Abstract
Enzymes are able to perform reactions under mild conditions, e.g., pH and temperature, with remarkable chemo-, regio-, and stereoselectivity. Because of this feature, the number of biocatalysts used in organic synthesis has rapidly increased during the last decades, especially for the production of chiral compounds. The present review highlights biotechnological processes for the production of chiral alcohols by reducing prochiral ketones. These reactions can be catalyzed by either isolated enzymes or whole cells that exhibit ketone-reducing activity. The use of isolated enzymes is often preferred because of a higher volumetric productivity and the absence of side reactions. Both types of catalysts have also deficiencies limiting their use in synthesis of chiral alcohols. Because reductase-catalyzed reactions are dependent on cofactors, one major task in process development is to provide an effective method for regeneration of the consumed cofactors. In this paper, strategies for cofactor regeneration in biocatalytic ketone reduction are reviewed. Furthermore, different processes carried out on laboratory and industrial scales using isolated enzymes are presented. Attention is turned to process parameters, e.g., conversion, yield, enantiomeric excess, and process strategies, e.g., the application of biphasic systems or methods of in situ (co)product recovery. The biocatalytic production of chiral alcohols utilizing whole cells is presented in part II of this review (Goldberg et al., Appl Microbiol Biotechnol, 2007).
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
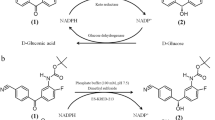
Today, chiral compounds are the most important building blocks in the chemical and pharmaceutical industry for the production of, for example, chemical catalysts, liquid crystals, flavors, agrochemicals, or drugs (Daußmann et al. 2006a). Optical active secondary alcohols are especially widely used as intermediates for the introduction of chiral information into the product. In the industry, usually well-established chemical methods are used (Blaser et al. 2003), but in the last decades, the interest for creating stereogenic centers by applying biocatalytic methods has risen (Honda et al. 2006). In particular, the number of industrial processes using alcohol dehydrogenases (ADHs) is increasing (Breuer et al. 2004; Buchholz and Gröger 2006; Liese et al. 2006). These biocatalysts, used as isolated enzymes or whole cells, catalyze the stereoselective reduction of prochiral ketones with remarkable chemo-, regio-, and stereoselectivity (Fig. 1; Wandrey 2004).
Biocatalytic production of chiral alcohols
The biocatalytic production of chiral alcohols is possible following several pathways. Examples with enzymes from the classes oxidoreductases (EC 1), hydrolases (EC 3), and lyases (EC 4) are described in literature (Fig. 2).
Oxidoreductases catalyze redox reactions; thus, the transfer of electrons from or to the substrate. The reduction of ketones by, for example, ADHs (Peters 1998; also referred to as carbonyl reductases [CR] or ketone reductases) is the issue of this review and will be explained in detail later. Moreover, alcohols can also be obtained by oxidation of CH bonds by monooxygenases (van Beilen et al. 2003).
Hydrolases play the most important role in industrial enzymatic processes (Liese et al. 2006). Between 1987 and 2003, around 60% of the published articles were dealing with enzymes of this class (Faber 2004). The lipases as a subgroup of the hydrolases catalyze the hydrolytic cleavage of the carbon–oxygen single bonds in esters or analogous carboxylic derivatives to produce acids and alcohols (Bornscheuer et al. 1994; Jaeger et al. 1997; Reetz et al. 1997; Jaeger and Eggert 2002; Detry et al. 2006; Elend et al. 2006). In most cases, chiral products are obtained by the (dynamic) kinetic resolution of racemic mixtures.
Alcohols can also be obtained by using phosphate or sulphate esters as a substrate for phosphatases or sulphatases (Wallner et al. 2005), respectively. However, these enzymes are only rarely used for the production of alcohols (Faber 2004).
Lyases catalyze the formation of carbon–carbon bonds. By setting ketones or aldehydes as a substrate, alcohols are available (Adam et al. 1999; Pohl and Liese 2006). A widespread catalyst for this reaction is the benzaldehyde lyase (BAL), which produces hydroxyketones from two aldehydes (Demir et al. 2002; Kihumbu et al. 2002; Kurlemann and Liese 2004; Domínguez de María et al. 2006; Hildebrand et al. 2006; Stillger et al. 2006).
Other examples are the pyruvate decarboxylase, which couples, for example, acetaldehyde and benzaldehyde to phenylacetylcarbinol (Rosche et al. 2002; Leksawasdi et al. 2004) or the deoxyriboaldolase that is able to catalyze the aldol condensation with chloroacetaldehyde with two molecules of acetaldehyde in the industrial production of statines (Liu et al. 2004; Müller 2005).
Another group of enzymes to be mentioned are the hydroxynitrile lyases. The coupling of ketones with hydrogen cyanide leads to nitrile substituted alcohols (Griengl et al. 1997; Avi et al. 2004; Fechter and Griengl 2004; Gaisberger et al. 2004).
Cofactor regeneration
The majority of ADHs are dependent on the nicotinamide cofactors β-1,4-nicotinamide adenindinucleotide (NADH) or β-1,4-nicotinamide adenindinucleotide phosphate (NADPH). Rarely, enzyme-bound cofactors from the group of the flavines (FAD; Walsh 1980) or methoxatines (pyrroloquinoline quinine, PQQ; McWhirter and Klapper 1990) are found. Because all these cofactors are too expensive to be used stoichiometrically—with prices ranging from €1,300 per mol for NAD+ to more than €2,700,000 per mol for PQQ (Leonida 2001)—there has been significant interest in developing efficient cofactor regeneration processes (Wichmann and Vasic-Racki 2005; Lütz 2006). FAD- and PQQ-dependent enzymes are seldomly used in preparative synthesis (Liese et al. 2006) and thus not considered in this review.
Cofactor regeneration is carried out parallel to the conversion of substrate to product (Fig. 3). In the case of the ADHs, the production of NADPH can be carried out by means of chemical, electrochemical, photochemical, and enzymatic methods. A further approach to cofactor regeneration in whole-cell biotransformation processes is the usage of the metabolism of cultivated or resting cells utilizing glucose or other compounds as nutrient (Haberland et al. 2002).
Electrochemical regeneration
Cofactors only switch between the oxidized and reduced state, so electrochemical methods are appealing. Direct cathodic reduction (Simon et al. 1985; Biade et al. 1992) of NADP+ suffer from low regioselectivity and side reactions because of high overpotentials (Hollmann and Schmid 2004). Therefore, organic and metal containing electron shuttles for the transfer of electrons between electrode and NADP+ or NADPH, respectively, were developed (Steckhan et al. 1990, 1991).
Typical organic electron shuttles (Fig. 4) are based on large conjugated systems. In the case of methyl viologen (MV, 1), enzymes like ferredoxin NADP+ reductase for NADPH or diaphorase for NADH are necessary to transfer the reduction equivalents from the viologen to NADP+ (Yuan et al. 1997).
In the glycerol dehydrogenase-catalyzed oxidative resolution of racemic 1,2-diols 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonate (2) is applied to regenerate NAD+ (Schröder et al. 2003; Degenring et al. 2004). Further examples are 1,10-phenanthroline-5,6-dione (3) or its N-monomethylated derivatives (Hilt et al. 1997).
Besides the application of organic reduction equivalents, metal-containing electron shuttles were described: for example, [Cp*Rh(bpy)(H2O)]2+ (Cp* = C5Me5, bpy = 2,2′-bipyridine) performs fast and quantitative reductions of NADP+ to NADPH (Steckhan et al. 1990; Vuorilehto et al. 2004; Hollmann et al. 2006).
