparged into fine bubbles at the bottom of the column for CO2 supply and culture mixing (Kumar et al. 2011). Typical area-to-volume ratios in these systems are in the range of 10 to 30 m−1 (Sánchez Mirón et al. 1999). If the liquid flow is essentially random, the vertical PBR is a bubble column (Fig. 5b). On the other hand, if a concentric tube is installed inside the vertical column (namely, riser) and the CO2-enriched gas is sparged inside the riser creating a recirculating flow pattern (upwards in the riser and downwards in the downcomer), the PBRs are named airlift PBRs (Fig. 5c).
Vertical tubular PBRs are compact and low-cost alternatives in terms of investment cost, and some PBRs include dome or annular configurations in order to minimize the dark zones, since the low illuminated surface area is their main drawback (Morweiser et al. 2010). The energy input required ranges from 50 to 70 W m−3 (Béchet et al. 2012). Vertical tubular PBRs provide an average potential for CO2 capture higher than open ponds, but lower than that of their horizontal counterparts. A recent study reported a maximum CO2 fixation rate of 0.97 g L−1 day−1 in a 3-L bubble column PBR using the microalga Scenedesmus obtusiusculus (Toledo-Cervantes et al. 2013), while CO2 capture rates of up to5.4 g L−1 day−1 were recorded for Aphanothece microscopica Nägeli in the same type of PBR (Jacob-Lopes et al. 2009). The high variability of these results confirms the high impact of the strain and the operational conditions on the PBR potential. Furthermore, estimated CO2 capture rates of up to 1.45 g CO2 L−1 day−1 have been achieved in column PBRs operated with the cyanobacteria Anabaena (González López et al. 2009) (Table 3). At these CO2 fixation rates in vertical tubular PBRs, the ratio of kilograms of CO2 fixed to kilograms of CO2 emitted can be estimated to be 1.9 (Covenant of Mayors 2012).
-
2.
Flat-plate PBRs are similar to the vertical tubular PBRs described previously but are designed to minimize the light path and to provide a homogeneous light penetration. Flat prisms between 3 and 10 cm deep with a surface area of ≈1–2 m2 made of a transparent material are employed, which provide the high illuminated area-to-volume ratios (20–40 m−1) needed to achieve high photosynthetic efficiencies and, therefore, high CO2 removal rates (Fig. 5d) (Tredici and Zittelli 1998; Barbosa et al. 2005). Gas sparging at the bottom is employed for both pneumatic mixing and CO2 supply, with energy consumptions similar to those of vertical tubular configurations, although alternative agitation methods consisting of panel rotation have been implemented (Kumar et al. 2011). Flat-plate PBRs are mainly illuminated on one side and can be arranged in optimal angles facing solar irradiation to maximize microalgae growth (Xu et al. 2009). Recently, new configurations such as alveolar and V-shaped panels have been implemented, resulting in increased microalgae productivities (Carvalho et al. 2006). On the other hand, flat-plate PBRs require high land areas and supporting structures when many replicate units are installed in full-scale applications (Ugwu et al. 2008). The productivities and, therefore, the CO2 capture rates in flat-plate PBRs are similar to those recorded in vertical tubular reactors (≈1 g CO2 L−1 day−1) but 10 times higher than that in HRAPs (Posten 2009; Morita et al. 2000) (Table 3). Besides, flat-plate PBRs possess ratios of 1.46 kg CO2 fixed kg−1 CO2 emitted (Covenant of Mayors 2012).
-
3.
Horizontal tubular PBRs consist of a set of horizontal transparent tubes arranged in parallel, closed loop, coiled around a hollow cylinder, or α-shape, acting as light collector (Carvalho et al. 2006; Merchuk et al. 2007; Kumar et al. 2011). They are usually equipped with a centrifugal pump or an airlift system to provide adequate flow and mixing, with the airlift systems being employed for the cultivation of shear-sensitive microalgae (Fig. 5e). Gas exchange takes place in the tubes or in especially dedicated CO2 absorption units. The high illuminated area-to-volume ratios (45–70 m−1) result in high biomass productivities (Tredici and Zittelli 1998). The large pH, CO2, and O2 gradients in long tubes and fouling have been pointed out as the major causes of process failures in industrial-scale facilities, while the need to maintain adequate liquid velocities in the tubes implies high energy consumption (≥100 W m−3). Strategies such as water spraying over the tubes, installation of heat exchangers, tube overlapping, and immersion of the tubes inside a pool are the most cost-effective alternatives for temperature control in outdoor facilities (Ugwu et al. 2008; Kumar et al. 2011; Singh and Sharma 2012). Horizontal tubular PBRs support the highest biomass productivities and, thus, CO2 capture potential, with average values higher than 2 g CO2 L−1 day−1 (Acién Fernández et al. 2001; Morita et al. 2002; Hall et al. 2003), although capture values higher than 4.5 g CO2 L−1 day−1 have been occasionally achieved in a tubular airlift bioreactor (Sobczuk et al. 2000). CO2 capture rates of ≈1 g CO2 L−1 day−1 have been recently recorded outdoors in a 220-L external loop airlift PBR with Anabaena sp. ATCC 33047 (Sánchez Fernández et al. 2012). Finally, ratios of up to 4.1 kg CO2 fixed kg−1 CO2 emitted for these PBRs have been recorded, which represent the best results so far reported among enclosed PBRs (Covenant of Mayors 2012).
Despite CO2 solubility in water being relatively high compared to that of O2, CH4, or N2 (Henry’s law constant of 3.4 × 10−2 mol L−1 atm−1), its cost-efficient supply to the microalgae broth still constitutes a large-scale technical challenge, affecting the pH and mixing in the system. Three main CO2 supply systems have been implemented in the PBR configurations previously described: independent gas exchangers, direct gas injection to the cultivation broth, and membrane-based transfer. Among them, membrane transfer deserves special attention when CO2 capture is the primary objective of the PBR since mass transfer in membrane units is no longer affected by the boundary layer between the gas and the aqueous phase, but a gas-permeable membrane allows CO2 diffusion from the gas and O2 desorption from the microalgal cultivation medium. This approach allows for reduced operating costs, since CO2 absorption is decoupled from culture mixing, which represents a degree of freedom for process optimization (Morweiser et al. 2010). Theoretically, membrane transfer avoids CO2 losses to the atmosphere and allows for an accurate control of the transfer rates. The use of hollow fiber membranes allows for operation at low pressure drops since no water column pressure has to be overcome by the CO2-laden emission. On the other hand, porous membranes provide high transfer areas but require high pressures to force the CO2 through the membrane (especially in highly salty media), the latter implying thicker membranes and high-performance materials (Carvalho et al. 2006). A recent study reported CO2 removal efficiencies of 85 % at a CO2 concentration of 2 % in a hollow fiber membrane PBR (Kumar et al. 2010a). Finally, another recently proposed strategy to efficiently capture CO2 from flue gases consists of absorbing the CO2 in an aqueous solution, which is further purified by microalgae in a PBR. Alkaline sodium carbonate and bicarbonate solutions have been proposed as suitable solutions that can be biologically regenerated as an alternative to methyl ethyl amine, diethyl amine, or NaOH, which are used as chemical absorbents and must be regenerated by heating in highly energy-demanding processes (González López et al. 2012).
Limitations in the biological treatment of GHGs and future research needs
Despite the promising potential of biotechnologies for the abatement of CH4, N2O, and CO2 emissions, their cost-efficient application is often limited by the poor mass transport of CH4, and in a lower extent of N2O and CO2, from the gas to the aqueous phase due to the low aqueous solubility of these GHGs (dimensionless Henry’s law constants, H, H CH4 = 30, H N2O = 2, and H CO2 = 1.2). Thus, these high H values result in low concentration gradients (low driving forces) for GHG mass transport from the gas to the aqueous phase containing or surrounding the microbial communities and, therefore, in a reduced GHG biodegradation performance (Muñoz et al. 2007). In the particular case of CH4 and N2O, this low mass transport entails process operation at high EBRTs, which significantly increases both the investment and operating costs of conventional biotechnologies. In PBRs, this limited mass transport restricts the implementation of a direct bubbling of the CO2-laden streams in HRAPs and increases the volume of external CO2 transfer units or in situ pumps. In this context, the development of innovative high-performance mass transfer approaches is mandatory. However, unlike the classical approach of improving mass transport by increasing the energy input to the bioreactor, the high-performance transport should be based on increased GHG concentration gradients (e.g., absence of aqueous phase, high cell hydrophobicity, high pH, or presence of a nonaqueous organic phase) and larger gas–cell interfacial areas.
Under mass transfer-limiting conditions, the poor knowledge of the GHG biodegradation kinetics at the trace level concentrations (almost equal to micrograms per liter) typically present in the aqueous phase when treating real emissions has also limited the correct design of bioreactors for GHG abatement (Estrada et al. 2012a). For instance, for typical emissions containing 10–200 mg m−3 of CH4 and 50–1,000 mg m−3 of N2O, microorganisms would be exposed to maximum aqueous concentrations ranging from 0.3 to 7 μg CH4 L−1 and from 25 to 500 μg N2O L−1, respectively (Shimizu et al. 2000; Kampschreur et al. 2008; Girard et al. 2011; Gustavsson and la Cour Jansen 2011). In this context, it must be stressed that microbial characteristics and, in particular, microbial affinity for the target pollutant quantified as K M and K S play a key role on the performance of biotechnologies treating poorly soluble gaseous pollutants (Hernández et al. 2010; Rocha-Rios et al. 2011). Despite the empirical evidence for the fact that microorganisms can mineralize pollutants down to the picograms per liter level, the number of studies evaluating pollutant biodegradation at the micrograms per liter to nanograms per liter level is unfortunately scarce (Roch and Alexander 1997). Besides, the few kinetic studies reported for methanotrophs were carried out using a methodology based on the estimation of the aqueous CH4 concentration by CH4 headspace analyses in closed systems inoculated at high biomass concentrations and under gentle agitation, conditions which do not ensure the absence of mass transfer limitations and, therefore, the validity of the experimental data (Yoon et al. 2009). As a matter of fact, while mechanistic model predictions using the available kinetic data for methanotrophs show that CH4 biodegradation would not be feasible at atmospheric concentrations (1.8 ppmv), there are empirical evidence of CH4 removal from 1.8 to 0.1 ppmv (Whalen et al. 1990). Likewise, in the particular case of N2O-degrading bacteria or photosynthetic microorganisms, the number of studies conducted to date is scarce. In light of the current limitations of microbiology, novel methodologies for the accurate determination of microbial GHG biodegradation kinetics at trace level concentrations must be developed. The quantification of microbial growth and the measurement of the GHG concentrations at the micrograms per liter level, in both the aqueous phase and the biofilm, constitute key analytical challenges.
