Abstract
An efficient method of formulating serum-free medium (SFM) for production of therapeutic antibodies by recombinant CHO (rCHO) cells was developed using two rCHO cell lines producing a therapeutic antibody. In this method, ten kinds of SFM were prepared by supplementing the basal SFM with statistically designed mixtures (total 5 g L−1) of three non-animal-derived hydrolysates: yeastolate, soy hydrolysate, and wheat gluten hydrolysate. When the two rCHO cell lines were cultivated, the mixtures of soy hydrolysate and wheat gluten hydrolysate showed a positive effect on cell growth. On the other hand, the mixtures including a high portion of yeastolate significantly enhanced specific antibody productivity. To reconstitute the mixture ratios of the three hydrolysates for high growth and antibody production, the effect of each medium was analyzed by the statistical program Design-Expert®. The resulting medium gave a 1.9–3.3-fold increase in the maximum antibody concentration, compared to the basal SFM. Taken together, the supplementation of hydrolysates to the basal SFM with the help of statistical analysis is an efficient means of developing SFM for therapeutic antibody production by rCHO cells.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Chinese hamster ovary (CHO) cells have been most widely used for the commercial production of therapeutic antibodies. The popularity of CHO cells is likely to persist as the demand for therapeutic antibodies continues to increase (Chiou et al. 2009; Majors et al. 2008; Nam et al. 2008).
For large-scale suspension cultures of recombinant CHO (rCHO) cells, a serum-free medium (SFM) is desirable because the use of serum increases the risk of contamination of infectious agents such as mycoplasma, viruses, and prions and complicates the subsequent purification process (Glassy et al. 1988; Keen and Rapson 1995; Schrőder et al. 2004; Huang et al. 2007). However, no universal SFM applicable to all cell lines is available. Thus, a specific SFM for each individual cell line needs to be developed to maximize cell growth and antibody production.
Although a chemically-defined SFM is desirable, it often results in decreased cell growth and protein production, compared to serum-containing medium. Furthermore, due to the complexity of nutritional requirements of CHO cells, it is time-consuming to formulate the chemically defined SFM. In this regard, SFM supplemented with hydrolysates, which contain oligopeptides, amino acids, iron salts, lipids, vitamins, and other low molar mass substances in trace elements, is a useful alternative (Chun et al. 2007; Wang et al. 2007). In fact, SFM supplemented with hydrolysates is the most widely used medium in the biopharmaceutical industry. However, the composition of this SFM supplemented with hydrolysates is unrevealed to the public.
Among various raw materials, the trend of avoiding animal-derived materials during the biopharmaceutical process has led to the extensive use of non-animal hydrolysates such as plant hydrolysate and yeast hydrolysate (Donaldson and Shuler 1998; Franek et al. 2000; Heidemann et al. 2000). The source of the hydrolysates affects cell growth and production (Sung et al. 2004). Accordingly, careful consideration should be taken when determining the kind and quantity of hydrolysates to be used.
In the present study, we describe a simple and rapid method of developing SFM supplemented with non-animal-derived hydrolysates for suspension cultures of rCHO cell lines producing therapeutic antibodies.
Materials and methods
Cell lines
Two antibody-producing rCHO cell lines were used in this study. One rCHO cell line produces a recombinant antibody against human platelets and was established by transfection of a vector containing dihydrofolare reductase (dhfr) and the heavy chain and light chain of the antibody into dhfr-deficient CHO cells (DG44) and which underwent subsequent dhfr/methotrexate (MTX)-mediated gene amplification (Jun et al. 2005). The stable rCHO cell line (C11-08) was selected at 80 nM MTX. The other rCHO cell line produces a humanized anti-4-1BB antibody for active suppression of antibody-mediated autoimmune reactions. It was established by transfection of a vector containing the dhfr gene and the heavy chain of the humanized antibody and another vector containing the light chain into dhfr-deficient CHO cells (DUKX-B11, ATCC CRL-9096) (Hong et al. 2000). The stable rCHO cell line (H-1-5) was selected at 1 μM MTX.
