Abstract
In this study, we assessed various leaf structural and chemical features as possible predictors of the size of the phyllosphere bacterial population in the Mediterranean environment. We examined eight perennial species, naturally occurring and coexisting in the same area, in Halkidiki (northern Greece). They are Arbutus unedo, Quercus coccifera, Pistacia lentiscus, and Myrtus communis (evergreen sclerophyllous species), Lavandula stoechas and Cistus incanus (drought semideciduous species), and Calamintha nepeta and Melissa officinalis (nonwoody perennial species). M. communis, L. stoechas, C. nepeta, and M. officinalis produce essential oil in substantial quantities. We sampled summer leaves from these species and (1) estimated the size of the bacterial population of their phyllosphere, (2) estimated the concentration of different leaf constituents, and (3) studied leaf morphological and anatomical features and expressed them in a quantitative way. The aromatic plants are on average more highly colonized than the other species, whereas the nonwoody perennials are more highly colonized than the woody species. The population size of epiphytic bacteria is positively correlated with glandular and nonglandular trichome densities, and with water and phosphorus contents; it is negatively correlated with total phenolics content and the thickness of the leaf, of the mesophyll, and of the abaxial epidermis. No correlation was found with the density of stomata, the nitrogen, and the soluble sugar contents. By regression tree analysis, we found that the leaf-microbe system can be effectively described by three leaf attributes with leaf water content being the primary explanatory attribute. Leaves with water content >73% are the most highly colonized. For leaves with water content <73%, the phosphorus content, with a critical value of 1.34 mg g−1 d.w., is the next explanatory leaf attribute, followed by the thickness of the adaxial epidermis. Leaves higher in phosphorus (>1.34 mg g−1 d.w.) are more colonized, and leaves with the adaxial epidermis thicker than 20.77 μm are the least colonized. Although these critical attributes and values hold true only within the Mediterranean ecosystem studied and the range of observations taken, they are important because they provide a hypothesis to be tested in other Mediterranean ecosystems and other biomes. Such comparative studies may give insight as to the general properties governing the leaf-microbe system.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Leaf microbial communities are very diverse. Bacteria are the most abundant inhabitants, often found in numbers averaging 106–107 CFU cm−2 (up to 108 CFU g−1) of leaf [3, 4, 27]. Bacterial populations on the phyllosphere vary in size both among and within species and also over short time scales. At the species level, they vary greatly not only among individuals, even in close proximity to each other, but also among leaves of the same individual [26, 35, 75]. Leaf age and season greatly influence the epiphytic bacterial population [14, 66].
Studies involving leaf imprints have shown that bacteria do not occur in a uniform pattern across leaf surfaces [38, 72]. As revealed by scanning electron microscopy, the most common sites of bacterial colonization are the base of trichomes, stomata, and the epidermal cell wall junctions, especially in the grooves along the veins [41, 42, 56]. Bacteria have been found not only in depressions in the cuticle [41], but also beneath the cuticle [6] and near hydathodes [47]. In the Mediterranean species oleander and olive, Surico [65] found stomatal pits and shields of pectate hairs to be sites of bacterial colonization, respectively.
Leaf surface is a nutrient-limited environment [2]. It has been reported that bacteria in the phyllosphere are primarily carbon- [45, 73, 74] and secondarily nitrogen-limited [29]. Experimenting with Pseudomonas fluorescens, Mercier and Lindow [45] argue that the initial sugar abundance on uncolonized leaves is the primary determinant of the total bacterial population size that leaves can support after inoculation. Exogenous addition of nutrient sources on the leaf surface, such as aphid honeydew and pollen, has been associated with increase in the size of the epiphytic microbial community [11, 17, 63]. It has been also found that nutrient concentration in plant tissues and leaf surfaces are positively correlated (loglinearly) [10]. Leaf structural traits, such as the cuticle covering epidermal cell walls of higher plants, play a role in making the leaf surface a nutrient-limited environment. Because of the hydrophobic nature of cutin and waxes, its lipid components, the cuticle forms an effective transport barrier for water and polar substances. Reduced leaf wetting inhibits leaching of substances from the leaf interior, and consequently, low nutrient levels are available to microorganisms living on leaf surfaces [36]. Despite the barriers and limitations, high numbers of bacteria usually colonize plants, which suggests that nutrient- and energy-rich molecules on the leaf surface are adequate to support large microbial populations.
Secondary metabolites such as alkaloids, isoprenoids, and phenolic acids are leached or exuded from the leaf interior onto the leaf surface [15, 16, 46]. Several of them have been shown to inhibit growth of fungi, bacteria, and viruses [22, 54]. For instance, essential oils that are produced in leaf surface glandular trichomes are reported to be active against a wide spectrum of microorganisms [9, 28, 32, 61]. Still, essential-oil effects are not always negative. In some cases, they enhance microbial growth [68]. How all the different nutrients, primary and secondary metabolites of the leaf surface influence the phyllosphere microbial colonization remains unresolved.
The question we address in this study is which of the leaf structural and chemical features determine the microbial abundance on the leaf surfaces of perennial species, native to the Mediterranean environment. In particular, we examine the size of the phyllosphere bacterial population in relation to (1) leaf anatomical and morphological traits (glandular and nonglandular trichomes, stomata, thickness of the leaf and of individual leaf tissues, and specific leaf mass) and (2) leaf constituents (content in water, N, P, soluble sugars, phenolic compounds, and essential oils). We also examine whether species groupings, defined by the species life form or ability to produce essential oil, are also distinguishable by the level of the microbial colonization of their phyllosphere.
Materials and Methods
Study Area, Plant Material, and Sampling
We examined eight perennial species, woody and nonwoody, naturally occurring and coexisting in the same area, in Halkidiki (northern Greece). The woody species are Arbutus unedo L., Quercus coccifera L., Pistacia lentiscus L., Myrtus communis L., Lavandula stoechas L., and Cistus incanus L., all commonly found in the mediterranean-type ecosystems of Greece. The two nonwoody perennials, Calamintha nepeta (L.) Savi and Melissa officinalis L., occur in less arid microsites of the study area; the first occurs in open spaces, whereas the latter is found exclusively along a seasonal stream. A. unedo, Q. coccifera, P. lentiscus, and M. communis, which are tall evergreen shrubs, are major components of maquis, one of the two types of mediterranean-type ecosystems. C. incanus and L. stoechas are low, drought semideciduous species, commonly found in phrygana, the other mediterranean-type ecosystem. C. incanus is also common in maquis and in the shrub layer of Mediterranean conifer forests. Four species, M. communis, L. stoechas, C. nepeta, and M. officinalis, produce in substantial quantities essential oils, which are mixtures of isoprenoid compounds of low molecular weight giving the producing plants their characteristic odor.
The climate of the area is Mediterranean with rather mild and wet winters and hot, dry summers. The temperature of the coldest month is 5.6°C, whereas that of the hottest month is 26.7°C. August is the driest month of the year with an average rainfall of 14.3 mm (data of the meteorological station of Aristotle University of Thessaloniki, the nearest to the study area).
