Abstract
Introduction
Patients with oropharyngeal or hypopharyngeal squamous cell carcinoma (SCC) have a high risk of having distant metastases or second primary tumors. We prospectively evaluate the clinical usefulness of 18F-fluoro-2-deoxyglucose positron emission tomography (18F-FDG PET), extended-field multi-detector computed tomography (MDCT), and their side-by-side visual correlation for the detection of distant malignancies in these two tumors at presentation.
Materials and methods
A total of 160 patients with SCC of the oropharynx (n = 74) or hypopharynx (n = 86) underwent 18F-FDG PET and extended-field MDCT to detect distant metastases or second primary tumors. Suspected lesions were investigated by means of biopsy, clinical, or imaging follow-up.
Results
Twenty-six (16.3%) of our 160 patients were found to have distant malignancy. Diagnostic yields of 18F-FDG PET and MDCT were 12.5% and 8.1%, respectively. The sensitivity of 18F-FDG PET for detection of distant malignancies was 1.5-fold higher than that of MDCT (76.9% vs. 50.0%, P = 0.039), while its specificity was slightly lower (94.0% vs. 97.8%, P = 0.125). Side-by-side visual correlation of MDCT and 18F-FDG PET improved the sensitivity and specificity up to 80.8% and 98.5%, respectively, leading to alteration of treatment in 13.1% of patients. A significant difference in survival rates between its positive and negative results was observed.
Conclusion
18F-FDG PET and extended-field MDCT had acceptable diagnostic yields for detection of distant malignancies in untreated oropharyngeal and hypopharyngeal SCC. 18F-FDG PET was 1.5-fold more sensitive than MDCT, but had more false-positive findings. Their visual correlation improved the diagnostic accuracy, treatment planning, and prognosis prediction.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The prognosis of patients with head and neck cancer varies substantially. Survival is the lowest in patients with oropharyngeal or hypopharyngeal tumors, who are at a high risk of developing distant metastases and second primary tumors [1–7]. Thorough physical examination and endoscopy with biopsy sampling generally serve as the main diagnostic tools in patients with oropharyngeal and hypopharyngeal squamous cell carcinoma (SCC). Computed tomography (CT) or MRI of the head and neck is routinely done to assess the locoregional status, whereas chest radiography, liver sonography, and whole-body bone scanning are performed to exclude the possibility of distant malignant lesions. However, conventional work-up may be inadequate for the assessment of small distant lesions, resulting in misclassification of clinical stage and subsequent treatment strategy and prognosis.
Chest and abdominal CT has been shown to have a higher accuracy than chest radiography and liver sonography for detection of lung and liver metastases [7–11]. However, some controversy still exists regarding the accuracy and utility of screening CT for detecting distant lesions in patients with head and neck SCC (HNSCC). These discrepancies are not surprising since head and neck tumors are a heterogeneous group of neoplasms that display a wide range of biologic behaviors. In this regard, some authors have suggested that all patients with HNSCC should undergo additional CT for accurate staging [7–9]. In contrast, other studies have concluded that CT is not generally warranted due to diagnostic yields of less than 5% [11–14]. It has been also suggested that CT should only be performed in selected high-risk patients. An overall diagnostic yield of 10.8–37.5% has been reported in this patient group [3, 6, 10, 15, 16].
With the continuous advancement of CT technology, multi-detector row computed tomography (MDCT) with thin collimation (0.5–0.75 mm) has become widely used in clinical practice. The submillimeter configuration of MDCT detector allows for multi-planar reformation with high spatial resolution. Moreover, multiple detector channels lead to substantial increase in scanning speed and volume coverage. With the use of MDCT in patients with head and neck cancer, the scan volume can be easily extended from head and neck to chest and abdomen. In patients with oropharyngeal and hypopharyngeal SCC, extended-field MDCT can be used to assess the distant conditions along with the locoregional status in a single examination. However, the potential clinical usefulness of this technique in this scenario has not yet been investigated.
18F-Fluorodeoxyglucose positron emission tomography (18F-FDG PET) is a functional imaging technique that provides information about tissue metabolism in the whole body. Several studies have previously shown that 18F-FDG PET may add to the diagnostic utility of anatomic imaging as a screening method for distant metastases in patients with head and neck cancer. An incremental diagnostic advantage with a wide range of 1–17% has been reported [17–23]. Since tumors arising from different sites of the head and neck area have different clinical behaviors, they should be analyzed separately to produce comparable results. Oropharyngeal and hypopharyngeal SCC can be grouped together because they have the same risk factors, lymphatic drainage, and are at higher risk for distant malignancies [1, 4–6]. It follows that detailed imaging evaluation of distant sites may have a greater clinical impact of this patient group.
