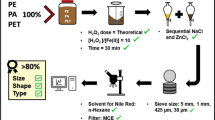
Abstract
The extraction of plastic microparticles, so-called microplastics, from sludge is a challenging task due to the complex, highly organic material often interspersed with other benign microparticles. The current procedures for microplastic extraction from sludge are time consuming and require expensive reagents for density separation as well as large volumes of oxidizing agents for organic removal, often resulting in tiny sample sizes and thus a disproportional risk of sample bias. In this work, we present an improved extraction method tested on return activated sludge (RAS). The treatment of 100 ml of RAS requires only 6% hydrogen peroxide (H2O2) for bleaching at 70 °C, followed by density separation with sodium nitrate/sodium thiosulfate (SNT) solution, and is completed within 24 h. Extracted particles of all sizes were chemically analyzed with confocal Raman microscopy. An extraction efficiency of 78 ± 8% for plastic particle sizes 20 µm and up was confirmed in a recovery experiment. However, glass shards with a diameter of less than 20 µm remained in the sample despite the density of glass exceeding the density of the separating SNT solution by 1.1 g/cm3. This indicates that density separation may be unreliable for particle sizes in the lower micrometer range.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
In recent years, microplastics have been found in virtually all types of aquatic environments worldwide. Microplastics were discovered in abundance on shorelines (Browne et al. 2011), in fresh water systems (Corcoran et al. 2015; Eerkes-Medrano et al. 2015; Klein et al. 2015; Yonkos et al. 2014; Zhang et al. 2015), in the marine environment (Barboza and Gimenez 2015), and even embedded in arctic sea ice (Obbard et al. 2014). Thus, the quantification of microplastics in different environmental matrices has become a crucial task. Moreover, wastewater treatment plants (WWTP) are suspected to disseminate microplastics into marine and fresh water systems (McCormick et al. 2014; Browne et al. 2011; Dubaish and Liebezeit 2013). In addition, land application of sewage sludge is a common practice in many countries (Singh and Agrawal 2008). Sewage sludge contains organic and inorganic plant nutrients. Its application in agriculture substitutes for chemical fertilizers but may introduce undesirable substances, for example, heavy metals and microplastics. Therefore, assessment of the microplastic content of sludge has become highly relevant. Methods for quantifying microplastics, originally implemented for marine samples, are increasingly adapted to be used on wastewater and even sludge samples.
The quantification of microplastics requires first the extraction of the microparticles from the environmental matrix followed by the identification of the microplastics among the extracted particles. The most common method for extraction is by density separation as initially proposed for seawater by Thompson et al. (2004); i.e., the density of a given sample is raised by adding saturated salt solution to the sample so that lighter particles, including most plastics, float to the top to be removed by draining the supernatant. Several modifications for adapting the method for marine sediments were proposed, for example, elutriation of the sample (Claessens et al. 2013), organic matter oxidization with H2O2 (Imhof et al. 2012; Nuelle et al. 2014), hot acid digestion with HNO3 (Claessens et al. 2013; Dubaish and Liebezeit 2013), and enzymatic digestion (Cole et al. 2014). For extracting high-density particles, different salts were used, including sodium poly-tungstate (SPT; 1.4 g/ml) (Corcoran et al. 2009), zinc chloride (ZC; 1.5–1.7 g/ml) (Imhof et al. 2012; Dubaish and Liebezeit 2013), and sodium iodite (NaI; 1.8 g/ml) (Nuelle et al. 2014). However, the amount of salts and acids required for sample treatment is a prohibitive factor in these modifications, even more so when treating samples with very high organic content, such as activated sludge.
Studies on extracting microplastics from environmental matrices used either sieving or density separation (Van Echelpoel et al. 2014) for extraction, while relying mostly on visual inspection with optical microscopy for microplastics identification (Carr et al. 2016). However, visual inspection alone cannot provide reliable information about the type of particles and is even unreliable with respect to separating plastics from nonplastics at particle sizes <500 µm (Löder and Gerdts 2015). Therefore, chemical characterization of extracted particles is crucial to identify microplastics reliably and thus assessing the efficacy of the extraction process.