Media and reagents
Iscove's modified Dulbecco's medium (IMDM), dialyzed fetal bovine serum (dFBS), and yeastolate ultrafiltrate were purchased from Invitrogen (Carlsbad, CA, USA). Soy hydrolysate (HyPep® 1510) and wheat gluten hydrolysate (HyPep® 4601) were obtained from Kerry Bio-science (Cork, Ireland). The other medium supplements used in this work were purchased from Sigma-Aldrich (St. Louis, MO, USA).
A basal SFM was developed in our laboratory and its components are given in Table 1. For the SFM formulation, medium supplements, except for insulin, phosphatidylcholine, and ferric citrate, were dissolved in water before addition to the culture media. Insulin was dissolved in 1 M acetic acid, phosphatidylcholine in absolute ethanol, and ferric citrate in boiling water.
Supplementation of hydrolysates to the basal SFM was determined by the mixture design which consists of three components, yeastolate ultrafiltrate (A), soy hydrolysate (B), and wheat gluten hydrolysate (C). It contains the ten blends of media with different ratios of the hydrolysates (Table 2). Blends were made so that the total amount of all three components always equaled 5 g L−1.
Cell maintenance and culture
The two rCHO cell lines were maintained in suspension in 125-mL Erlenmeyer shake flasks (Corning, NY, USA) on an orbital shaker (Vision, Korea) rotating at 110 rpm, which was placed in a 5% CO2/air, humidified at 37 °C, incubator. Unless otherwise specified, the working volume in the 125-mL Erlenmeyer shake flasks was 30 mL. The medium for culture maintenance of the C11-08 cell line was IMDM supplemented with 10% dFBS. For the H-1-5 cell line, IMDM supplemented with 5% dFBS was used. Neither antibiotics nor MTX was added to the media.
For batch cultures, exponentially growing cells in shake flasks containing the maintenance medium were used as an inoculum. After centrifugation at 1,200 ×g for 5 min at 4 °C, the cells were washed with phosphate-buffered saline (PBS). The cells were pelleted again by centrifugation and were resuspended in the SFM under examination. MTX was not included in this SFM. Cells were then inoculated at a concentration of 2 × 105 cells/mL in 125-mL Erlenmeyer shake flasks containing the SFM under examination and were sampled everyday. Aliquots of spent media, after centrifugation, were taken and kept frozen at −70 °C for further enzyme-linked immunosorbent assay (ELISA) analysis.
Statistical analysis of the responses of rCHO cell lines to ten different mixtures of hydrolysates
DOE software Design-Expert® (version 7.0, Stat-Ease Inc., Minneapolis, MN, USA) was used for the analysis of responses to the hydrolysate mixtures. The program provided a mixture matrix, a fitted linear or quadratic mixture model, and a contour plot of the predicted elongation values of the two responses, maximum cell density (or time integral value of cell concentration) and maximum antibody concentration. It was then possible to find the optimal ratios of the three hydrolysates for these two responses. The numerical and graphical optimization section of this program allowed the determination of the ratio of hydrolysates that would give the best combination of cell and antibody concentrations.
Cell concentration, viability, and antibody assay
Cell concentration was estimated using a hemacytometer. Viable cells were distinguished from dead cells using the trypan blue dye exclusion method. Secreted antibody concentration was quantified by an ELISA as described previously (Jun et al. 2005; Yoon et al. 2003). For quantification of anti-4-1BB antibody, 96-well plates (Nunc) were coated with bacterially expressed 4-1BB.
Evaluation of specific growth rate and antibody productivity
The specific growth rate (μ) was based on data collected during the exponential growth phase (from day 1 to day 4). Specific antibody productivity (q) was evaluated as described earlier (Renard et al. 1988). When the antibody concentrations are plotted against the time integral values of the growth curve, the slope represents q.