The biological abatement of N2O is also limited by the poor understanding of the microbiology governing N2O biodegradation under aerobic conditions. The few existing studies on biological NO x treatment (mainly NO and NO2) were based on heterotrophic denitrification using O2-free synthetic emissions (conditions which are far from those typically found in real N2O-laden emissions) and autotrophic nitrification and, in most cases, were mass transfer limited (Flanagan et al. 2002). In addition, several authors have concluded that the implementation of nitrogen oxide removal based on denitrification might be limited by the need for low O2 concentrations and the use of an exogenous carbon source (Kalkowski and Conrad 1991; Sakurai and Sakurai 1997). In this regard, aerobic denitrification might constitute the cornerstone for the development of cost-efficient N2O abatement processes. Thus, despite the controversy that has surrounded aerobic denitrification for years, there are nowadays consistent research findings to support the constitutive nature of some NO3 −, NO2 −, and N2O reductases in bacteria such as P. stutzeri or Thiosphaera pantotropha (Robertson et al. 1995). As a matter of fact, N2O reduction coupled to the oxidation of an electron donor (e.g., acetate) has been reported even at O2 concentrations twice that of air saturation (Miyahara et al. 2010). However, apart from this proof of concept carried out in test tubes, aerobic denitrification has never been evaluated in a continuous bioreactor configuration, which constitutes a technological challenge in terms of electron donor supply and competition with obligate aerobic heterotrophs.
Finally, microalgae-based CO2 capture is also limited by the low PE achieved in conventional PBRs (2–5 %) during outdoors cultivation (which significantly increases both investment and operation costs) and also by the perishable nature of microalgal biomass, which does not allow for the long-term storage of CO2 (Acién Fernández et al. 2012b). In this context, the development of both innovative PBRs capable of minimizing the detrimental effects of photorespiration, photoinhibition, photosaturation, and temperature on PE and of low-cost microalgae harvesting technologies is crucial. Today, CO2 capture using microalgae-based processes still exhibit high operating costs, despite being lower than those of their physical/chemical counterparts. In a recent literature review published by Acién Fernández et al. (2012b), the cost of CO2 capture in a 100-ha facility of HRAP was estimated at 0.23 € kg−1CO2 by using wastewater as free nutrient source and flue gases as free CO2 source. Microalgae harvesting constitutes a significant part of the overall operating costs and is often carried out by centrifugation, filtration, coagulation–flocculation, flotation, or gravity sedimentation (Packer 2009). Full-scale algal mass production plants are often constructed with centrifugation or filtration units, despite not being recommended for small species like Chlorella or Scenedesmus (Molina Grima et al. 2003). In this context, Acién Fernández et al. (2012b) estimated the energy requirements associated to microalgae harvesting by flocculation–sedimentation followed by centrifugation in 0.1 kWh m−3. In brief, most of the CO2 mitigation of microalgae-based processes will actually come from the production of bioenergy from the biomass produced (with the subsequent reduction in fossil fuel consumption) and from the energy-efficient carbon and nutrients removal during wastewater treatment in PBRs (Muñoz and Guieysse 2006; Kumar et al. 2010b; Acién Fernández et al. 2012b).
Conclusions
To date, biotechnologies for GHG abatement have shown promising results in terms of elimination capacity and environmental impact. The prohibitive treatment costs and the CO2 footprint of physical/chemical techniques, especially when dealing with large volumes of air with low GHG concentration, certainly encourage the development and application of high-performance biological methods to mitigate this global environmental problem. Despite the fact that some technical and microbiological limitations must still be overcome, the potential of biological techniques for GHG abatement has been consistently demonstrated, high removal efficiencies being so far reached mainly for CH4 and CO2 and in a lower extent for N2O. In this context, the understanding of the microbiology underlying GHG biodegradation was identified as one of the most important knowledge gaps requiring further research. In addition, the development of innovative bioreactor configurations to improve both GHG mass transfer from the gas phase to the microorganisms and light supply (in the case of CO2 abatement processes) constitutes a key research niche. Finally, the feasibility of coupling GHG biodegradation with the production of high added-value products such as PHB, single-cell protein, pharmaceuticals, or biofuels can impact positively on the overall cost-effectiveness of biological treatment processes.
References
Acién Fernández FG, Fernández Sevilla JM, Sánchez Pérez JA, Molina Grima E, Chisti Y (2001) Airlift-driven external-loop tubular photobioreactors for outdoor production of microalgae: assessment of design and performance. Chem Eng Sci 56:2721–2732. doi:10.1016/S0009-2509(00)00521-2
Acién FG, Fernández JM, Magán JJ, Molina E (2012) Production cost of a real microalgae production plant and strategies to reduce it. Biotechnol Adv 30:1344–1353. doi:10.1016/j.biotechadv.2012.02.005
Acién Fernández FG, González-López CV, Fernández Sevilla JM, Molina Grima E (2012) Conversion of CO2 into biomass by microalgae: how realistic a contribution may it be to significant CO2 removal? Appl Microbiol Biotechnol 96:577–586. doi:10.1007/s00253-012-4362-z
Ahn JH, Kim S, Park H, Rahm B, Pagilla K, Chandran K (2010) N2O emissions from activated sludge processes, 2008–2009: results of a national monitoring survey in the United States. Environ Sci Technol 44:4505–4511. doi:10.1021/es903845y
Akdeniz N, Janni KA, Salnikov IA (2011) Biofilter performance of pine nuggets and lava rock as media. Biores Technol 102:4974–4980. doi:10.1016/j.biortech.2011.01.058
Amaral JA, Knowles R (1995) Growth of methanotrophs in methane and oxygen counter gradients. FEMS Microbiol Lett 126:215–220. doi:10.1111/j.1574-6968.1995.tb07421.x
Anthony C (1982) The biochemistry of methylotrophs. Academic, New York
Apel WA, Turick CE (1992) The use of denitrifying bacteria for the removal of nitrogen oxides from combustion gases. Fuel 72:1715–1718. doi:10.1016/0016-2361(93)90360-E
Arcangeli JP, Arvin E (1999) Modeling the growth of a methanotrophic biofilm: estimation of parameters and viability. Biodegrad 10:177–191
Avalos A, Jones PJ, Heitz M (2012) Methane treatment in biotrickling filters packed with inert materials in presence of a non-ionic surfactant. J Chem Technol Biotechnol 87:848–853. doi:10.1002/jctb.3811
Balasubramanian R, Kenney GE, Rosenzweig AC (2011) Dual pathways for copper uptake by methanotrophic bacteria. J Biol Chem 286:37313–37319. doi:10.1074/jbc.M111.284984
Barbosa MJ, Zijffers JW, Nisworo A, Vaes W, van Schoonhoven J, Wijffels RH (2005) Optimization of biomass, vitamins, and carotenoid yield on light energy in a flat-panel reactor using the A-stat technique. Biotechnol Bioeng 89:233–242. doi:10.1002/bit.20346
Barsanti L, Gualtieri P (2006) Algae: anatomy, biochemistry and biotechnology. CRC, Boca Raton
Basu P, Katterle B, Andersson KK, Dalton H (2003) The membrane-associated form of methane mono-oxygenase from Methylococcus capsulatus (Bath) is a copper/iron protein. Biochem J 369:417–429. doi:10.1042/BJ20020823
Beal EJ, House CH, Orphan VJ (2009) Manganese- and iron-dependent marine methane oxidation. Science 325:184–187. doi:10.1126/science.1169984
Béchet Q, Muñoz R, Shilton A, Guieysse B (2012) Outdoor cultivation of temperature-tolerant Chlorella sorokiniana in a column photobioreactor under low power-input. Biotechnol Bioeng n/a-n/a. doi:10.1002/bit.24603
Bédard C, Knowles R (1989) Physiology, biochemistry and specific inhibitors of CH4, NH4 and CO oxidation by methanotrophs and nitrifiers. Microbiol Rev 53:68–84
