Abstract
Objective
Carvedilol is a nonselective β- and α1-receptor antagonist with additional antioxidant properties in vitro. In this study, we assessed the antioxidative potential of carvedilol in cell culture and in antihypertensive doses in healthy men.
Methods
In vitro, human cultured endothelial cells were treated with native low-density lipoprotein (LDL), oxidized LDL or tumor necrosis factor (TNF)α in the absence and in the presence of carvedilol (40 µM); 8-iso-prostaglandin (PG)F2α, as parameter of oxidative stress, was determined in the supernatants. In a double-blind, randomized, cross-over study, 17 healthy men received 25 mg carvedilol b.i.d., 100 mg metoprolol b.i.d. or placebo for 6 days. After each treatment, systemic oxidative stress was assessed by measuring urinary excretion of 8-iso-PGF2α and 2,3-dinor-5,6-dihydro-8-iso-PGF2α, and the plasma concentration of 3-nitrotyrosine by means of gas chromatography-tandem mass spectrometry. In addition, thiobarbituric acid-reactive substances (TBARS) in plasma were assessed using spectrophotometry.
Results
Native LDL and oxidized LDL induced 8-iso-PGF2α production in endothelial cells. Carvedilol significantly reduced this effect (e.g., for oxidized LDL: 2.66±0.22 pg vs 1.46±0.14 pg 8-iso-PGF2α per µg protein, P<0.05). In healthy volunteers, carvedilol and metoprolol markedly decreased blood pressure and heart rate, but had no statistically significant effect on any indicator of oxidative stress measured. Remarkably, a trend toward reduction of urinary isoprostanes and 3-nitrotyrosine in plasma by both active treatments was observed, suggesting a non-specific antioxidative effect by β blockade.
Conclusions
In vitro, the antioxidative potential of carvedilol was confirmed. In healthy men, antihypertensive doses of carvedilol exert no specific inhibition of oxidative stress.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Carvedilol, a vasodilating and nonselective β- and α1-adrenoreceptor antagonist, has been associated with additional antioxidative effects. Carvedilol and its metabolites, especially BM 91.0228, act as antioxidatives in vitro with a potency 30 to 80 times higher than vitamin E or probucol [1]. Carvedilol has been shown to inhibit lipid peroxidation of myocardial cell membranes and thus protects endothelial and vascular smooth muscle cells from free radical-mediated injury [2]. Carvedilol has also been shown to scavenge peroxy radicals in chemical systems in vitro [3]. Studies in humans have suggested that these in vitro findings may translate into antioxidative effects in vivo [4, 5, 6, 7]. However, the methods applied so far to assess generation of free radicals, i.e., measurement of thiobarbituric acid-reactive substances (TBARS), LDL or DNA oxidation, are doubtful [8, 9]. Now, with the discovery of the isoprostanes and 3-nitrotyrosine and the recognition of their usefulness as biomarkers of oxidative stress, more reliable analytical parameters are available to assess oxidative status in vivo [9, 10].
Isoprostanes are prostaglandin (PG)-like compounds that are produced by nonenzymatic free-radical-catalyzed peroxidation of esterified arachidonic acid [9]. Formation of 8-iso-PGF2α, one of the most abundant isoprostanes formed in vivo, was found to be elevated in animals exposed to experimentally induced oxidative stress as well as in certain cardiovascular diseases [11, 12]. 8-Iso-PGF2α and its main metabolite 2,3-dinor-5,6-dihydro-8-iso-PGF2α are excreted into urine, thus providing a non-invasive parameter to assess oxidative stress in vivo [13].
3-Nitrotyrosine, a novel marker of reactive nitrogen species mediated oxidative stress, is formed from the nitration of tyrosine and tyrosine residues in proteins by the strong oxidant peroxynitrite (ONOO−) [10, 14]. Peroxynitrite itself is formed from the diffusion-controlled reaction of nitric oxide (NO) with superoxide (O2 −). 3-Nitrotyrosine occurs physiologically in plasma and is found at increased amounts in atherosclerotic lesions and early fatty streaks [15].