In August 2001, we sampled leaves from each of these species to (1) estimate the size of the epiphytic bacterial population, (2) examine leaf morphological and anatomical features, and (3) estimate the concentration of different leaf constituents. Sampling took place early in the morning. In all cases, samples consisted of mature leaves, collected at random from three individuals per species. Five samples were taken from each marked individual, thus making 15 samples per species. For essential oil estimation, bulkier samples had to be taken. Whenever the essential oil yield per plant was lower than the detection limit of the analytical method, we collected material from plants neighboring each of the three marked individuals. The essential oil yield was assigned to the marked individual contributing to the sample. For all species, sampling took place on the same day and at the same time (morning).
Estimation of the Total Bacterial Population
After sampling, leaves were placed in sterile plastic bags, were transported to the laboratory in an icebox, and were analyzed within 24 h. The serial dilution plating method [39] was used. Each sample was weighed and immersed in 25-mL sterile phosphate buffer (0.01 M, pH 7.3) supplemented with 0.1% bactopeptone, in a 100-mL Erlenmeyer flask. Flasks were sonicated in an ultrasonic cleaner for 10 min; the temperature of water did not exceed 20°C. Portions (100 μL) from the original wash and appropriate dilutions thereof prepared in 0.01 M phosphate buffer (pH 7.3) were plated onto nutrient agar (NAG) medium, supplemented with 2.5% (v/v) glycerol, and amended with 30 μg mL−1 natamycin to prevent fungal contamination. Bacterial populations were enumerated after incubation for 2–5 days at 24°C. Results are expressed as log(CFU + 1) per gram fresh weight, where CFU corresponds to colony forming units. The reasons for the log(CFU + 1) instead of the common log(CFU) transformation of data are described in detail in Yadav et al. [75].
Light Microscopy and Measurements
The detailed morphological and anatomical study of the leaves of the eight Mediterranean perennial species was the scope of a previous publication [76]. From that publication, we make use of some data that serve our goal to examine the relationship between the size of the phyllosphere bacterial population and leaf structural traits. The methods used for the study of features dealt with in this article are the following. Leaf samples were cut into small pieces and were fixed for 3 h with 5% glutaraldehyde in 0.05 M phosphate buffer (pH 7.2). They were then washed in buffer and postfixed for 4 h with similarly buffered 1% OsO4. The temperature in all solutions was kept at 0°C to avoid leaching of phenolics during fixation. For tissue dehydration, specimens were treated with graded ethanol series (50–100%) and propylene oxide. They were then infiltrated and embedded in Spurr's resin [62]. Semithin sections were cut with Reichert OM U2 ultramicrotome; they were stained with 1% toluidine blue O in 1% borax and photographed with a Zeiss III photomicroscope.
From light micrographs of leaf cross-sections (×800), we measured the thickness of the leaf, of the mesophyll, and of the epidermis (adaxial and abaxial) for each species. The density of stomata was estimated using micrographs of leaf paradermal sections. We estimated the specific leaf mass (SLM) as the ratio of leaf dry mass to leaf area [55]. Dry leaf mass was determined by oven drying the leaves at 70°C for 72 h [5], and leaf area was measured with a leaf area meter (Eijkelkamp, Agrisearch equipment, Netherlands).
From stereoscopic photographs of the adaxial and abaxial leaf surfaces, we estimated the density of glandular and nonglandular trichomes. They were counted over a defined area on the photograph. Their density was computed after the magnification of the photograph. In the case of the very pubescent L. stoechas, the density of nonglandular trichomes was estimated by counting them over a fixed area under the stereoscope.
Leaf Chemical Constituents
Leaf samples were oven dried at 60°C for 48 h and ground. Total nitrogen content was determined by the semimicro Kjeldahl method using Foss Tecator Kjeltec Auto 2200 Analyzer. Phosphorus was determined colorimetrically by the molybdenum blue method [1]. Soluble sugars were extracted from powdered dry material with 60% ethanol and were determined colorimetrically according to the phenol-sulfuric acid method [12]. For the estimation of total phenolics, dry leaf samples were crushed into powder in a test tube and extracted with 50% aqueous methanol. They were further measured colorimetrically by the Folin–Ciocalteu method [71]. Results are expressed in tannic acid equivalents (TAE) per amount of leaf extracted. Calibration was made with tannic acid solutions. Air-dried leaves (30–50 g) of the aromatic plants, M. communis, L. stoechas, C. nepeta, and M. officinalis, were water distilled in a Clevenger apparatus for 3 h. Their content in essential oil is expressed in mL per 100 g of dry leaves. Leaf water content (LWC) was computed after the following equation: LWC = [(leaf fresh weight − leaf dry weight)/leaf fresh weight] × 100 [21].
Statistical Analyses
Differences of the attributes examined were determined by analysis of variance (ANOVA) and Tukey's B test for multiple comparisons. ANOVA assumptions were checked and data were transformed when necessary. Pearson correlation was used to examine relationships among various leaf features. To further investigate the pattern of leaf attributes, a principal component analysis was run on the two data sets concerning (1) the leaf structural traits and (2) the leaf chemical constituents. In both correlation and principal component analysis, there are three points per attribute for each species representing the number of individual plants (three) examined per species. Each of the three points is the average of five values representing the number of samples taken per individual. In principal component analysis (PCA), since all species do not bear trichome (glandular or nonglandular) on their leaves, those not bearing were assigned the value 1 and those bearing them were assigned the value 2. All the above statistical analyses were performed by use of the software package SPSS for Windows (11.5.1, SPSS Inc., USA).
To relate the abundance of the phyllosphere bacterial population to the leaf chemical and structural variables, we used the technique of regression trees. Trees complement or represent an alternative to many traditional statistical techniques, including multiple regression, analysis of variance, loglinear models, etc. Among their main advantages are (1) the flexibility to handle a broad range of response types, (2) the ease and robustness of construction, (3) the ease of interpretation, and (4) the ability to handle missing values in both response and explanatory variables [8]. Trees can be used for data exploration, description, and prediction of patterns and processes and are best suited for complex ecological data requiring flexible and robust analytical methods, which can deal with nonlinear relationships, high-order interactions, and missing values, giving at the same time easily understandable and interpretable results. The suitability of trees in analyzing ecological data has been repeatedly explored and proved in the recent years [7, 8, 19, 33].
Regression trees predict the value of a response variable from a set of explanatory variables. The basic assumption of the method is that the functional dependency among system variables is not uniform in the whole domain, but can be approximated as such on smaller subdomains. Recursive partitioning is the technique used in tree construction. It works by repeatedly splitting data into homogeneous subsets (minimizing the sum of squares within groups). The most informative attribute is identified at each repetition, and the data set is divided according to the values of this attribute. A split is defined by values less than and greater than some chosen value, and, therefore, at each split, the data are partitioned into two mutually exclusive groups. The process is repeated for each subset until pure data sets (e.g., where all cases have the same value) are produced or data sets that cannot be divided further. The latter data sets are the terminal “leaves” of the tree. The relative lengths of the vertical lines associated with each split represent graphically the proportion of the total sum of squares explained by each split.