Chemical regeneration
Inorganic salts like sodium dithionite (Na2S2O4; Jones et al. 1972) offer easy handling but suffer at the same time from enzyme deactivation processes at higher salt concentrations (Raunio and Lilius 1971).
Since the 1980s, the first examples utilizing hydrogen and metal complexes (Abril and Whitesides 1982; Wagenknecht et al. 2003) were described. More often than not, late transition metals like rhodium, ruthenium, and platinum and their complexes are applied.
A similar approach is the combination of a platinum carbonyl cluster with the dye safranine (4, Fig. 5) in a two-phase system (Bhaduri et al. 1998; Lütz 2006).
Photochemical regeneration
Homogeneous photosensitizers such as ruthenium or zinc complexes, dyes like methylene blue, and heterogeneous semiconductor powders and colloids like cadmium sulfide or titanium dioxide have been used for the light-induced production of methyl viologen and subsequent regeneration of NADPH in the presence of dihydrolipoamide dehydrogenase or ferredoxin reductase, respectively (Julliard 2004; Willner et al. 1990; Rickus et al. 2002).
Enzymatic regeneration
For this method, there are two different approaches: the enzyme-coupled and the substrate-coupled process (Hummel and Kula 1989).
The enzyme-coupled approach uses a sacrificial cosubstrate that is converted by a second enzyme in the opposite redox direction. Several methods are established in literature, some examples are explained in detail:
-
Formate oxidation by formate dehydrogenase (FDH)
A widespread NADPH regeneration system is the oxidation of formate to carbon dioxide by FDH (Hummel and Kula 1989; Seelbach et al. 1996; Tishkov et al. 1999). A process for the production and purification of FDH from Candida boidinii in large scale has been developed (Weuster-Botz et al. 1994), and the enzyme is used in the industrial production of l-tert-leucine (Bommarius et al. 1995). Advantages are inertness, readily removal of the coproduct carbon dioxide and thereby a favorable thermodynamic equilibrium, good availability, low cost of the FDH, and the cheap cosubstrate formate. In some cases, the change of pH during the cofactor regeneration causes inhibitory effects (Nidetzky et al. 1996); this can be overcome by applying a fed-batch process (Neuhauser et al. 1998). Most of the FDHs are NADH dependent, while many ADHs are NADPH dependent. In most cases, the lower activity of the FDH towards NADPH is tolerated (Ernst et al. 2005). An approach to overcome this drawback is the use of transhydrogenases that can move reduction equivalents between NADH and NADPH (Weckbecker and Hummel 2004).
-
Glucose dehydrogenase (GDH) or glucose-6-phosphate dehydrogenase (G6PDH)
The GDH or G6PDH oxidizes the cosubstrates glucose or glucose-6-phosphate, respectively, and is suited for the regeneration of NADPH (Kizaki et al. 2001; Weckbecker and Hummel 2005). Both enzymes are rather inexpensive, highly active, and stable. Because of the hydrolysis of the coproducts gluconolactone or 6-phosphogluconolactone to the corresponding acids, the reaction is nearly irreversible. The natural preference for NADP+ is a benefit for the use of these enzymes as cofactor-regenerating enzymes. A drawback is the high cost of glucose-6-phosphate and the demand for permeabilization when whole cells are applied as biocatalysts (Makino et al. 1989; Kataoka et al. 1998).
-
Alcohol oxidation by ADH
For the enzyme-coupled cofactor regeneration applying a second ADH, only few examples are known (Leonida 2001). The reduction of pyruvate to l-lactate can also be used for the cofactor regeneration in enzyme-coupled processes (Kim and Whitesides 1988; Liese et al. 1996).
-
Direct reduction of NADP+ with hydrogen/hydrogenase
Hydrogenases are bidirectional enzymes that catalyze the production and oxidation of molecular hydrogen. The direct regeneration of NADPH using this enzyme is probably the most elegant solution, as the molecule is consumed completely and no coproduct is formed (Klibanov and Puglisi 1980; Wong et al. 1981; Greiner et al. 2003; Mertens et al. 2003; Mertens and Liese 2004).
-
Phosphite dehydrogenase (PTDH)
The oxidation of phosphite to phosphate with the concomitant reduction of NAD+ to NADH is another approach to cofactor recycling (Vrtis et al. 2002). This enzyme can also use NADP+ as a cofactor but with a lower activity (Costas et al. 2001). PTDH has been combined with several dehydrogenases such as lactate dehydrogenase, horse liver alcohol dehydrogenase (HLADH), malate dehydrogenase (Vrtis et al. 2002), xylose reductase, and ADH (Johannes et al. 2007).
The enzyme-coupled process requires the application of two enzymes at the same time. The substrate-coupled approach, thus applying only one enzyme for the production of the desired compound and the cofactor regeneration, is a powerful alternative in comparison to the enzyme-coupled approach (Tishkov et al. 1999; Stillger et al. 2002).
The auxiliary cosubstrate in the substrate-coupled process for the production of chiral alcohols is in most cases 2-propanol, which is oxidized to the coproduct acetone. Because there is a competition between substrate, product, cosubstrate, and coproduct, a thermodynamic equilibrium is present; hence, the maximum conversion is limited by the thermodynamics of the system. In situ (co)product removal (ISPR) processes like organophilic pervaporation, gassing out, reduced pressure, destillation, or crystallization can be applied to shift the equilibrium (Lye and Woodley 1999; Stark and von Stockar 2003; Takors 2004; von Scala et al. 2005; Buque-Taboada et al. 2006; Goldberg et al. 2006).
Reduction of prochiral ketones catalyzed by isolated enzymes
The use of isolated enzymes as biocatalysts offers some advantages in comparison to whole cells. Because of the presence of only one or two enzymes (in case of enzyme-coupled cofactor regeneration approaches), side reactions can be avoided and thus the reduction of enantioselectivity. Therefore, downstream processing can be simplified. In comparison to processes catalyzed by whole cells, diffusion limitations do not occur. On the other hand, there is a need for an addition of cofactors to the process when isolated ADHs are applied. Furthermore, isolated enzymes may show high sensibility towards high concentrations of substrates and organic solvents (Liese et al. 1998; Schmid et al. 2001; Villela et al. 2003). Advantages of isolated dehydrogenases over their natural whole-cell biocatalysts have been reviewed several times (Kula and Kragl 2000; Hummel et al. 2003; Faber 2004). The decision on the application of isolated enzymes or whole cells depends on the specific requirements of the industrial reaction system.
The following section shows processes carried out in industrial and laboratory scales using isolated ADHs in a substrate-coupled approach or in combination with a second enzyme for cofactor regeneration. Different strategies for overcoming limitations and improving process parameters like conversion and space–time yield as well as for increasing the stability of biocatalysts should be pointed out.
Enzyme-coupled cofactor regeneration
Bristol-Myers Squibb uses an ADH from Acinetobacter calcoaceticus as catalyst for the production of 6-benzyloxy-(3R,5S)-dihydroxy-hexanoic acid ethyl ester, which is a key chiral intermediate for anticholesterol drugs. Cofactor NAD+ is added to the reaction medium together with GDH that catalyzes the regeneration of NADH. The producing ADH is applied as crude cell extracts. The process is carried out in a batch process with a yield of 92% and an enantiomeric excess (ee) of 99% (Patel et al. 1993). Furthermore, Codexis published the use of ADH and GDH, for example, for the production of ethyl-(S)-4-chloro-3-hydroxybutyrate. The process is carried out with almost complete conversion (>99.5%) and an ee greater than 99.9%. In this case, the enzymes have been optimized by directed evolution to achieve the process goals (Davis et al. 2005).
