Abstract
Objective
The clinical relevance of the CYP2D6 oxidation polymorphism in the treatment of depression with desipramine (DMI) was studied prospectively in depressed outpatients.
Methods
After CYP2D6 phenotype determination with dextromethorphan, 31 patients were treated with oral DMI at a dosage of 100 mg per day for 3 weeks. At the end of the 3rd week of treatment, severity of depressive symptoms was assessed by the Hamilton Depression Rating Scale and steady-state plasma concentrations of DMI and its metabolite 2-hydroxydesipramine (2-OH-DMI) were measured by high-performance liquid chromatography (HPLC).
Results
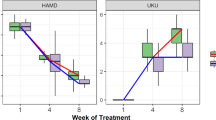
Plasma DMI levels were significantly correlated with dextromethorphan metabolic ratio. The two patients with the poor metabolizer phenotype showed the highest plasma concentrations of DMI and complained of severe adverse effects, requiring dosage reduction. No significant correlation was found between plasma levels of either DMI or DMI plus 2-OH-DMI and antidepressant effect.
Conclusion
These findings indicate that the dextromethorphan metabolic ratio has a great impact on steady-state plasma levels of DMI in depressed patients and may identify subjects at risk for severe concentration-dependent adverse effects. On the other hand, this index of CYP2D6 activity does not seem to predict the degree of clinical amelioration.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Dahl ML, Bertilsson L (1993) Genetically variable metabolism of antidepressants and neuroleptic drug in man. Pharmacogenetics 3: 61–70
Spina E, Caputi AP (1994) Pharmacogenetic aspects in the metabolism of psychotropic drugs: pharmacokinetic and clinical implications. Pharmacol Res 29: 121–137
Bertilsson L, Dahl ML (1996) Polymorphic drug oxidation: relevance to the treatment of psychiatric disorders. CNS Drugs 5: 200–223
Bertilsson L, Aberg-Wistedt A (1983) The debrisoquine hydroxylation test predicts steady-state plasma levels of desipramine. Br J Clin Pharmacol 15: 388–390
Nordin C, Siwers B, Benitez J, Bertilsson L (1985) Plasma concentrations of nortriptyline and its 10-hydroxymetabolite in depressed patients: relation to polymorphic debrisoquine hydroxylation metabolic ratio. Br J Clin Pharmacol 19: 832–835
Brosen K, Klysner R, Gram LF, Otton SV, Bech P, Bertilsson L (1986) Steady-state concentrations of imipramine and its metabolities in relation to the sparteine/debrisoquine polymorphism. Eur J Clin Pharmacol 30: 679–684
Spina E, Arena A, Pisani F (1987) Urinary desipramine hydroxylation index and steady-state plasma concentrations of imipramine and desipramine. Ther Drug Monit 9: 129–133
Bertilsson L, Mellstrom B, Sjoqvist F, Martensson B, Asberg M (1981) Slow hydroxylation of nortriptyline and concomitant poor debrisoquine hydroxylation: clinical implications. Lancet i: 560–561
Bertilsson L, Aberg-Wistedt A, Gustaffson LL, Nordin C (1985) Extremely rapid hydroxylation of debrisoquine: a case report with implicaton for treatment with nortriptyline and other tricyclic antidepressants. Ther Drug Monit 7: 478–480
Bluhm RE, Wilkinson GR, Shelton R, Branch RA (1993) Genetically determined drug-metabolizing activity and desipramine-associated cardiotoxicity: a case report. Clin Pharmacol Ther 53: 89–95
Meyer JW, Woggon B, Kupfer A (1988) Importance of oxidative polymorphism on clinical efficacy and side-effects of imipramine: a retrospective study. Pharmacopsychiatry 21: 365–366
Spina E, Steiner E, Ericsson O, Sjoqvist F (1987) Hydroxylation of desmethylimipramine: dependence on the debrisoquine hydroxylation phenotype. Clin Pharmacol Ther 41: 314–319
Hamilton M (1967) Development of a rating scale for primary depressive illness. Br J Soc Clin Psychol 6: 278–296
Spina E, Campo GM, Avenoso A, Caputi AP, Zuccaro P, Pacifici R, Gatti G, Strada G, Bartoli A, Perucca E (1994) CYP2D6-related oxidation polymorphism in Italy. Pharmacol Res 29: 281–289
Silverman G, Braithwaite RA (1973) Benzodiazepines and tricyclic antidepressant plasma levels. BMJ 3: 18–20
Sutfin TA, Jusko WJ (1979) High-performance liquid chromatography assay for imipramine, desipramine, and their 2-hydroxylated metabolites. J Pharm Sci 68: 703–705
Reisby N, Gram LF, Bech P, Nagy A, Petersen GO, Ortmann J, Ibsen I, Dencker SJ, Jacobsen O, Krautwald O, Sondergaard I, Christiansen J (1977) Imipramine: clinical effects and pharmacokinetic variability. Psychopharmacology 54: 263–272
Glassman A. Perel JM, Shostak M, Kantor SJ, Fleiss JL (1977) Clinical implications of imipramine plasma levels for depressive illness. Arch Gen Psychiatry 34: 197–204
Asberg M, Cronholm B, Sjoqvist F, Tuck D (1971) Relationship between plasma level and therapeutic effect of nortriptyline. BMJ 3: 331–334
Nelson JC, Jatlow P, Quinlan DM, Bowers MB (1982) Desipramine plasma concentration and antidepressant response. Arch Gen Psychiatry 39: 1419–1422
Amsterdam JD, Brunswick DJ, Potter L, Winokur A, Rickels K (1985) Desipramine and 2-hydroxydesipramine plasma levels in endogenous depressed patients: lack of correlation with therapeutic response. Arch Gen Psychiatry 42: 361–364
Potter WZ, Calil HM, Sutfin TA, Zavadil AP, Jusko WJ, Rapoport J, Goodwin FK (1982) Active metabolites of imipramine and desipramine in man. Clin Pharmacol Ther 32: 393–401
Bertilsson L, Dahl ML, Sjoqvist F, Aberg-Wistedt A, Humble M, Johannson I, Lundqvist E, Ingelman-Sundberg M (1993) Molecular basis for rational megaprescribing in ultrarapid hydroxylators for debrisoquine. Lancet 341: 63
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Spina, E., Gitto, C., Avenoso, A. et al. Relationship between plasma desipramine levels, CYP2D6 phenotype and clinical response to desipramine: a prospective study. Eur J Clin Pharmacol 51, 395–398 (1997). https://doi.org/10.1007/s002280050220
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s002280050220