The aim of the present study was to test the putative antioxidative potential of carvedilol and its metabolite BM 91.0228 in cultured human endothelial cells and in vivo in healthy humans by assessment of oxidative stress caused by reactive oxygen species (ROS) as well as reactive nitrogen species (RNS) as reflected by the isoprostanes 8-iso-PGF2α and 2,3-dinor-5,6-dihydro-8-iso-PGF2α, 3-nitrotyrosine, and TBARS.
Methods
In vitro experiments
Preparation of native and oxidized LDL
Native and oxidized low density lipoprotein (LDL and oxLDL, respectively) were prepared as described by Esterbauer et al. [16]. Briefly, LDL was isolated by density-gradient ultracentrifugation of normal human fresh frozen plasma. oxLDL was prepared by incubating native LDL with 2.5 µM CuSO4 at 37°C for 24 h.
Cell culture
The permanent human endothelial cell line, EA·hy 926, which was derived by fusing human vein endothelial cells with the permanent human cell line A549, was generously supplied by Dr. Cora-Jean Edgell, University of North Carolina. Cells were cultured in Dulbecco’s modified Eagle medium (GIBCO BRL) containing 10 ml of HAT supplement (100 µM hypoxanthine, 0.4 µM aminopterine, 16 µM thymidine; GIBCO BRL) and 50 ml of heat-inactivated fetal bovine serum (GIBCO BRL) on plastic ware at 37°C in humidified air with 5% CO2. After dissociation of the endothelial cells with trypsin-EDTA (GIBCO 784BRL), cells were plated into 6-well culture plates (Corning Inc.).
After 3 days, cells were washed with phosphate buffered saline (PBS) before they were incubated with native LDL (2000 mg/l), oxLDL (300 mg/l), tumor necrosis factor α (TNFα; 200 U/ml, Sigma) or PBS as control for 4 h in the presence or absence of 40 μM carvedilol or 40 μM BM 91.0228. Experiments were carried out in triplicate.
In vivo study
Subjects
Seventeen healthy men, mean (±SD) age 24.2±2.5 years (age range 20–27 years) were included in this study. Inclusion criteria were: male, age 18–35 years, healthy, written informed consent. Exclusion criteria were: acute or chronic diseases, intake of legal or illegal drugs or alcohol, smoking, systolic blood pressure at rest below 120 mmHg. Inclusion and exclusion criteria were controlled by clinical history, physical examination, laboratory screening, and 12-lead electrocardiography. The study was approved by the ethics committee of the Medizinische Hochschule Hannover.
Study protocol
The subjects received in a double-blind, randomized, placebo-controlled, cross-over study design either carvedilol 25 mg b.i.d. (12.5 mg b.i.d. on day 1 and day 2), metoprolol 100 mg b.i.d. or placebo b.i.d. for 6 days. The study periods were separated by a wash-out period of 6 days, each. Compliance was controlled by observation of drug intake at 0800 hours in the study center; drug intake at 2000 hours was controlled by telephone. Blood pressure and heart rate were assessed on each day at 0750 hours, i.e., before drug intake.
On day 6 of each medication phase, the subjects collected a 24-h urine for analysis of 8-iso-PGF2α and 2,3-dinor-5,6-dihydro-8-iso-PGF2α. Urine was collected in polypropylene bottles containing 1 mM each of 4-hydroxy-tempo (HTMP) and EDTA as antioxidants. Urine samples were divided into 3×50-ml aliquots and stored at −20°C until analysis. The same day, venous blood was collected at 0755 hours using syringes containing ethylene diamine tetraacetic acid (EDTA) and placed immediately on ice. Blood was centrifuged (800 g, 10 min) within 15 min after collection, and plasma was stored at −78°C until analysis of 3-nitrotyrosine and TBARS.
