Abstract
Travel distance to surgical centers may be increased when coverage restrictions prevent children with congenital heart disease (CHD) from receiving care at out-of-state congenital heart surgery centers. We estimated the minimum travel distance to congenital heart surgery centers among publicly insured infants with time-sensitive CHD surgical needs, under two different scenarios: if they were and were not restricted to in-state centers. Using 2012 Medicaid Analytic eXtract data from 40 states, we identified 4598 infants with CHD that require surgery in the first year of life. We calculated the minimum travel distance between patients’ homes and the nearest cardiac surgery center, assuming patients were and were not restricted to in-state centers. We used linear regression to identify demographic predictors of distance under both scenarios. When patients were not restricted to in-state centers, mean minimum travel distance was 43.7 miles, compared to 54.1 miles when they were restricted. For 5.9% of patients, the difference in travel distance under the two scenarios exceeded 50 miles. In six states, the difference in mean minimum travel distance exceeded 20 miles. Under both scenarios, distance was positively predicted by rural status, residence in middle-income zip codes, and white/non-Hispanic or American Indian/Alaskan Native race/ethnicity. For some publicly insured infants with severe CHD, facilitating the receipt of out-of-state care could mitigate access barriers. Existing efforts to regionalize care at fewer centers should be designed to avoid exacerbating access barriers among publicly insured CHD patients.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Congenital heart disease (CHD) represents the most common and resource-intensive class of birth defects in the US. Approximately 6 in every 1000 live-born children require cardiac heart surgery for CHD [1], and half of these patients are covered by public insurance programs such as Medicaid or the Children’s Health Insurance Program, CHIP [2,3,4,5,6,7,8,9]. Given the number and vulnerability of publicly insured CHD patients who require surgery, minimizing barriers to accessing surgical care is an important clinical and policy goal [10, 11].
For many publicly insured CHD patients who require early surgery, such as infants with Tetralogy of Fallot, a potentially important access barrier may be the distances they must travel to congenital heart surgery centers. These patients may have limited financial resources to travel long distance for surgery, or to stay for extended periods in hotels during hospitalizations [12,13,14]. Additionally, travel distance may be high due to barriers to accessing out-of-state care. By federal law, state Medicaid programs must pay for out-of-state care if there is an emergency, the service is not available in the state, or the standard of care in the area is to receive care across state lines. When these conditions are not met, some patients may have to travel to in-state centers even when an out-of-state center is much closer [15]. Even when a state Medicaid program is willing to pay for out-of-state care, patients may still be unable to access this care if the out-of-state provider is not enrolled in the Medicaid program of the patient’s state [16,17,18]. For example, some providers do not enroll in out-of-state Medicaid programs when reimbursement rates are lower than that of their home state [19, 20].
A recent study described the distances that children with CHD patients traveled for surgery, and showed that approximately 25% of patients traveled greater than 100 miles [21]. This study was important and highly rigorous, but did not focus on the publicly insured, included patients with both time-sensitive and less urgent surgical needs, and did not assess the degree to which access to out-of-state surgical centers could decrease travel distance. To address these gaps, we used national 2012 Medicaid claims to estimate the magnitude and predictors of travel distance for care among publicly insured infants requiring surgery in the first year of life, assuming these patients were or were not restricted to in-state centers. We focused on this population because their time-sensitive surgical needs may make them particularly vulnerable to access barriers from high travel distance.
Methods
Primary Data Sources
Congenital heart surgical centers were identified using the 2012 directory of 119 centers released by Congenital Cardiology Today (Online Appendix 1) [22]. This directory lists providers and includes addresses at all centers in North America that perform open heart surgeries in children. We chose this as our source of provider supply, rather than surgeon-level data (such as the Society of Thoracic Surgeons-Congenital Heart Surgery Registry or the American Medical Association Physician Masterfile) because some congenital heart surgeons operate at multiple centers in different locations but submit data under a single tax identifier associated with one primary location.
Data on patient characteristics and diagnoses were obtained from the 2012 Medicaid Analytic eXtract (MAX) database from 40 “states” (39 states and the District of Columbia). Each of these 40 states contained a congenital heart surgical center. Seven states without a congenital heart surgical center (Alaska, Montana, New Hampshire, North Dakota, South Dakota, Vermont, Wyoming) were excluded as patients in these states could not be “restricted” to in-state care. We also excluded patients living in Hawaii, given the questionable validity of using straight-line distance across a large body of water to reflect distance to out-of-state care. Finally, we lacked access to MAX data for three states at the time of analysis (Colorado, Idaho, Kansas). Based on 2010 data, these 39 states plus the District of Columbia collectively account for > 95% of Medicaid births [23].