Western blot analysis
Culture supernatants, including 0.2 μg of produced antibody, from serum-free and serum-supplemented cultures were electrophoresed on an SDS-PAGE gel and transferred onto a polyvinylidene fluoride membrane (Bio-Rad, Hercules, CA, USA). Membranes were probed with an anti-human whole IgG antibody-peroxidase conjugate (Sigma). After washing the blots with PBS-T buffer (phosphate-buffered saline containing 0.1% Tween-20), bands were visualized by the ECL Western blotting system (Amersham, Uppsala, Sweden).
Results
Cell growth and antibody production in SFM supplemented with a mixture of hydrolysates
To determine the effect of hydrolysates in regard to cell growth and antibody production, C11-08 and H-1-5 cells were cultivated in SFM supplemented with different ratios of hydrolysates as summarized in Table 2, respectively. Cells were also cultivated in the basal SFM as control.
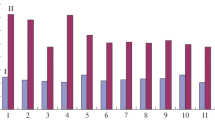
Figure 1 shows cell growth, cell viability, and antibody production profiles during batch cultures of C11-08 cells. Supplementation of soy hydrolysate, in general, showed a positive effect on cell growth (Fig. 1a). When only wheat gluten hydrolysate was supplemented to basal SFM, cell growth was not improved. However, supplementation of wheat gluten hydrolysate with other hydrolysates, particularly soy hydrolysate, improved cell growth significantly. The highest maximum viable cell concentration was achieved in media #5 which was supplemented with 50% soy hydrolysate and 50% wheat gluten hydrolysate. Compared to the basal SFM, a 1.6-fold increase in maximum viable cell concentration was achieved in media #5. Supplementation of yeast hydrolysate did not improve cell growth and rather inhibited cell growth at high concentration (≥ 3.35 g/L). Compared to the basal SFM, the maximum viable cell concentration achieved in media #1 and media #7 was decreased by 36% and 16%, respectively. Supplementation of hydrolysates, regardless of hydrolysates used, improved cell viability (Fig. 1b). Furthermore, supplementation of any combination of hydrolysates, except for wheat gluten hydrolysate only, significantly enhanced antibody production. Supplementation of yeastolate significantly enhanced q Ab. Compared with q Ab in the basal SFM, a 2.3-fold increase in q Ab was achieved in media #1 which was supplemented with yeastolate only. Accordingly, supplementation of yeastolate, despite its inhibitory effect on cell growth, resulted in improved antibody production. The highest antibody concentration was achieved in media #8 which was supplemented with 17% yeastolate, 67% soy hydrolysate, and 17% wheat gluten hydrolysate. Compared to the basal SFM, a 2.8-fold increase in maximum antibody concentration was achieved in media #8.
Batch culture of C11-08 using various SFM supplemented with the 10 mixtures of hydrolysates. a Cell growth. b Cell viability. c Antibody production. ● media 1; ○ media 2; ▼ media 3; ▽ media 4; ■ media 5; □ media 6; ◆ media 7; ◇ media 8; ▲ media 9; △ media 10;  SFM without supplementation. The compositions of the hydrolysates for the ten media are shown in Table 2. Each data point represents the mean of duplicate experiments
SFM without supplementation. The compositions of the hydrolysates for the ten media are shown in Table 2. Each data point represents the mean of duplicate experiments
The μ, maximum viable cell concentration, q Ab, and maximum antibody concentration in SFM supplemented with different ratios of hydrolysates were summarized in Table 3.
The effect of supplementation of hydrolysates on cell growth and antibody production of H-1-5 cells was similar to that of C11-08 cells as shown in supplementary Fig. S1. Supplementation of soy hydrolysate, in general, showed a beneficial effect on cell growth. Supplementation of yeastolate showed an inhibitory effect on cell growth, but a beneficial effect on antibody production. As summarized in Table 3, the highest maximum viable cell concentration of H-1-5 cells, like C11-08 cells, was achieved in media #5. The high antibody concentration was achieved in media #7, #8, and #10 which are supplemented with different ratios of all three hydrolysates.