We used the program S-Plus 6.1 for windows (Insightful Corp., 2002) for tree construction. The response variable of the regression tree is the size of the phyllosphere bacterial population. The variables examined as to their explanatory value are the following: the thickness of the adaxial epidermis, of the abaxial epidermis, of the mesophyll, and of the leaf, the density of stomata, of glandular, and of nonglandular trichomes, SLM, and water, nitrogen, phosphorus, soluble sugar, essential oil, and phenolics contents. The effectiveness of the regression tree to explain the variation of the population size of epiphytic bacteria was examined by the Pearson correlation coefficient.
Results
Phyllosphere Bacterial Population
The size of the total epiphytic bacterial population varies significantly among species (Fig. 1). Overall, A. unedo and P. lentiscus are the least populated. Although members of different groups do not always differ regarding the size of the epiphytic bacterial population, the aromatic plants are on average more colonized than the other species, and the nonwoody perennials are more colonized than the evergreen sclerophyllous and the drought semideciduous species. There is no difference between the two groups of the woody Mediterranean species.
Total bacterial populations [log (CFU + 1)] on the leaves of the Mediterranean species Arbutus unedo (Au), Quercus coccifera (Qc), Pistacia lentiscus (Pl), Myrtus communis (Mc), Lavandula stoechas (Ls), Cistus incanus (Ci), Calamintha nepeta (Cn), and Melissa officinalis (Mo). In the inset, given are the averages per species group, evergreen-sclerophyllous (ES), drought semideciduous (DS), nonwoody perennials (NW), aromatic plants (A), and nonaromatic plants (NA). Bars represent standard errors of the means (n = 15). Different letters at the top of the columns show significant differences among species at p < 0.05.
Leaf Structural and Chemical Traits
Thickness-related variables differ significantly among species (Fig. 2). The evergreen sclerophyllous species are clearly distinguished from the rest by having thicker leaves and mesophyll (Figs. 2c, d). Leaf and mesophyll thickness are highly correlated (Table 1). The same holds true for the thickness of the adaxial and abaxial epidermis. In all species, the epidermis is thicker in the adaxial part of the leaf (p < 0.05; Figs. 2a, b).
Average thickness of (a) the adaxial epidermis, (b) the abaxial epidermis, (c) the mesophyll, and (d) the leaf of the species studied. Species abbreviations are as in Fig. 1. Bars represent standard errors of the means (n = 15). Different letters at the top of the columns show significant differences among species at p < 0.05.
Specific leaf mass (SLM) corresponding to the ratio of leaf dry mass to leaf area differs significantly among species (Table 2). It is lowest in the two nonwoody perennials, but also in the woody M. communis. In all species, stomata are present on the abaxial leaf surface. In L. stoechas, C. incanus, C. nepeta, and M. officinalis, a few stomata are also present on the adaxial surface, but their extremely low number makes density calculation impractical. Stomatal density differs significantly among species, but there is no apparent group-related trend (Table 2).
Trichomes were not found on the leaves of the evergreen sclerophyllous species. The drought semideciduous and the nonwoody species bear both glandular and nonglandular trichomes, except for C. incanus, in which glandular trichomes were not detected. The density of both types of trichomes is highest on the abaxial leaf surface; only in M. officinalis the density of nonglandular trichomes is similar on both surfaces. Over all species and for either leaf surface, the density of both glandular and nonglandular trichomes is highest in L. stoechas (Table 2).
The concentration of the leaf constituents examined varies significantly among species (Fig. 3). Nitrogen and phosphorus contents are highest in the nonwoody perennials. The representatives of the evergreen sclerophyllous species are clearly differentiated from the rest by having considerably lower phosphorus content. Levels of leaf soluble sugars vary among species but without any discernible group-related trend; the highest value was recorded in A. unedo (53.4 mg g−1) and the lowest in P. lentiscus and C. incanus (21.1 mg g−1). The concentration of total phenolics ranges from 36 mg TAE g−1 (in M. communis) to 131.3 mg TAE g−1 (in C. incanus); it is remarkably lower in essential oil-producing species (Fig 3d). The highest essential oil yield (2.01 mL/100 g) was recorded in C. nepeta (Fig. 3e), whereas the lowest (0.1 mL/100 g) in M. officinalis. Leaf water content varies among species in a group-consistent way; it is highest in the two nonwoody perennials and lowest in the evergreen sclerophyllous species (Fig. 3f).
(a) Nitrogen, (b) phosphorus, (c) soluble sugar, (d) total phenolics, (e) essential oil, and (f) water contents of the leaves of the species studied. Species abbreviations are as in Fig. 1. Bars represent standard errors of the means (n = 15). Different letters at the top of the columns show significant differences among species at p < 0.05.
Correlation analysis of the leaf constituents gave significant results in a number of cases. Highly correlated are the nitrogen and phosphorus contents (positively) and the essential oil and the total phenolics contents (negatively) (Table 3). The leaf water content is correlated with all leaf constituents examined, except with soluble sugars; it is most highly correlated with phosphorus content.
Results of PCA of the leaf structural traits are presented in Fig. 4. The first two components of the PCA explain 77.5% of the total variance. Evergreen sclerophyllous species are clearly separated from the drought semideciduous and the nonwoody species along the first axis, which explains 48.4% of the total variance. The density of the glandular and nonglandular trichomes, and the thickness of the leaf and of the mesophyll, are the traits primarily responsible for this separation. Along the second axis, explaining 29.1% of the total variance, species are separated after the epidermis thickness.
Projection of the eight Mediterranean species on the first two principal components on the basis of their structural attributes. The first axis accounts for 48.4% of the total variance and the second for 29.1%. Also inserted are the original attributes, with their vectors intersecting at (0, 0). The length of each attribute vector is proportional to its contribution to the principal component axis. The attributes are thickness of the adaxial epidermis (AdE), thickness of the abaxial epidermis (AbE), leaf thickness (LT), mesophyll thickness (MT), specific leaf mass (SLM), density of stomata (ST), density of glandular trichomes (GH), and density of nonglandular trichomes (NGH).
Results of PCA of the leaf constituents are presented in Fig. 5. The first two components of the PCA explain 70.9% of the total variance. All evergreen sclerophyllous species and C. incanus are located along the negative side and the nonwoody species and L. stoechas along the positive side of the first axis, which explains 49.9% of the total variance. Water, phosphorus, nitrogen, and essential oil contents have positive scores, whereas total phenolics content has a high negative score in the first component axis. The position of species along the second axis, which explains 21% of the total variance, is determined by the soluble sugar content.
Projection of the eight Mediterranean species on the first two principal components on the basis of their chemical attributes. The first axis accounts for 49.9% of the total variance and the second for 21.0%. Also inserted are the original attributes, with their vectors intersecting at (0, 0). The length of each attribute vector is proportional to its contribution to the principal component axis. The attributes are nitrogen content (N), phosphorus content (P), soluble sugar content (SU), total phenolics content (PH), essential oil yield (EO), and water content (WC).