MAX data are research-friendly, publicly accessible versions of Medicaid claims, submitted by states to the Centers for Medicare and Medicaid Services (CMS) on annual bases. Data include claims from children enrolled in both fee-for-service and managed Medicaid, as well as those enrolled in the CHIP [24, 25]. MAX data are an important source of data for national studies of pediatric care [26], with more than 40% of children covered by public insurance at some point during each year [27]. Unlike procedure-based registries or state inpatient administrative databases that only include CHD patients who undergo surgery, MAX data can be used to identify all children with selected CHD diagnoses, regardless of whether they underwent surgery. We used data from 2012 as it is the most recent year for which MAX data are available from the majority of states.
Sample
Our sample included infants who were born during 2012 and who had one or more inpatient or outpatient claim during 2012 with one of the 11 ICD-9-CM diagnosis codes (Table 1). These codes corresponded to 10 CHD conditions that always or almost always require surgical repair within the first year of life. Conditions were not mutually exclusive (e.g., patients could have both Tetralogy of Fallot and pulmonary valve atresia).
Study Variables
The main outcome variable was minimum travel distance for care, defined as the straight-line distance (in miles) between zip code centroids of a patient’s home and the nearest congenital heart surgical center. Distances between zip code centroids have been found to be a good proxy for driving distance and driving time [28,29,30]. Although our cohort only included patients from 40 states, we assumed patients could travel to centers in any state. Of note, our outcome does not represent the distance patients actually traveled for care, which cannot be reliably assessed in MAX data and may be affected by considerations such as a center’s reputation or volume. Rather, our outcome represented a lower bound of travel distance for care. We calculated our outcome under two scenarios. First, we assumed that patients could travel to the nearest center even if it was out-of-state. Second, we assumed that patients could not receive out-of-state care [13,14,15,16,17,18].
From the MAX data, we obtained information on patient age in months at the time of their first claim with one of the 11 CHD diagnosis codes, sex, zip code, and race/ethnicity. The MAX race/ethnicity variable indicates whether patients are Hispanic or belong to the following non-Hispanic categories: white, black, Asian/Pacific Islander, American Indian/Alaskan Native, Native Hawaiian/Pacific Islander, more than one race, or unknown race.
From 2012 Internal Revenue Service data, we obtained mean-adjusted gross income by patient zip code of residence [31]. From 2010 US Census Bureau data, we obtained the urban/rural designation of patient zip code of residence. The Census Bureau defines urban areas as zip codes with ≥ 50,000 people, urban–rural areas as zip codes with 2500–49,999 people, and rural areas as zip codes with < 2500 people [32].
Statistical Analysis
For both scenarios of travel distance (restricting and not restricting patients to in-state centers), we used standard descriptive statistics to calculate the national mean and median of minimum travel distance. We also calculated travel distance by each demographic characteristic and diagnosis. For both scenarios, we also determined the proportion of patients with travel distance of 0–24.9, 25–49.9, 50–99.9, and ≥ 100 miles.
We calculated the difference in minimum travel distance for each patient assuming they were or were not restricted to in-state centers, then calculated the proportion of patients for whom this difference exceeded 50 miles (corresponding to roughly 1 h of additional travel time). We averaged this difference over all patients residing in each state to calculate the mean difference at the state level, and used a non-parametric test (Wilcoxon signed-rank test) to assess the significance of this difference for each state. We calculated the number of states for which the difference exceeded 20 miles (corresponding to roughly 1 standard deviation above the mean of the 39 state differences). To assess predictors of minimum travel distance under both scenarios, we used linear regression to model log-transformed travel distance as a function of patient age in months, sex, race/ethnicity, mean-adjusted gross income by zip code (by quartiles), and urban/rural status of zip code of residence. For ease of interpretation, we exponentiated coefficients from regressions to obtain the multiplicative changes in distances associated with changes in predictors.
Data on race/ethnicity, sex, and mean-adjusted gross income by zip code were missing for 29.1%, 0.3%, and 2.1% of the cohort, respectively. Missingness of race/ethnicity was strongly predicted by state, supporting the assumption that these data were missing at random. We imputed missing data using multiple imputation by chained equations with 20 imputations. Race/ethnicity, sex, mean-adjusted gross income by zip code, patient age, urban/rural status, and state were used in imputation models.
