Abstract
Chinese alligator (Alligator sinensis) is a critically endangered species endemic to China. Concentrations of heavy metals (As, Fe, Mn, Cu, Pb, Cd, Cr, Zn, and Hg) were examined in the tissues of Chinese alligators to elucidate the background distribution of these metals in the alligator body. Generally, within the body compartments, metal concentrations were high in liver, kidney, and heart, and low in pancreas and gonad. Study of heavy metal levels in the feces and eggs of Chinese alligator suggested that Chinese alligators could reduce body burden of toxic substances by excreting them to feces and/or sequestrating them into eggs to a lesser extent. In addition, to test whether eggshell or egg membrane could be used as surrogates to measure heavy metal load in egg contents, the correlation of metal concentrations between three egg compartments was determined. Of the nine elements analyzed, concentrations of iron, copper, and zinc in the shell membrane were highly correlated with the levels in egg contents, whereas no metal was significantly correlated between eggshell and egg contents. This suggested that the shell membrane could be a useful bioindicator for Fe, Cu, and Zn contaminations in the eggs of Chinese alligator. In a comparison of metal contents in the eggs of individuals from the Anhui captive population, the wild population in Anhui Province, and those of the Changxing captive population, higher Cu, Zn, and Cd levels and a lower Pb level were found in the Changxing individuals, indicative of specific pollutants in different areas. In addition, the majority of metal elements in the muscles of Chinese alligators and American alligators are in the same ranges. As a result of the data found in the eggs of the two alligator species, the Chinese alligators may be exposed to a higher level of metal pollutants. The study provided measurement of the heavy metal distribution in the endangered Chinese alligator for the first time and could serve as the background for the monitoring of possible heavy metal contaminations in the alligator habitats.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
The Chinese alligator (Alligator sinensis) is endemic to China and is one of the most endangered species among the world’s 23 species of crocodilians. The remaining wild individuals are now restricted to a few small, isolated areas in southern Anhui Province and the adjacent Zhejiang Province (Thorbjarnarson and Wang 1999). A survey concluded in 2002 found that the total population of this species in the wild is probably fewer than 130 and is declining at a rate of 4–6% annually. The largest population found in the wild was composed of only 9–11 individuals (Thorbjarnarson et al. 2002). To save this species, Anhui Xuanzhou and Zhejiang Changxing captive-bred populations were established in 1979. Releases from the two captive populations are probably the only viable option for restoring this species to the wild, and reintroduction and habitat restoration are currently being planned.
Previous studies showed that application of large amounts of chemical fertilizers and insecticides might affect the Chinese alligator (Thorbjarnarson and Wang 1999). For example, Zhou (1997) reported that in 1984 two alligators died from the effects of insecticides in a farm in Jinxian County (Anhui Provience) and alligators were found dying from eating poisoned rodents (Thorbjarnarson et al. 2002). So far, the survey of the heavy metal contaminations in the field where the two Chinese alligator captive-bred centers are located as well as in the habitats of the remaining wild populations out has not been carried out.
Heavy metals have been recognized as one category of environmental contaminant affecting crocodilians (Brisbin et al. 1998), and crocodilians could be used as the bioindicators of environment pollution (Manolis et al. 2002a). Many researchers have reported the presence of mercury in crocodilian species (Hord et al. 1990; Heaton-Jones et al. 1997; Elsey et al. 1999; Rainwater et al. 2002; Rumbold et al. 2002). Using some readily-obtained tissues, such as scutes or claws, interrelationships of mercury concentrations among a variety of tissues were also surveyed, which provided non-destructive screenings of Hg in alligator populations (Yanochko et al. 1997; Jagoe et al. 1998). However, only a limited number of studies regarding the exposure and impacts of heavy metals in internal tissues have been conducted on crocodilian species. Delany et al. (1988) measured 7 metals in tail muscle from 32 American alligators (Alligator mississippiensis) at eight Florida lakes; Jeffree et al. (2001) provided the baseline concentrations of 18 elements in the flesh and osteoderms of estuarine crocodiles (Crocodylus porosus). Burger et al. (2000) examined the bioaccumulation of 8 metals in different tissues of American alligators; different concentrations were detected in internal tissues, and the possibility of using skin and tail tissues as bioindicators was also assessed.