Relationships of the Epiphytic Bacterial Population with Leaf Structural and Chemical Traits
The size of the phyllosphere bacterial population is correlated with a number of traits, both chemical and structural. It is positively correlated with water and phosphorus content and negatively with total phenolics content (Table 4). Of the structural traits, it is positively correlated with the trichome density (both glandular and nonglandular) and negatively with the thickness of the leaf, of the mesophyll, and of the abaxial epidermis. For trichome density, the nonparametric Spearman's correlation analysis was used because the evergreen sclerophyllous species do not bear trichomes.
Regressions were run between the size of the phyllosphere bacterial population and (1) the first component scores of the species, after their structural attributes, and (2) the first component scores of the species, after their chemical attributes. Regressions for both analyses are significant (Table 5). This means that microbial abundance in the phyllosphere of the species examined is influenced by the patterns of both the structural and the chemical attributes. Results showed that 35% of the variation in the size of the phyllosphere bacterial population is explained by the leaf structural profile, whereas 38% of the variation is explained by the leaf chemical profile. Given these values as well as the fact that the first component of the species structural and chemical attributes explains 48.4 and 49.9% of the total variation, respectively, it derives that the chemical attributes have slightly higher contribution in determining the size of the phyllosphere bacterial population.
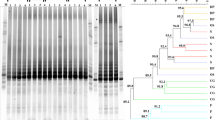
The regression tree explaining the abundance of the phyllosphere bacteria in terms of leaf structural and chemical attributes is four-leaved (Fig. 6). The first split is based on the leaf water content, which is the primary explanatory attribute. The resulting right branch is strongly homogeneous and is not subsequently divided, forming a leaf with mean rating 4.43. This means that whenever the water content exceeds 73%, the bacterial population size is high, with a mean value of 4.43 log(CFU + 1) g−1 f.w. The left branch, comprising samples with leaf water content <73%, is further divided after leaf phosphorus content that separates samples containing phosphorus in concentrations above and below 1.34 mg g−1 d.w. When phosphorus content is above this value, the mean population size of epiphytic bacteria is 4.03 log(CFU + 1) g−1 f.w. When it is lower, a third variable, the thickness of the epidermis, explains the size of the bacterial population. Low bacterial abundance is associated with thicker epidermis (>20.77 μm). This means that the abundance of the phyllosphere microbial population can be explained in terms of only 3 of the 14 leaf structural and chemical variables examined. The Pearson correlation coefficient between observed and predicted values of the regression tree is equal to 0.78. It is noteworthy that neither the nitrogen nor the sugar contents are among the explanatory attributes.
Regression tree analysis of the size of the phyllosphere microbial population. Each of the three splits (nonterminal nodes) is labeled with the variable and its values that determine the split. Each of the four leaves (terminal nodes) is labeled with the mean rating and (in parenthesis) the number of observations in the group. The vertical length of each split is proportional to the variation explained.
Discussion
A number of features varying among species distinguish the groups examined. Evergreen sclerophyllous species (A. unedo, Q. coccifera, P. lentiscus, and M. communis) are separated from the rest by their thick leaves and mesophyll, low water and phosphorus contents, and absence of trichomes. The nonwoody perennials (C. nepeta and M. officinalis) differ from all other species in having high nitrogen, phosphorus, and water contents. Essential oil-producing plants (M. communis, L. stoechas, C. nepeta, and M. officinalis) have the lowest total phenolics content. The different leaf structural and chemical profiles of the species examined are reflected in the size of epiphytic bacterial populations; the aromatic plants are on average more highly colonized than the other species, and the nonwoody perennials are more highly colonized than the woody species.
Regarding the chemical traits of the species that we studied, we must note the following. The leaf nitrogen levels of A. unedo, Q. coccifera, and P. lentiscus are similar to those reported for summer leaves from different locations in Greece [37, 43]. The low foliar phosphorus concentration of the evergreen sclerophyllous species is in agreement with previous findings regarding members of this group from other countries [24, 25]. The soluble sugar content of P. lentiscus leaves is comparable to that reported previously [43], but this is not the case for A. unedo and Q. coccifera; in their leaves, the soluble sugar content is considerably lower than that reported by Meletiou-Christou et al. [43]. The high soluble sugar content of M. communis is comparable to that reported for both sun and shade summer leaves of the species from Portugal [44]. Comparisons regarding the structural traits of the species studied with previous reports have been made in Yadav et al. [76].
The evergreen sclerophyllous species (except for the essential oil-producing M. communis) and C. incanus are characterized by high total phenolics content. In contrast, all essential oil-producing species have low total phenolics content. Among them, M. officinalis has the highest concentration of total phenolics, while having the lowest essential oil yield. The inverse accumulation patterns of phenolics and isoprenoids seem to reflect a trade-off in carbon allocation among the different types of secondary metabolites.
Results of correlation analysis revealed significant relationships with 8 out of the 14 leaf traits examined, four positive (with both glandular and nonglandular trichome density, phosphorus and water contents) and four negative (total phenolics content and thickness of the leaf, of the mesophyll, and of the lower epidermis). Among the structural traits, the density of glandular trichomes is the most highly correlated, followed by the density of nonglandular trichomes. As suggested earlier [41, 65], these structures provide conducive habitats for bacterial colonization because they facilitate adherence of bacterial cells on the leaf surface [67], they insure free water availability on the leaf surface by increasing the boundary layer and concomitantly decreasing water loss because of transpiration [20], they regulate leaf heat load by reflecting the visible and infrared radiations [13], and they protect the leaf from UV-B exposure [31]. Our findings agree with recent observations of Monier and Lindow [49]. These authors found that Pseudomonas syringae preferentially forms aggregates at the base of glandular trichomes and argue that glandular trichomes probably offer optimal conditions for microbial growth because of their ability to retain water droplets and secrete a diversity of chemical compounds that promote bacterial growth.
Although stomata have been found to be sites of bacterial colonization of leaf surfaces [18, 57, 65], no significant correlation was found between the size of the epiphytic bacterial population and stomatal density. This also agrees with Monier and Lindow [49], who found that aggregates of P. syringae were associated with all anatomical features of the leaf surface except stomata.
The population size of epiphytic bacteria is negatively correlated with the thickness of the leaf, and also of its mesophyll and of its epidermis (abaxial), but not with SLM. Leaf constituents have been found correlated with leaf structural traits [50, 52]. On this ground, we can argue that these negative relationships are due to the resulting low availability of compounds necessary for microbial growth, when the length of the diffusion path and the associated barriers influence their diffusion to the leaf surface. There are a number of barriers in the movement of molecules from the leaf interior to the leaf surface. Molecule flow, which has variable duration, depends on specific factors associated with their physicochemical properties, the plant species, and tissue densities [10]. For instance, a thick and compact mesophyll, as is the case with the evergreen sclerophyllous species [76], may hinder free movement of compounds. By limiting diffusion of nutrients and reducing wetting of leaf surfaces [36, 40], cuticular waxes constitute a barrier in metabolite transport, and this might affect the phyllosphere microbial colonization.