Analyses were performed using Stata 15 (College Station, TX). The Institutional Review Boards of the University of Chicago and Columbia University Irving Medical Center exempted this study from review.
Sensitivity Analyses
To assess the robustness of findings to changes in patient cohort selection, we repeated our analyses with the following modifications: (1) We only included infants with two or more instances of the 11 ICD-9-CM diagnosis codes, separated by a day or more (n = 3266) (2) we additionally included 1337 infants with CHD conditions that often, but do not always, require surgical repair in the first year of life, such as endocardial cushion defect (745.6, n = 147), atresia and stenosis of the aorta (747.22, n = 172), and pulmonary artery coarctation and atresia (747.31, n = 1018), and (3) we excluded infants with missing sex, race/ethnicity, or zip code income data (n = 1420).
We also calculated travel distance (assuming that patients could travel outside their state) when additionally including data from seven states that lacked a congenital heart surgical center (Online Appendix 2).
Results
The sample included 4598 infants. Of all infants, 21% were less than 3 months old at the time of identification, 45% less than 6 months, and 71% less than 9 months. The majority of patients lived in urban–rural zip codes (57%) or urban zip codes (35%). The three most common diagnoses were Tetralogy of Fallot (31%), hypoplastic left heart syndrome (23%), and transposition of the great vessels (17%, Table 1).
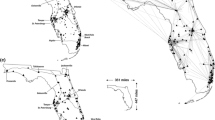
The 119 congenital heart surgical centers are listed in Online Appendix 1. Figure 1a and Table 2 display the number of patients in each state. Patient density generally correlated with changes in the population of each state. Figure 1b shows the mean distances by state, assuming that patients could cross state lines.
Distance
Assuming that patients could cross state lines, mean minimum travel distance was 43.7 (SD 46.3) miles, and median minimum travel distance was 24.7 (25–75th percentile 7.8–68.7) miles. The proportion of patients traveling 0–24.9, 25–49.9, 50–99.9, and 100 + miles was 50.2%, 15.9%, 20.8%, and 13.1%, respectively.
Assuming that patients could not cross state lines, mean minimum travel distance was 54.1 (SD 86.2) miles, and median minimum travel distance was 27.4 (25–75th percentile 8.2–79.3) miles. The proportion of patients traveling 0–24.9, 25–49.9, 50–99.9, and 100 + miles was 48.2%, 14.6%, 19.5%, and 17.7%, respectively.
The difference in minimum travel distance when patients were and were not restricted to in-state centers ranged from 0 to 621.5 miles (mean and median 9.2 and 0.0 miles). The median was 0.0 because for 87.0% of patients, the in-state option was the closest option. For 5.9% of patients in the sample, the difference in minimum travel distance exceeded 50 miles. This proportion varied widely between states, ranging from 0.6% in New York to 25.3% in Illinois.
Among the 39 states, the mean difference in minimum travel distance when patients could and could not cross state lines ranged from 0.0 to 33.4 miles (mean and median 10.1 and 8.5 miles, Table 2). For six states, this difference exceeded 20 miles—Illinois, West Virginia, Indiana, Nevada, Tennessee, and Utah. The difference was statistically significant for 26 states. Northeastern states demonstrated the smallest increase in travel distance. In a few states (e.g., Minnesota), the difference was zero, implying that for patients in these states, the nearest center would be the same regardless of whether there was an out-of-state restriction.
Patient Demographic Predictors of Distance
Assuming that patients could cross state lines, non-Hispanic black and Hispanic race/ethnicity was associated with 21% (CI 13–28%) and 16% (CI 7–22%) lower distance compared to white/non-Hispanic race/ethnicity, respectively. Residence in a rural area was associated with a 689% greater distance compared to residence in an urban area (CI 593–799%). Residence in zip codes in the second and third highest quartile of mean-adjusted gross income was associated with a 30% (CI 19–43%) and 31% (19–44%) increase in distance compared to the poorest quartile, respectively. Sex and age were not significant predictors of distance (Table 3). When assuming that patients were restricted to in-state centers, demographic predictors of distance were similar.