The accumulation of heavy metal in the crocodilian’s eggs has received little attention. Metal levels in the eggs of American crocodile (Crocodylus acutus) and American alligator have been quantified (Ogden et al. 1974; Stoneburner and Kushlan 1984; Heinz et al. 1991). Phelps et al. (1986) examined heavy metals contaminations in the eggs of Nile crocodile (Crocodylus niloticus) sampled from several localities in Zimbabwe. Rainwater et al. (2002) surveyed the heavy metal concentrations in the eggs of Morelet’s crocodile (Crocodylus moreletti) from three locations in nonalluvival lagoons. Manolis et al. (2002b) detected various trace element concentrations in wild saltwater crocodile eggs and revealed the habitat pollution. Ding et al. (2001) presented the first study on heavy metal concentrations in the egg of Chinese alligator in Anhui Province. These studies laid the foundations for further heavy metal contamination surveys for the crocodilian species.
Heavy metal distribution in internal tissues and eggs is not clear in Chinese alligators. And there are no reports, to our knowledge, of mercury pollution in this species. In this study, the concentrations of As, Fe, Mn, Cu, Pb, Cd, Cr, Zn, and Hg in various tissue types of Chinese alligator were measured. One of our objectives was to reveal the metal distribution among internal organs and elucidate the background distribution of these metals in the alligator body.
Similar to the freshwater turtle eggs (Nagle et al. 2001), high contaminant levels in crocodilian eggs might increase embryo mortality and decrease hatching fitness. Heavy metal concentrations in the egg may reflect long-term contamination, as some egg contents are drawn from the skeleton (Manolis et al. 2002b). The eggshell or shell membranes were surveyed to be used as indicators of pollutants in egg contents in some studies (Morera et al. 1997). Accordingly, heavy metal distributions within the three egg compartments (eggshells, shell membranes, and egg contents) of the Chinese alligator were investigated in this study. The relationships of metal concentrations within and among the three compartments were examined in order to assess the reliability of eggshell or shell membrane as substitutes for egg contents in measurement. Chinese alligators are critically endangered; intentional killing and harmful sampling of wild alligators is strictly prohibited. If the levels of metals in eggshell or shell membrane show a significant correlation with the levels in egg content, the easily collectable eggshell and shell membrane could, therefore, be used as non-invasive indicators of contaminants in alligator eggs.
Materials and Methods
Sample Collection and Preparation
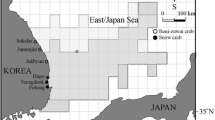
The Changxing Nature Reserve and Breeding Research Center for Chinese alligator, located in Changxing County, Zhejiang Province, is centrally within the current geographical distribution range of the Chinese alligators (29° 40’∼31° 30’N, 160° 00’∼120° 00’E).
Two alligator samples (a mature male and a mature female) were collected on February 2004 after death of unidentified causes. Animals were transported to the laboratory fresh and dissected to collect tissues and organs including heart, lung, liver, stomach, kidney, intestine, tracheas, pancreas, gonad, and muscle. Ten Chinese alligator eggs from 3 different clutches (1 egg each from two different clutches and 8 eggs from a third clutch) were collected on September 2003. Eggshell, shell membrane, and egg content (yolk plus albumen) were carefully separated. Six feces samples from different alligator individuals were collected. Two species of fishes used as the feeding food of the alligators in the Changxing Breeding Center were randomly sampled. All the samples were gently washed in distilled water, left to dry at room temperature for 24 hours, and oven-dried at 60°C to constant weight, and then digested to a transparent solution with a mixture of nitric, perchloric, and sulphuric acids.
Water and sediment samples were collected from 10 breeding ponds. Five water and 5 sediment samples from different sites were collected from each pond. The water samples were then transferred to reagent bottles and acidified by a measured volume of concentrated nitric acid and were filtered through a 0.45-μm micropore membrane filter. Sediment samples were air dried and sieved through a 2-mm nylon sieve, then digested with concentrated nitric and hydrochloric acid to extract the metals.