Biochemical analyses
Isoprostanes and 3-nitrotyrosine
8-Iso-PGF2α, 2,3-dinor-5,6-dihydro-8-iso-PGF2α and 3-nitrotyrosine were quantified by previously described fully validated gas chromatography-tandem mass spectrometry (GC-MS/MS) methods using [2H4]-8-iso-PGF2α (Cayman, Ann Arbor, MI, USA), [18O2]-2,3-dinor-5,6-dihydro-8-iso-PGF2α, and 3-nitro-L-[2H3]tyrosine as internal standards, respectively [17, 18]. Real samples were accompanied by quality control samples as described previously [17, 18]. Urinary excretion rates of 8-iso-PGF2α and 2,3-dinor-5,6-dihydro-8-iso-PGF2α were corrected for excretion of creatinine which was determined spectrophotometrically by the alkaline picric acid reaction with an automatic analyzer (Beckmann 6641, Galway, Ireland).
Aliquots (1.8 ml) of cell supernatants were spiked with 0.5 ng [2H4]-8-iso-PGF2α. Urine samples (5 ml) were spiked with 5 ng of [2H4]-8-iso-PGF2α and 5 ng [18O2]-2,3-dinor-5,6-dihydro-8-iso-PGF2α. Samples were acidified to pH 3.5, analytes were solid-phase extracted on octadecylsilica cartridges and eluted from the columns with ethyl acetate. After solvent evaporation, the pentafluorobenzyl esters were prepared, which were subsequently separated by thin-layer chromatography and further converted to their trimethylsilyl ether derivatives, and analyzed by GC-MS/MS in the selected reaction mode exactly as described elsewhere [17].
Plasma samples (2 ml) were spiked with 6.5 nM 3-nitro-l-[2H3]tyrosine, ultrafiltered by centrifugation at 1500 g using 20-kDa cut-off Centrisart ultrafiltration cartridges (Sartorius, Germany), and aliquots (200 µl) of the ultrafiltrate samples were analyzed by HPLC. Endogenous 3-nitrotyrosine and 3-nitro-l-[2H3]tyrosine eluted in a single HPLC fraction; they were isolated by solid-phase extraction, derivatized to their n-propyl ester-pentafluoropropionyl amide-trimethylsilyl ether derivatives and quantified by GC-MS/MS in the selected reaction monitoring mode as described [18].
Quantification of plasma TBARS
TBARS in plasma were determined by the method of Cordova et al. [19]. To the plasma sample (1 ml) or a 1,1,3,3-tetraethoxypropane (TEP) (Sigma) standard solution were added 100 µl of 2% 2,6-tert-butyl-4-methylphenol (BHT) (Sigma) in ethanol and 1 ml of 20% trichloroacetic acid/0.6 M HCl solution. After centrifugation, a 0.2-ml aliquot of a thiobarbituric acid (TBA) solution (0.12 M TBA/0.26 M Tris buffer, pH 7) was added to the supernatant. The mixture was heated at 95°C for 60 min, then cooled to and stored at 0°C until analysis by spectrophotometry at 532 nm. Reagent blanks, TEP working standard solution, plasma specimens and a quality control specimen were processed in duplicate alongside with real samples.
Protein measurement
Protein concentration in the cell culture was measured by the method of Lowry [20]. Cells were resuspended in 1 ml of distilled water and lysed by rapid freezing and thawing. Cells were collected from culture plates with a cell scraper. Cells and cell lysates were centrifuged and supernatants were used for Lowry assay.
Statistical analyses
Data were analyzed with SPSS for Windows (release 11.0.1., SPSS Inc., Chicago, Illinois). All data are given as the mean ±SD. Statistical significance of differences for the in vitro experiments was determined by analysis of variance (ANOVA) followed by Scheffé’s F-test. For the in vivo study, student’s paired two-tailed t-test was used. Spearman’s correlation coefficients were calculated between hemodynamic parameters (diastolic and systolic blood pressure, heart rate) and the markers of oxidative stress. Carry over effects were analyzed as described by Hills and Armitage [21]. A P value <0.05 was considered significant.
