Abstract
Background
While MR enterography (MRE) is commonly used to identify segments of bowel affected by Crohn disease in children, there is little data to support this practice.
Objective
To assess the ability of MRE to identify segments of small bowel and colon affected by pediatric Crohn disease using histopathology as the reference standard.
Materials and methods
Pediatric MRE examinations performed between April 2009 and December 2010 were identified. Subjects were excluded if they did not have Crohn disease, if they had Crohn disease but no correlative histopathology within 2 months of imaging, or if they had undergone prior bowel resection. Pertinent MRE and histopathology reports were reviewed, and normal and abnormal bowel segments were documented.
Results
Thirty-two pediatric MRE examinations were identified with correlative histopathology. MRE had an overall sensitivity of 94% for detecting the presence of Crohn disease, in general. At the bowel segmental level, MRE had a sensitivity of 66%, specificity of 90%, positive predictive value of 85% and negative predictive value of 76%. The terminal ileum was abnormal by MRE in 11 of 15 (73%) subjects lacking a diagnostic biopsy of this bowel segment.
Conclusion
MRE successfully identifies small bowel and colon segments affected by pediatric Crohn disease.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Crohn disease is relatively common in the pediatric population, with up to 20–30% of affected individuals presenting during either childhood or adolescence [1, 2]. Its incidence in children appears to be increasing, having doubled during a recent 10-year period [1], and it may present in children younger than 3 years of age [3]. While ileocolonoscopy with biopsy is the primary tool for diagnosing and following this condition in both pediatric and adult patients [4], radiological assessment is commonly performed for a variety of reasons, including to identify segments of bowel affected by Crohn disease at the time of initial diagnosis, to assess changes in small bowel and colon disease activity over time, to determine disease response to medical management and to evaluate for disease-related complications.
While the imaging assessment of pediatric Crohn disease was limited primarily to endoluminal barium studies (such as small bowel follow-through, small bowel enteroclysis and contrast enema) in the past, cross-sectional imaging modalities, such as CT enterography (CTE) [5], MR enterography (MRE) [6–11], and, on occasion, ultrasound [12–18] are regularly utilized today. During the past few years, MRE has become the de facto imaging test of choice for assessing pediatric Crohn disease at many institutions, including ours, due to its lack of ionizing radiation [19] and its ability to assess for both intestinal (small bowel and colon) and extra-intestinal disease manifestations. This change in practice pattern, however, is based on limited medical evidence, as there are very few studies comparing MRE findings to histopathology specimens in children with Crohn disease.
The purpose of this study was to assess retrospectively the ability of MRE to identify segments of small bowel and colon affected by Crohn disease in children using histopathology as the reference standard. Additionally, we wanted to establish how frequently unequivocal terminal ileitis is diagnosed by MRE in the setting of a nondiagnostic terminal ileal biopsy specimen.
Methods and materials
Institutional review board (IRB) approval was obtained prior to performing this retrospective HIPAA-compliant investigation. The requirement for subject informed consent was waived.
All pediatric (patients younger than 18 years of age) MRE examinations performed between April 1, 2009, and December 31, 2010, within the University of Michigan Health System were identified. Subjects were excluded if they did not have or were not eventually diagnosed with Crohn disease, if they had Crohn disease but no histopathological specimen available for review within 2 months of their imaging study, or if they had Crohn disease and had undergone prior bowel resection. All MRE examinations included in this study were performed at a field strength of 1.5 Tesla (Achieva; Philips Healthcare, Best, The Netherlands) using a standard protocol for all patients, including axial and coronal T2-weighted single-shot turbo spin-echo (SSTSE), axial and coronal two-dimensional (2-D) steady-state free precession (SSFP), axial T2-weighted turbo spin-echo (TSE) with fat saturation, axial diffusion-weighted, coronal dynamic postcontrast three-dimensional (3-D) T1-weighted spoiled gradient recalled echo (SPGR) with fat saturation, and coronal/axial delayed postcontrast 3-D T1-weighted SPGR with fat-saturation pulse sequences. Oral contrast material (VoLumen; E-Z-EM/Bracco Imaging, Princeton, N.J.) was consumed by all children prior to imaging using a weight-based protocol. Intravenous glucagon was administered to all children prior to postcontrast imaging to minimize bowel peristalsis-related artifacts, also using a weight-based protocol.