Heavy Metal Analysis
The resultant solutions for all the samples were diluted to a known volume with deionized water and transferred to acid-washed sample tubes. The samples were analyzed for As, Fe, Mn, Cu, Pb, Cd, Cr, Zn, and Hg. As, Fe, Mn, and Zn were directly determined by a flame atomic absorption spectrophotometer (F-AAS, Perkin Elmer). Cu, Pb, and Cd were measured by F-AAS after diethyl-dithiocarbamate-methyl isobutyl ketone treatment (Honda et al. 1986). The concentration of Cr for all the samples was examined by colorimetry. The concentration of Hg in sediment samples was determined by cold vapor AAS (Akagi and Nishimura 1991), while water and animal samples were measured by atomic fluorescence spectroscopic analysis. Accuracy of these analyses was controlled using a standard reference material, NIES No. 1 (Okamoto et al. 1978). The standard error from triplicate analysis was less than 5% for each element. Concentrations in tissues and eggs were given on a dry-weight basis.
Statistical Analysis
All statistical calculations were performed using the SPSS 11.5 software package for Windows (SPSS Inc., Microsoft Co.). Tabulated values represent mean ± SD for all the samples. A significance level of p < 0.05 was chosen for all statistical tests. Differences in concentrations between eggshells and egg contents, shell membranes and egg contents of Chinese alligators were determined by paired t-tests. The relationship between concentrations of heavy metals within and between compartments (eggshell, shell membrane, and egg contents) was assessed by nonparametric Spearman’s correlations.
Results
Heavy Metal Distribution in Tissues
Concentrations of heavy metals (As, Fe, Mn, Cu, Pb, Cd, Cr, Zn, and Hg) in various tissues and organs of Chinese alligators are given in Table 1. All the elements were detected in all of the samples analyzed. In general, concentrations were higher in liver, kidney, and heart, and lower in pancreas and gonad for Chinese alligators. Liver and kidney had a higher accumulation of heavy metals, which was similar to the reports from America alligators, marine turtles, and many other animals (Burger et al. 2000; Storelli and Marcotrigiano 2003; Ikemoto et al. 2004).
The highest concentrations of Fe, Mn, and Cu were found in the liver for both female and male individuals. The highest rate of Hg was found in the kidney, which was dissimilar to some reptiles where the highest concentration of Hg was generally found in the liver (Yanochko et al. 1997; Jagoe et al. 1998; Storelli et al. 1998; Godley et al. 1999; Sakai et al. 1995, 2000). The highest levels of As, Pb, Cd, and Zn differed between female and male individuals. Overall, in 63.3% of measurements, the female samples had higher heavy metal concentrations than the males, while 36.7% cases were the opposite.
Heavy Metal Distribution in Eggs
Heavy metal concentrations in eggshell, shell membrane, and egg contents are given in Table 2. All the elements were detected in the three egg compartments. Levels were significantly higher in the eggshells than in the egg contents for Mn, Pb, and Cr, whereas the concentrations were significantly higher in the egg contents for As, Fe, Zn, and Hg, with no difference for Cu and Cd between the two compartments. Except for As, Cu, and Cd, concentrations of other elements were significantly (1.6–6.1 times) greater in shell membranes than in egg contents. In contrast, the levels of As and Cd in egg contents were higher than in shell membranes, with Cu showing no significant difference.
Significant negative correlation was found between Fe and Pb in the eggshells, while significant positive correlation was deduced between Cu and Pb in the shell membranes. Further, significant positive correlations existed in the egg contents among concentrations of As and Cu, Pb and Cr (Table 3).
One of our objectives was to determine if eggshells or shell membranes could be used as bioindicators of the metal level in eggs of the Chinese alligators. Of the nine elements analyzed in the three compartments, Fe, Cu, and Zn showed significant correlations between shell membranes and egg contents. Two other metals (As and Hg) showed a significant correlation between eggshells and shell membranes. No metal showed a significant correlation between eggshells and egg contents (Table 3).
