Abstract
We investigated the effects of propofol and propofol–ketamine on hemodynamics, sedation level, and recovery period in pediatric patients undergoing cardiac catheterization. We performed a prospective, randomized, double-blind study. The study included 60 American Society of Anesthesiologists physical status II or III (age range, 1 month–13 years) undergoing cardiac catheterization for evaluation of congenital heart disease. Propofol and ketamine were prepared in 5% glucose solution to a final concentration of 5 and 1 mg/ml, respectively; similar injectors containing 5% glucose solution only were prepared. Fentanyl (1 μg/kg) and propofol (1.5 mg/kg) were given to both groups. Then, group 1 received 0.5 ml/kg of 5% glucose and group 2 0.5 ml/kg of ketamine solution by an anesthesiologist who was unaware of the groups of patients. Local anesthesia with 1% lidocaine was administered before intervention in all patients. The noninvasively measured mean arterial pressure, heart rate, respiratory rate, and peripheral oxygen saturation were recorded at the baseline, following drug administration, at 3, 5, 10, 15, 20, and 30 minutes and then at 15-minute intervals until the end of the procedure. Additional drug and fentanyl requirements to maintain a sedation level of 4 or 5 were recorded. After the procedure, the time to a Steward recovery score of 6 and adverse effects in the first 24 hours were recorded. The number of patients with more than a 20% decrease in mean arterial pressure was 11 in group 1 and 3 in group 2 (p < 0.05). The number of patients who experienced more than a 20% decrease in heart rate was 12 in group 1 and 5 in group 2 (p = 0.054). Ten patients in group 1 and 3 patients in group 2 required additional fentanyl doses (p = 0.057). The number of additional propofol doses was lower in group 2 (p < 0.05). Propofol combined with low-dose ketamine preserves mean arterial pressure better without affecting the recovery and thus is a good option in pediatric patients undergoing cardiac catheterization.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
There is no ideal anesthetic technique that can be applied universally in children undergoing cardiac catheterization. The goals for anesthetic management of children undergoing cardiac catheterization include adequate analgesia, sedation, and immobility, with minimal depression of cardiac function and respiratory drive. Oral ketamine–midazolam, intramuscular meperidine, promethazine, intravenous ketamine, remifentanil, and propofol have been used for this purpose [1, 7, 9].
Ketamine is frequently preferred for cardiac catheterization in pediatric patients [4, 13]. However, it has significant disadvantages, including hemodynamic side effects such as hypertension and tachycardia, psychomimetic effects, and long recovery period [18]. Although propofol allows smooth induction and rapid recovery, it causes cardiorespiratory depression, which may pose a significant risk in pediatric cardiac patients undergoing catheterization. However, recent studies have shown that the combination of ketamine with propofol prevents the cardiorespiratory depression of propofol as well as the psychomimetic effects of ketamine, and the low-dose ketamine and propofol combination provides adequate sedation and analgesia [3, 14].
We investigated the effects of propofol and propofol–ketamine on hemodynamics, sedation level, and recovery period in pediatric patients undergoing cardiac catheterization.
Patients and Methods
After institutional review approval and informed parental consent, 60 children, American Society of Anesthesiologists physical status II or III, aged 3 months–13 years, who were scheduled for elective cardiac catheterization for evaluation of congenital heart disease were studied. Premedication was not given. Procedures were performed after 4–6 hours of fasting. Patients requiring ventilatory treatment or intravenous inotropic support were excluded. The patients were admitted to the angiography unit; an intravenous cannula was placed and an infusion of one-third saline solution was started. The patients were randomly assigned to one of two treatment groups to receive either propofol or ketamine–propofol. Propofol and ketamine were diluted to final concentrations of 5 and 1 mg/ml, respectively, in 5% dextrose, and similar injectors containing only 5% were also prepared. In all patients, local anesthesia with 1% lidocaine was administered after administration of fentanyl (1 μg/kg) and propofol (1.5 mg/kg). Then, patients in group 1 were given 0.5 ml/kg of 5% glucose and group 2 0.5 ml/kg of ketamine solution by an anesthesiologist unaware of the medications. Patients in group 1 were not given ketamine. The noninvasively measured mean arterial pressure (MAP), heart rate (HR), respiratory rate (RR), and peripheral oxygen saturation (SpO2) were measured at baseline, following drug administration, at 3, 5, 10, 15, 20, and 30 minutes, and then at 15-minute intervals until the end of the procedure. The Ramsey sedation score was used for assessing sedation level [17] (Table 1). Half of the initial drug dose was repeated to maintain a sedation level of 4–5. If the patient still showed discomfort (moved or cried), fentanyl (1 μg/kg) was added. Respiratory depression was defined as a respiratory rate less than 8 breaths/minute or an apnea lasting longer than 15 seconds. Decreases in oxygen saturation of more than 5 from the initial value and larger than 20% changes in the MAP and HR were recorded. The patients were observed until the Steward recovery score [20] became higher than 6 and then referred to the pediatric intensive care unit (Table 2). All adverse effects during the procedure and the following 24 hours were recorded.