MRE/histopathology correlation for Crohn disease
Pertinent MRE reports (rather than MRE images) were retrospectively reviewed. Both normal (unaffected by Crohn disease) and abnormal (affected by Crohn disease) small bowel (excluding the duodenum) and colon segments were documented based upon the “Findings” and “Impressions” sections of the imaging reports. No specific imaging report finding or constellation of imaging report findings was used to strictly define abnormal bowel segments. The small bowel and colon were divided into the following eight segments: jejunum, ileum, cecum, ascending colon, transverse colon, descending colon, sigmoid colon and rectum. If the imaging report failed to mention a portion of the bowel, this segment (or segments) was excluded from our results and statistical analysis. Severity of disease by MRE was not considered.
Next, histopathology reports from either endoscopic biopsy or surgical bowel resection within 2 months of MRE were separately and retrospectively reviewed. Again, both small bowel (excluding the duodenum) and colon segments were documented as being either normal or abnormal. The small bowel and colon were divided into the following seven segments: ileum, cecum, ascending colon, transverse colon, descending colon, sigmoid colon and rectum. If the histopathology report failed to mention a portion of the bowel, this segment (or segments) was excluded from our results and statistical analysis. Severity of disease by histopathology was not considered.
Assessment of jejunal/ileal Crohn disease in the absence of available histopathology
Regarding MRE examinations with reported evidence of jejunal or ileal Crohn disease and no available diagnostic histopathological data, images were reviewed retrospectively by three pediatric radiologists. Crohn disease was considered to be present only if there was unanimous consensus opinion regarding the abnormality of the bowel segment (or segments) in question. These radiologists only were asked to document whether a bowel segment was normal or abnormal; they were not asked to detail specific MRE findings (such as bowel wall thickening, abnormal bowel wall postcontrast hyperenhancement, etc.).
Demographic information at the time of MRE, including the child’s age and gender, was documented by reviewing our electronic medical record system.
Statistical analysis
Continuous data were summarized using means and ranges, while categorical data were summarized by counts and percentages. The sensitivity of MRE for detecting the presence of pediatric Crohn disease involving the small bowel and/or colon, in general, was calculated with 95% exact confidence interval using histopathology as the reference standard. The sensitivity, specificity, positive predictive value and negative predictive value of MRE were also determined with 95% exact confidence intervals at the bowel segmental level (segments of jejunum and ileum confirmed to be positive for Crohn disease by consensus of three pediatric radiologists were not included in these calculations, as no correlative histopathological data were available). A two-sample t-test was used to assess for any significant difference in subject ages between genders.
Results
One-hundred and ninety-one MRE examinations were performed in pediatric patients at our institution during the study period (Fig. 1). A total of 32 MRE examinations (from 31 subjects) were identified with relevant correlative histopathological data following the application of exclusion criteria. Eighteen (56%) imaging studies were performed on male subjects, while 14 (44%) were performed on female subjects. At the time of imaging, mean subject age was 13.2 years (range, 6–17 years). Mean age was slightly higher for female subjects (14.1 years, range 8–17 years) compared to male subjects (12.5 years, range 6–17 years), although this difference was not statistically significant (P = 0.2). Histopathology data was acquired, on average, 9.6 days prior to MRE imaging (range, 51 days prior to MRE to 34 days after MRE).
MRE/histopathology correlation for Crohn disease
MRE had an overall sensitivity of 94% (95% CI [79%, 99%]; 30 true-positive and 2 false-negative examinations) for detecting the presence of pediatric Crohn disease, in general, involving the small bowel and/or colon.
A total of 149 (out of a possible 224) diagnostic ileocolorectal bowel segments were identified from the pertinent 32 MRE examinations. Seventy-five bowel segments were excluded due to either incomplete MRE or histopathological data. At the bowel segmental level, MRE had a sensitivity of 66% (95% CI [53%, 77%]; 44 true-positive and 23 false-negative bowel segments), specificity of 90% (95% CI [82%, 96%]; 74 true-negative and 8 false-positive bowel segments), positive predictive value of 85% (95% CI [72%, 93%]), and negative predictive value of 76% (95% CI [68%, 84%]). MRE demonstrated a sensitivity of 93% (14 true-positive and 1 false-negative examinations) for the detection of terminal ileitis, however. True-positive (Fig. 2), true-negative, false-positive (Fig. 3) and false-negative (Fig. 4) results by bowel segment are provided in Table 1.