There are several examples for the use of FDH as a cofactor-regenerating enzyme in industrial biocatalysis. Ciba Spezialitätenchemie AG uses (R)-lactate-NAD oxidoreductase from Staphylococcus epidermis together with FDH from C. boidinii for the synthesis of (R)-2-hydroxy-4-phenyl-butyric acid. The process is carried out in a continuously operated stirred tank reactor equipped with an ultrafiltration membrane to retain the enzymes. To avoid degassing of the side product CO2, which causes mechanical force, a pressure of 3 bar is applied. The reaction is carried out in a scale of 0.2 L with a space–time yield of 410 g L−1 day−1 and an ee of 99.9% (Schmidt et al. 1992). The company Pfizer makes use of the same combination of enzymes to produce (R)-3-(4-fluorophenyl)-2-hydroxypropanoate in an industrial scale. This reaction is also carried out in a continuously operated enzyme membrane reactor (EMR) with a scale of 2.2 L. This process is also characterized by a good space–time yield of 560 g L−1 day−1 and an ee of 99.9% (Tao and McGee 2002).
The two described industrial processes with enzyme-coupled regeneration of cofactors via FDH are both characterized by a simple reactor setup and an easy reaction strategy. Some interesting processes dealing with enzyme-coupled cofactor regeneration via FDH and two phase systems had been published, which are described in detail below. The main goal of applying biphasic systems is the feasibility of increasing the amounts of hydrophobic substrates to reach higher productivities. Beside biphasic systems, other methods like the usage of cyclodextrin-containing buffers as reaction medium have been applied to achieve higher concentrations of poorly soluble substrates (Zelinski et al. 1999).
A simple way to create an aqueous/organic biphasic system is the formation of microemulsions. Depending on the mixture and temperature, small droplets of the nonaqueous phase are dispersed in water, or in case of an oil-rich emulsion, reverse micelles of water can be dispersed in oil. Orlich et al. (2000) reported the application of reverse micelles for ADH-catalyzed reduction of ketones in a enzyme-coupled approach (Fig. 6). The microemulsion contained water, cyclohexane, and Marlipal 013-16 as the surfactant. The stability of the investigated ADHs and FDH where both increased in comparison to former aqueous/organic biphasic systems known from literature so far. Furthermore, it was possible to perform successful semibatch experiments reducing a prochiral ketone (2-butanone) in full conversion and enantioselectivity.
Liese et al. (1998) reported the enantioselective reduction of 2-octanone catalyzed by Candida parapsilosis CR (CPCR). To increase the substrate solubility, an emulsion membrane reactor was developed, which consists of two reactor units. The first one was a stirred emulsion vessel in which the aqueous phase was separated from the organic phase by a hydrophilic ultrafiltration membrane. The organic phase was pure substrate. The substrate saturated the aqueous phase then entered an EMR where the substrate 2-octanone was reduced to (S)-2-octanol by the CPCR (Fig. 7). By this reactor design, the product can by extracted into the organic phase, which is beneficial for the driving force of the reaction equilibrium and for downstream processing. Furthermore, there is no direct contact between the enzyme and an organic interphase. By applying this reactor design, poorly soluble ketones can be supplied in higher concentrations compared to reactor systems with only one aqueous phase. In the emulsion membrane reactor, conversion of 91%, a space–time yield of 11 g L−1 day−1, and an ee greater than 99.5% were obtained. In contrast to the latter two-phase reactor system, a continuously operated EMR could be operated over a period of more than 4 months at a space–time yield of 21.1 g L−1 day−1 (97% conversion, total turnover number [ttn]=13.6) in the range of solubility of the reactants. In the case of the emulsion reactor, the ttn could be increased to 124, because of the higher concentration of substrate that can be applied. Furthermore, downstream processing was simplified because the product is concentrated in the organic phase.
To avoid direct contact between the aqueous and the organic phase, biphasic systems can be operated with membrane modules. Kruse et al. (1996) developed a continuously operated reactor system of three main cycles. The EMR cycle represents a continuously operated loop reactor supplied with a hollow-fiber ultrafiltration membrane. The hydrophobic product of the enzyme-catalyzed process is extracted by means of a hydrophobic membrane into the hexane cycle, making the recycling of the hydrophilic cofactor possible in the NAD+ cycle. Thus, the residence time of the cofactor can be decoupled from the residence time of the substrate (Fig. 8). Chiral hydrophobic alcohols like (S)-1-phenylpropan-2-ol, (S)-4-phenylbutan-2-ol, and (S)-6-methylhept-5-en-2-ol have been produced in this reactor setup by using ADH from Rhodococcus erythropolis together with FDH from C. boidinii. For the three processes, conversions of 72, 80, and 65% could be achieved. The ee was higher than 99% in all cases. The described reactor setup led to ttns for NAD+ up to 1,350 \( {\text{mol}}_{{\text{P}}} \;{\text{mol}}^{{ - 1}}_{{{\text{NAD}}}} , \) which means a 25-fold increase in comparison to standard techniques without cofactor retention (Kruse et al. 1996).
In a similar reactor performance the reduction of acetophenone was carried out. The reaction was catalyzed by CPCR in a enzyme-coupled approach with FDH. The process has been compared to chemical borane reduction using a homogeneously soluble polymer-bound oxazaborolidine catalyst. The biological method yielded (S)-phenylethanol in ee >99% with a space–time yield of 88 g L−1 d−1. Although the chemical methods provided higher space–time yield (1,400 g L−1 d−1), it became obvious, that the chemical method offers some disadvantages in terms of ee, which was only 94%, and stability of the catalyst (Rissom et al. 1999).
Mertens et al. (2003) reported direct regeneration of NADPH with a hydrogenase from Pyrococcus furiosus. The enzyme is capable of generating the NADPH directly from the oxidized NADP+ without producing any by-products other than protons. Thus, the enzyme not only offers the feasibility to catalyze the enzyme-coupled in situ regeneration of NADPH but also the synthesis of NADPH. By applying an ADH from Thermoanaerobium sp. together with the hydrogenase from P. furiosus, acetophenone and (2S)-hydroxy-1-phenyl-propanone have been reduced enantioselectively to the corresponding alcohol with complete conversion and an ee greater than 99.5%.
Substrate-coupled cofactor regeneration
For the use of isolated enzymes in biotransformation processes with substrate-coupled cofactor regeneration, it is essential that the enzyme can withstand elevated concentrations of cosubstrate. HLADH and ADH from Lactobacillus brevis (LbADH; Hummel 1997) are known to be able to operate at rather high alcohol concentrations. For that reason, these enzymes were used as catalysts for the enantioselective preparation of propargylic alcohols in a substrate-coupled approach (Schubert et al. 2002). Recently, Kosjek et al. (2004) reported the purification and characterization of a new chemotolerant ADH-‘A’ from Rhodococcus ruber DSM 44541. This enzyme can withstand acetone concentrations up to 50% v/v and 2-propanol concentrations even up to 80% v/v. Because of this feature, this enzyme is a very interesting biocatalyst for synthesis in a substrate-coupled process.