The total phenolics content is negatively correlated with the size of the epiphytic bacterial population. This may be related to the antimicrobial activity of phenolic compounds [22, 54, 60]. However, no such relationship was found with essential oils, despite their well-known antimicrobial activity [9, 28, 32, 48, 61]. In fact, the density of glandular trichomes is positively correlated with the size of the epiphytic bacterial population. In another study with Mediterranean aromatic plants, other than the ones used in the current work [32], we have also found that the richest in essential oil species were not always the least colonized. It seems, therefore, that the nature of the relationship between essential oils and microbes is not a simple one. We should also note that although the antimicrobial activity of essential oils is very well known, it is not universal against all microbes. Some soil bacteria can use them as carbon and energy source [23, 69, 70]. It is possible that some phyllosphere bacteria also have such a property.
Correlation analysis gives some insight as to which of the leaf traits examined are related with the abundance of microbes on leaf surfaces. However, in cases of complex systems, there are a number of variable interdependencies. To have a comprehensible description of the leaf-microbe system's behavior, we used regression tree analysis. As a result, we found that three parameters can explain to a large degree the size of the epiphytic bacterial population. The leaf water content proved by far the best explanatory variable, followed by the phosphorus content and to a less extent by the thickness of the adaxial epidermis.
The effects of leaf wettability, relative humidity, free moisture, and water potential of various tissues on epiphytic microbial colonization have been repeatedly examined [30, 53, 57, 78]. Abundance of water is the most important factor limiting microbial growth on interfacial surfaces [78] like the leaf surface. Water supply on the phyllosphere is of crucial importance for germination and growth of the leaf-associated microbial populations. Under arid climatic conditions, passive movement of water molecules down a water potential gradient supplies water on leaf surfaces. In absence of rain, fog, or dew, water in the leaf is the only source of water on the leaf surface, and this explains why the leaf water content proved the primary attribute explaining the size of the phyllosphere bacterial population in the Mediterranean ecosystem that we studied.
When the leaf water content is less than 73%, the phosphorus content plays a major role in explaining the population size of epiphytic bacteria, which suggests that bacterial abundance on leaf surfaces is primarily P-limited. Neither the nitrogen nor the sugar contents are important in this respect. These results are in agreement with Schönherr and Baur [58], who state that (1) only relatively lipophilic molecules can be expected to penetrate cuticles in significant amounts, (2) monosaccharides do not diffuse across intact cuticles in rates required to sustain growth of epiphytic microorganisms, (3) epicuticular waxes are a potential source of organic carbon, especially since epicuticular waxes can be regenerated if removed, (4) nitrogen is not expected to be a growth-limiting factor particularly because there is ample deposition of nitrogenous compounds from the atmosphere, and (5) for ATP or other polar P-containing compounds, being unlikely to penetrate cuticles in significant amounts, availability of phosphorus may well be growth-limiting for epiphytic microbes. Results regarding phosphorus, nitrogen, and soluble sugar contents, obtained through regression tree analysis, are in agreement with the conclusions and predictions of these authors, made after their observations and estimations at the physiological level. We must also note that in a study with Q. coccifera from Halkidiki (one of the species that we examined and from the same area), Papatheodorou and Stamou [51] reported that it is a low-P species and also that it is P- rather than N-limited. Our results, besides showing that Q. coccifera is a low-P species (as all evergreen sclerophyllous species that we studied are), provide evidence that the growth of microbes on its leaves is also P-limited.
For values of water content less than 73% and of phosphorus content less than 1.34 mg g−1 d.w., the population size of the epiphytic bacteria can be predicted after a leaf structural trait, the thickness of the upper leaf epidermis; leaves with an upper epidermis thicker than 20.77 are less colonized. The importance of a thick epidermis in limiting epiphytic microbial growth could be related to barriers that this leaf trait imposes for transport and release of compounds, necessary for microbial growth. Cuticular waxes constitute the main transport barrier in plant cuticles; cuticle permeability to water increases by up to 1500-fold and to organic compounds by up to 9200-fold, when waxes are extracted [58]. Yet, they amount to only a few percent of the mass of cuticles so that cuticle thickness is not related to permeability [34, 59]. Given the above, the relationship between the epidermis thickness and the population size of the epiphytic bacterial population cannot be fully explained.
As final remarks, we must stress the following points. (1) We quantified the population sizes of culturable bacteria only. However, stress has been found to affect culturability of bacteria, and many plant-associated bacterial species have not been cultured yet [77]. Therefore, the bacterial population size and composition of the leaves of plants growing under dry and hot conditions may be more complex than what plating onto culture media can reveal. (2) Because this study was carried out with samples from a single time point, it cannot be clearly established that the factors found to control the population sizes of epiphytic bacteria are indeed related to bacterial growth rather than to bacterial survival under the arid conditions of Mediterranean summer. Also, instantaneous measures of constituents only provide a snapshot of variable leaf constituents over time and may fail to distinguish between leaf constituents that differ in their importance to phyllosphere bacterial populations [64]. For these reasons, we are currently conducting a long-term study with parallel estimations of microbial abundance and leaf constituents over regular time intervals to have clearer answers regarding these concerns. (3) Regression trees are descriptive models, and thus, their predictions are valid only within the range of the observed values of the attributes examined. This means that the dependence of bacterial abundance primarily on the water and phosphorus contents of the leaves holds true only within the Mediterranean ecosystem and the range of plant species that we studied. Despite this, the importance of this finding is that it provides a hypothesis to be tested in other Mediterranean ecosystems and in other biomes. As the background information is limited, comparative studies of this type may convey new knowledge, reveal unknown patterns, and give insight as to the general properties, particularly regarding critical leaf attributes and their values, governing the leaf-microbe system.