Behrenfeld M, Prasil O, Kolber Z, Babin M, Falkowski P (1998) Compensatory changes in photosystem II electron turnover rates protect photosynthesis from photoinhibition. Photosynth Res 58:259–268. doi:10.1023/A:1006138630573
Ben-Amotz A (1999) Production of β-carotene in Dunaliella. In: Cohen Z (ed) Chemicals from microalgae. Taylor and Francis, London, pp 196–204
Bender M, Conrad R (1992) Kinetics of CH4 oxidation in oxic soils exposed to ambient air or high CH4 mixing ratios. FEMS Microbiol Ecol 101:261–270
Bender M, Conrad R (1994) Methane oxidation activity in various soils and freshwater sediments: occurrence, characteristics, vertical profiles, and distribution on grain size fractions. J Geophys Res 99:16531–16540
Bender M, Conrad R (1995) Effect of CH4 concentrations and soil conditions on the induction of CH4 oxidation activity. Soil Biol Biochem 27:1517–1527. doi:10.1016/0038-0717(95)00104-M
Benemann JR (1997) CO2 mitigation with microalgae systems. Energy Convers Manag 38:S475–S479. doi:10.1016/S0196-8904(96)00313-5
Berger J, Fornés LV, Ott C, Jager J, Wawra B, Zanke U (2005) Methane oxidation in a landfill cover with capillary barrier. Waste Manag 25:369–373. doi:10.1016/j.wasman.2005.02.005
Bodrossy L, Kovács KL, McDonald IR, Murrell JC (1999) A novel thermophilic methane-oxidizing γ-Proteobacterium. FEMS Microbiol Lett 170:335–341. doi:10.1016/S0378-1097(98)00552-7
Börjesson G (1997) Methane oxidation in landfill cover soils. Doctoral thesis, Swedish University of Agricultural Sciences, Uppsala, Sweden
Börjesson G, Sundh I, Tunlid A, Svensson BH (1998) Methane oxidation in landfill cover soils as revealed by potential oxidation measurements and phospholipid fatty acid analyses. Soil Biol Biochem 30:1423–1433. doi:10.1016/S0038-0717(97)00257-5
Born M, Dörr H, Levin I (1990) Methane consumption in aerated soils of the temperate zone. Tellus 42:2–8. doi:10.1034/j.1600-0889.1990.00002.x
Boswell J (2001) Understand the capabilities of bio-oxidation. Chem Eng Prog 98:48–53
Boussiba S, Vonshak A, Cohen Z, Avissar I, Richmond A (1987) Lipid and biomass production by the halotolerant microalga Nannochloropsis salina. Biomass 12:37–47. doi:10.1016/0144-4565(87)90006-0
Bowman JP, McCammon SA, Skerratt JH (1997) Methylosphaera hansonii gen. nov., sp. nov., a psychrophilic, group I methanotroph from Antarctic marine-salinity, meromictic lakes. Microbiol 143:1451–1459. doi:10.1099/00221287-143-4-1451
Bowman JP (2006) The methanotrophs—the families Methylococcaceae and Methylocystaceae Ch. 3.1.14. Prokaryotes 5:266–289. doi:10.1007/0-387-30745-1_15
Bratina BJ, Brusseau GA, Hanson RS (1992) Use of 16S rRNA analysis to investigate phylogeny of methylotrophic bacteria. Int J Syst Bacteriol 42:645–648. doi:10.1099/00207713-42-4-645
Brusseau GA, Bulygina E, Hanson RS (1994) Phylogenetic analysis and development of probes for differentiating methylotrophic bacteria. Appl Environ Microbiol 60:626–636
Buchholz LA, Valklump J, Collins MLP, Brantner CA, Remsen CC (1995) Activity of methanotrophic bacteria in Green-Bay sediments. FEMS Microbiol Ecol 16:1–8. doi:10.1111/j.1574-6941.1995.tb00262.x
Calvin M (1989) Forty years of photosynthesis and related activities. Photosynth Res 21:3–16. doi:10.1007/BF00047170
Carlsson AS, van Beilen JB, Möller R, Clayton D (2007) Micro and macro-algae: utility for industrial applications. CPL, Newbury
Carvalho AP, Meireles LA, Malcata FX (2006) Microalgal reactors: a review of enclosed system designs and performances. Biotechnol Progr 22:1490–1506. doi:10.1021/bp060065r
Cerveny J, Setlik I, Trtilek M, Nedbal L (2009) Photobioreactor for cultivation and real-time, in situ measurement of O2 and CO2 exchange rates, growth dynamics, and of chlorophyll fluorescence emission of photoautotrophic microorganisms. Eng Life Sci 9:247–253. doi:10.1002/elsc.200800123
Chaumont D (1993) Biotechnology of algal biomass production: a review of systems for outdoor mass culture. J Appl Phycol 5:593–604. doi:10.1007/BF02184638
Chen CY, Yeh KL, Su HM, Lo YC, Chen WM, Chang JS (2010) Strategies to enhance cell growth and achieve high-level oil production of a Chlorella vulgaris isolate. Biotechnol Prog 26:679–686. doi:10.1002/btpr.381
Chida K, Shen G, Kodoma T, Minoda Y (1983) Acidic polysaccharide production from methane by a new methane-oxidizing bacterium H-2. Agric Biol Chem 47:275–280
Chini G, Rodolfi L, Biondi N, Tredici MR (2006) Productivity and photosynthetic efficiency of outdoor cultures of Tetraselmis suecica in annular columns. Aquacult 261:932–943. doi:10.1016/j.aquaculture.2006.08.011
Chisti Y (2007) Biodiesel from microalgae. Biotechnol Adv 25:294–306. doi:10.1016/j.biotechadv.2007.02.001
Chistoserdova L (2011) Modularity of methylotrophy, revisited. Environ Microbiol 13:2603–2622. doi:10.1111/j.1462-2920.2011.02464.x
Choi DW, Kunz RC, Boyd ES, Semrau JD, Antholine WE, Han JI, Zahn JA, Boyd JM, De La Mora AM, DiSpirito AA (2003) The membrane-associated methane monooxygenase (pMMO) and pMMO-NADH: quinone oxidoreductase complex from Methylococcus capsulatus Bath. J Bacteriol 185:5755–5764. doi:10.1128/JB.185.19.5755-5764.2003
Choi DW, Antholine WA, Do YS, Semrau JD, Kisting CJ, Kunz RC, Campbell D, Rao V, Hartsel SC, DiSpirito AA (2005) Effect of methanobactin on methane oxidation by the membrane-associated methane monooxygenase in Methylococcus capsulatus Bath. Microbiol 151:3417–3426. doi:10.1099/mic.0.28169-0
Chou MS, Lin JH (2000) Biotrickling filtration of nitric oxide. J Air Waste Manage Assoc 50:502–508. doi:10.1080/10473289.2000.10464033
Christophersen M, Linderød L, Jensen PE, Kjeldsen P (2000) Methane oxidation at low temperatures in soil exposed to landfill gas. J Environ Qual 29:1989–1997
Colliver BB, Stephenson T (2000) Production of nitrogen oxide and dinitrogen oxide by autotrophic nitrifiers. Biotechnol Adv 18:219–232. doi:10.1016/S0734-9750(00)00035-5
Costa JAV, Linde GA, Atala DIP, Mibielli GM, Krüger RT (2000) Modeling of growth conditions for cyanobacterium Spirulina platensis in microcosms. World J Microbiol Biotechnol 16:15–18. doi:10.1023/A:1008992826344
Covenant of Mayors (2012) Technical annex to the SEAP template instructions document: the emission factors. Available at http://www.covenantofmayors.eu/support/faq_en.html?id_faq=32. Accessed 11 January 2013
Craggs R, Sutherland D, Campbell H (2012) Hectare-scale demonstration of high rate algal ponds for enhanced wastewater treatment and biofuel production. J Appl Phycol 24:329–337. doi:10.1007/s10811-012-9810-8
Dalton H, Prior SD, Leak DJ, Stanley SH (1984) Regulation and control of methane monooxygenase. In: Crawford RL, Hanson RS (eds) Microbial growth on C1 compounds. American Society for Microbiology, Washington, pp 75–82
Dalton H (1992) Methane oxidation by methanotrophs: physiological and mechanistic implications. In: Murrell JC, Dalton H (eds) Methane and methane utilizers. Plenum, New York, pp 85–114
Davies SL, Whittenbury R (1970) Fine structure of methane and other hydrocarbon oxidizing bacteria. J Gen Microbiol 61:227–232. doi:10.1099/00221287-61-2-227
de Godos I, Blanco S, García-Encina PA, Becares E, Muñoz R (2009) Long-term operation of high rate algal ponds for the bioremediation of piggery wastewaters at high loading rates. Biores Technol 100:4332–4339. doi:10.1016/j.biortech.2009.04.016
de Morais MG, Costa JAV (2007a) Biofixation of carbon dioxide by Spirulina sp. and Scenedesmus obliquus cultivated in a three-stage serial tubular photobioreactor. J Biotechnol 129:439–445. doi:10.1016/j.jbiotec.2007.01.009
de Morais MG, Costa JAV (2007b) Carbon dioxide fixation by Chlorella kessleri, C. vulgaris, Scenedesmus obliquus and Spirulina sp. cultivated in flasks and vertical tubular photobioreactors. Biotechnol Lett 29:1349–1352. doi:10.1007/s10529-007-9394-6
de Morais MG, Costa JAV (2007c) Isolation and selection of microalgae from coal fired thermoelectric power plant for biofixation of carbon dioxide. Energy Convers Manage 48:2169–2173. doi:10.1016/j.enconman.2006.12.011
Dedysh SN, Panikov NS, Tiedje JM (1998) Acidophilic methanotrophic communities from Sphagnum peat bogs. Appl Environ Microbiol 64:922–929