Successful efforts have been done to make biocatalysts more stable against organic solvents. HLADH was crystallized in the presence of NADH. The resulting crystals were then treated with glutardialdehyde to yield cross-linked enzyme crystals. These crystals exhibited high stability towards organic solvents like 2-propanol in comparison to solubilized HLADH (St. Clair et al. 2000).
A good operating stability can also be achieved by immobilizing the ADHs. Hildebrand and Lütz (2006) reported the reduction of acetophenone by immobilized LbADH in a plug flow reactor. The enzyme has been immobilized on Sepabeads®. By optimizing the immobilization procedure, a 60-fold increase in stability in comparison to soluble enzyme could be achieved. With the immobilized LbADH, a plug-flow-reactor could be operated for more than 10 weeks. During that time, conversion could be held constant at 60% with an ee greater than 99.5% and a space–time yield of 30 g L−1 day−1.
Isolated LbADH was also applied for the reduction of more complex ketones in a substrate-coupled cofactor regeneration approach. 5-Hydroxy-3-oxocarboxylates are valuable intermediates in the synthesis of chiral building blocks such as 3,5-dihydroxycarboxylates and β-keto δ-lactones. A preparative-scale reduction of tert-butyl 3,5-dioxohexanoate resulted in 99.4% ee, complete regioselectivity, and 77% isolated yield of the corresponding alcohol. Furthermore, a chloroderivate of the same compound was reduced by LbADH. The product tert-butyl-(S)-6-chloro-5-hydroxy-3-oxohexanoate could be produced with greater than 99.5% ee and 72% isolated yield (Wolberg et al. 2000).
Nevertheless, some biotransformation processes with substrate-coupled cofactor regeneration demand removal of the coproduct acetone because acetone not only affects the activity of the enzyme but also causes a thermodynamic limitation. Stillger et al. (2002) used two different strategies for in situ acetone removal in the biocatalytic reduction of ethyl 5-oxohexanoate to (S)-ethyl 5-hydroxyhexanoate catalyzed by CPCR. In a first reactor setup, acetone has been stripped out from the reactor system by gassing the aqueous solution with humidified compressed air. Because of its low volatility, acetone can be removed effectively from a reaction system by this method. Compared to batch reactions without acetone removal, the conversion could be increased from 75% to more than 97%. In both cases, an ee of 99.5% could be achieved. In another approach, acetone was removed by a pervaporation step (Fig. 9) resulting in a conversion of 95% with an ee of 99.5% (Stillger et al. 2002).
Acetone removal is not restricted to biotransformation processes on laboratory scale. It is also realized as an important element in industrial processes. Wacker Fine Chemicals produces several β-keto esters on ton scale (Daußmann et al. 2006b). Isolated LbADH is used as biocatalyst for the synthesis of (R)-ethyl-3-hydroxybutyrate in a process with substrate-coupled cofactor regeneration via oxidation of 2-propanol. The coproduct acetone is removed by continuous stripping. This strategy shifts the equilibrium reaction towards complete conversion and simplifies downstream processing. Furthermore, it allows a continuous reuse of the aqueous phase in standard reactor vessels. The process is carried out with a yield of 96%, an ee of 99.8%, and a space–time yield of 92 g L−1 day−1 (Rosen et al. 2004). The synthesis of (R)-methyl-3-hydroxybutyrate is also catalyzed by LbADH in a substrate-coupled approach. The process is performed in a stirred batch reactor with a yield of 94% and an ee of 99.8%. Acetone is evaporated continuously under reduced pressure (Daußmann et al. 2006c).
The main problem of using enzymes as biocatalysts for the reduction of prochiral ketones is the low solubility of many ketones in aqueous solutions. For that reason, the creation of a biphasic system is preferable for the reduction of some hydrophobic substrates. As the occurrence of liquid–liquid interphases and presence of residual amounts of organic solvent in water can lead to deactivation of the biocatalyst, the low enzyme stability is a major drawback in biphasic enzymatic reactions. Villela et al. (2003) investigated the stability of three different ADHs in biphasic systems. Several solvents with different properties have been used as organic phase, e.g., nonane, cyclohexane, toluene, dichloromethane, tert-butyl methyl ether (MTBE) and ethyl acetate. It has been investigated if the logP value, which characterizes the polarity of an organic solvent, can be used as parameter to guide the solvent choice. It could be shown that ADHs show good stability in biphasic systems with MTBE as the organic solvent; in systems containing dichloromethane as the organic solvent, the stabilities of the enzymes were rather low. There was no correlation found between the polarity of the organic solvent and the influence on the stability of an enzyme. In addition to the hydrophobicity of a solvent, its functionality should be considered as well when screening for a solvent that should be applied in a biphasic system.
When a biphasic system is applied for enzymatic reactions, the nonaqueous phase cannot only be used as a substrate reservoir but also for the continuous extraction of products and the coproduct acetone. A novel approach is the use of ionic liquids as the nonaqueous phase in enzymatic biphasic systems (Eckstein et al. 2004). The partition coefficients of 2-propanol and acetone in a biphasic system containing buffer and the ionic liquid [BMIM][(CF3SO2)2N] (1-butyl-3-methylimidazolium bis((trifluoromethyl)sulfonyl)amide) significantly differ from their partitioning behavior in buffer/MTBE (see Table 1). LbADH has been used as a biocatalyst in biphasic systems with buffer/[BMIM][(CF3SO2)2N] and buffer/MTBE, respectively. The enzyme catalyzed the conversion of 2-octanone to (R)-2-octanol with an ee greater than 99%. Cofactor regeneration was carried out in the substrate-coupled approach using 2-propanol as cosubstrate. In the biphasic system with buffer/[BMIM][(CF3SO2)2N], the produced acetone is continuously removed from the buffer phase leading to lower acetone concentrations in the buffer phase. Because of faster cofactor regeneration, a higher reaction rate could be observed. Within the first 180 min, the reduction is much faster in the biphasic system containing the ionic liquid, leading to a conversion of 88%, while the reduction reaches a conversion of only 61% in the presence of MTBE. Other applications of ionic liquids in biocatalysis, which mainly deals with isolated lipases, have recently been reviewed (Kragl et al. 2002; van Rantwijk et al. 2003).
Enzymatic gas-phase reactions involve the conversion of a gaseous substrate to a gaseous product using a dry enzyme as the catalyst. This method offers some advantages in comparison to biocatalysis in aqueous media. Poorly water-soluble substrates can be applied as volatile compounds. The dilute product then can easily be recovered using fractionated condensation (Lamare et al. 2004). LbADH and ADH-T from Thermoanaerobacter sp. (Findrik et al. 2005) were investigated regarding their ability to catalyze the reduction of acetophenone in a gas-phase reaction with substrate-coupled cofactor regeneration via oxidation of 2-propanol. By optimization of the reaction conditions water activity, cofactor-to-protein molar ratio, and reaction temperature, which all affected the initial reaction rate and also the stability of the biocatalyst, productivities of 1,000 g L−1 day−1 for LbADH and 600 g L−1 day−1 for ADH-T could be achieved (Trivedi et al. 2006). Other works are dealing with the effects of immobilization and drying the biocatalysts. It could be shown that the addition of sucrose to the cell extract before immobilization of the enzyme led to a longer half-life time of the biocatalyst (Ferloni et al. 2004). Furthermore, the method for drying the enzyme has an impact to the enzyme stability (Trivedi et al. 2005).