Raman spectroscopy is a standard method for the detailed analysis of polymers, including but not limited to characterization (Sato et al. 2002), polymerization kinetics (Parnell et al. 2003; Brun et al. 2013), and degradation processes (Lippert et al. 1999; Oldak et al. 2005). In addition, in the form of confocal Raman microscopy, it is increasingly used for in vivo and in situ studies of complex biological materials, such as aquatic biofilms with embedded microparticles (Kniggendorf and Meinhardt-Wollweber 2011) and even protic function (Kniggendorf et al. 2014), allowing to examine not only extracted microparticles but also those still embedded in the environmental matrix (Kniggendorf and Meinhardt-Wollweber 2011). However, removing organic contaminants from the extracted particles significantly reduces the time needed for analyzing a given sample. Previously reported purification techniques using mineral acids, such as HNO3, have proven to be effective. However, the required volume of the chemicals may damage several widely used polymers (e.g., polyamide (PA), polyoxymethylene (POM), polycarbonate (PC)) and thus may lead to significantly biased results (Löder and Gerdts 2015).
In this work, we present an improved method for the extraction of microplastics from return activated sludge (RAS). The heat bleach method reduces the H2O2 volume needed for organic removal to approximately 6% of the sample volume. An elevated temperature of 70 °C increases the degradation of organic matter, reducing the time for purification to 24 h or less. A subsequent density separation using saturated sodium nitrate/sodium thiosulfate (SNT) solution extracted the microplastics from the remainder. SNT solution with a density of 1.46 g/ml is sufficient for extracting the most common types of synthetic polymers, including polycarbonate (PC; 1.20 g/cm3), polyurethane (PUR; 1.25 g/cm3), and even polyethylene terepththalate (PET; 1.38 g/cm3). The extraction efficiency of 78 ± 8% for particles of 20 µm and larger was determined in a subsequent recovery experiment, using pigmented polyethylene (PE) particles extracted from a commercially available personal care product. Compared with previously reported methods for the extraction of microplastics from sediment (Claessens et al. 2013; Imhof et al. 2012; Nuelle et al. 2014), the reduced volume of chemicals results in an estimated cost reduction of 75% or more for treating 100-ml samples.
To the best of our knowledge, this is the first time that SNT solution was used for the extraction of microplastics from sludge.
Materials and Methods
Only laboratory-grade chemicals and equipment were used in this work. The following standard operation procedures were adopted to avoid background contamination with microplastics: distilled deionized water (DDW) was used for the SNT solution and the PE particle suspension in the recovery experiment. No plastic vessels were used throughout the experiments. Glassware was cleaned in a laboratory dishwasher (Miele Professional G8793, Miele GmbH, Germany) using microplastics free laboratory glassware detergent (neodisher® LaboClean A 8, Carl Roth GmbH, Germany), subsequently rinsed with pressurized DDW, and inspected for adhering particles before use. Spot checks of surfaces were done microscopically. Vacuum filters were washed with DDW before and after use to eliminate wall-adhering particles. Sample solutions were always kept in glass containers and covered with glass plates or aluminum foil.
Return Activated Sludge
The sludge was procured from the Herrenhausen municipal wastewater treatment plant (Klärwerk Herrenhausen, Seelze, Germany) with a treatment capacity of 50,000 population equivalents and an average flow of 65,000 m3/day. The sludge had a total of 4503 g/ml suspended solids, a chemical oxygen demand of 5,260 mg/l, and contained 0.243 mg/l of NH4 +-N, 5.44 mg/l of NO3 −-N, and 0.032 mg/l of NO2 −-N.