Thus far, no study has specifically compared the diagnostic yields of 18F-FDG PET and extended-field MDCT for detection of distant malignancies in patients with oropharyngeal and hypopharyngeal SCC. Under these circumstances, we conducted a prospective study to determine the clinical usefulness of 18F-FDG PET, extended-field MDCT, and their side-by-side visual correlation for detection of distant metastases or synchronous second primary tumors in untreated oropharyngeal and hypopharyngeal SCC.
Materials and methods
Patient population
The present study was approved by the Institutional Review Board of our hospital. Written informed consent was obtained from all study participants. Between September 2003 and March 2006, a total of 160 patients (148 males and 12 females; mean age 52.9 years, age range 26–87 years) with SCC of the oropharynx or hypopharynx underwent both MDCT and 18F-FDG PET for pretreatment evaluation. The studies were not acquired on the same day but were performed within 1 week of each other. Inclusion criteria comprised the following: (1) a pathological diagnosis of SCC in the oropharynx or hypopharynx, (2) negative results from chest radiography, liver sonography, and whole-body bone scanning, and (3) no prior treatment to the head or neck region. Exclusion criteria were pregnancy and poorly controlled diabetes with a fasting serum glucose level of >200 mg/dL.
18F-FDG PET and extended-field MDCT
Images were obtained with an ECAT EXACT HR+ scanner (Siemens/CTI, Knoxville, TN, USA) with an in-plane spatial resolution of 4.5 mm and a transaxial field of view of 15 cm. All patients fasted for at least 6 h before PET imaging. The serum glucose level was measured before the intravenous administration of 370 MBq (10 mCi) of FDG. After intravenous injection of FDG, the patients relaxed in a quiet room for 40–60 min. The patient was scanned while lying supine along the central axis of the PET table. Seven sequential emission images were obtained from the head to the upper thigh, and transmission scans were obtained with 68Ge rod sources for attenuation correction. The emission and transmission scans were obtained in an alternating sequence per bed position. The reconstruction of transmission and emission scans used accelerated maximum-likelihood reconstruction and ordered-subset expectation maximization.
CT examinations were performed with a 16-detector row CT scanner (Somatom Sensation 16, Simens, Erlangen, Germany). Collimation was 16 × 0.75 mm and gantry rotation time was 0.5 s. All patients were scanned in the supine position from the skull base to the lower abdomen after intravenous administration of 100 mL contrast medium (Omnipaque, 350 mgI/mL, Amersham Health, Cork, Ireland). Tube voltage of 120 kVp and mAseff of 180–250 were used. Both the section thickness and reconstruction increment were 5 mm. All the images were electronically sent to a picture archiving and communication system for interpretation.
Image interpretation
18F-FDG PET and CT images were interpreted blindly and independently of each other. Three experienced nuclear medicine physicians independently interpreted the 18F-FDG PET images. Apart from being informed about the study protocol, no information on other imaging results was given to the nuclear medicine physicians.
Interpretation was based on visual evaluation. Any focus of FDG activity that was greater than surrounding background and not attributable to normal FDG biodistribution was evaluated. A list of potential distant lesions was recorded. The FDG uptake was evaluated with reference to normal tissue and scored the intensity of the FDG uptake on a five-point scale used by Ng et al. [24]: 0, definitely benign; 1, probably benign; 2, equivocal; 3, probably malignant; and 4, definite malignant. Both grade 3 and grade 4 were considered to be positive results. Discrepancies were resolved by consensus. Two experienced radiologists reviewed separately the CT findings. The criteria for lung malignancy were solitary lung nodule greater than or equal 1 cm if well defined, and nodular lesions ≥0.5 cm if irregular or multiple. Calcified nodules and segmental distributed lung disease were considered as benign process. Distant lymph node was considered metastatic if its short axial diameter was ≥1 cm.