Optically active diols are interesting building blocks in asymmetric synthesis. They can be obtained via chemical routes, but only one of the diol stereoisomers is accessible in good stereoisomeric purity from a given starting material (Koike et al. 2000; Choudary et al. 2001). Furthermore, the enzymatic and microbial reduction of 1,2-diketones and α-hydroxyketones does not afford the synthesis of all four possible stereoisomers (Mochizuki et al. 1995; Bortolini 1997). By employing a combination of enantioselective lyases, e.g., thiamine diphosphate-dependent benzoylformate decarboxylase or BAL, and diastereoselective ADHs, it has became possible to synthesize all four 1-phenylpropane-1,2-diol stereoisomers separately in a reaction cascade (Fig. 10). As substrates, simple molecules like benzaldehyde and acetaldehyde were used (Kihumbu et al. 2002).
Conclusion and outlook
The given examples point out that oxidoreductases namely, ADHs or CRs, have become a powerful tool in the synthesis of chiral alcohols. It could be shown that by developing appropriate process engineering strategies, it became possible to remarkably optimize a couple of biocatalytic processes catalyzed by isolated enzymes, e.g., in terms of space–time yield and catalyst usage. Successful attempts have been reported for the use of ADHs in nonconventional media like biphasic systems or gas phases. Such methods offer the possibility to apply remarkable high concentrations of substrate to the biocatalyst and thus increase volumetric productivities. Furthermore, thermodynamic limitations can be overcome by applying adequate ISPR techniques. These examples clearly demonstrate that by applying reaction engineering, ADHs can be successfully used as catalysts in preparative organic chemistry and on an industrial scale for the synthesis of chiral alcohols.
References
Abril O, Whitesides GM (1982) Hybrid organometallic/enzymatic catalyst systems: regeneration of NADH using dihydrogen. J Am Chem Soc 104:1552–1554
Adam W, Lazarus M, Saha-Möller CR, Schreier P (1999) Biocatalytic synthesis of optically active α-oxyfunctionalized carbonyl compounds. Acc Chem Res 32:837–845
Avi M, Fechter MH, Gruber K, Belaj F, Pöchlauer P, Griengl H (2004) Hydroxynitrile lyase catalyzed synthesis of heterocyclic (R)- and (S)-cyanohydrins. Tetrahedron 60:10411–10418
Bhaduri S, Mathur P, Payra P, Sharma K (1998) Coupling of catalyses by carbonyl clusters and dehydrogenases: reduction of pyruvate to l-lactate by dihydrogen. J Am Chem Soc 120:12127–12128
Biade AE, Bourdillon C, Lava JM, Mairesse G, Moiroux J (1992) Complete conversion of l-lactate into d-lactate. A generic approach involving enzymatic catalysis, electrochemical oxidation of NADH, and electrochemical reduction of pyruvate. J Am Chem Soc 114:893–897
Blaser HU, Malan C, Pugin B, Spindler F, Steiner H, Studer M (2003) Selective hydrogenation for fine chemicals: recent trends and new developments. Adv Synth Catal 345:103–151
Bommarius AS, Schwarm M, Stingl K, Kottenhahn M, Huthmacher K, Drauz K (1995) Synthesis and use of enantiomerically pure tert-leucine. Tetrahedron Asymmetry 6:2851–2888
Bornscheuer U, Reif OW, Lausch R, Freitag R, Scheper T, Kolisis FN, Menge U (1994) Lipase of Pseudomonas cepacia for biotechnological purposes: purification, crystallization and characterization. Biochim Biophys Acta 1201:55–60
Bortolini OMS (1997) An easy approach to the synthesis of optically active vic-diols: a new single-enzyme system. J Org Chem 62:1854–1856
Breuer M, Ditrich K, Habicher T, Hauer B, Keßeler M, Stürmer R, Zelinski T (2004) Industrielle Verfahren zur Herstellung von optisch aktiven Zwischenprodukten. Angew Chem 116:806–843 (also in Angew Chem Int Ed 43:788–824)
Buchholz S, Gröger H (2006) Enantioselective biocatalytic reduction of ketones for the synthesis of optically active alcohols. In: Patel RN (ed) Biocatalysis in the pharmaceutical and biotechnology industries. Taylor & Francis, New York, pp 757–790
Buque-Taboada EM, Straathof AJJ, Heijnen JJ, van der Wielen LAM (2006) In situ product recovery (ISPR) by crystallization: basic principles, design, and potential applications in whole-cell biocatalysis. Appl Microbiol Biotechnol 71:1–12
Choudary BM, Chowdari NS, Madhi S, Kantam ML (2001) A trifunctional catalyst for the synthesis of chiral diols. Angew Chem 113:4755–4759 (also in Angew Chem Int Ed 40:4655–4759)
Costas AMG, White AK, Metcalf WW (2001) Purification and characterization of a novel phosphorus-oxidizing enzyme from Pseudomonas stutzeri WM88. J Biol Chem 276:17429–17436
Daußmann T, Hennemann HG, Rosen TC (2006a) Enzymatische technologien zur synthese chiraler alkohol-derivate. Chem Ing Tech 78:249–255
Daußmann T, Dünkelmann P, Lütz S (2006b) Chiral alcohols. CHEManager (Europe) 3:8
Daußmann T, Rosen TC, Dünkelmann P (2006c) Oxidoreductases and hydroxynitrilase lyases: complementary enzymatic technologies for chiral alcohols. Eng Life Sci 6:125–129
Davis C, Grate J, Gray D, Gruber J, Huismann G, Ma S, Newman L, Sheldon R (2005) Enzymatic processes for the production of 4-substituted 3-hydroxybutyric acid derivatives. Codexis, Patent no. WO04015132
Degenring D, Schröder I, Wandrey C, Liese A, Greiner L (2004) Resolution of 1,2-diols by enzyme-catalyzed oxidation with anodic, mediated cofactor regeneration in the extractive membrane reactor: gaining insight by adaptive simulation. Org Process Res Dev 8:213–218
Demir AS, Şeşenoglu Ö, Eren E, Hosrik B, Pohl M, Janzen E, Kolter D, Feldmann R, Dünkelmann P, Müller M (2002) Enantioselective synthesis of α-hydroxy ketones via benzaldehyde lyase-catalyzed C–C bond formation reaction. Adv Synth Catal 344:96–103
Detry J, Rosenbaum T, Lütz S, Hahn D, Jaeger KE, Müller M, Eggert T (2006) Biocatalytic production of enantiopure cyclohexane-trans-1,2-diol using extracellular lipases from Bacillus subtilis. Appl Microbiol Biotechnol 72:1107–1116
Domínguez de María P, Stillger T, Pohl M, Wallert S, Drauz K, Groger H, Trauthwein H, Liese A (2006) Preparative enantioselective synthesis of benzoins and (R)-2-hydroxy-1-phenylpropanone using benzaldehyde lyase. J Mol Catal B Enzym 38:43–47
Eckstein M, Villela M, Liese A, Kragl U (2004) Use of an ionic liquid in a two-phase system to improve an alcohol dehydrogenase catalysed reduction. Chem Commun 9:1084–1085