Dedysh SN, Khmelenina VN, Suzina NE, Trotsenko YA, Semrau JD, Liesack W, Tiedje JM (2002) Methylocapsa acidiphila gen. nov., sp. nov., a novel methane-oxidizing and dinitrogen-fixing acidophilic bacterium from Sphagnum bog. Int J Syst Evol Microbiol 52:251–261
Delhoménie MC, Nikiema J, Bibeau L, Heitz M (2009) A new method to determine the microbial kinetic parameters in biological air filters. Chem Eng Sci 63:4126–4136. doi:10.1016/j.ces.2008.05.020
Desloover J, Puig S, Virdis B, Clauwaert P, Boeckx P, Verstraete W, Book N (2011) Biocathodic nitrous oxide removal in bioelectrochemical systems. Environ Sci Technol 45:10557–10566. doi:10.1021/es202047x
Desloover J, Vlaeminck SE, Clauwaert P, Verstraete W, Boon N (2012) Strategies to mitigate N2O emissions from biological nitrogen removal systems. Curr Opin Biotechnol 23:474–482. doi:10.1016/j.copbio.2011.12.030
Devinny JS, Deshusses MA, Webster TS (1999) Biofiltration for air pollution control. Lewis, Boca Raton
Doucha J, Straka F, Livansky K (2005) Utilization of flue gas for cultivation of microalgae (Chlorella sp.) in an outdoor open thin-layer photobioreactor. J Appl Phycol 17:403–412. doi:10.1007/s10811-005-8701-7
du Plessis CA, Strauss JM, Sebapalo EMT, Riedel KHJ (2003) Empirical model for methane oxidation using a composted pine bark biofilter. Fuel 82:1359–1365. doi:10.1016/S0016-2361(03)00040-1
Dunfield P, Knowles R, Dumont R, Moore TR (1993) Methane production and consumption in temperate and subarctic peat soils: response to temperature and pH. Soil Biol Biochem 25:321–325
Dunfield PF, Yuryev A, Senin P, Smirnova AV, Stott MB, Hou S, Ly B, Saw JH, Zhou A, Ren Y, Wang J, Mountain BW, Crowe MA, Weatherby TM, Bodelier PLE, Liesack W, Feng L, Alam M (2007) Methane oxidation by an extremely acidophilic bacterium of the phylum Verrucomicrobia. Nature 450:879–883. doi:10.1038/nature06411
Edwards GE, Walker DA (2004) Photosynthetic carbon assimilation. In: Archer MD, Barber J (eds) Molecular to global photosynthesis. Imperial College Press, London, pp 189–220
Einola JKM, Karhu AE, Rintala JA (2008) Mechanically–biologically treated municipal solid waste as a support medium for microbial methane oxidation to mitigate landfill greenhouse emissions. Waste Manag 28:97–111. doi:10.1016/j.wasman.2007.01.002
Enebo L (1967) A methane-consuming green algae. Acta Chem Scand 21:625–632
Environmental Protection Agency (2011) Inventory of US greenhouse gas emissions and sinks: 1990–2009 (April 2011), EPA 430-R-11-005. Available at http://www.epa.gov/climatechange/emissions/usinventoryreport.html. Accessed 1 June 2012
Eriksen NT (2008) The technology of microalgal culturing. Biotechnol Lett 30:1525–1536. doi:10.1007/s10529-008-9740-3
Estrada J, Kraakman B, Muñoz R, Lebrero R (2011) A comparative analysis of odour treatment technologies in wastewater treatment plants. Environ Sci Technol 45:1100–1106. doi:10.1021/es103478j
Estrada J, Rodríguez E, Quijano G, Muñoz R (2012a) Influence of gaseous VOC concentration on the biodiversity and biodegradation performance of microbial communities. Bioprocess Biosyst Eng 35:1477–1488. doi:10.1007/s00449-012-0737-x
Estrada JM, Kraakman NJR, Lebrero R, Muñoz R (2012b) A sensitivity analysis of process design parameters, commodity prices and robustness on the economics of odour abatement technologies. Biotechnol Adv 30:1354–1363. doi:10.1016/j.biotechadv.2012.02.010
Ettwig KF, Butler MK, Le Paslier D, Pelletier E, Mangenot S, Kuypers MMM, Schreiber F, Dutilh BE, Zedelius J, De Beer D, Gloerich J, Wessels HJ, van Alen T, Luesken F, Wu ML, van de Pas-Schoonen KT, Op den Camp HJ, Janssen-Megens EM, Francoijs KJ, Stunnenberg H, Weissenbach J, Jetten MS, Strous M (2010) Nitrite-driven anaerobic methane oxidation by oxygenic bacteria. Nature 464:543–548. doi:10.1038/nature08883
European Environment Agency (2011) Annual European Union greenhouse gas inventory 1990–2009 and inventory report 2011. Available at http://www.eea.europa.eu/publications/european-union-greenhouse-gas-inventory-2011. Accessed 1 June 2012
Figueroa RA (1993) Methane oxidation in landfill top soils. In: Christensen TH, Cossu R, Stegmann R (eds) Proceedings Sardinia ’93, vol. I, Fourth International Landfill Symposium, CISA, Environmental Sanitary Engineering Center, Cagliari, Italy, pp 701–716
Flanagan WP, Apel AA, Barnes JM, Lee BD (2002) Development of gas phase bioreactors for the removal of nitrogen oxides from synthetic flue gas streams. Fuel 81:1953–1961. doi:10.1016/S0016-2361(02)00130-8
Foley J, de Hass D, Yuan Z, Lant P (2010) Nitrous oxide generation in full-scale biological nutrient removal wastewater treatment plants. Water Res 44:831–844. doi:10.1016/j.watres.2009.10.033
García Camacho F, Gallardo Rodríguez JJ, Sánchez Mirón A, Belarbi EH, Chisti Y, Molina Grima E (2011) Photobioreactor scale-up for a shear-sensitive dinoflagellate microalga. Process Biochem 46:936–944. doi:10.1016/j.procbio.2011.01.005
Gebert J, Gröngröft A, Miehlich G (2001) Microbial reduction of methane and trace gas emissions in a biofilter. In: Proceedings from the 8th International Waste Management and Landfill Symposium, S Margherita di Pula, Cagliari, Italy, pp 585–593; SWANA-Solid Waste Association of North America, Silver Spring, MD, USA
Gebert J, Gröngröft A, Miehlich G (2003) Kinetics of microbial landfill methane oxidation in biolfilters. Waste Manag 23:609–619. doi:10.1016/S0956-053X(03)00105-3
Gebert J, Gröngröft A (2006) Performance of a passively vented field-scale biofilter for the microbial oxidation of landfill methane. Waste Manag 26:399–407. doi:10.1016/j.wasman.2005.11.007
Gioardano M, Beardall J, Raven JA (2005) Mechanisms in algae: mechanisms, environmental modulation, and evolution. Annu Rev Plant Biol 56:99–131. doi:annurev.pp.08.060157.001521/annurev.arplant.56.032604.144052
Girard M, Avalos Ramirez A, Buelna G, Heitz M (2011) Biofiltration of methane at low concentrations representative of the piggery industry—influence of the methane and nitrogen concentrations. Chem Eng J 168:151–158. doi:10.1016/j.cej.2010.12.054
Gladue RM, Maxey JE (1994) Microalgal feeds for aquaculture. J Appl Phycol 6:131–141. doi:10.1007/BF02186067
Glass JB, Orphan VJ (2012) Trace metal requirements for microbial enzymes involved in the production and consumption of methane and nitrous oxide. Front Microbiol 3:61. doi:10.3389/fmicb.2012.00061
Golden TC, Taylor FW, Salter EH, Ali Kalbassi M, Raiswell CJ (2004) Process for nitrous oxide removal. US Patent 6719827 B2
González López CV, Acién Fernández FG, Fernández Sevilla JM, Sánchez Fernández JF, Cerón García MC, Molina Grima E (2009) Utilization of the cyanobacteria Anabaena sp. ATCC 33047 in CO2 removal processes. Biores Technol 100:5904–5910. doi:10.1016/j.biortech.2009.04.070
González López CV, Acién Fernández FG, Fernández Sevilla JM, Sánchez Fernández JF, Molina Grima E (2012) Development of a process for efficient use of CO2 from flue gases in the production of photosynthetic microorganisms. Biotechnol Bioeng 109:1637–1650. doi:10.1002/bit.24446
González López CV, Cerón García MC, Fernández Sevilla JM, González Céspedes AM, Camacho Rodríguez J, Molina Grima E (2013) Medium recycling for Nannochloropsis gaditana cultures for aquaculture. Bioresour Technol 129:430–438. doi:10.1016/j.biortech.2012.11.061
Gouveia L, Veloso V, Reis A, Fernandes H, Novais J, Empis J (1996) Evolution of pigment composition in Chlorella vulgaris. Bioresour Technol 57:157. doi:10.1016/0960-8524(96)00058-2
Graham DW, Chaudhary JA, Hanson RS, Arnold RG (1993) Factors affecting competition between type I and type II methanotrophs in two-organism, continuous-flow reactors. Microb Ecol 25:1–17. doi:10.1007/BF00182126
Granger J, Ward BB (2003) Accumulation of nitrogen oxides in copper-limited cultures of denitrifying bacteria. Limnol Oceanogr 48:313–318
Gustavsson DJ, la Cour Jansen J (2011) Dynamics of nitrogen oxides emission from a full-scale sludge liquor treatment plant with nitritation. Water Sci Technol 63:2838–2845
Hall DO, Rao K (1999) Photosynthesis, 6th edn. Cambridge University Press, Cambridge
Hall DO, Acién Fernández FG, Cañizares Guerrero E, Rao KK, Molina Grima E (2003) Outdoor helical tubular photobioreactors for microalgal production: modeling of fluid-dynamics and mass transfer and assessment of biomass productivity. Biotechnol Bioeng 82:62–73. doi:10.1002/bit.10543