In statistical analysis, the hemodynamic data were evaluated by unpaired Student's t-test—gender, adverse effects, and the number of patients requiring additional fentanyl, the number of patients showing larger than 20% variation of hemodynamic data was determined by Chi-square, Fisher's exact test. The Mann–Whitney test used to analyze the number of additional propofol doses. Probability values less than 0.05 were considered significant.
Results
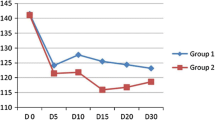
The groups did not differ significantly with respect to demographic characteristics and the period of catheterization (Table 3). The congenital heart diseases of the patients are shown in Table 4. The changes in MAP, HR, SpO2, and RR are depicted in Figs. 1 and 2.
The number of patients with more than a 20% decrease in blood pressure was 11 in group 1 and 3 in group 2 (p < 0.05). The number of patients who experienced more than a 20% decrease in HR with respect to the baseline value was 12 in group 1 and 5 in group 2 (p = 0.054). No patients experienced more than a 20% increase in HR and MAP (Table 5). Ten patients in group 1 and 3 patients in group 2 required additional fentanyl doses (p = 0.057). The number of additional propofol doses was lower in group 2 (Table 6) (p < 0.05). The time to a Steward score of 6 or higher was 6.76 ± 3.84 minutes for group 1 and 6.56 ± 2.7 minutes for group 2 (p > 0.05). None of the patients required postprocedure ventilation or intubation. The groups did not differ significantly with respect to adverse effects during the 24 hours following the procedure (Table 7).
Discussion
Our study shows that in pediatric patients undergoing cardiac catheterization, the combination of ketamine with propofol decreases the propofol dose and maintains MAP better without prolonging the recovery period.
Ketamine is widely used for sedation in pediatric patients undergoing cardiac catheterization [5, 8]. It provides good sedation and analgesia without causing respiratory depression. However, the long recovery period, delirium, and side effects such as tachycardia and hypertension limit its use [13, 16]. Morray et al. [13] reported that 2 mg/kg of ketamine does not affect hemodynamics in pediatric patients undergoing cardiac catheterization. Berman et al. [4] reported that ketamine increases pulmonary artery pressure and O2 consumption, and this may pose a risk in patients with increased pulmonary arterial pressure. Nguyen et al. [15], in a comparative study on etomidate, sodium γ-hydroxybutyrate, and ketamine, found no differences in hemodynamic and respiratory parameters but observed prolonged stupor and involuntary movements even 30 minutes after the end of the procedure in the ketamine group. However, these investigators used ketamine at 4 mg/kg bolus and 0.083 mg/kg/min infusion doses as a single agent. Because we used a lower dose and combined it with propofol, we did not observe prolonged recovery and involuntary movements.
Propofol has been recommended for pediatric cardiac catheterization for rapid recovery and smooth induction. However, the diminishing effect of systemic vascular resistance and MAP may limit the use of propofol for children with congenital heart disease [12, 19]. In patients without congenital heart disease, intravenous propofol administration decreases systolic arterial pressure by 30% and HR by 10–20% [2]. Williams et al. [22] reported that the ratios are similar in children with congenital heart disease and concluded that propofol should be used only in congenital heart disease patients with good ventricular function and in whom rapid recovery is desired. Gozal et al. [10], in their study on cardiac catheterization in pediatric patients with intracardiac shunts, reported that although systemic vascular resistance and pulmonary artery pressure decrease, the intracardiac shunt does not change to a certain extent and propofol is adequate for sedation in these patients.