Elend C, Schmeisser C, Leggewie C, Babiak P, Carballeira JD, Steele HL, Reymond JL, Jaeger KE, Streit WR (2006) Isolation and biochemical characterization of two novel metagenome-derived esterases. Appl Environ Microbiol 72:3637–3645
Ernst M, Kaup B, Müller M, Bringer-Meyer S, Sahm H (2005) Enantioselective reduction of carbonyl compounds by whole-cell biotransformation, combining a formate dehydrogenase and a (R)-specific alcohol dehydrogenase. Appl Microbiol Biotechnol 66:629–634
Faber K (2004) Biotransformations in organic chemistry (5th edn.). Springer, Berlin
Fechter MH, Griengl H (2004) Hydroxynitrile lyases: biological sources and application as biocatalysts. Food Technol Biotechnol 42:287–294
Ferloni C, Heinemann M, Hummel W, Daußmann T, Büchs J (2004) Optimization of enzymatic gas-phase reactions by increasing the long-term stability of the catalyst. Biotechnol Prog 20:975–978
Findrik Z, Vasic-Racki D, Lütz S, Daußmann T, Wandrey C (2005) Kinetic modelling of acetophenone reduction catalyzed by alcohol dehydrogenase from Thermoanaerobacter sp. Biotechnol Lett 27:1087–1095
Gaisberger RP, Fechter MH, Griengl H (2004) The first hydroxynitrile lyase catalysed cyanohydrin formation in ionic liquids. Tetrahedron Asymmetry 15:2959–2963
Goldberg K, Edegger K, Kroutil W, Liese A (2006) Overcoming the thermodynamic limitation in asymmetric hydrogen transfer reactions catalyzed by whole cells. Biotechnol Bioeng 95:192–198
Goldberg K, Schroer K, Lütz S, Liese A (2007) Biocatalytic ketone reduction—a powerful tool for the production of chiral alcohols—part II: whole cell reductions. Appl Microbiol Biotechnol (in this issue)
Greiner L, Müller DH, van den Ban ECD, Wöltinger J, Wandrey C, Liese A (2003) Membrane aerated hydrogenation: enzymatic and chemical homogeneous catalysis. Adv Synth Catal 345:679–683
Griengl H, Hickel A, Johnson DV, Kratky C, Schmidt M, Schwab H (1997) Enzymatic cleavage and formation of cyanohydrins: a reaction of biological and synthetic relevance. Chem Commun 1933–1940
Haberland J, Hummel W, Daußmann T, Liese A (2002) New continuous production process for enantiopure (2R,5R)-hexanediol. Org Process Res Dev 6:458–462
Hildebrand F, Lütz S (2006) Immobilisation of alcohol dehydrogenase from Lactobacillus brevis and its application in a plug-flow reactor. Tetrahedron Asymmetry 17:3219–3225
Hildebrand F, Kühl S, Pohl M, Vasic-Racki D, Müller M, Wandrey C, Lütz S (2006) The production of (R)-2-hydroxy-1-phenyl-propan-1-one derivatives by benzaldehyde lyase from Pseudomonas fluorescens in a continuously operated membrane reactor. Biotechnol Bioeng 96(5):835–843
Hilt G, Jarbawi T, Heineman WR, Steckhan E (1997) An analytical study of the redox behavior of 1,10-phenanthroline-5,6-dione, its transition-metal complexes, and its N-monomethylated derivative with regard to their efficiency as mediators of NAD(P)+ regeneration. Chem Eur 3:79–88
Hollmann F, Schmid A (2004) Electrochemical regeneration of oxidoreductases for cell-free biocatalytic redox reactions. Biocatal Biotransform 22:63–88
Hollmann F, Kleeb A, Otto K, Schmid A (2006) Corrigendum to “Coupled chemoenzymatic transfer hydrogenation catalysis for enantioselective reduction and oxidation reactions”. Tetrahedron Asymmetry 17:867–868
Honda K, Ishige T, Kataoka M, Shimizu S (2006) Microbial and enzymatic processes for the production of chiral compounds. In: Patel RN (ed) Biocatalysis in the pharmaceutical and biotechnology industries. Taylor & Francis, New York, pp 529–546
Hummel W (1997) New alcohol dehydrogenases for the synthesis of chiral compounds. Adv Biochem Eng Biotechnol 58:145–184
Hummel W, Kula MR (1989) Dehydrogenases for the synthesis of chiral compounds. Eur J Biochem 184:1–13
Hummel W, Abokitse K, Drauz K, Rollmann C, Gröger H (2003) Towards a large-scale asymmetric reduction process with isolated enzymes: expression of an (S)-alcohol dehydrogenase in E. coli and studies on the synthetic potential of this biocatalyst. Adv Synth Catal 345:153–159
Jaeger KE, Eggert T (2002) Lipases for biotechnology. Curr Opin Biotechnol 13:390–397
Jaeger KE, Schneidinger B, Rosenau F, Werner M, Lang D, Dijkstra BW, Schimossek K, Zonta A, Reetz MT (1997) Bacterial lipases for biotechnological applications. J Mol Catal B Enzym 3:3–12
Johannes TW, Woodyer RD, Zhao H (2007) Efficient regeneration of NADPH using an engineered phosphite dehydrogenase. Biotechnol Bioeng 96:18–26
Jones JB, Sneddon DW, Higgins W, Lewis A (1972) Preparative-scale reductions of cyclic ketone and aldehyde substrates of horse liver alcohol dehydrogenase with in situ sodium dithionite recycling of catalytic amounts of NAD. J Chem Soc Chem Commun 1972:856–857
Julliard M, Le Petit J, Ritz P (2004) Regeneration of NAD+ cofactor by photosensitized electron transfer in an immobilized alcohol dehydrogenase system. Biotechnol Bioeng 28:1774–1779
Kataoka M, Rohani LPS, Wada M, Kita K, Yanase H, Urabe I, Shimizu S (1998) Escherichia coli transformant expressing the glucose dehydrogenase gene from Bacillus megaterium as a cofactor regenerator in a chiral alcohol production system. Biosci Biotechnol Biochem 62:167–169
Kihumbu D, Stillger T, Hummel W, Liese A (2002) Enzymatic synthesis of all stereoisomers of 1-phenylpropane-1,2-diol. Tetrahedron Asymmetry 13:1069–1072
Kim MJ, Whitesides GM (1988) l-Lactate dehydrogenase: substrate specificity and use as a catalyst in the synthesis of homochiral 2-hydroxy acids. J Am Chem Soc 110:2959–2964
Kizaki N, Yasohara Y, Hasegawa J, Wada M, Kataoka M, Shimizu S (2001) Synthesis of optically pure ethyl (S)-4-chloro-3-hydroxybutanoate by Escherichia coli transformant cells coexpressing the carbonyl reductase and glucose dehydrogenase genes. Appl Microbiol Biotechnol 55:590–595
Klibanov AM, Puglisi AV (1980) The regeneration of coenzymes using immobilized hydrogenase. Biotechnol Lett 2:445–450
Koike T, Murata K, Ikariya T (2000) Stereoselective synthesis of optically active α-hydroxy ketones and anti-1,2-diols via asymmetric transfer hydrogenation of unsymmetrically substituted 1,2-diketones. Org Lett 2:3833–3836
Kosjek B, Stampfer W, Pogorevc M, Goessler W, Faber K, Kroutil W (2004) Purification and characterization of a chemotolerant alcohol dehydrogenase applicable to coupled redox reactions. Biotechnol Bioeng 86:55–62
Kragl U, Eckstein M, Kaftzik N (2002) Enzyme catalysis in ionic liquids. Curr Opin Biotechnol 13:565–571