Hanagata N, Takeuchi T, Fukuju Y, Barnes DJ, Karube I (1992) Tolerance of microalgae to high CO2 and high temperature. Phytochem 31:3345–3348. doi:10.1016/0031-9422(92)83682-O
Hanson RS (1980) Ecology and diversity of methylotrophic organisms. Adv Appl Microbiol 26:3–39. doi:10.1016/S0065-2164(08)70328-9
Hanson RS, Hanson TE (1996) Methanotrophic bacteria. Microbiol Rev 60:439–471
Haubrichs R, Widmann R (2006) Evaluation of aerated biofilter systems for microbial methane oxidation of poor landfill gas. Waste Manag 26:408–416. doi:10.1016/j.wasman.2005.11.008
Håvelsrud OE, Haverkamp THA, Kristensen T, Jakobsen KS, Rike AG (2011) A metagenomic study of methanotrophic microorganisms in Coal Oil Point seep sediments. BMC Microbiol 11:221. doi:10.1186/1471-2180-11-221
Henckel T, Roslev P, Conrad R (2000) Effects of O2 and CH4 on presence and activity of the indigenous methanotrophic community in rice field soil. Environ Microbiol 2:666–679. doi:10.1046/j.1462-2920.2000.00149.x
Hernández M, Quijano G, Thalasso F, Daugulis AJ, Villaverde S, Muñoz R (2010) A comparative study of solid and liquid non-aqueous phases for the biodegradation of hexane in two-phase partitioning bioreactors. Biotechnol Bioeng 106:731–740. doi:10.1002/bit.22748
Herzog H (2001) What future for carbon capture and sequestration? Environ Sci Technol 35:148A–153A
Hettiarachchi VC, Hettiaratchi PJ, Mehrotra AK, Kumar S (2011) Field-scale operation of methane biofiltration systems to mitigate point source methane emissions. Environ Pollut 159:1715–1720. doi:10.1016/j.envpol.2011.02.029
Higgins IJ, Best DJ, Hammond RC, Scott D (1981) Methane-oxidizing microorganisms. Microbiol Rev 45:556–590
Hill R, Bendall F (1960) Function of two cytochrome components in chloroplasts: a work hypothesis. Nature 186:136–137. doi:10.1038/186136a0
Ho SH, Chen CY, Lee DJ, Chang JS (2011) Perspectives on microalgal CO2-emission mitigation systems—a review. Biotechnol Adv 29:189–198. doi:10.1016/j.biotechadv.2010.11.001
Hoehler TM, Alperin MJ, Albert DB, Martens CS (1995) Field and laboratory studies of methane oxidation in an anoxic marine sediment: evidence for a methanogen-sulfate reducer consortium. Glob Biogeochem Cycles 8:451–463. doi:10.1029/94GB01800
Hood MC (2011) Design and operation of a biofilter for treatment of swine house pit ventilation exhaust. Thesis from Faculty of North Carolina State University (Biological and Agricultural Engineering)
Hooper AB, Terry KR (1979) Hydroxylamine oxidoreductase of Nitrosomonas: production of nitric oxide from hydroxylamine. Biochem Biophys Acta 571:12–20. doi:10.1016/0005-2744(79)90220-1
Hou CT, Laskin AI, Patel RN (1978) Growth and polysaccharide production by Methylocystis parvus OBBP on methanol. Appl Environ Microbiol 37:800–804
Hou S, Makarova KS, Saw JHW, Senin P, Ly BV, Zhou Z, Ren Y, Wang J, Galperin MY, Omelchenko MV, Wolf YI, Yutin N, Koonin EV, Stott MB, Mountain BW, Crowe MA, Smirnova AV, Dunfield PF, Feng L, Wang L, Alam W (2008) Complete genome sequence of the extremely acidophilic methanotroph isolate V4, Methylacidiphilum infernorum, a representative of the bacterial phylum Verrucomicrobia. Biol Direct 3:26. doi:10.1186/1745-6150-3-26
Hu Q, Guterman H, Richmond A (1996) A flat inclined modular photobioreactor for outdoor mass cultivation of photoautotrophs. Biotechnol Bioeng 51:51–60. doi:10.1002/(SICI)1097-0290(19960705)51:1<51::AID-BIT6>3.0.CO;2-#
Hu Q, Zarmi Y, Richmond A (1998) Combined effects of light intensity, light-path and culture density on output rate of Spirulina platensis (Cyanobacteria). Eur J Phycol 33:165–171. doi:10.1080/09670269810001736663
Humer M, Lechner P (1999) Alternative approach to the elimination of greenhouse gases from old landfills. Waste Manag Res 17:443–452. doi:10.1034/j.1399-3070.1999.00064.x
Intergovernmental Panel on Climate Change (2007) Climate change 2007: synthesis report. Available at http://www.ipcc.ch/publications_and_data/publications_ipcc_fourth_assessment_report_synthesis_report.htm. Accessed 12 January 2013
Iverson TM (2006) Evolution and unique bioenergetic mechanisms in oxygenic photosynthesis. Curr Opin Chem Biol 10:91–100. doi:10.1016/j.cbpa.2006.02.013
Jacob-Lopes E, Revah S, Hernández S, Shirai K, Franco TT (2009) Development of operational strategies to remove carbon dioxide in photobioreactors. Chem Eng J 153:120–126. doi:10.1016/j.cej.2009.06.025
Joergensen L (1985) The methane mono-oxygenase reaction system studied in vivo by membrane-inlet mass spectrometry. Biochem J 225:441–448
Kalkowski I, Conrad R (1991) Metabolism of nitric-oxide in denitrifying Pseudomonas aeruginosa and nitrate-respiring Bacillus cereus. FEMS Microbiol Lett 82:107–111. doi:10.1111/j.1574-6968.1991.tb04848.x
Kallistova AY, Kevbrina MV, Nekrasova VK, Glagolev MV, Serebryanaya MI, Nozhevnikova AN (2005) Methane oxidation in landfill cover soil. Microbiology 74:608–614. doi:10.1007/s11021-005-0110-z
Kalyuzhnaya MG, Khmelenina VN, Kotelnikova S, Holmquist L, Pedersen K, Trotsenko YA (1999) Methylomonas scandinivica sp. nov., a new methanotrophic psychrotrophic bacterium isolated from deep igneous rock ground water of Sweden. Syst Appl Microbiol 22:565–572. doi:10.1016/S0723-2020(99)80010-1
Kampschreur MJ, van der Star WRL, Wielders HA, Mulder JW, Jetten MSM, van Loosdrecht MCM (2008) Dynamics of nitric oxide and nitrous oxide emission during full-scale reject water treatment. Water Res 42:812–826. doi:10.1016/j.watres.2007.08.022
Kampschreur MJ, Temmink H, Kleerebezem R, Jetten MSM, van Loosdrecht MCM (2009) Nitrous oxide emission during wastewater treatment. Water Res 43:4093–4103. doi:10.1016/j.watres.2009.03.001
Karlsson J, Clarke AK, Chen ZY, Park YI, Hugghins SY, Husic HD, Moroney JV, Samuelsson G (1998) A novel α-type carbonic anhydrase associated with the thylakoid membrane in Chlamydomonas reinhardtii is required for growth at ambient CO2. EMBO J 17:1208–1216. doi:10.1093/emboj/17.5.1208
Kightley D, Nedwell DB, Cooper M (1995) Capacity for methane oxidation in landfill cover soils measured in laboratory-scale soils microcosms. Appl Environ Microbiol 61:592–610
Kim HJ, Graham DW, DiSpirito AA, Alterman MA, Galeva N, Larive CK, Asunskis D, Sherwood PMA (2004) Methanobactin, a copper-acquisition compound from methane-oxidizing bacteria. Science 305:1612–1615. doi:10.1126/science.1098322
Knief C, Dunfield PF (2005) Response and adaptation of different methanotrophic bacteria to low methane mixing ratios. Environ Microbiol 7:1307–1317. doi:10.1111/j.1462-2920.2005.00814.x
Knittel K, Boetius A (2009) Anaerobic oxidation of methane: progress with an unknown process. Annu Rev Microbiol 63:311–334. doi:annurev.pp.08.060157.001521/annurev.micro.61.080706.093130
Knowles R (1993) Methane: process of production and consumption. Agricultural ecosystem effects on trace gases and global climate change. American Society of Agronomy, Madison, pp 145–156
Kodama M, Ikemoto H, Miyachi S (1993) A new species of highly CO2-tolerant fast-growing marine microalga suitable for high-density culture. J Marine Biotechnol 1:21–25
Koh SC, Bowman JP, Sayler GS (1993) Soluble methane monooxygenase production and trichloroethylene degradation by a type methanotroph, Methylomonas methanica 68-1. Appl Environ Microbiol 59:960–967
Kumar A, Yuan X, Sahu AK, Dewulf J, Ergas SJ, Van Langenhove H (2010a) A hollow fiber membrane photo-bioreactor for CO2 sequestration from combustion gas coupled with wastewater treatment: a process engineering approach. J Chem Technol Biotechnol 85:387–394. doi:10.1002/jctb.2332
Kumar A, Ergas S, Yuan X, Sahu A, Zhang Q, Dewulf J, Malcata FX, van Langenhove H (2010b) Enhanced CO2 fixation and biofuel production via microalgae: recent developments and future directions. Trends Biotechnol 28:371–380. doi:10.1016/j.tibtech.2010.04.004
Kumar K, Dasgupta CN, Nayak B, Lindblad P, Das D (2011) Development of suitable photobioreactors for CO2 sequestration addressing global warming using green algae and cyanobacteria. Bioresour Technol 102:4945–4953. doi:10.1016/j.biortech.2011.01.054
Lee YK, Ding SY (1994) Cell cycle and accumulation of astaxanthin in Haematococcus lacustris (Chlorophyta). J Phycol 30:445–449. doi:10.1111/j.0022-3646.1994.00445.x