Sodium Nitrate/Sodium Thiosulfate Solution
The SNT solution was prepared as reported in (Semensatto and Dias-Brito 2007) by dissolving 212.0 g of sodium nitrate (NaNO3) and 361.7 g of sodium thiosulfate (Na2O3S2.5H2O) in 300 ml of distilled water. A warm water bath was used to accelerate the dissolution of the sodium thiosulfate. After dissolution, the final volume of the solution was adjusted to 500 ml and screened microscopically to ensure that no microparticles were present. The density of the SNT solution as used in the subsequent experiments was 1.46 g/ml.
Microparticle Extraction
Microparticles were extracted from RAS in a three-step process, beginning with purification of 100 ml RAS, followed by density separation and filtration. The experiment was performed on two identical sample volumes.
Purification of the samples was achieved by adding 30% H2O2 solution until H2O2 forms 3% of the sample volume, and heating the sample to 70 °C. The samples were kept at 70 °C for 5 h after completion of the reaction. 70 °C is below the continuous operating temperatures (COT) of the polymers reported in microplastics worldwide [PC: 120 °C; PET: 120 °C; polypropylene (PP): 100 °C, PE: 80 °C; PA: 85 °C, polystyrene (PS): 80 °C] except for polymethylmethacrylate (PMMA) with a COT of 70 °C and a reported melting temperature of 102 °C (Elias 2003). This step was repeated with the samples being kept overnight at 70 °C until they were visibly clear. The clear samples were cooled to room temperature and subjected to density separation using 150 ml of SNT solution prepared as described above on each sample volume. The final density of the sample/SNT solution was 1.35 g/ml, due to dilution of the SNT solution by the sample. The samples were rested until a clear supernatant was observed after approximately 24 h. The supernatant was collected and vacuum filtered through cellulose acetate membrane filters with a pore size of 0.45 µm (Sartorius AG, Göttingen, Germany). The filter paper was dried and stored until further analysis with confocal Raman microscopy.
Sample Preparation for Confocal Raman Microscopy
To reduce the total examination time and to avoid combustion of the cellulose acetate paper in the laser focus in case biological material is trapped within its fabric, the contents of the filter paper were backwashed into distilled water. For this, each filter was placed in a 100-ml beaker with 20 ml of distilled deionized water. The resulting particle suspension was subjected to an ultrasonic bath for 15 min to homogenize particle distribution. An uncoated 1.2-mm deep indentation glass slide was filled with the homogenized particle suspension, sealed with a 0.17-mm coverslip to avoid evaporation during the measurement, and subjected to confocal Raman microscopy as described below.
Confocal Raman Microscopy
Raman microscopy was performed with a confocal Raman microscope (CRM 200 by WITec GmbH, Ulm, Germany), equipped with a standard objective (Nikon CFI LU Plan, 50×, NA 0.55). A frequency-stabilized, frequency-doubled continuous-wave Nd:YAG laser at 531.9 nm was used for excitation. A multimode fiber connected the Raman microscope and the spectrometer (UHTS 300 by WITec), resulting in an effective spectrometer pinhole of 50 µm. Laser intensity was set to 36 mW. The loss within the optics prior sample contact was 30%. The grating used in the spectrometer had 600 l/mm. Raman spectra were recorded with an electron multiplying charge-coupled device (emCCD) camera (Andor DU970 N-BV-353), electrically cooled to −69 °C. The system had a spatial resolution of 0.48 µm as confirmed in (Kniggendorf et al. 2016) and a spectral accuracy of 2 cm−1. The recorded Raman spectra covered the spectral range between −120 and 3500 rel. cm−1 with a spectral resolution of 5 cm−1. Standard integration time used was 0.5 s, in case spectral intensity was not sufficient for identification, the integration time was increased to 2 s. Particles were searched in a regular search pattern of equidistant rows and lines along the bottom of the indentation (for sinking particles) and along the underside of the coverslip (for floating particles). The distance between neighboring lines was 100 µm, equivalent to the height of the field of view so that the whole surface was covered. Every particle spotted was micrographed and a Raman spectrum was recorded from its center. Total measurement time per sample was approximately 4 h. In addition, a Raman chemical image with a resolution of 1 spectrum per µm was recorded for one randomly chosen fiber in each sample.