Results
In vitro study
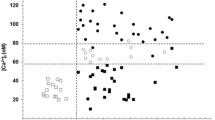
Incubation of cultured human endothelial cells with native LDL or oxLDL resulted in elevated concentrations of 8-iso-PGF2α in the supernatant (Fig. 1). The TNFα effect on 8-iso-PGF2α was not significant. Carvedilol and BM 91.0228 were equally effective in inhibiting 8-iso-PGF2α generation induced by LDL, oxLDL or TNFα (Fig. 1).
Concentration of 8-iso-PGF2α in the supernatant of human endothelial cell culture after incubation with phosphate buffered saline, low-density lipoprotein (LDL), oxidized LDL (Ox), or tumor necrosis factor α (TNF) for 4 h without (Contr) or with 40 μM carvedilol (Carv) or 40 μM BM 91.0228 (BM). Mean±SD of n=3, each. **P<0.001 vs PBS Contr; †P<0.05 and ††P<0.001 vs Contr of the same incubation group (LDL Contr, Ox Contr and TNF Contr, respectively)
In vivo study
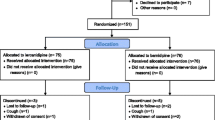
Seventeen volunteers participated in the study, 15 subjects completed the study in accordance with the study protocol. Two volunteers were withdrawn; one of them developed orthostatic hypotension during carvedilol treatment and wished to discontinue; the other volunteer had a common cold. No serious adverse events were observed.
Intake of carvedilol and metoprolol resulted in a statistically significant reduction in systolic and diastolic blood pressure as well as heart rate compared with placebo, proving compliance of the volunteers (Table 1).
Isoprostanes
Neither carvedilol nor metoprolol exerted a statistically significant effect on urinary excretion of 8-iso-PGF2α and 2,3-dinor-5,6-dihydro-8-iso-PGF2α compared with placebo (Table 2).
3-Nitrotyrosine
Plasma concentrations of 3-nitrotyrosine did not change significantly following carvedilol (0.87±0.31 pmol/ml) or metoprolol (0.91±0.50 pmol/ml) compared with placebo (1.10±0.56 pmol/ml).
TBARS
Plasma concentration of TBARS was 0.64±0.17 µM for placebo and did not change after administration of carvedilol (0.66±0.20 µM) or metoprolol (0.66±0.16 µM).
Correlations
Urinary excretion of 8-iso-PGF2α and plasma concentrations of TBARS were positively correlated with diastolic blood pressure (r=0.358, P=0.016 and r=0.345, P=0.020, respectively). No significant correlation was found between urinary 2,3-dinor-5,6-dihydro-8-iso-PGF2α or plasma 3-nitrotyrosine and hemodynamic parameters.
Quality control
Endogenous urinary 8-iso-PGF2α (439±15 pg/ml) and 2,3-dinor-5,6-dihydro-8-iso-PGF2α (501±42 pg/ml), plasma 3-nitrotyrosine (1.2±0.3 nM) and plasma TBARS (0.63±0.05 μmol/l) were determined in the quality control samples with an accuracy of 99.8±3.4%, 100.0±8.4%, 95.6±10.5% and 100.0±8.1%, respectively. The levels of these biomarkers measured in the present study are of the same order as those previously measured in normal healthy humans [17, 18, 19]. This confirmation and the high accuracy by which concomitantly processed quality control samples were determined in the present study underline the validity of the results observed for the isoprostanes, 3-nitrotyrosine and TBARS.
No carry-over effects were detected for any of the study parameters.