Outcome measures and data analysis
The head-and-neck cancer research team of our hospital (nuclear medicine physicians, radiologists, radiation oncologists, otolaryngologists, and medical oncologists) met weekly to view 18F-FDG PET and MDCT images side-by-side. The combined result was then made by discussing each case, and any disagreements were resolved by consensus. In the presence of suspected malignant lesions, biopsy was performed when possible. If a biopsy was not feasible or yielded negative results in patients with suspected imaging findings, close clinical and imaging follow-up was pursued. All patients were followed up for at least 12 months or until death. A minimum 12-month follow-up time was chosen since the majority of distant metastases become evident within this period [25]. Patients with negative imaging results at presentation who developed distant metastases within the subsequent 12-month period were considered as false negatives. Malignant lung lesions that cannot be distinguished between primary lesions or metastasis by histologic means were classified as distant metastases if appeared they as multiple, well-defined, or peripherally located masses. On the other hand, they were classified as second primary lung cancer if they appeared as a solitary, speculated, or centrally located mass [16, 22, 26].
We applied univariate and multivariate analyses to assess the relationship of the clinical characteristics and the occurrence of distant malignancies in patients with oropharyngeal and hypopharyngeal SCC. We also calculated the sensitivity, specificity, accuracy, positive predictive value, and negative predictive value of extended-field MDCT, 18F-FDG PET, and their visual correlation. Differences in sensitivity and specificity among these methods were assessed using the McNemar’s test. Follow-up was continued until August 2007 in order to evaluate the predictive potential of 18F-FDG PET and MDCT on patient outcome and survival. Survival time was recorded as the interval from the date of diagnosis to the date of death or last follow-up. Cumulative survival rate was calculated by using the Kaplan–Meier survival method. Differences in survival between patients with positive and negative results of different imaging methods were determined by means of the log-rank test.
Results
In 74 (46.3%) of our 160 patients, the tumors originated in the oropharynx, whereas in the remaining 86 (53.7%) patients, the tumors originated in the hypopharynx. T stages of the primary tumors were as follows: eight patients (5%) with T1, 39 patients (24.4%) with T2, 30 patients (18.8%) with T3, and 83 patients (51.8%) with T4. N stages of our patients were as follows: 29 patients (18.1%) with N0, 23 (14.4%) patients with N1, 92 (57.5%) patients with N2, and 16 (10%) patients with N3. Advanced nodal stage (N2–N3) occurred in 63 (73.3%) of 86 patients with hypopharyngeal SCC and in 45 (60.8%) of 74 patients with oropharyngeal SCC.
In the entire study cohort, a total of 26 patients (16.3%) were found to have distant malignant lesions. Specifically, half of them had distant metastases and another half had distant second primary tumors. Out of the 26 patients with distant malignancies, the diagnosis was confirmed pathologically in 18 individuals, whereas in the remaining eight patients definite progression was seen in follow-up imaging studies. A total of 27 distant sites were evident (14 distant metastases and 13 distant second primary tumors). The distant metastatic sites were lung (n = 8), mediastinal lymph nodes (n = 3), bone (n = 2), and liver (n = 1). The most common distant synchronous primary tumor was esophageal SCC (Fig. 1) that occurred in eight patients (seven patients with hypopharyngeal SCC and one patient with oropharyngeal SCC). Other distant primary tumors comprised colon adenocarcinoma (two patients with hypopharyngeal SCC), lung carcinoma (two patients with oropharyngeal SCC), and hepatocellular carcinoma (one patient with hypopharyngeal SCC).
The association between different variables (age, sex, location, T stage, and N stage) and the occurrence of synchronous distant malignancy in patients with oropharynx SCC and hypopharyngeal SCC is presented in Table 1. Univariate analysis showed that the risk of distant malignancies was significantly higher in patients with hypopharyngeal SCC or advanced nodal disease. The incidence of distant malignancy was 2.3-fold higher in patients with hypopharyngeal SCC compared to those with oropharyngeal SCC (22.1% vs. 9.5%, P = 0.031). On the other hand, the incidence of distant malignancy in patients with advanced N stage was 2.6-fold higher compared to those with early N stage (N0–N1; 20.2% vs. 7.8%, P = 0.042). Age, gender, and T stage were not significantly associated with risk of distant malignancy. After multivariate analysis with the logistic regression method, only hypopharyngeal location (hazard ratio 2.714, 95% confidence interval 1.071–6.882, P = 0.035) was the independent factor for distant malignancy.