References
SE Allen (Eds) (1989) Chemical Analysis of Ecological Materials Blackwell Science Oxford
JH Andrews (1992) ArticleTitleBiological control in the phyllosphere Annu Rev Phytopathol 30 603–635 Occurrence Handle10.1146/annurev.py.30.090192.003131 Occurrence Handle1:STN:280:DC%2BD1cvmvFehtQ%3D%3D Occurrence Handle18647102
JH Andrews RF Harris (2000) ArticleTitleThe ecology and biogeography of microorganisms on plant surfaces Annu Rev Phytopathol 38 145–180 Occurrence Handle10.1146/annurev.phyto.38.1.145 Occurrence Handle11701840
GA Beattie SE Lindow (1995) ArticleTitleThe secret life of foliar bacterial pathogens on leaves Annu Rev Phytopathol 33 145–172 Occurrence Handle10.1146/annurev.py.33.090195.001045 Occurrence Handle1:CAS:528:DyaK2MXosFWisL4%3D Occurrence Handle18294082
F Bussotti D Bettini P Grossoni S Mansuino R Nibbi C Soda C Tani (2002) ArticleTitleStructural and functional traits of Quercus ilex in response to water availability Environ Exp Bot 47 11–23 Occurrence Handle10.1016/S0098-8472(01)00111-3
WA Corpe S Rheem (1989) ArticleTitleEcology of the methylotrophic bacteria on leaving leaf surfaces FEMS Microbiol Ecol 62 243–250 Occurrence Handle10.1016/0378-1097(89)90248-6 Occurrence Handle1:CAS:528:DyaL1MXktlKmtrg%3D
A Dalaka B Kompare M Robnik-Šikonja SP Sgardelis (2000) ArticleTitleModelling the effects of environmental conditions on apparent photosynthesis of Stipa bromoides by machine learning tools Ecol Model 129 245–257 Occurrence Handle10.1016/S0304-3800(00)00237-4 Occurrence Handle1:CAS:528:DC%2BD3cXlt1yqtLo%3D
G De' ath KE Fabricius (2000) ArticleTitleClassification and regression trees: a powerful yet simple technique for ecological data analysis Ecology 81 3178–3192 Occurrence Handle10.1890/0012-9658(2000)081[3178:CARTAP]2.0.CO;2
SG Deans G Ritchie (1987) ArticleTitleAntibacterial properties of plant essential oils Int J Food Microbiol 5 165–180 Occurrence Handle10.1016/0168-1605(87)90034-1
S Derridj (1996) Nutrients on the leaf surface CE Morris PC Nicot C Nguyen (Eds) Aerial Plant Surface Microbiology Plenum Press New York 25–42
AJ Dik NJ Fokkema JA Vanpelt (1992) ArticleTitleInfluence of climatic and nutritional factors on yeast population-dynamics in the phyllosphere of wheat Microb Ecol 23 41–52 Occurrence Handle10.1007/BF00165906
M Dubois KA Gilles JK Hamilton PA Rebers F Smith (1956) ArticleTitleColorimetric methods for determination of sugars and related substances Anal Chem 28 350–356 Occurrence Handle10.1021/ac60111a017 Occurrence Handle1:CAS:528:DyaG28XjvFynsg%3D%3D
JR Ehleringer HA Mooney (1978) ArticleTitleLeaf hairs—effects on physiological activity and adaptive value to a desert shrub Oecologia 37 183–200 Occurrence Handle10.1007/BF00344990
GL Ercolani (1991) ArticleTitleDistribution of epiphytic bacteria on olive leaves and the influence of leaf age and sampling time Microb Ecol 21 35–48
V Fiala C Glad M Martin E Jolivet S Derridj (1990) ArticleTitleOccurrence of soluble carbohydrates on the phylloplane of maize (Zea mays L.): variations in relation to leaf heterogeneity and position on the plant New Phytol 115 609–615 Occurrence Handle1:CAS:528:DyaK3MXhtFyjtrk%3D
NJ Fokkema (1981) Fungal leaf saprophytes, beneficial or detrimental? JP Blakeman (Eds) Microbial Ecology of the Phylloplane Academic Press London 433–439
NJ Fokkema I Riphagen RJ Poot C Jong Particlede (1983) ArticleTitleAphid honeydew, a potential stimulant of Cochliobolus sativus and Septoria nodorum and the competitive role of saprophytic mycoflora Trans Br Mycol Soc 81 355–363 Occurrence Handle10.1016/S0007-1536(83)80087-4
AE Gau C Dietrich K Kloppstech (2002) ArticleTitleNon-invasive determination of plant-associated bacteria in the phyllosphere of plants Environ Microbiol 4 744–752 Occurrence Handle10.1046/j.1462-2920.2002.00359.x Occurrence Handle12460282
V Geissen C Kampichler (2004) ArticleTitleLimits to the bioindication potential of Collembola in environmental impact analysis: a case study of forest soil-limiting and fertilization Biol Fertil Soils 39 383–390 Occurrence Handle10.1007/s00374-003-0714-2 Occurrence Handle1:CAS:528:DC%2BD2cXls1Sgtrk%3D
G Grammatikopoulos Y Manetas (1994) ArticleTitleDirect absorption of water by hairy leaves of Phlomis fruticosa and its contribution to drought avoidance Can J Bot 72 1805–1811
L Gratani E Ghia (2002) ArticleTitleChanges in morphological and physiological traits during leaf expansion of Arbutus unedo Environ Exp Bot 48 51–60 Occurrence Handle10.1016/S0098-8472(02)00010-2
RJ Grayer JB Harborne (1994) ArticleTitleA survey of antifungal compounds from higher plants Phytochemistry 37 19–42 Occurrence Handle10.1016/0031-9422(94)85005-4 Occurrence Handle1:CAS:528:DyaK2cXmslKlt7Y%3D
ET Griffiths SM Bociek PC Harries R Jeffcoat DJ Sissions PW Trudgill (1987) ArticleTitleBacterial metabolism of alpha-pinene: pathway from alpha-pinene oxide to acyclic metabolites in Nocardia sp. strain P18.3 J Bacteriol 169 4972–4979 Occurrence Handle3667521 Occurrence Handle1:CAS:528:DyaL1cXntFyisA%3D%3D
EV Gutiérrez R Vallejo J Romana J Fons (1991) ArticleTitleThe subantarctic Nothofagus forests of Tierra del Fuego: distribution, structure and production Oecol Aquat 10 1–14
F Hevia M Loreto O Minoletti KLM Decker REJ Boerner (1999) ArticleTitleFoliar nitrogen and phosphorus dynamics of three Chilean Nothofagus (Fagaceae) species in relation to leaf lifespan Am J Bot 86 447–455 Occurrence Handle10077506
SS Hirano CD Upper (1989) ArticleTitleDiel variation in population size and ice nucleation activity of Pseudomonas syringae on snap bean leaflets Appl Environ Microbiol 55 623–630 Occurrence Handle16347871 Occurrence Handle1:STN:280:DC%2BC3crotFentg%3D%3D
SS Hirano CD Upper (2000) ArticleTitleBacteria in the leaf ecosystem with emphasis on Pseudomonas syringae—a pathogen, ice nucleus and epiphyte Microbiol Mol Biol Rev 64 624–653 Occurrence Handle10974129 Occurrence Handle1:STN:280:DC%2BD3cvntVOmsg%3D%3D
AM Janssen NLJ Chin JJC Scheffer B Svendsen (1987) ArticleTitleScreening for antimicrobial activity of some essential oils by the agar overlay technique Pharm Weekbl Sci 8 289–292
