Abstract
Background
Elevated plasma total homocysteine (tHcy) appeared in levodopa/dopadecarcoxylase inhibitor (DDI) treated patients with Parkinson’s disease (PD). One therapeutic approach for tHcy reduction is vitamine supplementation, since folic acid and cobalamine catalyse and enhance metabolism of tHcy to methionine. A further therapeutic alternative is inhibition of catechol-O-methyltransfrase (COMT) on a regular basis, when levodopa/DDI treatment is performed.
Methods
We measured the concentrations of S-adenosylmethionine (SAM), S-adenosylhomocysteine (SAH), tHcy, levodopa and 3-O-methyldopa in plasma of 13 levodopa treated PD patients before first drug intake at 0600 hours. Blood samples were taken before and after 2 days of additional application of the centrally acting catechol-O-methyltransferase inhibitor tolcapone 100 mg t.i.d.
Results
Plasma levels of SAH [day 1: 48.32±22.52, 23.92–98.25 (mean±SD, range; μmol/l); day 3: 37.72±15.84, 23.4–61.89; p=0.01] and tHcy (day 1: 13.88±5.62, 7.63–24.81; day 3: 11.38±4.44, 5.98–20.45; p=0.04) significantly reduced. Plasma levels of levodopa did not significantly (p=0.17) increase, whereas 3-OMD concentrations significantly (p=0.0002) reduced after additional tolcapone intake. There was no significant change of SAM plasma levels (p=0.22).
Conclusion
Our prospective trial shows, that COMT inhibition with tolcapone lowers tHcy synthesis. Tolcapone may also possess beside its proven, occasional, hepatotoxic potency also beneficial effects via decrease of SAH and tHcy. This may hypothetically reduce homocysteine mediated progress of neuronal degeneration and the risk for onset of dementia, vascular disease and polyneuropathy in levodopa treated PD patients in the long term.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
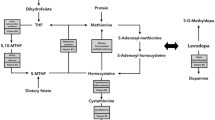
Levodopa is administered with dopa decarboxylase inhibitors (DDI), such as carbidopa or benserazide, to prevent its peripheral degradation. An other metabolic pathway is the conversion of levodopa to 3-O-methyldopa (3-OMD) by the enzyme catechol-O-methyltransferase (COMT). This increases conversion of levodopa to 3-O-methyldopa (3-OMD) by COMT in blood, peripheral tissue and nigrostriatal neurons. COMT requires Mg2+ as cofactor and as methyl donor S-adenosylmethionine (SAM), which is synthesised from adenosintriphosphate and methionine (MET). Therefore, O-methylation of levodopa to 3-OMD is associated with conversion of SAM to S-adenosyl-L-homocysteine (SAH) and subsequently total homocysteine (tHcy) [7]. Accordingly, elevated plasma tHcy appeared in levodopa treated patients with Parkinson’s disease (PD) compared to matched controls and correlated with their total daily levodopa dosage. Treated but not previously untreated PD patients showed augmented tHcy concentrations in plasma [10]. Several lines of evidence suggest that this increase of tHcy contributes to higher incidence of vascular disease, brain atrophy, peripheral sural neurodegeneration, dementia or all of them in the long run in PD patients [8, 13]. One therapeutic approach for tHcy reduction is vitamine supplementation, since folic acid and cobalamine catalyse and enhance metabolism of tHcy to MET. A further therapeutic alternative is inhibition of COMT on a regular basis, when levodopa/DDI treatment is performed. On the one hand, COMT inhibitors increase the bioavalibility of levodopa, but on the other hand, combination of levodopa/DDI with COMT inhibitors reduces O-methylation of levodopa and thus decreases homocysteine levels [12]. We performed a trial with the recently relaunched, also centrally acting, COMT inhibitor tolcapone (3,4-dihydroxy-4′-methyl-5-nitrobenzophenone) to investigate whether tolcapone application reduces SAH- and tHcy synthesis in treated PD patients.
Materials and methods
Subjects
Thirteen PD patients [6 men, 7 women, age: 65.23±10.26 (mean±SD) range: 48–83 years, Hoehn and Yahr stage: 2.89±1.08, range I-IV, oral levodopa intake: 457.69±272.22, range 125–900 mg, benserazide: n=11, carbidopa: n=2] participated in this study. They were on a stable dopaminergic substitution therapy, which mainly consisted of levodopa/DDI and one dopamine agonist. No participant was previously exposed to neuroleptic drugs or any treatment known to affect the dopaminergic system. Exclusion criteria were clinical signs of dementia, any electrophysiological or morphological evidence (cranial CT or MRI scan) of additional CNS pathology exceeding PD. All subjects fulfilled clinical diagnostic UK Brain bank criteria for PD. We excluded PD patients with metabolic disturbances, like diabetes mellitus, hypertension, reduced levels of vitamin B6, cobalamin and/or folic acid or neurological diseases other than PD. We did not determine the frequency of the MTHFR C677T genotype in our PD patients. This would have improved the validity of this trial.