Heavy Metal Concentrations in the Alligator Feces and in the Diet of the Alligators
The metal concentrations in two species of feeding fish were measured and the heavy metal concentrations of 6 fecal samples were also analyzed to investigate possible ways of heavy metal excretion. The concentrations of 9 heavy metals (As, Fe, Mn, Cu, Pb, Cd, Cr, Zn, and Hg) in the alligator feces and in the diet of the Chinese alligators in the Changxing Captive-bred Center are presented in Table 4. Concentrations of heavy metals in fecal samples were relatively higher than the diet fishes examined (Table 4).
Heavy Metal Concentration in Water and Sediments
The concentrations (mean ± SD) of 9 heavy metals (As, Fe, Mn, Cu, Pb, Cd, Cr, Zn, and Hg) in water and sediment samples from the Changxing Nature Reserve and Breeding Research Center are presented in Table 5. All the surveyed heavy metals were present in these samples. Results showed Fe ranked at the highest concentrations (126 ± 23 μg/L in water; 10,700 ± 800 μg/g in sediment), while As showed the lowest level in water (0.770 ± 0.396 μg/L) and Hg exhibited the lowest level in sediment (0.084 ± 0.008 μg/g).
Discussion
Heavy Metal Uptake and Elimination
Environmental contaminants such as metals, organochlorines, and other persistent chemicals would be expected to exert their toxic and teratogenic effects on crocodilians (Woodward et al. 1993; Guillette et al. 1994, 1996). A variety of heavy metals have been identified in water and sediments in the Changxing Nature Reserve and Breeding Research Center (Table 5), and the concentrations of these metals might be site-specific and variable. Our data showed that the environmental metals have been accumulated to various amounts in the tissues and eggs of the alligators (Table 1 and Table 2), with low levels of toxic heavy metals detectable in this captive population. Furthermore, Table 1 showed that large inter-organ variations in metal accumulations existed in the Chinese alligator body. Due to lack of adequate information on metal metabolism in Chinese alligators, it was difficult to establish a general pattern of progressive relationship between heavy metals in water and in the Chinese alligator body. The exact tolerable levels of various heavy metals in water and sediments for Chinese alligators need further investigations.
In addition to water and sediments containing metals, metals were also detected in alligator food items. How the Chinese alligators cope with these metals is an interesting issue. The metal concentrations in the two feeding species of fish and 6 feces samples were compared to gain a physiological pathway of heavy metal excretion in the alligators. Concentrations of heavy metals in fecal samples were higher compared to those in the diet fish and body compartments (Table 5). For example, the concentration of Cr in feces was about 58 times higher than in the food taken. Compared to the concentrations of heavy metal in the eggs, feces also showed higher concentrations for those elements (except Cu) (Tables 2 and 5). As for the body organs, only Cu and Hg in liver and kidney showed higher accumulations than in the feces (Tables 1 and 5). Thus, we confirmed that Chinese alligators could transfer toxic metals such as Cd, Cr, Hg, Pb, and Zn to the feces and excrete them.
Metal Concentration in Chinese Alligator Eggs
Heavy metal in eggs may affect the development and hatchling of offsprings and, thus, is an important aspect in determining the physiological and ecological effects of metal contaminations. However, there are currently only a few studies that have reported heavy metal contamination in crocodilian eggs (Campbell 2003; Ogden et al. 1974; Stoneburner and Kushlan 1984; Heinz et al. 1991; Phelps et al. 1986; Rainwater et al. 2002; Ding et al. 2001).
In this study, we found that heavy metals were present in eggshell, shell membrane, and egg contents (Table 2). Diverse animal species have been reported to excrete heavy metals in egg compartments, such as female birds in their egg contents (Burger and Gochfeld 1991) and eggshells (Burger 1994). The high concentrations of many types of metals in the egg compartments found in this study may suggest that female alligators also use the eggs as a mean of reducing body burden of toxic substances through maternal transfer to eggs. Similar situations were reported in other reptiles, including loggerhead turtles (Caretta caretta), green turtles (Chelonia mydas), and slider turtles (Trachemys scripta) (Sakai et al. 1995, 2000; Burger and Gibbons 1998).