Microplastics among the microparticles were confirmed by comparing their Raman spectra to a set of reference Raman spectra recorded with the same system from laboratory grade polymer granules (PA, PC, PE, PET, PMMA, PP, PS) purchased from Sigma Aldrich GmbH, Germany. Mineral microparticles were identified using the RRUFF database for spectroscopic data on minerals (Lafuente et al. 2015).
Extraction Efficiency
PE Particle Preparation
Pigmented PE microparticles, extracted by density separation from a commercial personal care product (Garnier skin naturals Pure Active 3-in-1 face cleanser), were used to determine the extraction efficiency. The density separation was repeated until the particles were visibly free of any other chemicals from the cosmetic, and the separated particles were analyzed with confocal Raman microscopy as described above to confirm their material as PE and the absence of other substances than blue pigmentation in their chemical composition. Particle sizes observed with the stereo microscope (Stemi-2000-C by Zeiss, Germany) ranged between 20 and 600 µm, with even smaller particles being spotted during the chemical analysis with the confocal Raman microscope (50× objective, CRM 200 by WITec GmbH, Germany). Nanoparticles smaller than the detection limit of the confocal Raman microscope of 0.48 µm may have been present. No microparticles other than PE microplastics were found in the suspension.
Recovery Experiment
One milliliter of PE particle suspension was added to 100 ml of distilled deionized water (preformed in duplicate) and filtered directly onto cellulose acetate filter paper. The initial particle concentration (c i ) was determined by counting the blue PE particles using a stereo-microscope (Stemi-2000-C by Zeiss), equipped with an objective with a magnification of 4 (Zeiss ACHROPLAN 4× (No. 440020)).
Subsequently, 1 ml of homogenized PE particle suspension was added to 100 ml of RAS (performed in triplicate). The spiked RAS samples were subjected to the extraction process as described in "Microparticle extraction". Blue particles on the filter paper were counted visually as described above to determine the final particle concentration (c f ). The extraction efficiency (ŋ) was calculated as ŋ = c f / c i * 100%.
Results and Discussion
Time and Cost Efficiency
The use of 6% H2O2 with an elevated temperature of 70 °C resulted in a considerable reduction in organic material within 24 h. This is a significant improvement to the method proposed by (Nuelle et al. 2014), requiring 7 days for treatment of sediment samples with 35% H2O2 at room temperature, and even less than the 15% H2O2 (Van Echelpoel et al. 2014) used for purifying sludge. In addition, the elevated temperature reduced the overall water content of the sample, and thus the required volume of SNT separation solution, by 50% through evaporation, achieving the same sample volume reduction as used by (Claessens et al. 2013; Imhof et al. 2012; Nuelle et al. 2014) with less effort than the combination of fluidization, sand bath, or passing air jets, respectively. The costs of treating 100-ml samples are reduced by 75% compared with previously reported methods used on sediment (Claessens et al. 2013; Imhof et al. 2012; Nuelle et al. 2014).
At 1.46 g/ml, the achievable separation density with SNT solution is within the range of other separation densities reported for different salt solutions, such as sodium poly-tungstate (1.4 g/ml) (Corcoran et al. 2009), zinc chloride (1.5–1.7 g/ml) (Imhof et al. 2012; Dubaish and Liebezeit 2013), and sodium iodite (1.8 g/ml) (Nuelle et al. 2014). This is sufficient to separate most of the lighter polymers, such as PE, PET, PS, PP, and PMMA, but like the other methods excludes the heavier polymers, such as PVC-C (1.64 g/cm3), which is only within the range of the most expensive sodium iodite solution, and PTFE (2.16 g/cm3), which to the best of our knowledge is not within the range of any reported density separation protocol to date.