Design of hydrolysate mixtures using statistical analysis
To determine the ratios of hydrolysates yielding the highest maximum viable cell concentration or the highest antibody concentration, the data in Table 3 were analyzed using DOE software.
Figure 2 shows contour plots of experimental region. In a contour plot, overall trends of response of cells to the media formulation can be seen through the line of the same response points predicted by the program. For both C11-08 and H-1-5 cells, mix S1, reconstituted for the highest maximum viable cell concentration, contains 60% of soy hydrolysate and 40% of wheat gluten hydrolysate. Mix S2, reconstituted for the highest antibody concentration, somewhat differed between the two cell lines. Mix S2 for C11-08 cells contains 52% of yeastolate, 18% of soy hydrolysate, and 30% of wheat gluten hydrolysate. For H-1-5 cells, mix S2 contains 70% of yeastolate and 30% of wheat gluten hydrolysate.
Contour plots analyzed using Design-Expert®. a Maximum viable cell concentration of C11-08. b Maximum antibody concentration of C11-08. c Maximum viable cell concentration of H-1-5. d Maximum antibody concentration of H-1-5. Numerical values on the line mean the predicted ratios to the responses of SFM
To confirm the effect of the supplementation of mix S1 and mix S2 on cell growth and antibody production, C11-08 and H-1-5 cells were cultivated in SFM supplemented with mix S1 and mix S2, respectively. Cells were also cultivated in the basal SFM as control.
Figure 3 shows cell growth and antibody production profiles during batch cultures. Both cell lines showed substantially higher cell growth in mix S1 than in mix S2 or the basal SFM. Furthermore, antibody production in mix S1 and mix S2 was also substantially higher than that in the basal medium, demonstrating the potential of DOE for determining the optimal ratio of different hydrolysates.
Batch cultures of two rCHO cell lines using SFM supplemented with mix S1 and mix S2. a Cell growth of C11-08. b Antibody production of C11-08. c Cell growth of H-1-5. d Antibody production of H-1-5. SFM supplemented with: ● mix S1; ○ mix S2; ▽ without supplementation. Each data point represents the mean of duplicate experiments
The μ, maximum viable cell concentration, q Ab, and maximum antibody concentration in mix S1, mix S2, and the basal SFM were summarized in Table 4.
Cell maintenance in SFM supplemented with mix S1 and mix S2
In experiments to design the SFM supplemented with the mixture of hydrolysates shown in Figs. 1 and 3, cells grown in a serum-containing medium were used as an inoculum. Accordingly, it is necessary to test whether the developed SFM supplemented with a mixture of hydrolysates supports cell growth during successive passages. To test this, cells were inoculated at a concentration of 2 × 105 cells mL−1 in 125-mL Erlenmeyer shake flasks containing the SFM supplemented with mix S1, the SFM supplemented with the mix S2, and the basal SFM, respectively. Cells were cultivated in a repeated batch mode by being re-inoculated at a concentration of 2 × 105 cells mL−1 every 3 days for six consecutive passages.
Figure 4 shows the cell growth profile of C11-08 cells during successive passages. Cell growth rate was not deteriorated during successive passages, suggesting that cells can be maintained in these SFM supplemented with hydrolysates. The average μ during six passages in SFM supplemented with mix S1, which was the highest, was 0.60 ± 0.04 day−1 (average ± standard deviation, n = 6). The average μs in the SFM supplemented with mix S2 and the basal SFM were 0.53 ± 0.05 and 0.43 ± 0.10 day−1, respectively.
Figure 5 shows the cell growth profile of H-1-5 cells during successive passages. Like the C11-08 cells, cell growth of H-1-5 cells could be maintained in the SFM supplemented with hydrolysates and the basal SFM. The average μs in the SFM supplemented with mix S1, mix S2, and the basal SFM were 0.64 ± 0.08, 052 ± 0.04, and 0.68 ± 0.06 day−1, respectively.