P Ji M Wilson (2003) ArticleTitleEnhancement of population size of a biological control agent and efficacy in control of bacterial speck of tomato through salicylate and ammonium sulfate amendments Appl Environ Microbiol 69 1290–1294 Occurrence Handle10.1128/AEM.69.2.1290-1294.2003 Occurrence Handle12571060 Occurrence Handle1:CAS:528:DC%2BD3sXhtF2itbo%3D
KB Johnson VO Stockwell TL Sawyer D Sugar (2000) ArticleTitleAssessment of environmental factors influencing growth and spread of Pantoea agglomerans on and among blossoms of pear and apple Phytopathology 90 1285–1294 Occurrence Handle18944433 Occurrence Handle1:STN:280:DC%2BD1cjjslOlsQ%3D%3D
G Karabourniotis A Kyparissis Y Manetas (1993) ArticleTitleLeaf hairs of Olea europaea L. protect underlying tissue against ultraviolet-B radiation damage Environ Exp Bot 33 341–345 Occurrence Handle10.1016/0098-8472(93)90035-E
K Karamanoli D Vokou U Menkissoglu H-I Constantinidou (2000) ArticleTitleBacterial colonization of phyllosphere of Mediterranean aromatic plants J Chem Ecol 26 2035–2048 Occurrence Handle10.1023/A:1005556013314 Occurrence Handle1:CAS:528:DC%2BD3cXotFaisbY%3D
AJ Kerkhoff SN Martens GA Shore BT Milne (2004) ArticleTitleContingent effects of water balance variations on tree cover density in semiarid woodlands Glob Ecol Biogeogr 13 237–246 Occurrence Handle10.1111/j.1466-822X.2004.00087.x
G Kerstiens (1995) ArticleTitleCuticular water permeability of European trees and shrubs grown in polluted and unpolluted atmospheres and its relation to stomatal response to humidity in beech (Fagus sylvatica L.) New Phytol 129 495–503
LL Kinkel M Wilson SE Lindow (1995) ArticleTitleEffects of scale on estimates of epiphytic bacterial populations Microb Ecol 29 282–297 Occurrence Handle10.1007/BF00164891
D Knoll L Schreiber (2000) ArticleTitlePlant–microbe interactions: wetting of ivy (Hedera helix L.) leaf surfaces in relation to colonization by epiphytic microorganisms Microb Ecol 40 33–42 Occurrence Handle10977875
M Kouki Y Manetas (2002) ArticleTitleToughness is less important than chemical composition of Arbutus leaves in food selection by Poecilimon species New Phytol 154 399–407 Occurrence Handle10.1046/j.1469-8137.2002.00375.x Occurrence Handle1:CAS:528:DC%2BD38XktFWnt70%3D
C Leben (1988) ArticleTitleRelative humidity and the survival of epiphytic bacteria with buds and leaves of cucumber plants Phytopathology 78 179–185
SE Lindow DC Arny CD Upper (1978) ArticleTitleDistribution of ice nucleation-active bacteria on plants in nature Appl Environ Microbiol 36 831–838 Occurrence Handle736541 Occurrence Handle1:STN:280:DyaE1M7gt1aqtg%3D%3D
SE Lindow MT Brandl (2003) ArticleTitleMicrobiology of the phyllosphere Appl Environ Microbiol 69 1875–1883 Occurrence Handle10.1128/AEM.69.4.1875-1883.2003 Occurrence Handle12676659 Occurrence Handle1:CAS:528:DC%2BD3sXivFKrur8%3D
EL Mansvelt MJ Hattingh (1987) ArticleTitleScanning electron microscopy of colonization of pear leaves by Pseudomonas syringae pv. syringae Can J Bot 65 2517–2522
RLR Mariano SM McCarter (1993) ArticleTitleEpiphytic survival of Pseudomonas viridiflava on tomato and selected weed species Microb Ecol 26 47–58 Occurrence Handle10.1007/BF00166029
MS Meletiou-Christou S Rhizopoulou S Diamantoglou (1994) ArticleTitleSeasonal changes of carbohydrates, lipids and nitrogen content in sun and shade leaves from four Mediterranean evergreen sclerophylls Environ Exp Bot 34 129–140 Occurrence Handle10.1016/0098-8472(94)90032-9 Occurrence Handle1:CAS:528:DyaK2cXltlSjs7g%3D
MM Mendes LC Gazarini ML Rodrigues (2001) ArticleTitleAcclimation of Myrtus communis to contrasting Mediterranean light environments—effects on structure and chemical composition of foliage and plant water relations Environ Exp Bot 45 165–178 Occurrence Handle10.1016/S0098-8472(01)00073-9 Occurrence Handle11275224 Occurrence Handle1:CAS:528:DC%2BD3MXitF2msrc%3D
J Mercier SE Lindow (2000) ArticleTitleRole of leaf surface sugars in colonization of plants by bacterial epiphytes Appl Environ Microbiol 66 369–374 Occurrence Handle10618250 Occurrence Handle1:CAS:528:DC%2BD3cXktlSrtg%3D%3D Occurrence Handle10.1128/AEM.66.1.369-374.2000
GT Merrall (1981) Physical factors that influence the behavior of chemicals on leaf surfaces JP Blakeman (Eds) Microbial Ecology of the Phylloplane Academic Press London 265–281
TW Mew IC Mew JS Huang (1984) ArticleTitleScanning electron microscopy of virulent and avirulent strains of Xanthomonas campestris pv. oryzae on rice leaves Phytopathology 74 635–641 Occurrence Handle10.1094/Phyto-74-635
N Mimica-Dukic B Bozin M Sokovic N Simin (2004) ArticleTitleAntimicrobial and antioxidant activities of Melissa officinalis L. (Lamiaceae) essential oil J Agric Food Chem 52 2485–2489 Occurrence Handle10.1021/jf030698a Occurrence Handle15113145 Occurrence Handle1:CAS:528:DC%2BD2cXivFemtb4%3D
J-M Monier SE Lindow (2004) ArticleTitleFrequency, size and localization of bacterial aggregates on bean leaf surface Appl Environ Microbiol 70 346–355 Occurrence Handle10.1128/AEM.70.1.346-355.2004 Occurrence Handle14711662 Occurrence Handle1:CAS:528:DC%2BD2cXmvFansQ%3D%3D
Ü Niinemets K Kull (2003) ArticleTitleLeaf structures vs. nutrient relationships vary with soil conditions in temperate shrubs and trees Acta Oecol 24 209–219 Occurrence Handle10.1016/S1146-609X(03)00094-8
E Papatheodorou GP Stamou (2004) ArticleTitleNutrient attributes of tissues in relation to grazing in an evergreen sclerophyllous shrub (Quercus coccifera L.) dominating vegetation in Mediterranean-type ecosystem J Arid Environ 59 217–227 Occurrence Handle10.1016/j.jaridenv.2003.12.012
PJ Peeters (2002) ArticleTitleCorrelation between leaf constituent levels and the densities of herbivorous insect guilds in an Australian forest Austral Ecol 27 658–671 Occurrence Handle10.1046/j.1442-9993.2002.01227.x
PL Pusey (2000) ArticleTitleThe role of water in epiphytic colonization and infection of pomaceous flowers by Erwinia amylovora Phytopathology 90 1352–1357 Occurrence Handle18943376 Occurrence Handle1:STN:280:DC%2BD1cjjslCktw%3D%3D