Sensitivity Analyses
Conclusions were not substantively different when excluding patients with only one instance of an ICD-9-CM code, when including infants with CHD that do not always require surgical repair in the first year of life, and when excluding infants with missing data (Online Appendices 3, 4, and 5).
Discussion
Using national data from 39 states and the District of Columbia, we assessed the minimum travel distance to CHD centers among publicly insured infants with CHD conditions requiring early surgery, assuming they were or were not restricted to in-state centers. The difference in minimum travel distance under these scenarios exceeded 50 miles for 5.9% of patients. This proportion varied widely between states, ranging from 0.6% in New York to 25.3% in Illinois. White/non-Hispanic or American Indian/Native race/ethnicity, with residence in rural areas, and with residence in middle-income zip codes were each associated with higher minimum travel distance. These findings suggest that many publicly insured infants with time-sensitive surgical needs must travel long distances for care, particularly infants in certain demographic and geographic groups.
This study used national data to estimate minimum travel distance in a cohort of CHD patients who may be particularly vulnerable to access barriers due to high travel distances. Previous studies examining travel distance to congenital heart surgical centers have focused on single institutions or have included adult CHD patients, patients without CHD, patients with CHD that does not require surgery, or privately insured patients [19, 33,34,35]. We also report for the first time that residence in a rural area was positively associated with travel distance, and that racial/ethnic minority status other than Native American was associated with lower travel distance compared to white race/ethnicity, likely due to the concentration of racial/ethnic minorities in urban areas.
Our findings have implications for the current debate about regionalization of CHD surgical care [36,37,38,39,40,41,42]. Studies have demonstrated up to sevenfold variation in risk-adjusted outcomes between congenital heart surgery centers [43,44,45,46,47,48]. Consequently, many advocate establishing “centers of excellence” for CHD care, which could divert patients to high-volume centers, minimize practice variation, and potentially improve outcomes and decrease costs [49,50,51,52]. Such a model assumes that patients can travel long distances and bypass their nearest surgical center. While studies demonstrate that some patients do bypass the nearest center [19, 33, 35], these studies included privately and publicly insured patients with both elective and emergent surgical needs. Our results demonstrate that certain demographic groups of publicly insured infants with time-sensitive surgical needs already live far from care. Closing centers under a regionalization model might exacerbate access barriers for these patients, depending on whether these centers disproportionately serve these patients and the availability of alternative nearby centers. Future research is needed to assess whether the benefits of regionalization may outweigh any increased access barriers from longer travel distances, using approaches such as simulation exercises [40], or cost-effectiveness analysis.
Our study is the first to quantify the degree to which minimum travel distance would differ if publicly insured infants with CHD were or were not always restricted to in-state centers. The magnitude of the difference in distance under these two scenarios varied widely. Our findings suggest that facilitating the receipt of out-of-state care may be an important step toward alleviating current access barriers, particularly for patients in certain states. Of note, facilitating such care would also be crucial if CHD surgical care continues to be regionalized. Because regionalization could decrease the number of available surgical centers, fewer states might have in-state options. Thus, policies that guarantee out-of-state Medicaid coverage, such as automatically enrolling providers in the Medicaid programs of nearby states, may be essential to minimize access barriers under a regionalization model [17,18,19].
Our study has some limitations. First, we calculated the minimum distances patients must travel for care, which may be lower than the distance they actually travel and which therefore may underestimate access barriers. There might be practical reasons why some patients elect to bypass their nearest center or be referred elsewhere. Some patients may select centers based on reputation or outcomes [53], prior relationships with providers, or established referral practices between their pediatric cardiologist (or pediatrician or insurance provider) and specific surgeons. Likewise, some patients might have family or other supports closer to other centers.
Second, we lacked information on the center at which patients ultimately received care. Therefore, we could not assess whether in-state restrictions might have impeded access to higher-quality out-of-state centers. Third, we did not assess whether in-state restrictions might worsen outcomes by increasing travel distance. MAX data lack detailed outcomes data (e.g., morbidity, quality of life), and the date-of-death fields are not always complete. Furthermore, there are many potential confounders of the relationship between outcomes and travel distance that cannot be controlled for using these data.
Finally, we used 2012 data. Currently, 2012 is the latest year in which MAX data from almost every state are available. Future studies should repeat our analyses using more recent data as they are released. Finally, MAX data submitted by comprehensive managed care organizations may not perfectly capture diagnoses, though the quality of these data is improving [54]. In sensitivity analyses, findings were not affected substantially by changes to our cohort. Finally, our study focused on inpatient surgical care and did not examine post-operative outpatient care that may be required throughout a patient’s lifetime.