Extraction Efficiency
The extraction efficiency was determined to be 78 ± 8% for PE particles of the same size and shape spectrum as found in the analyzed RAS samples (see “RAS analysis and environmental impact of detected microplastic concentrations” section). This is up to par or even better than previously reported results obtained with single-step density separation using salt solutions for particles of the same size spectrum, albeit lower than reported density separation preceded by elutriation of the sample. For example, Imhof et al. (2012) recovered only 39.8% of microplastics when relying solely on density separation with saturated NaCl solution for treating sediment samples while recovering 95.5% with elutriation, and Claessens et al. (2013) recovered up to 99% when using a combination of elutriation and saturated NaI solution on sediments. However, both treatments are more time and cost intensive than density separation with SNT solution. In addition, sludge is a more challenging type of sample than sediment, because it is comprised of a cohesive matrix of organic material, microorganisms, and inorganic particles held together by biopolymers with a high affinity for most polymer surfaces (Hori and Matsumoto 2010; Garrett et al. 2008).
RAS Analysis and Environmental Impact of Detected Microplastic Concentrations
On average, a sample of 100 ml of RAS contained 223 microparticles or 495 particles per g dry weight of RAS. Particle sizes were highly variant; sizes ranged from the lower detection limit (0.48 µm) to 500 µm with most of the particles having sizes between 20 and 100 µm for the largest diameter. Particle shapes also were highly variant, including irregular fragments, spheres, thin films, and filaments (fibers). A selection of white light micrographs of small microparticles characterized with confocal Raman microscopy is given in Fig. 1. All fibers found in the analyzed sample volume had an average thickness of 10 µm and consisted of several PET filaments without any detectable additives or attached contaminants (see Fig. 2 for a white light micrograph of a typical PET fiber and the corresponding Raman chemical image). The corresponding Raman spectra are given in Fig. 3. In addition, the Raman spectra belonging to identified microplastics were compared to laboratory grade plastic samples to confirm that the extraction procedure did not affect the chemical composition of the respective plastics. An example of a Raman spectra comparison for PET (sample fiber 4; poly(ethylene terephthalate) pellets, PCode 1002025386 Lot # MKBV4092 V, Sigma-Aldrich Chemie GmbH, Steinheim, Germany) is shown in Fig. 4.
White light micrographs of randomly chosen microparticles in one of the samples. We dare our readers to identify the microplastics among them without referring to the respective Raman spectra given in Fig. 3. The white bar measures 20 µm
White light micrograph of a PET fiber. Inset Raman chemical image of the fiber, for the corresponding Raman spectrum, see Fig. 3 (spectrum 2)
Raman spectra of the microparticles presented in previous figures. Spectrum 1: polyacetylated cellulose (particles b, g); spectrum 2: polyethylene terephthalate (fiber); spectrum 3: TiO2 in rutile conformation (particle l); spectrum 4: polypropylene (particles c, i, j); spectrum 5: polyamide (particle h); spectrum 6: carotenoid-containing biomatter (particle d); spectrum 7: unidentified substance (particle f); spectrum 8: glass (particles e, k)
Comparison of the Raman spectrum recorded from the fourth fiber (2) found in the sample with laboratory grade polyethylene terephthalate (PET) (spectrum 1). All Raman lines of PET are present at expected positions and relative intensities. Small additional lines in the fingerprint region (500–1500 cm−1) of the fiber spectrum (2) correspond to well-known Raman lines seen in most biological materials and indicate small biological contaminants left on the sample after extraction
Of the characterized microparticles, 46% discounting fibers were strongly fluorescent and could not be photobleached without destroying the respective particle. Because synthetic polymers are known to withstand the laser wavelength and intensity applied, these particles are unlikely to have been microplastics. Another 8% of the particles yielded good quality Raman spectra that could not be positively matched yet and thus are listed as “unknown.” Of the remaining particles, the majority (36%) were beads of approximately 30 µm in diameter, identified by their Raman spectrum as semisynthetic polymers originating from acetylated cellulose (Zhang et al. 2011), followed by irregular shaped polypropylene (23%) and glass shards [RRUFF ID: R110082] (22%), as well as fully bleached microalgae cells containing carotenoids (De Gelder et al. 2007) as the most specific markers (14%) and TiO2 in rutile phase [RRUFF ID: R040049] (5%). No microplastics smaller than 20 µm in diameter were found. All particles smaller than 20 µm in diameter were identified as nonpolymers, most often glass (diameters > 5 µm) and TiO2 (>0.5 µm).