Discussion
In our in vitro study, LDL and oxLDL augmented the generation of 8-iso-PGF2α in cultured human endothelial cells. Carvedilol and its metabolite BM 91.0228 blunted this response suggesting that these agents possess antioxidative activity. The carvedilol metabolite BM 91.0228 is characterized by a lower adrenergic receptor affinity, but by a threefold higher antioxidative potency than carvedilol [1]. Thus, the results for BM 91.0228 suggest that the antioxidative activity in vitro is not related to β-receptor blocking properties. Our findings in vitro confirm previous in vitro studies using TBARS, the activity of sarcoplasmic reticulum Ca2+-ATPase or apoptosis in endothelial cells as parameters for oxidative stress [22, 23, 24, 25]. In our and the other studies mentioned, carvedilol was tested in vitro in concentrations ranging from 2.6–40 µM [22, 23, 24, 25]. The plasma peak concentration of carvedilol in therapeutic dosage has been reported to be one to two orders of magnitude smaller, i.e., 0.16–0.29 µM [26]. Only one ex vivo study in isolated rabbit hearts showed anti-oxidant activity of 0.1 µM carvedilol, a concentration that led neither to β-blockage nor negative inotropism [27].
To examine the relevance of the in vitro findings in vivo, we investigated the antioxidative effects of carvedilol at standard antihypertensive dosage in healthy volunteers. While treatment with carvedilol 25 mg b.i.d. for 6 days reduced heart rate and blood pressure, it did not decrease significantly systemic oxidative stress in healthy young men. However, there was a trend toward reduction of urinary isoprostane excretion and plasma 3-nitrotyrosine concentrations by carvedilol. Remarkably, also for metoprolol a trend for a reduced excretion of 2,3-dinor-5,6-dihydro-8-iso-PGF2α and plasma 3-nitrotyrosine concentrations was observed. Retrospective sample size calculation based on the actual data revealed that 262, 129 and 48 participating volunteers would have been needed to show a significant difference of 8-iso-PGF2α, 2,3-dinor-5,6-dihydro-8-iso-PGF2α and 3-nitrotyrosine, respectively. For the comparison, metoprolol versus placebo, the corresponding values are 3524 (8-iso-PGF2α), 33 (2,3-dinor-5,6-dihydro-8-iso-PGF2α) and 72 (3-nitrotyrosine).
A similar observation was reported for TBARS, which were too insensitive to show any effect in our study, by Kukin et al. in patients with stable heart failure treated with carvedilol or metoprolol for 6 months [28]. In the study by Kukin and co-workers, TBARS levels were elevated at base line and declined during therapy in both treatment groups in parallel to clinical improvement [28]. Since TBARS levels are correlated to NYHA functional class [29], the authors explained the reduction of oxidative stress by clinical improvement as therapeutic effect of β blockers [28]. In our study, the reduced heart work by carvedilol and metoprolol may account for the trend toward reduction of oxidative stress. This speculation is supported by the positive correlation between 8-iso-PGF2α and diastolic blood pressure as well as TBARS and diastolic blood pressure in our study.
In contrast to the above-described findings, two other in vivo studies showed a specific antioxidative effect of carvedilol. In patients with chronic heart failure, carvedilol 25 mg b.i.d. significantly reduced erythrocyte superoxide dismutase and glutathione peroxidase activity after 12 weeks of therapy [7]; likewise, in patients with non-insulin-dependent diabetes mellitus and hypertension, TBARS were reduced by a 24-week therapy with carvedilol [5]. The active comparators in these studies, metoprolol and atenolol, respectively, had no effect on the parameters of oxidative stress measured. Two further studies demonstrated an inhibited generation of reactive oxygen species by leukocytes and reduced LDL oxidation in patients with hypertension and healthy men, respectively, treated with carvedilol [4, 6]. Remarkably, the antioxidative effects in the latter study by Dandona et al. were observed at the low dose of 3.125 mg carvedilol b.i.d. given to eight healthy volunteers for 7 days [6]. However, in both studies no β blockers as active controls were employed.