Concentrations of Cu, Zn, Cd, and Pb in a single infertile Chinese alligator egg from Anhui Chinese alligator Breeding Center (Anhui) and from the wild in Anhui Province (wild) were quantified by Ding et al. (2001). Comparisons between Anhui Province (Ding et al.’s study) and the Changxing Center (current study) can, therefore, be made. Cu, Zn, and Cd were higher and Pb was much lower in the eggs of the Changxing population than the Anhui Center and the wild alligators (Table 6). Pollution is one of the threats to the remaining wild Chinese alligator populations (Thorbjarnarson et al. 1999). The higher Cu, Zn, and Cd levels and the lower Pb levels in the eggs from the Changxing individuals are possibly indicative of specific pollutants in different areas, namely, higher Cu, Zn, and Cd content in the Changxing area, and heavier Pb pollution in the Anhui Captive Center and the surrounding wild. Unfortunately, the data by Ding et al.’s study was very much limited due to the small sampling. Another comparison that can be drawn is between the American alligators from Florida, USA, and the Chinese alligators. It has been reported that the eggs of American alligators showed no contamination of As, Cd, and Hg (Heinz et al. 1991). This implied that the critically endangered Chinese alligators experience a more hazardous environment and this should be a factor to consider in the reintroduction program.
For some endangered species such as Chinese alligator, deliberate killing and harmful sampling are strictly prohibited. Thus, non-lethal monitoring and non-destructive sampling methods for heavy metal estimations are needed. In this study, eggshell and shell membrane were investigated as indicators for monitoring heavy metal levels in Chinese alligator eggs.
We determined if there was a correlation between metal levels among the three egg compartments to test the utility of eggshell and/or shell membrane as non-invasive bioindicators of heavy metal levels in the entire alligator egg. After hatching, the Chorioallantoic membranes (CAMs) are usually retained within the eggshell and can be used for chemical residue analysis. CAMs have been used successfully to examine contaminant exposure and predict chemical concentrations in multiple species of birds and reptiles (Pepper et al. 2004). As shown in Table 3, Fe, Cu, and Zn showed significant correlations between shell membrane and egg contents, while no metal was significantly correlated between eggshell and egg contents, suggesting that shell membranes could be used as a bioindicator of the Chinese alligator egg for the three metals: Fe, Cu, and Zn.
Comparisons with American Alligator
The data from the present study indicated various levels of heavy metal accumulation in tissues and eggs of Chinese alligator, suggesting heavy metal exposure in this captive population. To evaluate the extent of heavy metal contamination in this population, we compared the mean concentrations of several elements to those of the American alligators (Table 7). As shown in Table 7, similar concentrations of heavy metals were present in the muscles of the two species. The Concentrations of the majority of the metals in the Chinese alligators were within the ranges of those detected in American alligators, except the element Pb, which showed a higher concentration in the Chinese alligators.
This study provides documentation of metal distribution in captive Chinese alligators and an impetus for more in-depth studies on exposure and response of this critically endangered species to environmental contaminants for possible heavy metal contamination control for this critically endangered species. However, a paucity of information on the effects of heavy metals on crocodilians makes it difficult to speculate on the biological significance of these concentrations (Stoneburner and Kushlan 1984). Whether the heavy metal concentrations in the alligator tissues were within the normal range has not been clear so far. Controlled laboratory studies and further investigations concerning heavy metal effects on alligators are warranted.