Lidstrom ME, Stirling DI (1990) Methylotrophs: genetics and commercial applications. Annu Rev Microbiol 44:27–58. doi:annurev.pp.08.060157.001521/annurev.mi.44.100190.000331
Lipscomb JD (1994) Biochemistry of the soluble methane monooxygenase. Annu Rev Microbiol 48:371–399. doi:annurev.pp.08.060157.001521/annurev.mi.48.100194.002103
Löffler G, Wargadalam VJ, Winter F, Hofbauer H (2002) Decomposition of nitrous oxide at medium temperatures. Combust Flame 120:427–438. doi:10.1016/S0010-2180(99)00106-6
Lontoh S, Semrau JD (1998) Methane and trichloroethylene degradation by Methylosinus trichosporium OB3b expressing particulate methane monooxygenase. Appl Environ Microb 64:1106–1114
Lundquist T, Woertz I, Quinn NW, Benemann J (2010) A realistic technology and engineering assessment of algae biofuel production. Energy Biosciences Institute, University of California, Berkeley
Madigan MT, Martinko JM, Parker J (2006) Brock, biology of microorganisms, 10th edn. Pearson, New Jersey
Mandeno G, Craggs R, Tanner C, Sukias J, Webster-Brown J (2005) Potential biogas scrubbing using a high rate pond. Water Sci Technol 51:253–256
Massey University (2011) Algal raceway simulator. Available at http://algae.massey.ac.nz/. Accessed 1 November 2012
Matsuda Y, Bozzo GG, Colman B (1998) Regulation of dissolved inorganic carbon transport in green algae. Can J Bot 76:1072–1083. doi:10.1139/b98-072
Matsumoto H, Shioji N, Hamasaki A, Ikuta Y, Fukuda Y, Sato M, Endo N, Tsukamoto T (1995) Carbon dioxide fixation by microalgae photosynthesis using actual flue gas discharged from a boiler. Appl Biochem Biotechnol 51:681–692. doi:10.1007/BF02933469
Melis A (1999) Photosystem-II damage and repair cycle in chloroplasts: what modulates the rate of photodamage in vivo? Trends Plant Sci 4:130–135. doi:10.1016/S1360-1385(99)01387-4
Melse RW, Van der Werf AW (2005) Biofiltration for mitigation of methane emission from animal husbandry. Environ Sci Technol 39:5460–5468. doi:10.1021/es048048q
Merchuk JC, García-Camacho F, Molina-Grima E (2007) Photobioreactor design and fluid dynamics. Chem Biochem Eng Q 21:345–355
Metting FB (1996) Biodiversity and application of microalgae. J Ind Microbiol Biotechnol 17:477–489. doi:10.1007/BF01574779
Miller AG, Espie G, Canvin DT (1990) Physiological-aspects of CO2 and HCO3 − transport by cyanobacteria—a review. Can J Bot Rev Can Bot 68:1291–1302. doi:10.1139/b90-165
Mitra M, Melis A (2008) Optical properties of microalgae for enhanced biofuels production. Opt Expr 16:21807–21820. doi:10.1364/OE.16.021807
Miyahara M, Kim SW, Fushinobu S, Takaki K, Yamada T, Watanabe A, Miyauchi K, Endo G, Wakagi T, Shoun H (2010) Potential of aerobic denitrification by Pseudomonas stutzeri TR2 to reduce nitrous oxide emissions from wastewater treatment plants. Appl Environ Microbiol 76:4619–4625. doi:10.1128/AEM.01983-09
Miyairi S (1995) CO2 assimilation in a thermophilic cyanobacterium. Energy Convers Manag 36:763–766. doi:10.1016/0196-8904(95)00116-U
Miziorko HM, Lorimer GH (1983) Ribulose-1,5-bisphosphate carboxylase-oxygenase. Annu Rev Biochem 52:507–535. doi:annurev.pp.08.060157.001521/annurev.bi.52.070183.002451
Mohanty SR, Bodelier PLE, Floris V, Conrad R (2006) Differential effects of nitrogenous fertilizers on methane-consuming microbes in rice field and forest soils. Appl Environ Microbiol 72:1346–1354. doi:10.1128/AEM.72.2.1346-1354.2006
Molina Grima E, Belarbi EH, Acién Fernández FG, Robles Medina A, Chisti Y (2003) Recovery of microalgal biomass and metabolites: process options and economics. Biotechnol Adv 20:491–515. doi:10.1016/S0734-9750(02)00050-2
Morita M, Watanabe Y, Saiki H (2000) Investigation of photobioreactor design for enhancing the photosynthetic productivity of microalgae. Biotechnol Bioeng 69:693–698. doi:10.1002/1097-0290(20000920)69:6<693::AID-BIT14>3.0.CO;2-0
Morita M, Watanabe Y, Saiki H (2002) Photosynthetic productivity of conical helical tubular photobioreactor incorporating Chlorella sorokiniana under field conditions. Biotechnol Bioeng 77:155–162. doi:10.1002/bit.10119
Moroney JV, Somanchi A (1999) How do algae concentrate CO2 to increase the efficiency of photosynthetic carbon fixation. Plant Physiol 119:9–16. doi:10.1104/pp.119.1.9
Morweiser M, Kruse O, Hankamer B, Posten C (2010) Developments and perspectives of photobioreactors for biofuel production. Appl Microbiol Biotechnol 87:1291–1301. doi:10.1007/s00253-010-2697-x
Muñoz R, Guieysse B (2006) Algal–bacterial processes for the treatment of hazardous contaminants: a review. Water Res 40:2799–2815. doi:10.1016/j.watres.2006.06.011
Muñoz R, Villaverde S, Guieysse B, Revah S (2007) Two-phase partitioning bioreactors for treatment of volatile organic compounds. Biotechnol Adv 25:410–422. doi:10.1016/j.biotechadv.2007.03.005
Muñoz R, Alzate ME, Bahr M, Díaz I, Díaz M, Dominguez A (2012) Microalgae bring an opportunity to reduce the CO2 footprint of WWTPs by coupling biogas upgrading and nutrients removal from centrates. Proceedings of the IWA international conference EcoTechnologies for Wastewater Treatment, Santiago de Compostela, pp 25–27
Muramatsu H, Tokura K, Mori T, Akahori R, Watanabe K, Satsuma T, Hattori T, Murakami Y (1997) Inhibitory effect of oxygen on catalytic removal of nitrous oxide with methane. Energy Convers Manag 38:1399–1403. doi:10.1016/S0196-8904(96)00169-0
Nagase H, Eguchi K, Yoshihara K, Hirata K, Miyamoto K (1998) Improvement of microalgal NO x removal in bubble column and airlift reactors. J Ferment Bioeng 86:421–423. doi:10.1016/S0922-338X(99)89018-7
Nakajima Y, Ueda R (1997) Improvement of photosynthesis in dense microalgal suspensions by reduction of light harvesting pigments. J Appl Phycol 9:503–510. doi:10.1023/A:1007920025419
Nascimento DM, Hudepohl NJ, Schroeder ED, Chang DPY (2000) Bio-oxidation of nitric oxide in a nitrifying aerobic filter. Proceedings of the 93rd Annual Meeting, Air and Waste Management Association, Salt Lake City, UT
Nelson DL, Cox MM (2005) Lehninger principles of biochemistry, 4th edn. WH Freeman and Company, New Jersey
Nielsen AK, Gerdes K, Murrell JC (1997) Cooper-dependent reciprocal transcriptional regulation of methane monooxygenase genes in Methylococcus capsulatus and Methylosinus trichosporium. Mol Microbiol 25:399–409. doi:10.1046/j.1365-2958.1997.4801846.x
Nikiema J, Brzezinski R, Heitz M (2007) Elimination of methane generated from landfills by biofiltration: a review. Rev Environ Sci Biotechnol 6:261–284. doi:10.1007/s11157-006-9114-z
Nikiema J, Heitz M (2009) The influence of the gas flow rate during methane biofiltration on an inorganic packing material. Can J Chem Eng 87:136–142. doi:10.1002/CJCE.20131
Norsker NH, Barbosa MJ, Vermuë MH, Wijffels RH (2011) Microalgal production—a close look at the economics. Biotechnol Adv 29:24–27. doi:10.1016/j.biotechadv.2010.08.005
O’Connor M (1981) Regulation and genetics in facultative methylotrophic bacteria. In: Dalton H (ed) Microbial growth on C1 compounds. Heyden, London, pp 294–300
Ogawa T, Aiba S (1981) Bioenergetic analysis of mixotrophic growth in Chlorella vulgaris and Scenedesmus acutus. Biotechnol Bioeng 3:1121–1132. doi:10.1002/bit.260230519
Ogbonna JC, Tanaka H (1998) Cyclic autotrophic/heterotrophic cultivation of photosynthetic cells: a method of achieving continuous cell growth under light/dark cycles. Biores Technol 65:65–72. doi:10.1016/S0960-8524(98)00018-2
Ogbonna JC, Tanaka H (2000) Light requirement and photosynthetic cell cultivation—development of processes for efficient light utilization in photobioreactors. J Appl Phycol 12:207–218. doi:10.1023/A:1008194627239
Omelchenko L, Savel’eva ND, Vasil’ev LV, Zavarin GA (1993) A psychrophilic methanotrophic community from a tundra soil. Microbiol 61:754–759. doi:10.1177/0734242X09339325
Ono E, Cuello JL (2007) Carbon dioxide mitigation using thermophilic cyanobacteria. Biosys Eng 96:129–134. doi:10.1016/j.biosystemseng.2006.09.010
Op den Camp HJM, Islam T, Sttot MB, Harhangi HR, Hynes A, Schouten S, Jetten MSM, Birkeland NK, Pol A, Dunfield PF (2009) Environmental, genomic and taxonomic perspectives on methanotrophic Verrucomicrobia. Environ Microbiol Rep 1:293–306. doi:10.1111/j.1758-2229.2009.00022.x