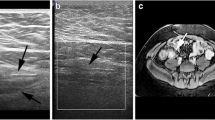
A 17-year-old girl with newly diagnosed ileocolonic Crohn disease. Both MRE and histopathology confirmed the presence of terminal ileitis. a Axial T2-W single-shot turbo spin-echo (SSTSE) image through the lower abdomen demonstrates marked wall thickening of the terminal ileum (arrows). b Axial T2-W turbo spin-echo (TSE) image with fat saturation demonstrates marked bowel wall edema (white arrows) and adjacent mesenteric inflammatory changes. An enlarged lymph node is noted adjacent to the terminal ileum (black arrow). c Postcontrast axial 3-D T1-weighted spoiled gradient recalled echo (SPGR) image with fat saturation reveals avid hyperenhancement of the terminal ileum (arrows) and adjacent mesentery
A 17-year-old boy with a history of colonic Crohn disease and increasing abdominal pain. MRE demonstrated marked terminal ileitis and apparent involvement of the cecum, while pancolonic endoscopic biopsies were normal and no diagnostic terminal ileal biopsy was obtained. Axial T2-W TSE image with fat saturation shows marked terminal ileal mural thickening and edema (white arrows). Inflammatory changes are noted in the adjacent mesentery. The cecal wall (black arrow) appears thickened and edematous
A 12-year-old girl with symptomatic Crohn disease and an ileocecectomy 4 weeks following MRE. The MRE was interpreted as normal, while surgical pathology suggested the presence of minimal active terminal ileitis. a, b Axial T2-W SSTSE and coronal 2-D balanced steady-state free precession (SSFP) images reveal excellent distention of the terminal ileum without appreciable wall thickening (arrows). c Postcontrast axial 3-D T1-W SPGR image with fat saturation demonstrates slight enhancement of the terminal ileum (arrow) that was interpreted as being within normal limits
Assessment of jejunal/ileal Crohn disease in absence of available histopathology
Fifteen of 30 (50%) MRE examinations lacked diagnostic histopathological evaluation of the terminal ileum by ileocolonoscopy and biopsy. In these 15 subjects, the terminal ileum was unable to be visualized or intubated, or the biopsy specimen provided for histopathological review labeled terminal ileum was either nondiagnostic or proved to be colon. The terminal ileum was unequivocally abnormal by MRE (confirmed by unanimous consensus opinion of three pediatric radiologists performing a retrospective review for this investigation) in 11 of 15 (73%) subjects lacking a diagnostic biopsy of this bowel segment (Figs. 3 and 5). Surgically resected bowel specimens provided the correlative histopathological data for two MRE examinations.
A 16-year-old girl with a history of ileocolic Crohn disease. MRE demonstrated marked terminal ileitis, while pancolonic endoscopic biopsies were normal and no diagnostic terminal ileal biopsy was obtained. a Coronal 2-D SSFP image shows abnormal bowel wall thickening of the terminal ileum (arrows) with adjacent mesenteric hypervascularity and fibrofatty proliferation. b Postcontrast 3-D T1-W SPGR image with fat saturation demonstrates marked terminal ileal hyperenhancement (arrows) and adjacent mesenteric vascular engorgement
Finally, previously unknown but unequivocal jejunal involvement by Crohn disease was diagnosed in 4 of 32 (13%) subjects by MRE (Figs. 6). These abnormal bowel segments were also confirmed to be abnormal based upon unanimous consensus opinion of three pediatric radiologists performing a retrospective review of these bowel segments for this investigation.
A 12-year-old boy with a 3-month history of abdominal pain, fevers, weight loss and elevated inflammatory markers. Endoscopic biopsies of the colon revealed scattered granulomas, while the terminal ileum was normal. MRE confirmed the presence of extensive small bowel Crohn disease affecting the jejunum. a Coronal 2-D SSFP image shows extensive jejunal wall thickening and nodularity in the left upper quadrant. Free fluid is present in the right lower quadrant (arrow). b Postcontrast axial 3-D T1-W SPGR image with fat saturation shows avid hyperenhancement of multiple thickened jejunal loops (arrows)
Discussion
Based on our results, MRE has a very high sensitivity for detecting the presence of pediatric Crohn disease, in general, involving the small bowel and/or colon, with a sensitivity greater than 90%. In our study, only two of 32 MRE examinations in children with Crohn disease failed to show evidence of disease. Our sensitivity is greater than that observed by Lipson et al. [20], who compared small bowel follow-through and biopsy for assessment pediatric Crohn disease of the terminal ileum. The exact sensitivity of CTE for detecting pediatric Crohn disease is unknown, though it is likely greater than that of endoluminal barium studies and similar to MRE based upon adult investigations [21]. It is also important to remember that the clinical information gained by MRE is acquired without using ionizing radiation, as compared to CTE and endoluminal barium studies. This fact is particularly important in children, who are at greater risk than adults for adverse effects related to radiation exposure [19].