Design
Each participant fasted and was off any medication for at least 8 h before taking of blood samples in the morning on day 1 and day 3. Thus, we tried to avoid an impact of acute levodopa/DDI intake. We added 100 mg tolcapone t.i.d. to the antiparkinsonian drug regimen on day 1. The other concomitant pharmacotherapy remained stable. Adverse effects, such as nausea or dyskinesia, did not appear.
Blood samples
Venous puncture was performed 20 min before the baseline investigation, to enable stable conditions and 3 ml of blood was drawn with a separate syringe and discarded before taking each 10-ml specimen. Blood samples were collected in EDTA-test tubes containing 100 μl of 0.5% sodium disulfite solution. The plasma obtained from rapid centrifugation was immediately frozen at −80°C until analysis within 14 days. Reversed-phase high performance liquid chromatography (HPLC) was employed in combination with electrochemical detection for the measurement of levodopa levels in plasma, which was diluted with a factor of 1:1.95 before assessment. 3-OMD-, SAH-, SAM-, tHcy- plasma levels were determined by HPLC [11].
Statistics
Non-parametric Wilcoxon’s signed rank test was used for comparisons. Level of significance was p<0.05.
Ethics
Informed consent was obtained from each subject. This study was approved by the local ethical commitee.
Results
Plasma levels of SAH [day 1: 48.32±22.52, 23.92–98.25 (mean±SD, range; μmol/l); day 3: 37.72±15.84, 23.4–61.89; Fig. 1; p=0.01] and tHcy (day 1: 13.88±5.62, 7.63–24.81; day 3: 11.38±4.44, 5.98–20.45; Fig. 2; p=0.04) significantly reduced. Plasma levels of levodopa did not significantly (day 1: 96.5±104.14, 5.22–365.2; day 3: 196.2±165.65, 13.1–467.9; p=0.17) increase, whereas 3-OMD concentrations significantly [day 1: 2452.49±1937.92, 654.1–6924.9; day 3: 998.23±862.95, 313.3–2,922.1 (ηg/ml); p=0.0002] reduced after additional tolcapone intake. There was no significant change of SAM plasma levels [day 1: 64.34±19.34, 35.26–99.73; day 3: 60.31±19.85, 29.66–97.13 (ηg/ml); p=0.22].
Discussion
Addition of tolcapone to a stable antiparkinsonian drug regime reduced both SAH and tHcy plasma levels. Thus, we provide further evidence in our prospective pivotal trial, that COMT inhibition lowers tHcy synthesis. To date, this was only shown with the peripherally acting COMT inhibitor entacapone. There are two other prospective studies published on this issue, using entacapone as the COMT inhibitor, in the USA. These studies may have been underpowered because of folate supplementation in the American diet, leading to a milder increase in homocysteine than expected [14, 16]. Observational European non-prospective studies showed lower tHcy levels in entacapone treated patients [4, 18, 19]. Thus, folate supplementation in the North American diet may also explain the heterogeneity of results. Our trial additionally measured SAH, SAM and levodopa levels in plasma in contrast to the others, and we used tolcapone, which has a stronger COMT inhibiting effect than entacapone [15]. Our sample size is quite small and we found three non-responders in terms of homocysteine decrease. From this point of view, determination of the frequency of the MTHFR C677T genotype would have been useful, but we were not able to perform this due to technical reasons.
Since tolcapone also acts centrally [1, 17], our outcomes hypothetically indicate that tolcapone may also reduce tHcys levels within the brain and thus the associated neurotoxic effects on neurons, as shown in sural peripheral nerves and neuronal cell cultures, in the long term [2, 3, 5, 8]. Thus, our results indicate, that central COMT inhibition may hypothetically be neuroprotective in the long term due to a reduction of putative levodopa induced centrally elevated homocysteine levels. Moreover administration of tolcapone in combination with central blocking of monoaminooxidase-B (MAO-B) with, i.e., selegiline or rasagiline may reduce free radical synthesis, which is enhanced by central metabolism of dopamine via the monoaminooxidase-B [9]. However, this combination may also have unforeseen consequences, since COMT has a broad detoxification potential within the brain. Thus, central COMT- and MAO-B inhibition may also force central dopamine metabolism further down the methylation path to N-methylation. This may result in an increased N-methylation of tetrahydroisoquinolines and other dopamine derivatives to PD symptoms, causing neurotoxic N-methyl-4-phenylpyridine-like substrates and a corresponding higher SAM consumption in the long term. SAM is the methyl donor for nicotinamide N-methylation and most other important methylation reactions, including that of DNA and RNA [6]. There was no significant change of SAM plasma concentrations in our trial, but SAM decrease is known in levodopa/DDI-treated PD patients [11].
A significant reduction of 3-OMD appeared despite its long half life in plasma on day 3, which demonstrates the potency of COMT inhibition by tolcapone. We found no significant increase of levodopa concentrations probably due to their short half life in plasma.