Ota M, Kato Y, Watanabe H, Watanabe M, Sato Y, Smith RLJ, Inomata H (2009) Effect of inorganic carbon on photoautotrophic growth of microalga Chlorococcum littorale. Biotechnol Prog 25:492–498. doi:10.1002/btpr.123
Packer M (2009) Algal capture of carbon dioxide; biomass generation as a tool for green house gas mitigation with reference to New Zealand energy strategy and policy. Energy Policy 37:3428–3437. doi:10.1016/j.enpol.2008.12.025
Park S, Lee CH, Ryu CR, Sung K (2009) Biofiltration for reducing methane emissions from modern sanitary landfills at the low methane generation stage. Water Air Soil Pollut 196:19–27. doi:10.1007/s11270-008-9754-4
Patt TE, Hanson RS (1978) Intracytoplasmatic membrane, phospholipid, and sterol content of Methylobacterium organophilum cells grown under different conditions. J Bacteriol 134:636–644
Pol A, Heijmans K, Harhangi HR, Tedesco D, Jetten MSM, Op den Camp HJM (2007) Methanotrophy below pH 1 by a new Verrucomicrobia species. Nature 450:874–878. doi:10.1038/nature06222
Posten C (2009) Design principles of photo-bioreactors for cultivation of microalgae. Eng Life Sci 9:165–177. doi:10.1002/elsc.200900003
Powelson DK, Chanton J, Abichou T, Morales J (2006) Methane oxidation in water-spreading and compost biofilters. Waste Manage Res 24:528–536. doi:10.1177/0734242X06065704
Price GD, Badger MR (1989) Expression of human carbonic anhydrase in the cyanobacterium Synechococcus PCC 7942 creates a high CO2-requiring phenotype. Evidence for a central role for carboxysomes in the CO2 concentrating mechanism. Plant Physiol 91:505–513
Price GD, Coleman JR, Badger MR (1992) Association of carbonic anhydrase activity with carboxysomes isolated from cyanobacterium Synechococcus PCC 7942. Plant Physiol 100:784–793
Pulz OP (2001) Photobioreactors: production systems for phototrophic microorganisms. Appl Microbiol Biotechnol 57:287–293. doi:10.1007/s002530100702
Quayle JR (1972) The metabolism of one-carbon compounds by micro-organisms. Adv Microbiol Physiol 7:119–203. doi:10.1016/S0065-2911(08)60078-8
Quijano G, Hernández M, Thalasso F, Muñoz R, Villaverde S (2009) Two-phase partitioning bioreactors in environmental biotechnology. Appl Microbiol Biotechnol 84:829–846. doi:10.1007/s00253-009-2158-6
Raja R, Hemaiswarya S, Kumar NA, Sridhar S, Rengasamy R (2008) A perspective on the biotechnological potential of microalgae. Crit Rev Microbiol 34:77–88. doi:10.1080/10408410802086783
Rassamee V, Sattayatewa C, Pagilla K, Chandran K (2011) Effect of oxic and anoxic conditions on nitrous oxide emissions from nitrification and denitrification. Biotechnol Bioeng 108:2036–2045. doi:10.1002/bit.23147
Raven JA, Cockell CS, De la Rocha CL (2008) The evolution of inorganic carbon concentrating mechanisms in photosynthesis. Phil Trans R Soc B 363:2641–2650. doi:10.1098/rstb.2008.0020
Ravishankara AR, Daniel JS, Portmann RW (2009) Nitrous oxide (N2O): the dominant ozone-depleting substance emitted in the 21st century. Science 326:123–125. doi:10.1126/science.1176985
Reeburgh WS (2007) Oceanic methane biogeochemistry. Chem Rev 107:486–513. doi:10.1021/cr050362v
Remsen CC, Minnich EC, Stephens RS, Buchholz L, Lidstrom ME (1989) Methane oxidation in Lake Superior sediments. J Great Lakes Res 5:141–196. doi:10.1016/S0380-1330(89)71468-4
Richmond A (1986) Microalgae of economic potential. In: Richmond A (ed) Handbook of microalgal mass culture. CRC, Boca Raton, pp 200–202
Richmond A (1990) Large scale microalgal culture and applications. In: Round FE, Chapman DJ (eds) Progress in phycological research, vol. 7. Biopress Ltd., Bristol, pp 239–320
Richmond A (2004) Biological principles of mass cultivation. In: Richmond A (ed) Handbook of microalgal culture: biotechnology and applied phycology. Blackwell Science Ltd., Oxford, pp 125–177
Robertson LA, Dalsgaard T, Revsbech NP, Hadioetomo R, Kuenen JG (1995) Confirmation of ‘aerobic denitrification’ in batch cultures, using gas chromatography and 15N mass spectrometry. FEMS Microbiol Ecol 18:113–120. doi:10.1111/j.1574-6941.1995.tb00168.x
Roch F, Alexander M (1997) Inability of bacteria to degrade low concentrations of toluene in water. Environ Technol Chem 16:1377–1383. doi:10.1002/etc.5620160707
Rocha-Rios J, Bordel S, Hernández S, Revah S (2009) Methane degradation in two-phase partition bioreactors. Chem Eng J 152:289–292. doi:10.1016/j.cej.2009.04.028
Rocha-Rios J, Quijano G, Thalasso F, Revah S, Muñoz R (2011) Methane biodegradation in a two-phase partition internal loop airlift reactor with gas recirculation. J Chem Technol Biotechnol 86:353–360. doi:10.1002/jctb.2523
Sakurai N, Sakurai T (1997) Isolation and characterization of nitric oxide reductase from Paracoccus halodenitrificans. Biochem 36:13809–13815. doi:10.1021/bi971070u
Sánchez Fernández JF, González López CV, Acién Fernández FG, Fernández Sevilla JM, Molina Grima E (2012) Utilization of Anabaena sp. in CO2 removal processes. Modelling of biomass, exopolysaccharides productivities and CO2 fixation rate. Appl Microbiol Biotechnol 94:613–624. doi:10.1007/s00253-011-3683-7
Sánchez Mirón A, Contreras Gómez A, García Camacho F, Molina Grima E, Chisti Y (1999) Comparative evaluation of compact photobioreactors for large-scale monoculture of microalgae. J Biotechnol 70:249–270. doi:10.1016/S0168-1656(99)00079-6
Sato T, Usui S, Tsuchiya Y, Kondo Y (2006) Invention of outdoor closed type photobioreactor for microalgae. Energy Convers Manage 47:791–799. doi:10.1016/j.enconman.2005.06.010
Satsuma A, Maeshima H, Watanabe K, Suzuki K, Hattori T (2000) Effects of methane and oxygen on decomposition of nitrous oxide over metal oxide catalysts. Catal Today 63:347–353. doi:10.1016/S0920-5861(00)00478-8
Savir Y, Noor E, Milo R, Tlusty T (2010) Cross species analysis traces adaptation of Rubisco toward optimality in a low-dimensional landscape. PNAS 107:3475–3480. doi:10.1073/pnas.0911663107
Scheutz C, Kjeldsen P, Bogner JA, De Visscher A, Gebert J, Hilger HA, Huber-Humer M, Spokas K (2009) Microbial methane oxidation processes and technologies for mitigation of landfill gas emissions. Waste Manage Res 27:409–455. doi:10.1177/0734242X09339325
Scott D, Brannan J, Higgins IJ (1981) The effect of growth conditions on intracytoplasmic membranes and methane monooxygenase activities in Methylosinus trichosporium OB3b. J Gen Microbiol 125:63–72. doi:10.1099/00221287-125-1-63
Seckbach J, Ikan R (1972) Sterols and chloroplast structure of Cyanidium caldarium. Plant Physiol 49:457–459
Semple KT, Cain RB, Schmidt S (1999) Biodegradation of aromatic compounds by microalgae. FEMS Microbiol Lett 170:291–300. doi:10.1111/j.1574-6968.1999.tb13386.x
Semrau JD, DiSpirito AA, Yoon S (2010) Methanotrophs and cooper. FEMS Microbiol Rev 34:496–531. doi:10.1111/j.1574-6976.2010.00212.x
Semrau JD (2011) Bioremediation via methanotrophy: overview of recent findings and suggestions for future research. Front Microbiol 2:209. doi:10.3389/fmicb.2011.00209
Shanmugasundram R, Lee CM, Sublette KL (1993) Reduction of nitric oxide by denitrifying bacteria. Appl Biochem Biotechnol 39–40:727–737. doi:10.1007/BF02919031
Shimizu A, Tanaka K, Fujimori M (2000) Abatement technologies for N2O emissions in the adipic acid industry. Chemosphere Glob Chang Sci 2:425–434
Singh RN, Sharma S (2012) Development of suitable photobioreactor for algae production—a review. Renew Sust Energ Rev 16:2347–2353. doi:10.1016/j.rser.2012.01.026
Skalska K, Miller JS, Ledakowicz S (2010) Trends in NO x abatement: a review. Sci Total Environ 408:3976–3989. doi:10.1016/j.scitotenv.2010.06.001
Sobczuk TM, García F, Camacho F, Acién FG, Molina E (2000) Carbon dioxide uptake efficiency by outdoor microalgal cultures in tubular airlift photobioreactors. Biotechnol Bioeng 67:465–475. doi:10.1002/(SICI)1097-0290(20000220)67:4<465::AID-BIT10>3.0.CO;2-9
Spolaore P, Joannis-Cassan C, Duran E, Isambert A (2006) Commercial applications of microalgae. J Biosci Bioeng 101:87–96. doi:10.1263/jbb.101.87
Staehelin A (1986) Chloroplast structure and supramolecular organization of photosynthetic membranes. In: Staehelin LA, Arntzen CA (eds) Photosynthesis III. Photosynthetic membranes and light-harvesting systems, vol 19. Springer, New York, pp 1–84