References
Akagi H, Nishimura H (1991) Speciation of mercury in the environment. In: Suzuki T, Imura N, Clarkson TW (eds) Advances in mercury toxicology. Plenum Press, New York, pp 53–76
Brisbin IL Jr, Jagoe CH, Gaines KF, Gariboldi JC (1998) Environmental contaminants as concerns for the conservation biology of crocodilians. In: Crocodiles. Proc 14th Working Meeting Croc Spec Grp, SSC-IUCN, pp 155–173
Burger J (1998) Heavy metals in avian eggshells: another excretion method. J Toxicol Environ Health 41:207–220
Burger J, Gibbons JW (1998) Trace elements in egg contents and egg shells of slider turtles (Tmchemys scripta) from the Savannah River Site. Arch Environ Contam Toxicol 34:382–386
Burger J, Gochfeld M (1991) Cadmium and lead in common terns (Aves: Sterna hirundd): relationship between levels in parents and eggs. Environ Monit Assess 16:253–258
Burger J, Gochfeld M, Rooney AA, Orlando EF, Woodward AR, Guillette LJ Jr (2000) Metals and metalloids in tissues of American alligators in three Florida lakes. Arch Environ Contam Toxicol 38:501–508
Campbell KR (2003) Ecotoxicology of crocodilians. Appl Herpetol 1:45–163
Delany MF, Bell JU, Sundlof SF (1988) Concentrations of contaminants in muscle of the American alligator in Florida. J Wildl Dis 24:62–66
Ding Y, Wang X, He L, Wang R, Xie W, Shao M, Thorbjarnarson J (2001) A preliminary study on heavy metal elements (Cu, Zn, Cd, and Pb) in different parts of the eggs of Chinese alligator (Alligator sinensis). Zool Res 22(3):253–256
Elsey RM, Lance VA, Campbell L (1999) Mercury levels in alligator meat in south Louisiana. Bull Environ Contain Toxicol 63:598–603
Godley BJ, Thompson DR, Furness RW (1999) Do heavy metal concentrations pose a threat to marine turtles from the Mediterranean Sea? Mar Pollut Bull 38:497–502
Guillette LJ Jr, Gross TS, Masson GR, Matter JM, Percival HF, Woodward AR (1994) Developmental abnormalities of the gonad and abnormal sex. hormone concentrations in juvenile alligators from contaminated and control lakes in Florida. Environ Health Perspect 102:680–688
Guillette LJ Jr, Pickford DB, Grain DA, Rooney AA, Percival HF (1996) Reduction r in penis size and plasma testosterone concentrations in juvenile alligators living in a contaminated environment. Gen Comp Endocrinol 101:32–42
Heaton-Jones TG, Homer BL, Heaton-Jones DL, Sundlof SF (1997) Mercury distribution in American alligators (Alligator mississippiensis) in Florida. J Zoo Wildl Med 28:62–70
Heinz GH, Percival HF, Jennings M (1991) Contaminants in American alligator eggs from Lake Apopka, Lake Griffin, and Lake Okeechobee, Florida. Environ Monit Assess 16:277–285
Honda K, Min BY, Tatsukawa R (1986) Distribution of heavy metals and their age related changes in the eastern great white egret, Egretta alba modesta, in Korea. Arch Environ Contam Toxicol 15:185–197
Hord LJ, Jennings ML, Brunell A (1990) Mercury contamination of Florida alligators. In: Crocodiles. Proc 10th Working Meeting Croc Spec Grp, SSC-IUCN, pp 229–240
Ikemoto T, Kunito T, Watanabe I, Yasunaga G, Baba N, Miyazaki N, Petrov EA, Tanabe S (2004) Comparison of trace element accumulation in Baikal seals (Pusa sibirica), Caspian seals (Pusa caspica) and northern fur seals (Callorhinus ursinus). Environ Pollut 127:83–97
Jagoe CH, Arnold-Hill B, Yanochko GM, Winger PV, Brisbin IL Jr (1998) Mercury in alligators (Alligator mississippiensis) in the southeastern United States. Sci Total Environ 21:255–262
Jeffree RA, Markich SJ, Twining JR (2001) Element concentrations in the flesh and osteoderms of estuarine crocodiles (Crocodylus porosus) from the Alligator Rivers Region, Northern Australia: biotic and geographic effects. Arch Environ Contam Toxicol 40:236–245
Manolis SC, Webb GJW, Britton AR (2002a) Crocodilian and other reptiles: Bioindicators of pollution. In: The Finniss River; a natural laboratory y of mining impacts: Past, present and future. ANSTO: Sydney, pp 65–69