Conclusion
Among publicly insured infants with CHD who require early surgery, many live far away from surgical centers that can provide definitive care, with some demographic and geographic groups at a particular disadvantage. For many of these infants, facilitating the receipt of out-of-state care could alleviate access barriers. Efforts to regionalize CHD surgical care at fewer centers should be designed to avoid exacerbating access barriers for patients, particularly those who already live far from care.
References
Hoffman JI, Kaplan S (2002) The incidence of congenital heart disease. J Am Coll Cardiol 39:1890–1900
Connor JA, Gauvreau K, Jenkins KJ (2005) Factors associated with increased resource utilization for congenital heart disease. Pediatrics 116:689–695
Finlayson SR, Birkmeyer JD, Tosteson AN, Nease RF Jr (1999) Patient preferences for location of care: implications for regionalization. Med Care 37:204–209
Smith PC, Powell KR (2002) Can regionalization decrease the number of deaths for children who undergo cardiac surgery? A theoretical analysis. Pediatrics 110:849–850
Quintessenza JA, Jacobs JP, Morrell VO (2003) Issues in regionalization of pediatric cardiovascular care. Prog Pediatr Cardiol 18:49–53
Lorch SA, Silber JH, Even-Shoshan O, Millman A (2009) Use of prolonged travel to improve pediatric risk-adjustment models. Health Serv Res 44:519–541
Burki S, Fraser CD (2016) Larger centers may produce better outcomes: is regionalization in congenital heart surgery a superior model? Semin Thorac Cardiovasc Surg Pediatr Card Surg Annu 19:10–13
Danton MH (2016) Larger centers produce better outcomes in pediatric cardiac surgery: regionalization is a superior model—the con prospective. Semin Thorac Cardiovasc Surg Pediatr Card Surg Annu 19:14–24
Anderson BR, Fieldston ES, Newburger JW, Bacha EA, Glied SA (2018) Disparities in outcomes and resource use after hospitalization for cardiac surgery by neighborhood income. Pediatrics 141:e20172432
Erickson LC, Wise PH, Cook EF, Beiser A, Newburger JW (2000) The impact of managed care insurance on use of lower-mortality hospitals by children undergoing cardiac surgery in California. Pediatrics 105:1271–1278
Chang R-K, Chen AY, Klitzner TS (2000) Factors associated with age at operation for children with congenital heart disease. Pediatrics 105:1073–1108
Kucik JE, Nembhard WN, Donohue P, Devine O, Wang Y, Minkovitz CS, Burke T (2014) Community socioeconomic disadvantage and the survival of infants with congenital heart defects. Am J Public Health 104(11):150–157
Werner H, Latal B, Valsangiacomo Buechel E, Beck I, Landolt MA (2014) The impact of an infant’s severe congenital heart disease on the family: a prospective cohort study. Congenit Heart Dis 9(3):203–210
Fixler DE, Nembhard WN, Xu P, Ethen MK, Canfield MA (2012) Effect of acculturation and distance from cardiac center on congenital heart disease mortality. Pediatrics 129(6):1118–1124
United States Code of Federal Regulations. Title 42 (Public Health), section 431.52
United States Social Security Act, Section 1932 (b)(2)
United States Congress Bill. Accelerating Kids’ Access to Care Act. H.R.6524. Published 25 July 2018. https://www.congress.gov/bill/115th-congress/house-bill/6524. Accessed 12 Dec 2018
Committee on Child Health Financing, American Academy of Pediatrics (2013) Policy statement: Medicaid policy statement. Pediatrics 131(5):1697–1706
Schneider A (2018) How to make proper payments to out-of-state providers for Medicaid children with special health care needs. Georgetown University Health Policy Institute. Published 3 May 2018. https://ccf.georgetown.edu/2018/05/03/how-to-make-proper-payments-to-out-of-state-providers-for-medicaid-children-with-special-health-care-needs. Accessed 12 Dec 2018
FamiliesUSA. Fact sheet: interstate Medicaid billing problems and helping Medicaid beneficiaries who get care out of state. Published 18 April 2014. https://familiesusa.org/sites/default/files/product_documents/Interstate%20Medicaid%20Billing%20Problems.pdf. Accessed 12 Dec 2018