Particle size is an important parameter to consider in density separation. Extraction efficiencies have been consistently reported lower for small particles with diameters <1 mm (Nuelle et al. 2014; Imhof et al. 2012), mostly due to increased particle adhesion to surfaces, and most microplastics found in the environment are <1 mm in size (Browne et al. 2011). Moreover, the glass particles found in the sample had sizes between 5 and 15 µm and thus were considerably smaller than the detected microplastics (Fig. 1e, k). We believe that the glass remained in the sample despite its density (2.5 g/cm3) exceeding that of the SNT separation solution due to the higher surface-to-volume ratio of these small particles, indicating that density separation techniques may have limited effectiveness for separating plastics from nonplastics in the range of smaller microparticles with diameters <20 µm. This is especially worrisome with respect to the fact that these very small microplastics are considered the most dangerous to wildlife as these particles are mistaken for cells and are ingested instead of planktonic food (Sanchez et al. 2014; Taylor et al. 2016) and thus need to be monitored closely.
As stated, the microplastic concentration found in RAS from Klärwerk Herrenhausen, Lower Saxony, Germany, was 495 particles per g dry weight. Given the agricultural application of sludge as fertilizer, this means that with an estimated sludge application rate of 1.6 tons per hectare per year in Lower Saxony, 80,000 microplastic particles per m2 end up in the soil each year, giving rise to the concern that not only the already reported aquatic organisms are affected. Given the low biodegradability of plastics under environmental conditions (Mohee and Unmar 2007), an accumulation of microplastics in the soil and subsequent leach into surface waters and even ground waters are highly likely, calling for more work on effective methods for assessing microplastic content of sludge and soil samples and decisive measures for preventing microplastics to enter the environment in the first place.
Conclusions
We developed an optimized method for the extraction of microplastics from sludge with a density of up to 1.34 g/cm3, achieving an extraction efficiency of 78 ± 8% for particles >20 µm. The Heat-Bleach method was tested on return activated sludge sampled from a wastewater treatment plant in Lower Saxony, Germany, revealing that sludge flocs are a sink of microplastics. The common practice of land application of sewage sludge as fertilizer may further spread microplastics into the environment, contaminating the soil in Lower Saxony with additional 80,000 microplastic particles per m2 per year. The Heat-Bleach method requires a treatment time of less than 24 h, reduces the amount of density separation solution by 50%, and cuts the costs of treating 100 ml samples by 75% compared with previously reported methods used on sediment.