Kruse W, Hummel W, Kragl U (1996) Alcohol-dehydrogenase-catalyzed production of chiral hydrophobic alcohols. A new approach leading to a nearly waste-free process. Recl Trav Chim Pays-Bas 115:239–243
Kula MR, Kragl U (2000) In: Patel RN (ed) Stereoselective biocatalysis. Marcel Dekker, New York
Kurlemann N, Liese A (2004) Immobilization of benzaldehyde lyase and its application as a heterogeneous catalyst in the continuous synthesis of a chiral 2-hydroxy ketone. Tetrahedron Asymmetry 15:2955–2958
Lamare S, Legoy MD, Graber M (2004) Solid/gas bioreactors: powerful tools for fundamental research and efficient technology for industrial applications. Green Chem 6:445–458
Leksawasdi N, Chow YYS, Breuer M, Hauer B, Rosche B, Rogers PL (2004) Kinetic analysis and modelling of enzymatic (R)-phenylacetylcarbinol batch biotransformation process. J Biotechnol 111:179–189
Leonida MD (2001) Redox enzymes used in chiral syntheses coupled to coenzyme regeneration. Curr Med Chem 8:345–369
Liese A, Karutz M, Kamphuis J, Wandrey C, Kragl U (1996) Enzymatic resolution of 1-phenyl-1,2-ethanediol by enantioselective oxidation: overcoming product inhibition by continuous extraction. Biotechnol Bioeng 51:544–550
Liese A, Zelinski T, Kula MR, Kierkels H, Karutz M, Kragl U, Wandrey C (1998) A novel reactor concept for the enzymatic reduction of poorly soluble ketones. J Mol Catal B Enzym 4:91–99
Liese A, Seelbach C, Wandrey C (2006) Industrial biotransformations (2nd edn.). GmbH, Weinheim
Liu J, Hsu CC, Wong CH (2004) Sequential aldol condensation catalyzed by DERA mutant Ser238Asp and a formal total synthesis of atorvastatin. Tetrahedron Lett 45:2439–2441
Lütz S (2006) Transition metal catalyzed regeneration of nicotineamide cofactors. In: de Vries JG, Elsevier CJ (eds) The handbook of homogeneous hydrogenation (vol III). GmbH, Weinheim, pp 1471–1482
Lye GJ, Woodley JM (1999) Application of in situ product-removal techniques to biocatalytic processes. Trends Biotechnol 17:395–402
Makino Y, Ding JY, Negoro S, Urabe I, Okada H (1989) Purification and characterization of a new glucose dehydrogenase from vegetative cells of Bacillus megaterium. J Ferment Bioeng 67:374–379
McWhirter RB, Klapper MH (1990) Semiquinone radicals of methylamine dehydrogenase, methoxatin, and related o-quinones: a pulse radiolysis study. Biochemistry 29:6919–6926
Mertens R, Liese A (2004) Biotechnological application of hydrogenases. Curr Opin Biotechnol 15:343–348
Mertens R, Greiner L, van den Ban ECD, Haaker HBCM, Liese A (2003) Practical applications of hydrogenase I from Pyrococcus furiosus for NADPH generation and regeneration. J Mol Catal B Enzym 24–25:39–52
Mochizuki N, Hiramatsu S, Sugai T, Ohta H, Morita H, Itokawa H (1995) Improved conditions for the production and characterization of 1-arylpropane-1,2-diols and related compounds. Biosci Biotechnol Biochem 59:2282–2291
Müller M (2005) Chemoenzymatische synthese von bausteinen für Statin-Seitenketten. Angew Chem 117:366–369 (also in Angew Chem Int Ed 44:362–365)
Neuhauser W, Steininger M, Haltrich D, Kulbe KD, Nidetzky B (1998) A pH-controlled fed-batch process can overcome inhibition by formate in NADH-dependent enzymatic reductions using formate dehydrogenase-catalyzed coenzyme regeneration. Biotechnol Bioeng 60:278–282
Nidetzky B, Neuhauser W, Haltrich D, Kulbe KD (1996) Continuous enzymatic production of xylitol with simultaneous coenzyme regeneration in a charged membrane reactor. Biotechnol Bioeng 52:387–396
Orlich B, Berger H, Lade M, Schomacker R (2000) Stability and activity of alcohol dehydrogenases in W/O-emulsions: enantioselective reduction including cofactor regeneration. Biotechnol Bioeng 70:638–646
Patel RN, Banerjee A, McNamee CG, Brzozowski D, Hanson RL, Szarka LJ (1993) Enantioselective Microbial reduction of 3,5-dioxo-6-(benzyloxy) hexanoic acid, ethyl-ester. Enzyme Microb Technol 15:1014–1021
Peters J (1998) Dehydrogenases – characteristics, design of reaction conditions, and applications. In: Rehm HJ, Reed G (eds) Biotechnology Vol 8a: Biotransformations I, 2nd edn. Wiley-VCH Verlag GmbH, Weinheim, pp 393–460
Pohl M, Liese A (2006) Industrial processes using lyases for C–C, C–N and C–O bond formation. In: Patel RN (ed) Biocatalysis in the pharmaceutical and biotechnology industries. Taylor & Francis, New York, pp 661–676
Raunio R, Lilius EM (1971) Effect of dithionite on enzyme activities in vivo. Enzymologia 40:360–368
Reetz MT, Zonta A, Schimossek K, Liebeton K, Jaeger KE (1997) Erzeugung enantioselektiver biokatalysatoren für die organische chemie durch in-vitro-evolution. Angew Chem 109:2961–2963 (also in Angew Chem Int Ed Engl 36:2830–2832)
Rickus JL, Tobin AJ, Zink JI, Dunn B (2002) Photochemical enzyme co-factor regeneration: towards continuous glutamate monitoring with a sol–gel optical biosensor. Mater Res Soc Symp Proc 723:155–160
Rissom S, Beliczey J, Giffels G, Kragl U, Wandrey C (1999) Asymmetric reduction of acetophenone in membrane reactors: comparison of oxazaborolodine and alcohol dehydrogenase catalyzed processes. Tetrahedron Asymmetry 10:923–928
Rosche B, Sandford V, Breuer M, Hauer B, Rogers PL (2002) Enhanced production of R-phenylacetylcarbinol (R-PAC) through enzymatic biotransformation. J Mol Catal B Enzym 19–20:109–115
Rosen TC, Daußmann T, Stohrer J (2004) Bioreduction forms optically active 3-hydroxyesters. Speciality Chemicals Magazine, April, pp 39–40
Schmid A, Dordick JS, Hauer B, Kiener A, Wubbolts M, Witholt B (2001) Industrial biocatalysis today and tomorrow. Nature 409:258–268
Schmidt E, Ghisalba O, Gygax D, Sedelmeier G (1992) Optimization of a process for the production of (R)-2-hydroxy-4-phenylbutyric acid—an intermediate for inhibitors of angiotensin converting enzyme. J Biotechnol 24:315–327
Schröder I, Steckhan E, Liese A (2003) In situ NAD+ regeneration using 2,2′-azinobis(3-ethylbenzothiazoline-6-sulfonate) as an electron transfer mediator. J Electroanal Chem 541:109–115
Schubert T, Hummel W, Müller M (2002) Highly enantioselective preparation of multifunctionalized propargylic building blocks. Angew Chem 114:656–659 (also in Angew Chem Int Ed 41:634–637)
Seelbach K, Riebel B, Hummel W, Kula MR, Tishkov VI, Egorov AM, Wandrey C, Kragl U (1996) A novel, efficient regenerating method of NADPH using a new formate dehydrogenase. Tetrahedron Lett 31:1377–1380