MRE also appears to perform quite well at the bowel segmental level, with very few (8 of 149) bowel segments falsely positive for pediatric Crohn disease in our study. Because of this, MRE has a high specificity (90%) and positive predictive value (85%). Regarding the bowel segments that were falsely positive at MRE, we believe that it is quite possible that Crohn disease may have been present. This is because endoscopic biopsy is known to have sampling error that can cause a false-negative histopathological evaluation [22]. Sampling error is particularly relevant in assessing Crohn disease due to its eccentric, noncontiguous nature. While endoscopic biopsy is likely a slightly imperfect reference standard, it is still the best method available by which to assess the presence or absence of Crohn disease without performing surgical bowel resection.
The sensitivity of MRE for detecting individual abnormal bowel segments in children and adolescents with Crohn disease is 66% based on our study. A total of 23 diagnostic bowel segments were false-negative by MRE. Interestingly, 22 of these 23 false-negative bowel segments were either colon or rectum, locations that are typically easily accessible to endoscopic evaluation and biopsy. A study by Schreyer et al. [23] in adults also demonstrated that dedicated MR-based colonography, even after a bowel cleansing regimen used for optical colonoscopy, has a limited sensitivity for detecting colonic Crohn disease and that mild inflammation in most cases cannot be recognized. Only a single terminal ileal bowel segment was a false-negative at MRE, however. Consequently, given the number of true-positive terminal ileal segments, MRE has an extremely high sensitivity (93% based on our study) for the detection of terminal ileitis related to pediatric Crohn disease. Additionally, MRE confirmed the presence of terminal ileitis in 11 of 15 subjects lacking a diagnostic biopsy specimen of the terminal ileum, based on consensus review by three pediatric radiologists.
An additional benefit of MRE is its ability to assess more proximal ileum and jejunum. Crohn disease affecting the jejunum is more common in children than adults [24], and up to 10–15% of affected children will have diffuse small bowel involvement [2]. Four of 32 (13%) subjects in our study had previously unknown jejunal Crohn disease. The extent of jejunal disease was quite long in all four children. While all four subjects had Crohn disease involving the rectum and/or colon, only two had known small bowel involvement affecting the terminal ileum. In two subjects, MRE provided the first evidence of small bowel Crohn disease.
Our study has limitations. First, it is retrospective in design and relies upon the reports issued by a number of pathologists and radiologists, who likely have differing experience (and differing sensitivities and specificities) for the diagnosis of pediatric Crohn disease. MRE images were not retrospectively reanalyzed for our study, and these imaging studies were originally reported by seven different fellowship-trained pediatric radiologists. Second, this investigation contains a relatively small number of MRE examinations with correlative histopathology within 2 months of imaging. Even with our busy institutional pediatric MRE program, it is difficult to find children with Crohn disease, no prior bowel surgical resection and relevant histopathology within 2 months of imaging. Third, as mentioned above, histopathology is an imperfect reference standard due to endoscopic biopsy sampling errors. For example, the proximal ascending colon near the ileocecal valve may have appeared abnormal by MRE, whereas the endoscopic biopsy specimen labeled ascending colon could have been taken more distally near the hepatic flexure. Similarly, establishing the boundaries of specific bowel segments can be difficult by both MRE and endoscopy (for example, identifying the exact location of the rectosigmoid junction) and may be variable between radiologists and endoscopists. Fourth, there was, on average, 9.6 days between the acquisition of histopathological specimens and MRE imaging. While it is possible that during this time period disease activity could change, we believe that it is unlikely that a segment of bowel would go from entirely uninvolved to involved by Crohn disease or vice versa. Fifth, it is impossible to determine whether a radiologist reviewed available histopathology data at the time of MRE interpretation or whether a pathologist reviewed available imaging reports. Such a review could ultimately bias a radiologist’s final MRE report or a pathologist’s histopathology report. Finally, our study did not assess additional benefits of MRE, such as its ability to appraise disease activity and identify disease-related complications and extra-intestinal manifestations. We hope to address these additional benefits in future studies.
Conclusion
MRE successfully identifies the presence of Crohn disease affecting the small bowel and/or colon in children and adolescents. Additionally, MRE has a high specificity and positive predictive value for detecting disease involvement at the bowel segmental level. Our study demonstrates that this imaging test is particularly valuable for establishing the presence of terminal ileitis in the setting of a nondiagnostic endoscopic biopsy of this bowel segment. Finally, based on our data, MRE commonly identifies previously unknown jejunal involvement in pediatric Crohn disease patients. This investigation provides essential evidence-based support for the recently observed shift away from imaging modalities that use ionizing radiation to MRE for the appraisal of pediatric Crohn disease.