Cell growth and antibody production in the developed SFM
To determine whether cells can retain their antibody production capability after long-term maintenance in the developed SFM supplemented with a mixture of hydrolysates, cells after six passages shown in Figs. 4 and 5 were cultivated in the corresponding culture media.
Figure 6 shows cell growth and antibody production profiles during batch cultures. After long-term maintenance in the developed SFM, cell growth and antibody production capability of both C11-08 and H-1-5 cells were retained. The μ, maximum viable cell concentration, q Ab, and maximum antibody concentration in the SFM supplemented with mix S1, mix S2, and the basal SFM were summarized in Table 5.
Batch culture of the two CHO cell lines using SFM supplemented with mix S1 and mix S2 after serum-free adaptation for 6 passages. a Cell growth of C11-08. b Antibody production of C11-08. c Cell growth of H-1-5. d Antibody production of H-1-5. SFM supplemented with: ● mix S1; ○ mix S2; ▽ without supplementation. Each data point represents the mean of duplicate experiments
Discussion
SFM is a regulatory requirement for the commercial production of therapeutic antibodies from rCHO cell culture. However, due to the diverse nutritional requirements of cell lines, there is a need to formulate a SFM for each particular cell line, which is labor-intensive and time-consuming. Furthermore, the use of SFM often results in a decrease of specific growth and productivity. To fortify SFM, non-animal-derived hydrolysates with low cost, such as soy hydrolysate, wheat gluten hydrolysate, and yeastolate, have been used as additives for the production of recombinant proteins in rCHO cell culture (Franek et al. 2000; Sung et al., 2004).
In this study, we investigated a time-efficient method of developing SFM for therapeutic antibody production from rCHO cells. In this method, statistically designed mixtures of three non-animal-derived hydrolysates, including soy hydrolysate, wheat gluten hydrolysate, and yeastolate, were added to chemically defined SFM, which had been developed in our laboratory, and their efficacy was assessed using DOE. It takes less than 1 month to develop the SFM supplemented with hydrolysates for a specific rCHO cell line producing a therapeutic antibody.
Optimized media formulations by DOE did not result in a dramatic increase in antibody production, compared to some of the test media. It is likely that some of the initial test media are already optimal. We made a significant effort in developing the basal SFM. The basal SFM, of which the complete composition is listed in Table 1, supports cell growth of many rCHO cell lines. When rCHO cells grown in serum-containing medium were inoculated into the test media, cells could grow well without a noticeable lag phase (refer to Fig. 1 and Fig. S1).
The major advantage of DOE used in this study, compared to full-factorial designed experiments, is that we can analyze the overall effect of three kinds of hydrolysates through a limited number of experiments. Depending on the purpose of the medium, the mixture ratios of the hydrolysates can be easily determined using the DOE, for example, the mixture ratios yielding the highest maximum viable cell concentration, the highest antibody concentration, the highest μ, the q, or the combination of the two parameters with equal importance, etc.
For establishment of a rCHO cell line producing antibodies, the two most frequently used dhfr-deficient CHO host cell lines are DXB11 and DG44. The DXB cell line, originally derived from CHO-K1, contains a single point mutant allele, and the other dhfr allele has been deleted (Urlaub and Chasin 1980). The DG44 cell line, derived from CHOpro3− cells, is a double deletion mutant that contains no copies of the hamster dhfr gene (Urlaub et al. 1986). In our experience, DXB11 is easier to grow in suspension than DG44, but we have observed a reversion of DXB11 during adaptation to serum-free suspension culture. By contrast, no revertants of DG44 have been seen.
In this study, two rCHO cell lines derived from these two different host cell lines, C11-08 and H-1-5, were used for the development of SFM supplemented with hydrolysates.
Both rCHO cell lines grew well in the basal SFM of which the individual component is listed in Table 1. This result suggests that the basal SFM is likely to support cell growth of most of rCHO cell lines. Fortification of the basal SFM by hydrolysates significantly improved cell growth and antibody production, although the effect of hydrolysate mixtures depends on their source and mix ratios. The mixture of soy hydrolysate and wheat gluten hydrolysate showed a positive effect on cell growth, whereas the mixture including a high portion of yeastolate enhanced product concentration by increasing the q Ab.
