Abstract.
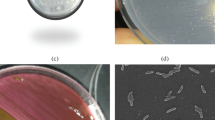
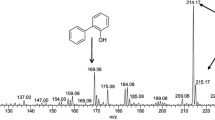
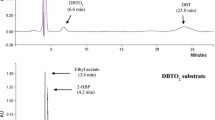
Naphtho[2,1-b]thiophene (NTH) is an asymmetric structural isomer of dibenzothiophene (DBT), and NTH derivatives can be detected in diesel oil following hydrodesulfurization treatment, in addition to DBT derivatives. Mycobacterium phlei WU-F1, which possesses high desulfurizing ability toward DBT and its derivatives over a wide temperature range (20–50 °C), could also grow at 50°C in a medium with NTH or 2-ethylNTH, an alkylated derivative, as the sole source of sulfur. At 50 °C, the resting cells of WU-F1 degraded 67% and 83% of 0.81 mM NTH and 2-ethylNTH, respectively, within 8 h. By GC-MS analysis, 2-ethylNTH-desulfurized metabolites were identified as 2-ethylNTH sulfoxide, 1-(2′-hydroxynaphthyl)-1-butene and 1-naphthyl-2-hydroxy-1-butene, and it was concluded that WU-F1 desulfurized 2-ethylNTH through a sulfur-specific degradation pathway with the selective cleavage of carbon–sulfur bonds. Therefore, M. phlei WU-F1 can effectively desulfurize asymmetric organosulfur compounds, NTH and 2-ethylNTH, as well as symmetric DBT derivatives under high-temperature conditions, and it may be a useful desulfurizing biocatalyst possessing a broad substrate specificity toward organosulfur compounds.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Furuya, .T., Kirimura, .K., Kino, .K. et al. Thermophilic biodesulfurization of naphthothiophene and 2-ethylnaphthothiophene by a dibenzothiophene-desulfurizing bacterium, Mycobacterium phlei WU-F1. Appl Microbiol Biotechnol 58, 237–240 (2002). https://doi.org/10.1007/s00253-001-0877-4
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s00253-001-0877-4