Lebovic et al. [12] compared the effects of ketamine and propofol in pediatric patients undergoing cardiac catheterization and reported that the frequency of patients with a 20% or more decrease in blood pressure was higher in the propofol group.
Propofol does not provide adequate analgesia at low doses and many patients make movements during the administration of local anesthesia [12]. Therefore, in this study we used fentanyl before local anesthesia. Another limiting factor is the high frequency of respiratory depression [19]. Mortero et al. [14] reported that the addition of low-dose ketamine to propofol preserves ventilation better. Although the end expiratory CO2 increased in the propofol group, it remained within the normal range in the ketamine–propofol group. They did not observe psychomimetic effects after surgery and cognitive function was better in the ketamine–propofol group.
Wathen et al. [21], in a study of children younger than 10 years of age, reported that among patients who received ketamine, the frequency of vomiting was 19.4% and that of psychomimetic effects (nystagmus, hallucination, and agitation) was 7.1%. Although the frequency of vomiting was reduced when ketamine was combined with midazolam, the frequency of psychomimetic effects did not change significantly. In children younger than 5 years of age, Green et al. [11] reported vomiting in 3.5% and agitation in 22.5%; in older children, the corresponding figures were 12.1% for both. Dachs and Innes [6] used 1–2 mg/kg ketamine in children between 18 months and 8 years of age and observed vomiting in only two children and agitation in only four. Badrinath et al. [3] combined different doses of ketamine with propofol; they observed that the frequency of nausea and vomiting increased with higher doses of ketamine. In contrast with the previous studies, we observed no patient with nausea or vomiting. This may be due to the low-dose combined with propofol.
In conclusion, the cardiorespiratory depressive effect of propofol limits its use as a single agent in patients undergoing cardiac catheterization. However, when propofol is combined with low-dose ketamine, it preserves MAP better without prolonging recovery, and this combination is a good option for patients undergoing cardiac catheterization.
References
SM Auden WL Sobezyk RE Solinger LJ Goldsmith (2000) ArticleTitleOral ketamine/midazolam is superior to intramuscular meperidine, promethazine, and chlorpromazine for pediatric cardiac catheterization Anesth Analg 90 299–305 Occurrence Handle10648310 Occurrence Handle1:CAS:528:DC%2BD3cXhtV2nsb4%3D
CS Aun RY Sung ME O'Meara TG Short TE Oh (1993) ArticleTitleCardiovascular effects of i.v. induction in children: comparison between propofol and thiopentone Br J Anaesth 70 647–653 Occurrence Handle8329259 Occurrence Handle1:STN:280:ByyA3MbosVc%3D
S Badrinath MN Avramov M Shadrick TR Witt AD Ivankovich (2000) ArticleTitleThe use of a ketamine–propofol combination during monitored anesthesia care Anesth Analg 90 858–862 Occurrence Handle10735789 Occurrence Handle1:CAS:528:DC%2BD3cXis1yisbw%3D
WJ Berman RR Fripp M Rubler L Alderete (1990) ArticleTitleHemodynamic effects of ketamine in children under cardiac catheterization Pediatr Cardiol 11 72–76 Occurrence Handle2349145 Occurrence Handle10.1007/BF02239565
DL Coppel JW Dundee (1972) ArticleTitleKetamine anesthesia for cardiac catheterization Anaesthesia 27 25–31 Occurrence Handle5007628 Occurrence Handle1:STN:280:CS2D1c3nsVc%3D
RJ Dachs GM Innes (1997) ArticleTitleIntravenous ketamine sedation of pediatric patients in the emergency department Ann Emerg Med 29 146–150 Occurrence Handle8998094 Occurrence Handle1:STN:280:ByiC2M3hs1Y%3D
A Dönmez A Kizilkan H Berksun B Varan K Tokel (2001) ArticleTitleOne center's experience with remifentanil infusions for pediatric cardiac catheterization J Cardiothorac Vasc Anesth 15 736–739 Occurrence Handle11748523