Welke KF, Pasquali SK, Lin P, Backer CL, Overman DM, Romano JC, Jacobs JP, Karamlou T (2019) Hospital distribution and patient travel patterns for congenital cardiac surgery in the United States. Ann Thorac Surg 107(2):574–581
Bhatt DR, Jue K, Stillwell J (2012) Congenital cardiac care providers in North America that offer open heart surgery for children. Congenit Cardiol Today (Partnersh Adult Congenit Pediatr Cardiol Sect Am Coll Cardiol). https://www.congenitalcardiologytoday.com/index_files/CCT-DIR-2012.pdf. Accessed 12 Jan 2017
Markus AR, Andres E, West KD, Garro N, Pellegrini C (2013) Medicaid covered births, 2008 through 2010, in the context of the implementation of health reform. Womens Health Issues 23:e273–e280
Medicaid analytic eXtract (MAX) general information. Centers for Medicare and Medicaid Services. Last modified 2 March 2018. https://www.cms.gov/research-statistics-data-and-systems/computer-data-and-systems/medicaiddatasourcesgeninfo/maxgeneralinformation.html. Accessed 7 Feb 2018
Harrington M, Kenney GM, Smith K, Clemans-Cope L, Trenholm C, Hill I, Orzol S, McMorrow S, Hoag S, Haley J, Zickafoose J, Waidmann T, Dye C, Benatar S, Qian C, Buettgens M, Fisher T, Lynch V, Hula L, Anderson N, Finegold K (2014) CHIPRA mandated evaluation of the Children’s Health Insurance Program: final findings. Mathematica Policy Research, Ann Arbor
Huybrechts KF, Palmsten K, Avorn J, Cohen LS, Holmes LB, Franklin JM, Mogun H, Levin R, Kowal M, Setoguchi S, Hernandez-Diaz S (2014) Antidepressant use in pregnancy and the risk of cardiac defects. N Engl J Med 370(25):2397–2407
Henry J. Medicaid State Fact Sheets. Kaiser Family Foundation. August 2013. https://kaiserfamilyfoundation.files.wordpress.com/2013/08/8050-06-medicaid-enrollment.pdf. Accessed 7 Feb 2019
Bazzoli GJ, Lee W, Hsieh HM, Mobley LR (2012) The effects of safety net hospital closures and conversions on patient travel distance to hospital services. Health Serv Res 47(1.1):129–150
Bliss RL, Katz JN, Wright EA, Losina E (2012) Estimating proximity to care: are straight line and zipcode centroid distances acceptable proxy measures? Med Care 50(1):99
Gregory PM, Malka ES, Kostis JB, Wilson AC, Arora JK, Rhoads GG (2000) Impact of geographic proximity to cardiac revascularization services on service utilization. Med Care 38(1):45–57
SOI tax stats—individual income tax 2012 zip code data. United States Internal Revenue Service. Last updated 18 May 2018. https://www.irs.gov/statistics/soi-tax-stats-individual-income-tax-statistics-2012-zip-code-data-soi. Accessed 21 Sep 2017
Geography: 2010 census urban and rural classification and urban area criteria. United States Census Bureau. Last updated 9 Feb 2015. https://www.census.gov/geo/reference/ua/urban-rural-2010.html. Accessed 23 Sep 2017
Pinto NM, Lasa J, Dominguez TE, Wernovsky G, Tabbutt S, Cohen MS (2012) Regionalization in neonatal congenital heart surgery: the impact of distance on outcome after discharge. Pediatr Cardiol 33:229–238
Mayer ML, Beil HA, von Allmen D (2009) Distance to care and relative supply among pediatric surgical subspecialties. J Pediatr Surg 44:483–495
Salciccioli K, Ermis P, Oluyomi A, Lopez K. Geographic disparities in access to care for adult congenital heart disease patients in the United States. In: Abstract presented at: 67th annual meeting of the American College of Cardiology, 9–11 March 2018, Orlando, Florida
Lorch SA, Myers S, Carr B (2010) The regionalization of pediatric health care. Pediatrics 126:2010–2019
Luft HS, Bunker JP, Enthoven AC (1979) Should operations be regionalized? The empirical relation between surgical volume and mortality. N Engl J Med 301:1364–1369
Moodie DS (2003) Regionalization of pediatric cardiac services. Prog Pediatr Cardiol 18:55–58