References
Malaty HM, Fan X, Opekun AR et al (2010) Rising incidence of inflammatory bowel disease among children: a 12-year study. J Pediatr Gastroenterol Nutr 50:27–31
Diefenbach KA, Breuer CK (2006) Pediatric inflammatory bowel disease. World J Gastroenterol 12:3204–3212
Heyman MB, Kirschner BS, Gold BD et al (2005) Children with early-onset inflammatory bowel disease (IBD): analysis of a pediatric IBD consortium registry. J Pediatr 146:35–40
Vucelic B (2009) Inflammatory bowel diseases: controversies in the use of diagnostic procedures. Dig Dis 27:269–277
Dillman JR, Adler J, Zimmermann EM et al (2010) CT enterography of pediatric Crohn disease. Pediatr Radiol 40:97–105
Paolantonio P, Ferrari R, Vecchietti F et al (2009) Current status of MR imaging in the evaluation of IBD in a pediatric population of patients. Eur J Radiol 69:418–424
Godefroy C, Pilleul F, Dugougeat F et al (2005) Value of contrast-enhanced MR enterography in pediatric Crohn’s disease: preliminary study. J Radiol 86:1685–1692
Alexopoulou E, Roma E, Loggitsi D et al (2009) Magnetic resonance imaging of the small bowel in children with idiopathic inflammatory bowel disease: evaluation of disease activity. Pediatr Radiol 39:791–797
Adler J, Higgins PD (2010) Getting the steak without the sizzle: is MR enterography as good as CT enterography? Inflamm Bowel Dis 16:712–713
Darbari A, Sena L, Argani P et al (2004) Gadolinium-enhanced magnetic resonance imaging: a useful radiological tool in diagnosing pediatric IBD. Inflamm Bowel Dis 10:67–72
Darge K, Anupindi SA, Jaramillo D (2008) MR imaging of the bowel: pediatric applications. Magn Reson Imaging Clin N Am 16:467–478
Dinkel E, Dittrich M, Peters H et al (1986) Real-time ultrasound in Crohn’s disease: characteristic features and clinical implications. Pediatr Radiol 16:8–12
Faure C, Belarbi N, Mougenot JF et al (1997) Ultrasonographic assessment of inflammatory bowel disease in children: comparison with ileocolonoscopy. J Pediatr 130:147–151
Haber HP, Busch A, Ziebach R (2000) Bowel wall thickness measured by ultrasound as a marker of Crohn’s disease activity in children. Lancet 355:1239–1240
Scholbach T, Herrero I, Scholbach J (2004) Dynamic color Doppler sonography of intestinal wall in patients with Crohn disease compared with healthy subjects. J Pediatr Gastroenterol Nutr 39:524–528
Bremner AR, Pridgeon J, Fairhurst J et al (2004) Ultrasound scanning may reduce the need for barium radiology in the assessment of small-bowel Crohn’s disease. Acta Paediatr 93:479–481
Alison M, Kheniche A, Azoulay R et al (2007) Ultrasonography of Crohn disease in children. Pediatr Radiol 37:1071–1082
Bremner AR, Griffiths M, Argent JD et al (2006) Sonographic evaluation of inflammatory bowel disease: a prospective, blinded, comparative study. Pediatr Radiol 36:947–953
Brenner DJ (2008) Should computed tomography be the modality of choice for imaging Crohn’s disease in children? The radiation risk perspective. Gut 57:1489–1490
Lipson A, Bartram CI, Williams CB et al (1990) Barium studies and ileoscopy compared in children with suspected Crohn’s disease. Clin Radiol 41:5–8
Lee SS, Kim AY, Yang SK et al (2009) Crohn disease of the small bowel: comparison of CT enterography, MR enterography, and small-bowel follow-through as diagnostic techniques. Radiology 251:751–761
Calhoun BC, Gomes F, Robert ME et al (2003) Sampling error in the standard evaluation of endoscopic colonic biopsies. Am J Surg Pathol 27:254–257
Schreyer AG, Rath HC, Kikinis R et al (2005) Comparison of magnetic resonance imaging colonography with conventional colonoscopy for the assessment of intestinal inflammation in patients with inflammatory bowel disease: a feasibility study. Gut 54:250–256
Ali SI, Carty HM (2000) Paediatric Crohn’s disease: a radiological review. Eur Radiol 10:1085–1094
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dillman, J.R., Ladino-Torres, M.F., Adler, J. et al. Comparison of MR enterography and histopathology in the evaluation of pediatric Crohn disease. Pediatr Radiol 41, 1552–1558 (2011). https://doi.org/10.1007/s00247-011-2186-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00247-011-2186-0