NS Faithfull R Haider (1971) ArticleTitleKetamine for cardiac catheterization Anaesthesia 26 318–323 Occurrence Handle5090227 Occurrence Handle1:STN:280:CS6B3s%2FmtVM%3D
RH Friesen M Alswang (1996) ArticleTitleChanges in carbon dioxide tension and oxygen saturation during deep sedation for paediatric cardiac catheterization Paediatr Anaesth 6 15–20 Occurrence Handle8839083 Occurrence Handle1:STN:280:BymH3snhvFM%3D
D Gozal AJ Rein A Nir Y Gozal (2001) ArticleTitlePropofol does not modify the hemodynamic status of children with intracardiac shunts undergoing cardiac catheterization Pediatr Cardiol 22 488–490 Occurrence Handle11894151 Occurrence Handle10.1007/s002460010280 Occurrence Handle1:STN:280:DC%2BD387mtVWrtg%3D%3D
SM Green N Kuppermann SG Rothrock CB Hummel M Ho (2000) ArticleTitlePredictors of adverse events with intramuscular ketamine sedation in children Ann Emerg Med J 35 35–42 Occurrence Handle1:STN:280:DC%2BD3c%2Fot1Onsg%3D%3D
S Lebovic DL Reich GL Steinberg FP Vela G Silvay (1992) ArticleTitleComparison of propofol versus ketamine for anesthesia in pediatric patients undergoing cardiac catheterization Anesth Analg 74 490–494 Occurrence Handle1554114 Occurrence Handle1:STN:280:By2B3cnmtlc%3D
JP Morray AM Lynn SJ Stamm et al. (1984) ArticleTitleHemodynamic effects of ketamine in children with congenital heart disease Anesth Analg 63 895–899 Occurrence Handle6486488 Occurrence Handle1:STN:280:DyaL2M%2FitVOltA%3D%3D
RF Mortero LD Clark MM Tolan et al. (2001) ArticleTitleThe effects of small-dose ketamine on propofol sedation: respiration, postoperative mood, perception, cognition, and pain Anesth Analg 92 1465–1469 Occurrence Handle11375826 Occurrence Handle1:CAS:528:DC%2BD3MXksVyht7g%3D
NK Nguyen S Magnier G Georget et al. (1991) ArticleTitleAnesthesia for heart catheterization in children, comparison of 3 techniques Ann Fr Anesth Reanim 10 522–528 Occurrence Handle1785701 Occurrence Handle1:STN:280:By2C2M3ks1Q%3D
RI Parker RA Mahan D Guigliano M Parker (1997) ArticleTitleEfficacy and safety of intravenous midazolam and ketamine as sedation for therapeutic and diagnostic procedures in children Pediatrics 99 427–431 Occurrence Handle9041300 Occurrence Handle10.1542/peds.99.3.427 Occurrence Handle1:STN:280:ByiC1MzntFE%3D
MAE Ramsey TM Savege BR Simpson R Goodwin (1974) ArticleTitleWith controlled sedation alphaxalone–alphadolone Br Med J 2 656–659
DL Reich G Silvay (1989) ArticleTitleKetamine: an update on the first 25 years of clinical experience Can J Anesth 1 387–400
I Smith PF White M Nathanson R Gouldson (1994) ArticleTitlePropofol Anesthesiology 81 1005–1043 Occurrence Handle7943815 Occurrence Handle1:STN:280:ByqD3s%2FktFA%3D
DJ Steward (1975) ArticleTitleA simplified scoring system for the postoperative recovery room Can Anaesth Soc J 22 111–113 Occurrence Handle1109700 Occurrence Handle1:STN:280:CSqD2szoslE%3D Occurrence Handle10.1007/BF03004827
JE Wathen MG Roback T Mackenzie JP Bothner (2001) ArticleTitleDoes midazolam alter the clinical effects of intravenous ketamine sedation in children? A double-blind, randomized, controlled, emergency department trial West Afr J Med 20 136–139
GD Williams TK Jones KA Hanson JP Morray (1999) ArticleTitleThe hemodynamic effects of propofol in children with congenital heart disease Anesth Analg 89 1411–1416 Occurrence Handle10589618 Occurrence Handle1:CAS:528:DC%2BD3cXhsF2jsA%3D%3D
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Akin, A., Esmaoglu, A., Guler, G. et al. Propofol and Propofol–Ketamine in Pediatric Patients Undergoing Cardiac Catheterization. Pediatr Cardiol 26, 553–557 (2005). https://doi.org/10.1007/s00246-004-0707-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00246-004-0707-4