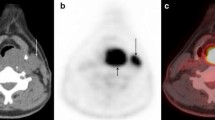
Out of 160 study participants, 26 patients (16.3%) had distant malignancy. Diagnostic yields of 18F-FDG PET and MDCT were 12.5% (20 of 160 patients) and 8.1% (13 of 160 patients), respectively. As shown in Table 2, the patient-based sensitivity of 18F-FDG PET for detection of distant malignancies was 1.5-fold higher than that of MDCT (76.9% vs. 50.0%, P = 0.039), while the patient-based specificity of 18F-FDG PET was slightly lower than that of MDCT (94.0% vs. 97.8%, P = 0.125). The sensitivity and specificity of the side-by-side visual correlation of 18F-FDG PET and MDCT were 3.9% and 4.5% greater than those of 18F-FDG PET alone, respectively (80.8% vs. 76.9%, P = 1; 98.5% vs. 94.0%, P = 0.031, respectively). Among 20 patients with distant malignancies disclosed by 18F-FDG PET, 8 had false-negative findings on MDCT. Out of these eight patients, five had synchronous second primary tumors (two thoracic esophageal SCCs, two lung carcinomas, and one colon adenocarcinoma, Fig. 2). On MDCT, the esophageal and colon lesions appeared as subtle wall thickening and were initially overlooked. One of the lung carcinomas manifested as a reticulonodular infiltrate in the left lower lobe, while the other manifested as two peripheral masses with focal pleural thickening in the right upper lobe and the left upper lobe, respectively. These lung lesions were initially interpreted as granulomatous inflammatory disease (score 2). On 18F-FDG PET, these lesions showed avid FDG uptake suggestive of malignancy (score 4). The remaining three patients had metastatic tumors (one bone metastasis, one mediastinal nodal metastasis, and one both liver and bone metastases, Fig. 3).
On the other hand, 18F-FDG PET missed distant malignancies in six patients (five patients with metastatic lung lesions and one patient with thoracic esophageal SCC). Out of these six patients, MDCT provided true-positive results in a single case. In this patient, an irregular nodule of 4.4 × 5.7 mm in size in the periphery of the lung was evident. The lung lesions in the remaining four patients were not visible on MDCT at presentation, but became visible on subsequent imaging after 3, 5, 6, and 7 months, respectively. Both 18F-FDG PET and MDCT missed an esophageal SCC disclosed as mucosal thickening by endoscopy. Table 2 also showed that the location-based sensitivity of 18F-FDG PET was highest in the colon, liver, bone, and mediastinum, followed by the esophagus and the lung.
In our series, both 18F-FDG PET and MDCT yielded false-positive results in two patients with enlarged mediastinal nodes. In addition, 18F-FDG PET produced eight false-positive results in other six patients (four lesions were reactive hyperplastic mediastinum lymph nodes, two lesions were due to pulmonary inflammatory disease (Fig. 4), and the remaining two lesions were misinterpretation of the ureters as abdominal metastatic nodes). The location-based specificity of 18F-FDG PET was the lowest in the mediastinum. On the other hand, MDCT produced a false-positive result in one patient with a granulomatous nodular lesion in the lung. This lesion was correctly classified as true-negative by 18F-FDG PET due to low FDG uptake.
As shown in Table 3, detection of distant malignancy by 18F-FDG PET, MDCT, or both at initial presentation led to positive clinical impact by altering clinical management in 13.1% (21/160) of our patients. Accordingly, patients with metastatic lesions were primarily treated with chemotherapy while most patients with second tumors underwent additional treatment for distant primary tumors. Table 4 lists the results of different imaging techniques and their corresponding survival rates at 0.5, 1, 1.5, and 2 years. Patients with positive 18F-FDG PET results or positive MDCT results had a poorer survival than those with negative results, albeit the differences failed to reach statistical significance. A significant difference in survival rates was observed between positive and negative results of visual correlation of 18F-FDG PET and MDCT (P = 0.017). Overall survival rates of patients with positive results of such visual correlation were 43% at 1 year and 32% at 2 years, while those with negative results were 72% at 1 year and 55% at 2 years. Figure 5 shows the Kaplan–Meier curves of survival according to imaging results.