Stanley SH, Prior SD, Leak D, Dalton H (1983) Copper stress underlies the fundamental change in intracellular location of methane mono-oxygenase in methane-oxidizing organisms: studies in batch and continuous cultures. Biotechnol Lett 5:487–492. doi:10.1007/BF00132233
Taiz L, Zeiger E (2002) Plant physiology, 3rd edn. Sinauer Associates Inc., Sunderland
Tallec G, Garnier J, Billen G, Gousailles M (2008) Nitrous oxide emissions from denitrifying activated sludge of urban wastewater treatment plants, under anoxia and low oxygenation. Biores Technol 99:2200–2209. doi:10.1016/j.biortech.2007.05.025
Tamiya H (1957) Mass culture of algae. Ann Rev Plant Physiol 8:309–334. doi:annurev.pp.08.060157.001521/annurev.pp.08.060157.001521
Thalasso F, Vallecillo A, García-Encina P, Fdz-Polanco F (1997) The use of methane as sole carbon source for wastewater denitrification. Water Res 31:55–60. doi:10.1016/S0043-1354(96)00228-X
Toledo-Cervantes A, Morales M, Novelo E, Revah S (2013) Carbon dioxide fixation and lipid storage by Scenedesmus obtusiusculus. Bioresour Technol. doi:10.1016/j.biortech.2012.12.081
Tredici MR, Zittelli GC (1998) Efficiency of sunlight utilization: tubular versus flat photobioreactors. Biotechnol Bioeng 57:187–197. doi:10.1002/(SICI)1097-0290(19980120)57:2<187::AID-BIT7>3.0.CO;2-J
Tredici MR (2009) Photobiology of microalgae mass cultures: understanding the tools for the next green revolution. Biofuels 1:143–162. doi:10.4155/bfs.09.10
Treude T, Ziebis W (2010) Methane oxidation in permeable sediments at hydrocarbon seeps in the Santa Barbara Channel, California. Biogeosci Discuss 7:1905–1933. doi:10.5194/bg-7-3095-2010
Tsubota J, Eshinimaev BT, Khmelenina VN, Trotsenko YA (2005) Methylothermus thermalis gen. nov., sp. nov., a novel moderately thermophilic obligate methanotroph from a hot spring in Japan. Int J Syst Evol Microbiol 55:1877–1884. doi:10.1099/ijs.0.63691-0
Uggetti E, García J, Lind SE, Martikainen PJ, Ferrer I (2012) Quantification of greenhouse gas emissions from sludge treatment wetlands. Water Res 46:1755–1762. doi:10.1016/j.watres.2011.12.049
Ugwu CU, Aoyagi H, Uchiyama H (2008) Photobioreactors for mass cultivation of algae. Biores Technol 99:4021–4028. doi:10.1016/j.biortech.2007.01.046
Utami TS, Hermansyah H, Nasikin M (2012) Biofiltration of nitrous oxide using cow-manure based compost as medium filter. J Environ Prot 3:584–588. doi:jep.2012.37070/jep.2012.37070
Veillette M, Girard M, Viens P, Brzezinski R, Heitz M (2012) Function and limits of biofilters for the removal of methane in exhaust gases from the pig industry. Appl Microbiol Biotechnol 94:601–611. doi:10.1007/s00253-012-3998-z
Viskari PJ, Colyer CL (2003) Rapid extraction of phycobiliproteins from cultured cyanobacteria samples. Anal Biochem 319:263–271. doi:10.1016/S0003-2697(03)00294-X
Wallar BJ, Lipscomb JD (2001) Methane monooxygenase component B mutants alter the kinetics of steps throughout the catalytic cycle. Biochem 40:2220–2233. doi:10.1021/bi002298b
Wang B, Li Y, Wu N, Lan C (2008) CO2 bio-mitigation using microalgae. Appl Microbiol Biotechnol 79:707–718. doi:10.1007/s00253-008-1518-y
Whalen SC, Reeburgh WS, Sandbeck KA (1990) Rapid methane oxidation in a landfill cover soil. Appl Environ Microbiol 56:3405–3411
Whalen SC, Reeburgh WS (1996) Moisture and temperature sensitivity of CH4 oxidation in boreal soils. Soil Biol Biochem 28:1271–1278. doi:10.1016/S0038-0717(96)00139-3
Whittenbury R (1981) The interrelationship of autotrophy and methylotrophy as seen in Methylococcus capsulatus (Bath). In: Dalton H (ed) Microbial growth on C1 compounds. Heyden, London, pp 181–190
Whittenbury R, Dalton H (1981) The methylotrophic bacteria. In: Starr MP, Stolp H, Truper HG, Balows A, Schlegel HG (eds) The procaryotes, a handbook on habits, isolation, and identification of bacteria, vol I. Springer, New York, pp 894–902
Wilshusen JH, Hettiaratchi JPA, De Visscher A, Saint-Fort R (2004) Methane oxidation and formation of EPS in compost: effect of oxygen concentration. Environ Pollut 129:305–314. doi:10.1016/j.envpol.2003.10.015
Winder R (2004) Methane to biomass. J Soc Chem Ind Lond 17:19
Woertz JR, Kinney KA, Szaniszlo PJ (2001) A fungal vapor phase bioreactor for the removal of nitric oxide from waste gas streams. J Air Waste Manage Assoc 51:895–902. doi:10.1080/10473289.2001.10464321
Wolf HJ, Hanson RS (1979) Isolation and characterization of methane-utilizing yeasts. J Gen Microbiol 114:187–194
Wood PM (1986) Nitrification as a bacterial energy source. In: Prosser JI (ed) Nitrification. Spec Publ Soc Gen Microbiol 20:39–62
Wrage N, Velthof GL, van Beusichem ML, Oenema O (2001) Role of nitrifier denitrification in the production of nitrous oxide. Soil Biol Biochem 33:1723–1732. doi:10.1016/S0038-0717(01)00096-7
Wu ML, Ettwig KF, Jetten MS, Strous M, Keltjens JT, van Niftrik L (2011) A new intra-aerobic metabolism in the nitrite-dependent anaerobic methane-oxidizing bacterium Candidatus ‘Methylomirabilis oxyfera’. Biochem Soc Trans 39:243–248. doi:10.1042/BST0390243
Wunderlin P, Mohn J, Joss A, Emmenegger L, Siegrist H (2012) Mechanisms of N2O production in biological wastewater treatment under nitrifying and denitrifying conditions. Water Res 46:1027–1037. doi:10.1016/j.watres.2011.11.080
Xu X, Xu H, Kapteijn F, Moulijn JA (2004) SBA-15 based catalysts in catalytic N2O decomposition in a model tail-gas from nitric acid plants. Appl Catal B 53:265–274. doi:10.1016/j.apcatb.2004.04.023
Xu L, Weathers PJ, Xiong XR, Liu CZ (2009) Microalgal bioreactors: challenges and opportunities. Eng Life Sci 9:178–189. doi:10.1002/elsc.200800111
Yoo C, Jun SY, Lee JY, Ahn CY, Oh HM (2010) Selection of microalgae for lipid production under high levels of carbon dioxide. Bioresour Technol 101:71–74. doi:10.1016/j.biortech.2009.03.030
Yoon S, Carey JN, Semrau JD (2009) Feasibility of atmospheric methane removal using methanotrophic biotrickling filters. Appl Microbiol Biotechnol 83:949–956. doi:10.1007/s00253-009-1977-9
Yoon S, Kraemer SM, DiSpirito AA, Semrau JD (2010) An assay for screening microbial cultures for chalkophore production. Environ Microbiol Rep 2:295–303. doi:10.1111/J.1758-2229.2009.00125.x
Yoshihara K, Nagase H, Eguchi K, Hirata K, Miyamoto K (1996) Biological elimination of nitric oxide and carbon dioxide from flue gas by marine microalga NOA-113 cultivation in a long tubular photobioreactor. J Ferment Bioeng 82:351–354. doi:10.1016/0922-338X(96)89149-5
Zamorano M, Pérez Pérez JI, Pavés IA, Ridao AR (2007) Study of the energy potential of the biogas produced by an urban waste landfill in Southern Spain. Renew Sust Energ Rev 11:909–922. doi:10.1016/j.rser.2005.05.007
Zhu XG, Long SP, Ort DR (2008) What is the maximum efficiency with which photosynthesis can convert solar energy into biomass? Curr Opin Biotechnol 19:153–159. doi:10.1016/j.copbio.2008.02.004
Zou N, Zhang CW, Cohen Z, Richmond A (2000) Production of cell mass and eicosapentaenoic acid (EPA) in ultrahigh cell density cultures of Nannochloropsis sp (Eustigmatophyceae). Eur J Phycol 35:127–133. doi:10.1080/09670260010001735711
Zúñiga C, Morales M, Le Borgne S, Revah S (2011) Production of poly-hydroxybutyrate (PHB) by Methylobacterium organophilum isolated from a methanotrophic consortium in a two-phase partition bioreactor. J Hazard Mater 190:876–882. doi:10.1016/j.jhazmat.2011.04.011
Acknowledgments
This research was supported by the Spanish Ministry of Economy and Competitiveness (RYC-2007-01667, JCI-2011-11009, and BES-2010-030994 contracts; CTQ2012-34949 and CONSOLIDER-CSD 2007-00055 projects). The Regional Government of Castilla y León is gratefully acknowledged (VA004A11-2 and GR76). The contributions of Esther Posadas (University of Valladolid) and Antonio Encina (University of León) during manuscript preparation are also acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
López, J.C., Quijano, G., Souza, T.S.O. et al. Biotechnologies for greenhouse gases (CH4, N2O, and CO2) abatement: state of the art and challenges. Appl Microbiol Biotechnol 97, 2277–2303 (2013). https://doi.org/10.1007/s00253-013-4734-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-013-4734-z