Simon H, Bader J, Gunther H, Neumann S, Thanos J (1985) Chirale verbindungen durch biokatalytische reduktionen. Angew Chem 97:541–555 (also in Angew Chem Int Ed Engl 24:539–553)
St. Clair N, Wang YF, Margolin AL (2000) Cofactor-bound cross-linked enzyme crystals (CLEC) of alcohol dehydrogenase. Angew Chem 112:388–391 (also in Angew Chem Int Ed 39:380–383)
Stark D, von Stockar U (2003) In situ product removal (ISPR) in whole cell biotechnology during the last twenty years. In: Scheper T (ed) Advances in biochemical engineering/biotechnology (vol 80). Springer, Berlin Heidelberg New York, pp 149–175
Steckhan E, Herrmann S, Ruppert R, Thömmes J, Wandrey C (1990) Kontinuierliche Erzeugung von NADH aus NAD+ und Formiat mit einem molekulargewichtsvergrößerten Homogenkatalysator in einem Membranreaktor. Angew Chem 102:445–447 (also in Angew Chem Int Ed Engl 29:388–390)
Steckhan E, Herrmann S, Ruppert R, Dietz E, Frede M, Spika E (1991) Analytical study of a series of substituted (2,2′-bipyridyl) (pentamethylcyclopentadienyl)rhodium and -iridium complexes with regard to their effectiveness as redox catalysts for the indirect electrochemical and chemical reduction of NAD(P)+. Organometallics 10:1568–1577
Stillger T, Bönitz M, Filho MV, Liese A (2002) Überwindung von thermodynamischen Limitierungen in substratgekoppelten Cofaktorregenerierungsverfahren. Chem Ing Tech 74:1035–1039
Stillger T, Pohl M, Wandrey C, Liese A (2006) Reaction engineering of benzaldehyde lyase from Pseudomonas fluorescens catalyzing enantioselective C–C bond formation. Org Process Res Dev 10:1172–1177
Takors R (2004) Ganzzell-ISPR-Prozessentwicklung: chancen und risiken. Chem Ing Tech 76:1857–1864
Tao JH, McGee K (2002) Development of a continuous enzymatic process for the preparation of (R)-3-(4-fluorophenyl)-2-hydroxy propionic acid. Org Process Res Dev 6:520–524
Tishkov VI, Galkin AG, Fedorchuk VV, Savitsky PA, Rojkova AM, Gieren H, Kula MR (1999) Pilot scale production and isolation of recombinant NAD+- and NADP+-specific formate dehydrogenases. Biotechnol Bioeng 64:187–193
Trivedi A, Heinemann M, Spieß AC, Daußmann T, Büchs J (2005) Optimization of adsoptive immobilization of alcohol dehydrogenase. J Biosci Bioeng 99:340–347
Trivedi A, Spieß AC, Daußmann T, Büchs J (2006) Study on mesophilic and thermophilic alcohol dehydrogenases in gas-phase reaction. Biotechnol Prog 22:454–458
van Beilen JB, Duetz WA, Schmid A, Witholt B (2003) Practical issues in the application of oxygenases. Trends Biotechnol 21:170–177
Van Rantwijk F, Lau RM, Sheldon RA (2003) Biocatalytic transformations in ionic liquids. Trends Biotechnol 21:131–138
Villela M, Stillger T, Müller M, Liese A, Wandrey C (2003) Is logP a convenient criterion to guide the choice of solvents for biphasic enzymatic reactions? Angew Chem 115:3101–3104 (also in Angew Chem Int Ed 42:2993–2996)
von Scala C, Fässler P, Gerla J, Maus E (2005) Kontinuierliche herstellung von kosmetischen fettsäureestern mittels reaktivdestillation und pervaporation. Chem Ing Tech 77:1809–1813
Vrtis JM, White AK, Metcalf WW, van der Donk WA (2002) Phosphite dehydrogenase: a versatile cofactor-regeneration enzyme. Angew Chem 114:3391–3393 (also in Angew Chem Int Ed 41:3257–3259)
Vuorilehto K, Lütz S, Wandrey C (2004) Indirect electrochemical reduction of nicotinamide coenzymes. Bioelectrochemistry 65:1–7
Wagenknecht PS, Penney JM, Hembre RT (2003) Transition-metal-catalyzed regeneration of nicotinamide coenzymes with hydrogen. Organometallics 22:1180–1182
Wallner SR, Bauer M, Würdemann C, Wecker P, Glöckner FO, Faber K (2005) Highly enantioselective sec-alkyl sulfatase activity of the marine planctomycete Rhodopirellula baltica shows retention of configuration. Angew Chem 117:6539–6542 (also in Angew Chem Int Ed 44:6381–6384)
Walsh C (1980) Flavin coenzymes: at the crossroads of biological redox chemistry. Acc Chem Res 13:148–155
Wandrey C (2004) Biochemical reaction engineering for redox reactions. Chem Rec 4:254–265
Weckbecker A, Hummel W (2004) Improved synthesis of chiral alcohols with Escherichia coli cells co-expressing pyridine nucleotide transhydrogenase, NADP+-dependent alcohol dehydrogenase and NAD+-dependent formate dehydrogenase. Biotechnol Lett 26:1739–1744
Weckbecker A, Hummel W (2005) Glucose dehydrogenase for the regeneration of NADPH and NADH. In: Barredo JL (ed) Microbial enzymes and biotransformations. Humana, Totowa, NJ, pp 225–238
Weuster-Botz D, Paschold H, Striegel B, Gieren H, Kula MR, Wandrey C (1994) Continuous computer controlled production of formate dehydrogenase (FDH) and isolation on a pilot scale. Chem Eng Technol 17:131–137
Wichmann R, Vasic-Racki D (2005) Cofactor regeneration at the lab scale. In: Kragl U (ed) Advances in biochemical engineering/biotechnology (vol 92). Springer, Berlin Heidelberg New York, pp 225–260
Willner I, Maidan R, Shapira M (1990) Thermal and photochemical regeneration of nicotinamide cofactors and a nicotinamide model Compound Using a water-soluble rhodium phosphine catalyst. J Chem Soc Perkin Trans 2:559–564
Wolberg M, Hummel W, Wandrey C, Müller M (2000) Highly regio- and enantioselective reduction of 3,5-dioxocarboxylates. Angew Chem 112:4476–4478 (also in Angew Chem Int Ed 39:4306–4308)
Wong CH, Daniels L, Orme-Johnson WH, Whitesides GM (1981) Enzyme-catalyzed organic synthesis: NAD(P)H regeneration using dihydrogen and the hydrogenase from Methanobacterium thermoautotrophicum. J Am Chem Soc 103:6227–6238
Yuan R, Watanabe S, Kuwabata S, Yoneyama H (1997) Asymmetric electroreduction of ketone and aldehyde derivatives to the corresponding alcohols using alcohol dehydrogenase as an electrocatalyst. J Org Chem 62:2494–2499
Zelinski T, Liese A, Wandrey C, Kula MR (1999) Asymmetric reductions in aqueous media: enzymatic synthesis in cyclodextrin containing buffers. Tetrahedron Asymmetry 10:1681–1687
Author information
Authors and Affiliations
Corresponding author
Additional information
Katja Goldberg and Kirsten Schroer did equally contribute to this review.
Rights and permissions
About this article
Cite this article
Goldberg, K., Schroer, K., Lütz, S. et al. Biocatalytic ketone reduction—a powerful tool for the production of chiral alcohols—part I: processes with isolated enzymes. Appl Microbiol Biotechnol 76, 237–248 (2007). https://doi.org/10.1007/s00253-007-1002-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-007-1002-0