The discrepancies may be explained by use of different assays to determine oxidative stress and different study populations. In this context, the limitations of our study are noteworthy. First, we studied the antioxidative effects of carvedilol in healthy, non-smoking men, who have no increased oxidative stress. Nevertheless, as consequence of aerobic life, even in healthy volunteers, the amount of oxidative stress is considerable [30, 31]. While this approach has the advantage of a well-controlled study population without concomitant diseases or medications, the effect of an antioxidative agent is more difficult to detect than under conditions of enhanced oxidative stress. However, our study was powered to detect meaningful differences of 23% (8-iso-PGF2α), 22% (2,3-dinor-5,6-dihydro-8-iso-PGF2α) and 22% (3-nitrotyrosine) between placebo and carvedilol (for n=15, alpha = 0.05, beta 0.8). In a previous study in 12 healthy volunteers, we were able to show a significant 18% decrease of 8-iso-PGF2α by 7 days of treatment with 5 mg/day nebivolol, a selective β1-receptor antagonist also associated with antioxidative properties [32]. Second, treatment duration was only 6 days. However, given the rapid onset of the antioxidative actions in vitro, the pronounced hemodynamic effects after 6 days of treatment and the findings of Dandona et al. (see above) [6], the duration of treatment should have been sufficiently long to show an antioxidative effect of carvedilol.
Altogether, our study adds a negative result to the already conflicting data on the antioxidative properties of carvedilol in vivo. However, it must be stressed that our findings in healthy volunteers do not rule out antioxidative properties of carvedilol in patients with enhanced oxidative stress, e.g., patients with chronic heart failure.
Whether such specific antioxidative actions of a drug are related to clinical benefit is a further question. Meta-analysis of the effects of vitamin E supplementation for 4–6 years on cardiovascular events indicates little, if any, benefit [29]. For β carotene, adverse effects on cardiovascular disease and the incidence of lung cancer were observed repeatedly [33, 34]. As far as carvedilol is concerned, even the maximum daily dosage failed in the EUROCARE trial to cause any reduction in re-stenosis after atherectomy, a complication in which oxidative stress has been invoked [35].
In conclusion, our results show that carvedilol exerts antioxidative actions in vitro at supra-therapeutic concentrations, whereas in vivo in healthy men carvedilol in standard antihypertensive dosage has no specific effects on systemic oxidative stress on the basis of reduction of urinary 8-iso-PGF2α and 2,3-dinor-5,6-dihydro-8-iso-PGF2α, circulating 3-nitrotyrosine and TBARS. Further studies investigating the effects of carvedilol on isoprostanes and 3-nitrotyrosine in patients with atherosclerotic cardiovascular disease, i.e., high oxidative stress, are encouraged.
References
Yue T-L, McKenns PJ, Lysko PG, Gu JL, Lysko KA, Ruffolo RR, Feuerstein GZ (1994) SB 211475, a metabolite of carvedilol, a novel antihypertensive agent, is a potent antioxidant. Eur J Pharmacol 251:237–243
Feuerstein G, Shusterman NH, Ruffolo RR (1997) Carvedilol update IV: prevention of oxidative stress, cardiac remodeling and progression of congestive cardiac failure. Drugs Today 33:453–473
Aruoma OI (1997) Scavenging of hypochlorous acid by carvedilol and ebselen in vitro. Gen Pharmacol 28:269–272
Maggi E, Marchesi E, Covini D, Negro C, Perani G, Bellomo G (1996) Protective effects of carvedilol, a vasodilating beta-adrenoceptor blocker, against in vivo low density lipoprotein oxidation in essential hypertension. J Cardiovasc Pharmacol 27:532–538