Manolis SC, Webb GJW, Britton AR, Jeffree RA, Markich SJ (2002b) Trace element concentrations of wild saltwater crocodile eggs. In: The Finniss River; a natural laboratory of mining impacts: Past, present and future. ANSTO: Sydney, ANSTO, pp 58–61
Morera M, Sanpera C, Crespo S, Jover L, Ruiz X (1997) Inter- and intraclutch variability heavy metals and selenium levels in Audouin’s gull eggs from the Ebro Delta, Spain. Arch Environ Contam Toxicol 33:71–75
Nagle RD, Rowe CL, Congdon JD (2001) Accumulation and selective maternal transfer of contaminants in the turtle Trachemys scripta associated with coal ash deposition. Arch Environ Contam Toxicol 40:531–536
Ogden JC, Robertson WB Jr, Davis GE, Schmidt TW (1974) South Florida Environmental Project. Pesticides, poly chlorinated biphenyls, and heavy metals in upper food chain levels, Everglades National Park and vicinity. Final report. Division of Natural Science and Research Management Studies, Everglades National Park, Florida
Okamoto K, Yamamoto Y, Fuwa K (1978) Pepperbush powder, a new standard reference material. Anal Chem 50:1950–1951
Pepper CB, Rainwater TR, Platt SG, Dever JA, McMurry ST, Anderson TA (2004) Organochlorine pesticides in chorioallantoic membranes of Morelet’s crocodile eggs from Belize. J Wildlife Dis 40:493–500
Phelps RJ, Focardi S, Fossi C, Leonzio C, Renzoni A (1986) Chlorinated hydrocarbons and heavy metals in crocodile eggs from Zimbabwe. Trans Zimbabwe Scient Assoc 63:8–15
Rainwater TR, Adair BM, Platt SG, Anderson TA, Cobb GP, MCMurry ST (2002) Mercury in Morelet’s crocodile eggs from Northern Belize. Arch Environ Contam Toxicol 42:319–324
Rumbold DG, Fink LE, Laine KA, Niemczyk SL, Chandrasekhar T, Wankel SD, Kendall C (2002) Levels of mercury in alligators (Alligator mississippiensis) collected along a transect through the Florida Everglades. Sci Total Environ 297:239–252
Sakai H, Ichihashi H, Suganuma H, Tatsukawa R (1995) Heavy metal monitoring in sea turtles using eggs. Mar Pollut Bull 30:347–353
Sakai H, Saeki K, Ichihashi H, Suganuma H, Tanabe S, Tatsukawa R (2000) Species specific distribution of heavy metals in tissues and organs of loggerhead turtle (Caretta caretta) and green turtle (Chelonia mydas) from Japanese coastal waters. Mar Pollut Bull 40:701–709
Stoneburner DL, Kushlan JA (1984) Heavy metal burdens in American crocodile eggs from Florida Bay, Florida, USA. J Herpetol 18:192–193
Storelli MM, Marcotrigiano GO (2003) Heavy metal residues in tissues of marine turtles. Mar Pollut Bull 46:397–400
Storelli MM, Ceci E, Marcotrigiano GO (1998) Distribution of heavy metal residues in some tissues of Caretta caretta (Linnaeus) specimens beached along the Adriatic Sea (Italy). Bull Environ Contam Toxicol 60:546–552
Thorbjarnarson J, Wang XM (1999) The conservation status of the Chinese alligator FFI. Oryx 33:152–159
Thorbjarnarson J, Wang XM, Ming S (2002) Wild populations of the Chinese alligator approach extinction. Biol Conserv 103:93–102
Woodward AR, Percival HF, Jennings ML, Moore CT (1993) Low clutch viability of American alligators on Lake Apopka. Florida Sci 56:52–63
Yanochko GM, Jagoe CH, Brisbin IL Jr (1997) Tissue mercury concentrations in alligators (Alligator mississippiensis) from the Florida Everglades and the Savannah River Site, South Carolina. Arch Environ Contam Toxicol 32:323–328
Zhou Y (1997) Analysis of the decline of the wild Alligator sinensis population. Sichuan J Zool 16:137 [in Chinese]
Acknowledgments
We thank the Changxing Nature Reserve and Breeding Research Center for providing all the samples used in this study. We are also indebted to the people who helped in the sample collection, Changjun Zeng and Yunfei Hao. This work was supported in part by a grant from the Department of Wildlife Conservation, State Forestry Administration of the P.R. of China.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xu, Q., Fang, S., Wang, Z. et al. Heavy Metal Distribution in Tissues and Eggs of Chinese Alligator (Alligator sinensis). Arch Environ Contam Toxicol 50, 580–586 (2006). https://doi.org/10.1007/s00244-005-1018-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00244-005-1018-3