It may be thought that the enhanced q Ab in SFM containing a high proportion of yeastolate was due to elevated osmolality. The osmolality of the basal SFM was 327 ± 2.8 mOsm/kg. Addition of hydrolysates (5 g L−1) increased the osmolality by about 11–31 mOsm/kg. Thus, the osmolality of all media used in this study was in the range of 327 to 358 mOsm/kg. Previously, it was reported that the growth and q of rCHO was not significantly affected in this range of osmolality (Kim et al. 2000; Ryu et al. 2000, 2001). Thus, elevated osmolality by the addition of yestolate is not a major factor for the enhanced q Ab.
The addition of 5 g L−1 of hydrolysates to the SFM resulted in an increase of total amino acid concentration. However, total amino acid concentrations in all SFM were not significantly different according to the product data sheet of the hydrolysates. When tested on mouse hybridoma culture, individual peptide fractions of soy and wheat gluten varied significantly in their growth-promoting and production-enhancing activities (Franek et al. 2000). The differences in the kind and concentration of peptides among hydrolysates may be the reason for the different effect on cell growth and antibody production.
The quality of the produced antibodies was not studied in depth. However, for quantification of anti-4-1BB antibody, we used an antigen-coated ELISA which showed the functional property of the antibody. In addition, Western blot analysis showed that the fully assembled antibody (H2L2) was the major portion of the antibodies produced in SFM supplemented with hydrolysates as shown in supplementary Fig. S2.
The application of the method of developing SFM supplemented with hydrolysates is not just limited to rCHO cells producing therapeutic antibodies. Using this method, we successfully developed SFM for rCHO cell lines producing various proteins such as erythropoietin, thrombopoietin, and β-interferon (data not shown).
In conclusion, SFM with an optimized composition of hydrolysates for a specific rCHO cell line, which is accomplished using DOE, can be made in 1 month. This time-efficient method of formulating SFM will be useful for the process development of antibody production from rCHO cells.
References
Chiou MJ, Chen LK, Peng KC, Pan CY, Lin TL, Chen JY (2009) Stable expression in a Chinese hamster ovary (CHO) cell line of bioactive recombinant chelonianin, which plays an important role in protecting fish against pathogenic infection. Dev Comp Immunol 33:117–126
Chun BH, Kim JH, Lee HJ, Chung NH (2007) Usability of size-excluded fractions of soy protein hydrolysates for growth and viability of Chinese hamster ovary cells in protein-free suspension culture. Bioresour Technol 98:1000–1005
Donaldson MS, Shuler ML (1998) Low-cost serum-free medium for the BTI-Tn5B1-4 insect cell line. Biotechnol Prog 14:573–579
Franek F, Hohenwarter O, Katinger H (2000) Plant protein hydrolysates: preparation of defined peptide fractions promoting growth and production in animal cells cultures. Biotechnol Prog 16:688–692
Glassy MC, Tharakan JP, Chau PC (1988) Serum-free media in hybridoma culture and monoclonal antibody production. Biotechnol Bioeng 32:1015–1028
Heidemann R, Zhang C, Qi H, Rule JL, Rozales C, Park S, Chuppa S, Ray M, Michaels J, Konstantinov K, Naveh D (2000) The use of peptones as medium additives for the production of a recombinant therapeutic protein in high density perfusion cultures of mammalian cells. Cytotechnology 32:157–167
Hong HJ, Lee JW, Park SS, Kang YJ, Chang SY, Kim KM, Murthy KK, Payne JS, Yoon SK, Park MJ, Kim IC, Kim JG, Kang CY (2000) A humanized anti-4-1BB monoclonal antibody suppresses antigen-induced humoral immune response in nonhuman primates. J Immunother 23:613–621