In conclusion, our results suggest that tolcapone may also possess, beside its proven, occasional, hepatotoxic potency, beneficial effects via decrease of SAH and tHcy. This may hypothetically reduce homocysteine mediated progress of neuronal degeneration and the risk for onset of dementia, vascular disease and polyneuropathy in levodopa-treated PD patients in the long term [8, 11, 13].
References
Ceravolo R, Piccini P, Bailey DL, Jorga KM, Bryson H, Brooks DJ (2002) 18F-dopa PET evidence that tolcapone acts as a central COMT inhibitor in Parkinson’s disease. Synapse 43:201–207
Huang G, Dragan M, Freeman D, Wilson JX (2005) Activation of catechol-O-methyltransferase in astrocytes stimulates homocysteine synthesis and export to neurons. Glia 51:47–55
Isobe C, Murata T, Sato C, Terayama Y (2005) Increase of total homocysteine concentration in cerebrospinal fluid in patients with Alzheimer’s disease and Parkinson’s disease. Life Sci 77:1836–1843
Lamberti P, Zoccolella S, Iliceto G, Armenise E, Fraddosio A, de Mari M et al (2005) Effects of levodopa and COMT inhibitors on plasma homocysteine in Parkinson’s disease patients. Mov Disord 20:69–72
Lee ES, Chen H, Soliman KF, Charlton CG (2005) Effects of homocysteine on the dopaminergic system and behavior in rodents. Neurotoxicology 26:361–371
Matsubara K, Aoyama K, Suno M, Awaya T (2002) N-methylation underlying Parkinson’s disease. Neurotoxicol Teratol 24:593–598
Miller JW, Shukitt-Hale B, Villalobos-Molina R, Nadeau MR, Selhub J, Joseph JA (1997) Effect of L-Dopa and the catechol-O-methyltransferase inhibitor Ro 41- 0960 on sulfur amino acid metabolites in rats. Clin Neuropharmacol 20:55–66
Müller T, Renger K, Kuhn W (2004) Levodopa associated homocysteine increase and sural axonal neurodegeneration. Arch Neurol 61:657–660
Müller T, Kuhn W, Przuntek H (1993) Therapy with central active catechol-O-methyltransferase (COMT)-inhibitors: is addition of monoamine oxidase (MAO)-inhibitors necessary to slow progress of neurodegenerative disorders? J Neural Transm Gen Sect 92:187–195
Müller T, Werne B, Fowler B, Kuhn W (1999) Nigral endothelial dysfunction, homocysteine, and Parkinson’s disease [letter]. Lancet 354:126–127
Müller T, Woitalla D, Hauptmann B, Fowler B, Kuhn W (2001) Decrease of methionine and S-adenosylmethionine and increase of homocysteine in treated patients with Parkinson’s disease. Neurosci Lett 308:54–56
Müller T, Woitalla D, Kuhn W (2003) Benefit of folic acid supplementation in parkinsonian patients treated with levodopa. J Neurol Neurosurg Psychiatry 74:549
Nakaso K, Yasui K, Kowa H, Kusumi M, Ueda K, Yoshimoto Y et al (2003) Hypertrophy of IMC of carotid artery in Parkinson’s disease is associated with L-DOPA, homocysteine, and MTHFR genotype. J Neurol Sci 207:19–23
O’Suilleabhain PE, Bottiglieri T, Dewey RB Jr, Sharma S, Diaz-Arrastia R (2004) Modest increase in plasma homocysteine follows levodopa initiation in Parkinson’s disease. Mov Disord 19:1403–1408
Onofrj M, Thomas A, Iacono D, Di Iorio A, Bonanni L (2001) Switch-over from tolcapone to entacapone in severe Parkinson’s disease patients. Eur Neurol 46:11–16
Ostrem JL, Kang GA, Subramanian I, Guarnieri M, Hubble J, Rabinowicz AL et al (2005) The effect of entacapone on homocysteine levels in Parkinson disease. Neurology 64:1482
Russ H, Müller T, Woitalla D, Rahbar A, Hahn J, Kuhn W (1999) Detection of tolcapone in the cerebrospinal fluid of parkinsonian subjects. Naunyn Schmiedebergs Arch Pharmacol 360:719–720
Valkovic P, Benetin J, Blazicek P, Valkovicova L, Gmitterova K, Kukumberg P (2005) Reduced plasma homocysteine levels in levodopa/entacapone treated Parkinson patients. Parkinsonism Relat Disord 11:253–256
Zoccolella S, Lamberti P, Armenise E, de Mari M, Lamberti SV, Mastronardi R et al (2005) Plasma homocysteine levels in Parkinson’s disease: role of antiparkinsonian medications. Parkinsonism Relat Disord 11:131–133
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Müller, T., Kuhn, W. Tolcapone decreases plasma levels of S-adenosyl-L-homocysteine and homocysteine in treated Parkinson’s disease patients. Eur J Clin Pharmacol 62, 447–450 (2006). https://doi.org/10.1007/s00228-006-0132-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-006-0132-0