Discussion
Distant metastases and second primary tumors are important prognosticators in patients with HNSCC [1, 2, 4, 5, 27–29]. Among HNSCC patients, the reported incidence of distant metastases at presentation is between 1.6% and 29% [7, 9–11, 15, 16, 26]. The incidence of distant metastases of the head and neck tumors is influenced by the location of the primary tumor and N stage of the neoplasm. The influence of T stage of the neoplasm, however, remains controversial [1, 4, 6, 28–30]. The reported incidence of synchronous second primary tumors in patients with HNSCC is between 2.3% and 12% [5, 7, 27, 30–32]. The risk of development of a second primary tumor in patients with HNSCC is 10- to 30-fold higher than in the general population. Notably, half of second primary tumors occur outside the head and neck area [5, 30, 33, 34]. In this prospective study of 160 patients with oropharyngeal or hypopharyngeal SCC, males accounted for the vast majority (>90%). The plausible explanation was that only a very small female population in Taiwan has heavy tobacco and alcohol consumption, which are the recognized major factors for the development of oropharyngeal or hypopharyngeal SCC. The incidence of distant malignancy at initial presentation was 16%. Specifically, the rate of distant metastases was 8%, whereas the frequency of second primary tumors was also 8%. Univariate analysis of our results showed that tumor location and nodal stage were significantly associated with the occurrence of distant malignancy. Multivariate analysis demonstrated that hypopharyngeal location was the independent risk factor for distant malignancy. This result suggests that the whole-body imaging technique may be more feasible in hypopharyngeal SCC.
Lung, liver, bone, and mediastinal nodes are the most frequent sites of distant metastasis in patients with HNSCC [4, 15, 28, 29]. In contrast, lung and esophagus are the most frequent sites of synchronous primary tumors outside the head and neck region [30, 31, 35]. In this study, a total of 27 distant malignancy sites were identified among our 26 patients with distant malignancies. The lung was the most frequent site, followed by the esophagus, mediastinal nodes, colon, bone, and liver. Nine of these 27 distant malignancies, including all the two bone lesions, one of the two liver lesions, one of the two colon lesions, one of three mediastinal nodal lesions, two of the eight esophageal lesions, and two of the ten lung lesions, were missed by MDCT but correctly identified by 18F-FDG PET. These findings might reflect the superiority of 18F-FDG PET over MDCT for identification of malignant lesions in the bone, colon, liver, mediastinum, esophageal, and, to a lesser extent, the lung. Although it is commonly thought that 18F-FDG PET is less sensitive than CT for the detection of small lung mass, 18F-FDG PET may be helpful to identify some lung malignancies with vivid FDG uptake but with atypical CT appearance, as seen in our two cases.
In this study, 18F-FDG PET missed distant malignancies in six patients (five patients with metastatic lung lesions and one patient with thoracic esophageal SCC). Among them, one patient with a small, irregular peripherally lung lesion was identified by MDCT. Side-by-side visual correlation of 18F-FDG PET with MDCT revised this false-negative result. Although our study showed no significant increment in sensitivity of visual correlation 18F-FDG PET and MDCT over 18F-FDG PET alone, this should not strictly imply that such visual correlation would not fare better. As shown in this case, the combined information helped disclose a small lung metastasis and subsequently led to spare fruitless aggressive local treatment. The remaining four pulmonary metastatic lesions were also false negative on initial MDCT and became visible on subsequent post-treatment images during the 12-month follow-up. These metastases were presumably present subclinically as micrometastases at initial presentation. The occurrence of such false-negative lesions is likely due to technical limitation of 18F-FDG PET. Specifically, since the threshold spatial resolution of PET is about 5 mm, it is possible that lesions with smaller size could be missed [36, 37]. Our study provides evidence that 18F-FDG PET, even when visually correlated with MDCT, is unable to detect all distant malignancies. The occurrence of lung micrometastases is the main shortcoming and occurred in 2.5% (4/160) of our patients. The presence of early superficial lesion is another pitfall. In this regard, a small second primary esophageal tumor was missed by both 18F-FDG PET and MDCT in one patient. Because early esophageal tumors can be cured, the precocious identification of these lesions may have great clinical relevance. Since the esophagus was the most common site of synchronous second primary tumors in our study, we suggest that negative 18F-FDG PET results cannot obviate the need for panendoscopy in patients who are treated with curative intent.
18F-FDG PET lacks precise anatomical resolution and may overdiagnose some inflammatory conditions. By virtue of its high spatial resolution, MDCT may serve as a cross-sectional imaging tool complementary to 18F-FDG PET in the evaluation of distant malignancies in patients with oropharyngeal and hypopharyngeal SCC. In this study, MDCT produced true-negative results in 80% of 18F-FDG PET false-positive findings. Accordingly, we found that visual correlation of PET with MDCT may be of aid to identify the exact nature of FDG avid areas at distant sites. Accordingly, it disclosed the presence of hilar fat in benign mediastinal nodes in FDG avid nodal hyperplasia, as well as the presence of segmental pulmonary infiltration in FDG avid inflammatory lung disease. It also corrected the misinterpretation of the ureter as abdominal metastatic lymph nodes due to spatial error. Notably, the specificity of visual correlation of 18F-FDG PET and MDCT was significantly higher than 18F-FDG PET alone.