Huang EP, Marquis CP, Gray P (2007) Development of Super-CHO protein-free medium based on a statistical design. J Chem Technol Biotechnol 82:431–441
Jun SC, Kim MS, Baik JY, Hwang SO, Lee GM (2005) Selection strategies for the establishment of recombinant Chinese hamster ovary cell line with dihydrofolate reductase-mediated gene amplification. Appl Microbiol Biotechnol 69:162–169
Keen MJ, Rapson NT (1995) Development of a serum-free culture medium for the large scale production of recombinant protein from a Chinese hamster ovary cell line. Cytotechnology 17:153–163
Kim TK, Ryu JS, Chung JY, Kim MS, Lee GM (2000) Osmoprotective effect of glycine betaine on thrombopoietin production in hyperosmotic Chinese hamster ovary cell culture: clonal variations. Biotechnol Prog 16:775–781
Majors BS, Betenbaugh MJ, Pederson NE, Chiang GG (2008) Enhancement of transient gene expression and culture viability using Chinese hamster ovary cells overexpressing Bcl-xL. Biotechnol Bioeng 101:567–578
Nam JH, Zhang F, Ermonval M, Linhardt RJ, Sharfstein ST (2008) The effects of culture conditions on the glycosylation of secreted human placental alkaline phosphatase produced in Chinese hamster ovary cells. Biotechnol Bioeng 100:1178–1192
Renard JM, Spagnoli R, Mazier C, Salles MF, Mandine E (1988) Evidence that monoclonal antibody production kinetics is related to the integral of viable cells in batch systems. Biotechnol Lett 10:91–96
Ryu JS, Kim TK, Chung JY, Lee GM (2000) Osmoprotective effect of glycine betaine on foreign protein in hyperosmotic recombinant Chinese hamster ovary cell cultures differs among cell lines. Biotechnol Bioeng 70:167–175
Ryu JS, Lee MS, Lee GM (2001) Effects of cloned gene dosage on the response of recombinant CHO cells to hyperosmotic pressure in regard to cell growth and antibody production. Biotechnol Prog 17:993–999
Schrőder M, Matischak K, Friedl P (2004) Serum- and protein-free media formulations for the Chinese hamster ovary cell line DUKXB11. J Biotechnol 108:279–292
Sung YH, Lim SW, Chung JY, Lee GM (2004) Yeast hydrolysate as a low-cost additive to serum-free medium for the production of human thrombopoietin in suspension culture of Chinese hamster ovary cells. Appl Microbiol Biotechnol 63:527–536
Urlaub G, Chasin LA (1980) Isolation of Chinese hamster ovary cell mutants deficient in dihydrofolate reductase activity. Proc Natl Acad Sci USA 77:4216–4220
Urlaub G, Mitchell PJ, Kas E, Chasin LA, Funanage VL, Myoda TT, Hamlin J (1986) Effect of gamma rays at the dihydrofolate reductase locus: deletion and inversions. Somat Cell Mol Genet 12:555–566
Wang JS, Zhao MM, Zhao QZ, Bao Y, Jiang YM (2007) Characterization of hydrolysates derived from enzymatic hydrolysis of wheat gluten. J Food Sci 72:C103–107
Yoon SK, Kim SH, Lee GM (2003) Effect of low culture temperature on specific productivity and transcription level of anti-4-1BB antibody in recombinant Chinese hamster ovary cells. Biotechnol Prog 19:1383–1386
Acknowledgment
This research was supported in part by the grant from the Ministry of Knowledge Economy, Korea.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Materials
Below is the link to the electronic supplementary material.
ESM 1
(DOC 397 kb)
Rights and permissions
About this article
Cite this article
Kim, S.H., Lee, G.M. Development of serum-free medium supplemented with hydrolysates for the production of therapeutic antibodies in CHO cell cultures using design of experiments. Appl Microbiol Biotechnol 83, 639–648 (2009). https://doi.org/10.1007/s00253-009-1903-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-009-1903-1