Lorch SA (2015) Ensuring access to the appropriate health care professionals: regionalization and centralization of care in a new era of health care financing and delivery. JAMA Pediatr 169:11–12
Chang R-K, Klitzner TS (2002) Can regionalization decrease the number of deaths for children who undergo cardiac surgery? A theoretical analysis. Pediatrics 109:173–181
New York State Department of Public Health. Pediatric congenital cardiac surgery in New York state, 2010–2013. Published Sep 2016. https://www.health.ny.gov/statistics/diseases/cardiovascular/heart_disease/docs/2010-2013_pediatric_congenital_cardiac_surgery.pdf. Accessed 8 Dec 2018
Cohen E. After CNN investigation, a push to halt child heart surgery at some hospitals. CNN, 13 Jan 2016. https://www.cnn.com/2016/01/13/health/jama-childrens-heart-surgery-hospitals/index.html. Accessed 8 Dec 2018
Jacobs JP, O’Brien SM, Pasquali SK, Jacobs ML, Jacour-Gayet FG, Tchervenkov CI, Austin EH III, Pizarro C, Pourmoghadam KK, Scholl FG, Welke KF, Mavroudis C (2011) Variation in outcomes for benchmark operations: an analysis of the Society of Thoracic Surgeons Congenital Heart Surgery Database. Ann Thorac Surg 92:2184–2192
Jenkins KJ, Newburger JW, Lock JE, Davis RB, Coffman GA, Iezzoni LI (1995) In-hospital mortality for surgical repair of congenital heart defects: preliminary observations of variation by hospital caseload. Pediatrics 95:323–330
Hirsch JC, Gurney JG, Donohue JE, Gebremariam A, Bove EL, Ohye RG (2008) Hospital mortality for Norwood and arterial switch operations as a function of institutional volume. Pediatr Cardiol 29:713–717
Chan T, Kim J, Minich LL, Pinto NM, Waitzman NJ (2015) Surgical Volume, hospital quality, and hospitalization cost in congenital heart surgery in the United States. Pediatr Cardiol 36:205–213
Anderson BR, Ciarleglio AJ, Cohen DJ, Lai WW, Neidell M, Hall M, Glied SA, Bacha EA (2016) The Norwood operation: relative effects of surgeon and institutional volumes on outcomes and resource utilization. Cardiol Young 26:683–692
Freeman CL, Bennett TD, Casper TC, Larsen GY, Hubbard A, Wilkes J, Bratton SL (2014) Pediatric and neonatal extracorporeal membrane oxygenation; does center volume impact mortality? Crit Care Med 42:512–519
Gardner W, Kelleher K (2014) A learning health care system for pediatrics. JAMA Pediatr 168:303–304
Pasquali SK, Dimick JB, Ohye RG (2015) Time for a more unified approach to pediatric health care policy? The case of congenital heart care. JAMA 314:1689–1690
Congenital Heart Public Health Consortium, CHPC (2012) CHPC FAQ fact sheet—long version. American Academy of Pediatrics. https://www.aap.org/en-us/Documents/chphc/chd_fact_sheet_long.pdf. Accessed 1 June 2018
Chua K, Conti RM, Freed GL (2018) Appropriately framing child health care spending: a prerequisite for value improvement. JAMA 319:1087–1088
The Society of Thoracic Surgeons. STS public reporting online: congenital heart surgery public reporting. https://publicreporting.sts.org/chsd. Accessed 12 Jan 2017
Byrd VL, Dodd AH (2015) Assessing the usability of encounter data for enrollees in comprehensive managed care 2010–2011. Mathematica Policy Research, Ann Arbor
Acknowledgements
The authors would like to thank Chuanhong Liao and Philip Schumm for their programming assistance.
Funding
Dr. Anderson receives Salary Support from the National Institutes of Health/National Heart Lung and Blood Institute (Grant Number K23 HL13343). The Medicaid Working Group at the University of Chicago provided funding for programming support. Dr. Chua is supported by a Career Development Award from the National Institute on Drug Abuse (Grant Number 1K08DA048110-01).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Woo, J.L., Anderson, B.R., Gruenstein, D. et al. Minimum Travel Distance Among Publicly Insured Infants with Severe Congenital Heart Disease: Potential Impact of In-state Restrictions. Pediatr Cardiol 40, 1599–1608 (2019). https://doi.org/10.1007/s00246-019-02193-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00246-019-02193-1