References
Barboza LGA, Gimenez BCG (2015) Microplastics in the marine environment: current trends and future perspectives. Mar Pollut Bull. doi:10.1016/j.marpolbul.2015.06.008
Browne MA, Crump P, Niven SJ, Teuten E, Tonkin A, Galloway T, Thompson R (2011) Accumulation of microplastic on shorelines worldwide: sources and sinks. Environ Sci Technol 45:9175–9179
Brun N, Youssef I, Chevrel M-C, Chapron D, Schrauwen C, Hoppe S, Bourson P, Durand A (2013) In situ monitoring of styrene polymerization using Raman spectroscopy: multi-scale approach of homogeneous and heterogeneous polymerization processes. J Raman Spectrosc 44:909–915
Carr SA, Liu J, Tesoro AG (2016) Transport and fate of microplastic particles in wastewater treatment plants. Water Res 91:174–182
Claessens M, Van Cauwenberghe LV, Vandegehuchte MB, Janssen CR (2013) New techniques for the detection of microplastics in sediments and field collected organisms. Mar Pollut Bull 70:227–233
Cole M, Webb H, Lindeque PK, Fileman ES, Halsband C, Galloway TS (2014) Isolation of microplastics in biota-rich seawater samples and marine organisms. Sci Rep 4:4528
Corcoran PL, Biesinger MC, Grifi M (2009) Plastics and beaches: a degrading relationship. Mar Pollut Bull 58(1):80–84
Corcoran PL, Norris T, Ceccanese T, Walzak MJ, Helm PA, Marvin CH (2015) Hidden plastics of Lake Ontario, Canada and their potential preservation in the sediment record. Environ Pollut 204:17–25
De Gelder J, De Gussem K, Vandenabeele P, Moens L (2007) Reference database of Raman spectra of biological molecules. J Raman Spectrosc 38:1133–1147
Dubaish F, Liebezeit G (2013) Suspended microplastics and black carbon particles in the Jade System, Southern North Sea. Water Air Soil Pollut 224:1–8
Eerkes-Medrano D, Thompson RC, Aldridge DC (2015) Microplastics in freshwater systems: a review of the emerging threats, identification of knowledge gaps and prioritization of research needs. Water Res 75:63–82
Elias H-G (2003) Makromoleküle: band 3: industrielle polymere und synthesen, 6th edn. WILEY-VCH Verlag GmbH & Co. KGaA, WILEY
Garrett TR, Bhakoo M, Zhang Z (2008) Bacterial adhesion and biofilms on surfaces. Prog Nat Sci 18:1049–1056
Hori K, Matsumoto S (2010) Bacterial adhesion: from mechanism to control. Biochem Eng J 48:424–434
Imhof HK, Schmid J, Niessner R, Ivleva NP, Laforsch C (2012) A novel, highly efficient method for the separation and quantification of plastic particles in sediments of aquatic environments. Limnol Oceanogr Methods 10:524–537
Klein S, Worch E, Knepper TP (2015) Occurrence and spatial distribution of microplastics in river shore sediments of the Rhine-Main Area in Germany. Environ Sci Technol 49:6070–6076
Kniggendorf A-K, Meinhardt-Wollweber M (2011) Of microparticles and bacteria identification—(resonance) Raman micro-spectroscopy as a tool for biofilm analysis. Water Res 45:4571–4582
Kniggendorf A-K, Meinhardt-Wollweber M, Yuan X, Roth B, Seifert A, Fertig N, Zeilinger C (2014) Temperature-sensitive gating of hCx26: high-resolution Raman spectroscopy sheds light on conformational changes. Biomed Opt Express 5(7):2054–2065
Kniggendorf A-K, Nogueira R, Kelb C, Schadzek P, Meinhardt-Wollweber M, Ngezahayo A, Roth B (2016) Confocal Raman microscopy and fluorescent in situ hybridization: a complementary approach for biofilm analysis. Chemosphere 161:112–118
Lafuente B, Downs RT, Yang H, Stone N (2015) The power of databases: the RRUFF project. In: Armbruster T, Danisi RM (eds) Highlights in mineral crystallography. de Gruyter, Berlin
Lippert T, Ortelli E, Raimondi J-C, Wambach J, Wei J, Wokaun A (1999) Imaging-XPS/Raman investigation on the carbonization of polyimide after irradiation at 308 nm. Appl Phys A. doi:10.1007/s003399900220