This study had some potential limitations. First, slice thickness of our PET scanner was 4.5 mm; therefore, some small lesions might be missed due to the partial volume effect. Application of the new generation scanners of reduced slice thickness may raise the sensitivity for lesion detection. Second, we used a slice thickness of 5 mm in MDCT and the size criteria of lung malignancy of ≥1 cm in well-defined, solitary nodule and of ≥0.5 cm if irregular or multiple. It follows that metastases smaller than 0.5 cm might be underdiagnosed. Currently, such small lesions can be detected with thin-slice MDCT, although the higher detection rate of small metastases has to be evaluated from a clinical perspective about the expanse of more false-positive results. Finally, the protocol for contrast material injection on MDCT affects detection of metastases, and it is recognized that multi-phase CT is more accurate than single-phase CT in the detection of liver metastasis. Since single-phase enhanced CT was performed to detect distant malignancy in this study, one might be concerned that such technique might not be optimal for the evaluation of liver malignancies. Nonetheless, our approach reflects the daily practice in most institutions. In this large prospective study, 18F-FDG PET and MDCT showed a diagnostic yield for distant malignancy in 12.5% and 8.1% of our patients, respectively. The visual correlation between 18F-FDG PET and MDCT led to a positive clinical impact by altering treatment in 13.1% of patients. Patients with distant metastases were offered systemic chemotherapy, whereas most patients with second primary tumors underwent additional surgery, chemoradiotherapy, or transarterial embolization. On the other hand, patients with positive imaging results had a poorer survival rate compared with patients with negative results. It is thus concluded that imaging results had prognostic implications. In the light of these findings, the use of these imaging methods to detect distant malignancy of oropharyngeal and hypopharyngeal SCC can be justified. 18F-FDG PET produced a 4.4% higher diagnostic yield than extended-field MDCT and thus should be preferred when available. Visual correlation of 18F-FDG PET with MDCT is highly recommended whenever available because this technique has a higher accuracy than PET alone [37–39]. This diagnostic advantage may facilitate the clinical management as well as the assessment of survival rates, as shown in our study.
In recent years, dual modality PET/CT has been used to provide accurately fused functional PET and morphological CT data in a single examination. Such hybrid machines may overcome the inherent limitations of PET due to poor anatomical resolution and has also been reported to have significantly better diagnostic accuracy than PET alone in the evaluation of HNSCC [40–42]. Thus far, however, no large-scale prospective study on the potential usefulness of PET/CT for pretreatment evaluation of distant sites has been conducted in patients with oropharyngeal or hypopharyngeal SCC. Our findings suggest that PET-CT would have a significant diagnostic yield in this clinical setting but a large designed prospective study is needed to confirm this. To our knowledge in this study we have examined the largest series of untreated oropharyngeal and hypopharyngeal SCC patients undergoing both 18F-FDG PET and MDCT. Specifically, our data may provide the basis for future comparisons of PET/CT with these imaging techniques for detection of distant metastases and second primary tumors in this patient group.
Conclusion
18F-FDG PET and extended-field MDCT appeared to have acceptable diagnostic yield for detection of distant malignancies in patients with newly diagnosed oropharyngeal and hypopharyngeal SCC. 18F-FDG PET was more sensitive but slightly less specific than MDCT. Visual correlation of 18F-FDG PET with MDCT improved the diagnostic accuracy of imaging findings, thereby facilitating treatment planning and prognosis prediction.