JP Rauha S Remes M Heinonen A Hopia M Kahkonen T Kujala K Pihlaja H Vuorela P Vuorela (2000) ArticleTitleAntimicrobial effects of Finnish plant extracts containing flavonoids and other phenolic compounds Int J Food Microbiol 56 3–12 Occurrence Handle10.1016/S0168-1605(00)00218-X Occurrence Handle10857921 Occurrence Handle1:CAS:528:DC%2BD3cXjt1art7o%3D
PB Reich MB Walter DS Ellisworth (1992) ArticleTitleLeaf life-span in relation to leaf, plant and stand characteristics among diverse ecosystems Ecol Monogr 62 365–392
IMM Roos MJ Hattingh (1983) ArticleTitleScanning electron microscopy of Pseudomonas syringae pv. morsprunorum on sweet cherry leaves Phytopathol Z 180 18–25
S Sabaratnam GA Beattie (2003) ArticleTitleDifferences between Pseudomonas syringae pv. syringae B728a and Pantoea agglomerans BRT98 in epiphytic and endophytic colonization of leaves Appl Environ Microbiol 69 1220–1228 Occurrence Handle10.1128/AEM.69.2.1220-1228.2003 Occurrence Handle12571050 Occurrence Handle1:CAS:528:DC%2BD3sXhtF2itLo%3D
J Schönherr P Baur (1996) Cuticle permeability studies: a model for estimating leaching of plant metabolites to leaf surfaces CE Morris PC Nicot C Nguyen (Eds) Aerial Plant Surface Microbiology Plenum Press New York 1–23
J Schönherr M Riederer (1989) ArticleTitleFoliar penetration and accumulations of organic chemicals in plant cuticles Rev Environ Contam Toxicol 108 1–70
JO Siqueira GN Muraleedharan R Hammerschmidt GR Safir (1991) ArticleTitleSignificance of phenolic compounds in plant–soil–microbial systems Crit Rev Plant Sci 10 63–121 Occurrence Handle1:CAS:528:DyaK3MXmtVehsrk%3D
A Sivropoulou S Kokkini T Lanaras M Arsenakis (1995) ArticleTitleAntimicrobial activity of mint essential oils J Agric Food Chem 43 2384–2388 Occurrence Handle10.1021/jf00057a013 Occurrence Handle1:CAS:528:DyaK2MXns1Cjt7c%3D
AR Spurr (1969) ArticleTitleA low viscosity epoxy resin embedding medium for electron microscopy J Ultrastruct Res 26 31–43 Occurrence Handle10.1016/S0022-5320(69)90033-1 Occurrence Handle4887011 Occurrence Handle1:CAS:528:DyaF1MXkvVahsLc%3D
B Stadler T Muller (2000) ArticleTitleEffects of aphids and moth caterpillars on epiphytic microorganisms in canopies of forest trees Can J For Ecol 30 631–638
NE Stamp MD Bowers (1990) ArticleTitlePhenology of nutritional differences between new and mature leaves and its effect on caterpillar growth Ecol Entomol 15 447–454
G Surico (1993) ArticleTitleScanning electron microscopy of olive and oleander leaves colonized by Pseudomonas syringae subsp. savastanoi J Phytopathol 138 31–40
IP Thompson MJ Baily JS Fenlon TR Fermor AK Lilley JM Lynch PJ McCormack MP McQuilken KJ Purdy PB Rainey JM Whipps (1993) ArticleTitleQuantitative and qualitative seasonal changes in the microbial community from the phyllosphere of sugar beet (Beta vulgaris) Plant Soil 150 177–191 Occurrence Handle10.1007/BF00013015
LW Timmer JJ Marois D Achor (1987) ArticleTitleGrowth and survival of Xanthomonads under conditions nonconductive to disease development Phytopathology 77 1341–1345
D Vokou (1999) Essential oils as allelochemicals: research advances in Greece SS Narwal (Eds) Allelopathy Update: vol. 2. Basic and Applied Aspects Science Publishers Inc. USA 47–63
D Vokou S Liotiri (1999) ArticleTitleStimulation of soil microbial activity by essential oils Chemoecology 9 41–45 Occurrence Handle10.1007/s000490050032 Occurrence Handle1:CAS:528:DyaK1MXisFSltLc%3D
D Vokou NS Margaris JM Lynch (1984) ArticleTitleEffects of volatile oils from aromatic shrubs on soil microorganisms Soil Biol Biochem 16 509–513 Occurrence Handle10.1016/0038-0717(84)90060-9 Occurrence Handle1:CAS:528:DyaL2MXlvFWksA%3D%3D
PG Waterman S Mole (1994) Analysis of Phenolic Plant Metabolites Blackwell Scientific Publication Oxford 66–103
DM Weller AW Saettler (1980) ArticleTitleColonization and distribution of Xanthomonas phaseoli ans X. phaseoli var. fuscans in field-grown navy beans Phytopathology 70 500–506
M Wilson SE Lindow (1995) ArticleTitleEnhanced epiphytic coexistence of near-isogenic salicylate-catabolizing and non-salicylate-catabolizing Pseudomonas putida strains after exogenous salicylate application Appl Environ Microbiol 61 1073–1076 Occurrence Handle1:CAS:528:DyaK2MXktV2rtrk%3D Occurrence Handle16534957
M Wilson MA Savka I Hwang SK Farrand SE Lindow (1995) ArticleTitleAltered epiphytic colonization of mannityl opine-producing transgenic tobacco plants by a mannityl opine-catabolizing strain of Pseudomonas syringae Appl Environ Microbiol 61 2151–2158 Occurrence Handle1:CAS:528:DyaK2MXlvFOgtrk%3D Occurrence Handle16535040
RKP Yadav JM Halley K Karamanoli H-I Constantinidou D Vokou (2004) ArticleTitleBacterial populations on the leaves of Mediterranean plants: quantitative features and testing of distribution models Environ Exp Bot 52 63–77 Occurrence Handle10.1016/j.envexpbot.2004.01.004
RK Yadav AM Bosabalidis D Vokou (2004) ArticleTitleLeaf structural features of Mediterranean perennial species: plasticity and life form specificity J Biol Res 2 21–34
C-H Yang DE Crowley J Borneman NT Keen (2001) ArticleTitleMicrobial phyllosphere populations are more complex than previously realized Proc Natl Acad Sci USA 98 3889–3894 Occurrence Handle11274410 Occurrence Handle1:CAS:528:DC%2BD3MXis1Khs7Y%3D
EI Zehr DP Shepard WC Bridges (1996) ArticleTitleBacterial spot of peaches as influenced by water congestion, leaf wetness duration and temperature Plant Dis 80 339–341 Occurrence Handle10.1094/PD-80-0339
Acknowledgments
R.K.P. Yadav is supported by a grant from the State Scholarships Foundation (IKY), Greece. He is currently on study leave from Tribhuvan University, Nepal. This project is also supported by the General Secretariat for Research and Technology, Ministry of Development, Greece (01 ED 317).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yadav, R.K.P., Karamanoli, K. & Vokou, D. Bacterial Colonization of the Phyllosphere of Mediterranean Perennial Species as Influenced by Leaf Structural and Chemical Features. Microb Ecol 50, 185–196 (2005). https://doi.org/10.1007/s00248-004-0171-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-004-0171-y