Löder M, Gerdts G (2015) Methodology used for the detection and identification of microplastics: a critical appraisal. In: Bergman M, Gutow L, Klages M (eds) Marine anthropogenic litter. Springer, Berlin, pp 447–448
McCormick A, Hoellein TJ, Mason SA, Schluep J, Kelly JJ (2014) Microplastic is an abundant and distinct microbial habitat in an urban river. Environ Sci Technol 48(2):11863–11871
Mohee R, Unmar G (2007) Determining biodegradability of plastic materials under controlled and natural composting environments. Waste Manag 27:1486–1493
Nuelle M-T, Dekiff JH, Remy D, Fries E (2014) A new analytical approach for monitoring microplastics in marine sediments. Environ Pollut 184:161–169
Obbard RW, Sadri S, Wong YQ, Khitun AA, Baker I, Thompson RC (2014) Global warming releases microplastic legacy frozen in Arctic Sea ice. Earth’s Future 2(6):315–320
Oldak D, Kaczmarek H, Buffeteau T, Sourisseau C (2005) Photo-and bio-degradation processes in polyethylene, cellulose and their blends studied by ATR-FTIR and Raman spectroscopies. J Mater Sci 40:4189–4198
Parnell S, Min K, Cakmak M (2003) Kinetic studies of polyurethane polymerization with Raman spectroscopy. Polymer 44:5137–5144
Sanchez W, Bender C, Porcher J-L (2014) Wild gudgeons (Gobio gobio) from French rivers are contaminated with microplastics: preliminary study and first evidence. Environ Res 128:98–100
Sato H, Shimoyama M, Kamiya T, Amari T, Sasic S, Ninomiya T, Siesler HW, Ozaki Y (2002) Raman spectra of high-density, low-density, and linear low-density polyethylene pellets and prediction of their physical properties by multivariate analysis. J Appl Polym Sci 86:443–448
Semensatto DL, Dias-Brito D (2007) Alternative saline solutions to float foraminiferal tests. J Foraminifer Res 37(3):265–269
Singh RP, Agrawal M (2008) Potential benefits and risks of land application of sewage sludge. Waste Manag 28:347–358
Taylor ML, Gwinnett C, Robinson LF, Woodall LC (2016) Plastic microfibre ingestion by deep-sea organisms. Sci Rep 6(33997):1–8
Thompson RC, Olsen Y, Mitchell RP, Davis A, Rowland SJ, John AWG, McGonigle D, Russel AE (2004) Lost at sea: where is all the plastic? Science 304(5672):838
Van Echelpoel W, Vandegehuchte M, De Gueldre G, Van Cauwenberghe L (2014) Microplastics in a biological wastewater treatment plant and the surrounding freshwater environment in Flanders: quantitative assessment. Master’s dissertation submitted in partial fulfillment of the requirements for the degree of Master of Science in “Technology for Integrated Water Management.” http://lib.ugent.de/catalog/rug01:002147111
Yonkos LT, Friedel EA, Perez-Reyes AC, Ghosal S, Arthur CD (2014) Microplastics in four estuarine rivers in the Chesapeake Bay, USA. Environ Sci Technol 48:14195–14202
Zhang K, Feldner A, Fischer S (2011) FT Raman spectroscopic investigation of cellulose acetate. Cellulose 18:995–1003
Zhang K, Gong W, Lv J, Xiong X, Wu C (2015) Accumulation of floating microplastics behind the Three Gorges Dam. Environ Pollut 204:117–123
Acknowledgements
The authors thank Karin Kock and Benjamin Schwanda for their technical assistance to their work. The work of Dr. Kniggendorf was funded by the Federal Ministry of Education and Research (BMBF) of Germany within the project of OPTIMUS (Project Number: 13N13811). The work of Surya Sujathan was funded by the DAAD Sandwich Model Scholarships for Master’s Students of the IITs and IIMs, 2015 (Personal Ref No: 91590631).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sujathan, S., Kniggendorf, AK., Kumar, A. et al. Heat and Bleach: A Cost-Efficient Method for Extracting Microplastics from Return Activated Sludge. Arch Environ Contam Toxicol 73, 641–648 (2017). https://doi.org/10.1007/s00244-017-0415-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00244-017-0415-8