References
Kotwall C, Sako K, Razack MS, Rao U, Bakamjian V, Shedd DP (1987) Metastatic patterns in squamous cell cancer of the head and neck. Am J Surg 154:439–442
Mohadjer C, Dietz A, Maier H, Weidauer H (1996) Distant metastasis and incidence of second carcinomas in patients with oropharyngeal and hypopharyngeal carcinomas. HNO 44:134–139
Halpern J (1997) The value of chest CT scan in the work-up of head and neck cancers. J Med 28:191–198
Ferlito A, Shaha AR, Silver CE, Rinaldo A, Mondin V (2001) Incidence and sites of distant metastases from head and neck cancer. ORL J Otorhinolaryngol Relat Spec 63:202–207
Erkal HS, Mendenhall WM, Amdur RJ, Villaret DB, Stringer SP (2001) Synchronous and metachronous squamous cell carcinomas of the head and neck mucosal sites. J Clin Oncol 19:1358–1362
Loh KS, Brown DH, Baker JT, Gilbert RW, Gullane PJ, Irish JC (2005) A rational approach to pulmonary screening in newly diagnosed head and neck cancer. Head Neck 27:990–994
Reiner B, Siegel E, Sawyer R, Brocato RM, Maroney M, Hooper F (1997) The impact of routine CT of the chest on the diagnosis and management of newly diagnosed squamous cell carcinoma of the head and neck. AJR Am J Roentgenol 169:667–671
Houghton DJ, Hughes ML, Garvey C et al (1998) Role of chest CT scanning in the management of patients presenting with head and neck cancer. Head Neck 20:614–618
Ong TK, Kerawala CJ, Martin IC, Stafford FW (1999) The role of thorax imaging in staging head and neck squamous cell carcinoma. J Craniomaxillofac Surg 27:339–344
Warner GC, Cox GJ (2001) Evaluation of chest radiography versus chest computed tomography in screening for pulmonary malignancy in advanced head and neck cancer. J Otolaryngol 32:107–109
Glynn F, Brennan S, O'Leary G (2006) CT staging and surveillance of the thorax in patients with newly diagnosed and recurrent squamous cell carcinoma of the head and neck: is it necessary? Eur Arch Otorhinolaryngol 263:943–945
Brown DH, Leakos M (1998) The value of a routine bone scan in a metastastic survey. J Otolaryngol 27:187–189
Nilssen EL, Murthy P, McClymont L, Denholm S (1999) Radiological staging of the chest and abdomen in head and neck squamous cell carcinoma—are computed tomography and ultrasound necessary? J Laryngol Otol 113:152–154
Tan L, Greener CC, Seikaly H, Rassekh CH, Calhoun KH (1999) Role of screening chest computed tomography in patients with advanced head and neck cancer. Otolaryngol Head Neck Surg 120:689–692
Jackel MC, Rausch H (1999) Distant metastasis of squamous epithelial carcinomas of the upper aerodigestive tract. The effect of clinical tumor parameters and course of illness. HNO 47:38–44
de Bree R, Deurloo EE, Snow GB, Leemans CR (2000) Screening for distant metastases in patients with head and neck cancer. Laryngoscope 110:397–401
Teknos TN, Rosenthal EL, Lee D, Taylor R, Marn CS (2001) Positron emission tomography in the evaluation of stage III and IV head and neck cancer. Head Neck 23:1056–1060
Wax MK, Myers LL, Gabalski EC, Husain S, Gona JM, Nabi H (2002) Positron emission tomography in the evaluation of synchronous lung lesions in patients with untreated head and neck cancer. Arch Otolaryngol Head Neck Surg 128:703–707
Schwartz DL, Ford EC, Rajendran J et al (2003) Staging of head and neck squamous cell cancer with extended-field FDG-PET. Arch Otolaryngol Head Neck Surg 129:1173–1178
Dresel S, Grammerstorff J, Schwenzer K et al (2003) [18F]FDG imaging of head and neck tumours: comparison of hybrid PET and morphological methods. Eur J Nucl Med Mol Imaging 30:995–1003
Schmid DT, Stoeckli SJ, Bandhauer F et al (2003) Impact of positron emission tomography on the initial staging and therapy in locoregional advanced squamous cell carcinoma of the head and neck. Laryngoscope 113:888–891
Brouwer J, Senft A, de Bree R et al (2006) Screening for distant metastases in patients with head and neck cancer: is there a role for (18F) FDG-PET? Oral Oncol 42:275–280
Keyes JW, Chen MY, Watson NE, Greven KM, McGuirt WF, Williams DW (2000) FDG PET evaluation of head and neck cancer: value of imaging the thorax. Head Neck 22:105–110
Ng SH, Chang JT, Chan SC et al (2004) Nodal metastases of nasopharyngeal carcinoma: patterns of disease on MRI and FDG PET. Eur J Nucl Med Mol Imaging 31:1073–1080
Leemans CR, Tiwari R, Nauta JJ, van der Waal I, Snow GB (1993) Regional lymph node involvement and its significance in the development of distant metastases in head and neck carcinoma. Cancer 71:452–456
Brouwer J, de Bree R, Hoekstra OS et al (2005) Screening for distant metastases in patients with head and neck cancer: is chest computed tomography sufficient? Laryngoscope 115:1813–1817
Brouwer J, de Bree R, Hoekstra OS et al (1994) Synchronous and metachronous head and neck carcinomas. Cancer 74:1933–1938
Calhoun KH, Fulmer P, Weiss R, Hokanson JA (1994) Distant metastases from head and neck squamous cell carcinomas. Laryngoscope 104:1199–1205
Leon X, Quer M, Orus C, del Prado Venegas M, Lopez M (2000) Distant metastases in head and neck cancer patients who achieved loco-regional control. Head Neck 22:680–686
Shaha AR, Hoover EL, Mitrani M, Marti JR, Krespi YP (1998) Synchronicity, multicentricity, and metachronicity of head and neck cancer. Head Neck Surg 10:225–228
Haughey BH, Gates GA, Arfken CL, Harvey J (1992) Meta-analysis of second malignant tumors in head and neck cancer: the case for an endoscopic screening protocol. Ann Otol Rhinol Laryngol 101:105–112
Jäckel MC, Reischl A, Huppert P (2007) Efficacy of radiologic screening for distant metastases and second primaries in newly diagnosed patients with head and neck cancer. Laryngoscope 117:242–247
Fijuth J, Mazeron JJ, Le Péchoux C et al (1992) Second head and neck cancers following radiation therapy of T1 and T2 cancers of the oral cavity and oropharynx. Int J Radiat Oncol Biol Phys 24:59–64
Parker RG, Enstrom JE (1988) Second primary cancers of the head and neck following treatment of initial primary head and neck cancers. Int J Radiat Oncol Biol Phys 14:561–564
Guardiola E, Pivot X, Dassonville O et al (2004) Is routine triple endoscopy for head and neck carcinoma patients necessary in light of a negative chest computed tomography scan? Cancer 101:2028–2033
Stoeckli SJ, Steinert H, Pfaltz M, Schmid S (2002) Is there a role for positron emission tomography with 18F-fluorodeoxyglucose in the initial staging of nodal negative oral and oropharyngeal squamous cell carcinoma. Head Neck 24:345–349
Ng SH, Yen TC, Liao CT et al (2005) 18F-FDG PET and CT/MRI in oral cavity squamous cell carcinoma: a prospective study of 124 patients with histologic correlation. J Nucl Med 46:1136–1143
Vansteenkiste JF, Stroobants SG, Dupont PJ et al (1998) FDG PET scan in potentially operable non-small cell lung cancer: do anatometabolic PET–CT fusion images improve the localization of regional lymph node metastases? The Leuven Lung Cancer Group. Eur J Nucl Med Mol Imaging 25:1495–1501
Ng SH, Yen TC, Chang JT et al (2006) Prospective study of [18F]fluorodeoxyglucose positron emission tomography and computed tomography and magnetic resonance imaging in oral cavity squamous cell carcinoma with palpably negative neck. J Clin Oncol 24:4371–4376
Schoder H, Yeung HW, Gonen M, Kraus D, Larson SM (2004) Head and neck cancer: clinical usefulness and accuracy of PET/CT image fusion. Radiology 231:65–72
Chen YK, Su CT, Ding HJ et al (2006) Clinical usefulness of fused PET/CT compared with PET alone or CT alone in nasopharyngeal carcinoma patients. Anticancer Res 26:1471–1477
Jeong HS, Baek CH, Son YI et al (2007) Use of integrated 18F-FDG PET/CT to improve the accuracy of initial cervical nodal evaluation in patients with head and neck squamous cell carcinoma. Head Neck 29:203–210
Acknowledgments
This work was supported by the National Science Council, grant no. 94-2314-B-182A-109 and by the Chang Gung Memorial Hospital, grant no. 95-0372B.
Conflict of interest statement
We declare that we have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ng, SH., Chan, SC., Liao, CT. et al. Distant metastases and synchronous second primary tumors in patients with newly diagnosed oropharyngeal and hypopharyngeal carcinomas: evaluation of 18F-FDG PET and extended-field multi-detector row CT. Neuroradiology 50, 969–979 (2008). https://doi.org/10.1007/s00234-008-0426-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00234-008-0426-2