Giugliano D, Acampora R, Marfella R, De Rosa N, Ziccardi P, Ragone R, De Angelis L, D’Onofrio F (1997) Metabolic and cardiovascular effects of carvedilol and atenolol in non-insulin-dependent diabetes mellitus and hypertension. A randomized, controlled trial. Ann Intern Med 126:955–959
Dandona P, Karne R, Ghanim H, Hamouda W, Aljada A, Magsino CH Jr (2000) Carvedilol inhibits reactive oxygen species generation by leukocytes and oxidative damage to amino acids. Circulation 101:122–124
Arumanayagam M, Chan S, Tong S, Sanderson JE (2001) Antioxidant properties of carvedilol and metoprolol in heart failure: a double-blind randomized controlled trial. J Cardiovasc Pharmacol 37:48–54
Gutteridge JMC, Halliwell B (1990) The measurement and mechanism of lipid peroxidation in biological systems. Trends Biochem Sci 15:129–135
Roberts LJ, Morrow JD (2002) Products of the isoprostane pathway: unique bioactive compounds and markers of lipid peroxidation. Cell Mol Life Sci 59:808–820
Halliwell B (1997) What nitrates tyrosine? Is nitrotyrosine specific as a biomarker of peroxynitrite in vivo? FEBS Lett 411:157–160
Davi G, Alessaridrini P, Mezetti A (1997) In vivo formation of 8-epi-prostaglandin F2 alpha is increased in hypercholesterolemia. Arterioscler Thromb Vasc Biol 17:3230–3235
Gniwotta C, Morrow JD, Roberts LJ, Kuhn H (1997) Prostaglandin F2-like compounds, F2-isoprostanes, are present in increased amounts in human atherosclerotic lesions. Arterioscler Thromb Vasc Biol 17:3236–3241
Roberts LJ, Moore KP, Zackert WE, Oates JA, Morrow JD (1996) Identification of the major urinary metabolite of the F2-isoprostane 8-iso-prostaglandine F2α in humans. J Biol Chem 271:20617–20620
Beckman JS, Ischiropoulos H, Zhu L, van der Woerd M, Smith C, Chen J, Harrison J, Martin JC, Tsai M (1992) Kinetics of superoxide dismutase and iron catalyzed nitration of phenolics by peroxynitrite. Arch Biochem Biophys 298:438–45
Beckmann JS, Ye YZ, Anderson PG, Chen J, Accavitti MA, Tarpey MM, White CR (1994) Extensive nitration of protein tyrosines in human atherosclerosis detected by immunohistochemistry. Biol Chem Hoppe Seyler 375:81–88
Esterbauer H, Striegl G, Puhl H, Rotheneder M (1989) Continuous monitoring of in vitro oxidation of human low density lipoprotein. Free Radic Res Commun 6:67–75
Schwedhelm E, Tsikas D, Durand T, Gutzki F-M, Guy A, Rossi J-C, Frölich JC (2000) Tandem mass spectrometric quantification of 8-iso-prostaglandin F2α and its metabolite 2,3-dinor-5,6-dihydro-8-iso-prostaglandin F2α in human urine. J Chromatogr B 744:99–112
Schwedhelm E, Tsikas D, Gutzki F-M, Frölich JC (1999) Gas chromatographic-tandem mass spectrometric quantification of free 3-nitrotyrosine in human plasma at the basal state. Anal Biochem 276:195–203
Cordova C, Musca A, Violi F, Alessandri C, Ghiselli A, Luliano L, Balsano F (1984) Influence of vitamin E on plasma malondialdehyde-like material in man. Thromb Haemostas 51:347–348
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with folin phenol reagent. J Biol Chem 193:265–275
Hills M, Armitage P (1979) The two-period cross-over clinical trial. Br J Clin Pharmacol 8:7-20
Lysko PG, Webb CL, Gu JL, Ohlstein EH, Ruffolo RR Jr, Yue TL (2000) A comparison of carvedilol and metoprolol antioxidant activities in vitro. J Cardiovasc Pharmacol 36:277–281
Rössig L, Haendeler J, Mallat Z, Hugel B, Freyssinet J-M, Tedgui A, Dimmeler S, Zeiher AM (2000) Congestive heart failure induces endothelial cell apoptosis: protective role of carvedilol. J Am Coll Cardiol 36:2081–2089
Flesch M, Maack C, Cremers B, Baumer AT, Sudkamp M, Bohm M (1999) Effect of β-blockers on free radical-induced cardiac contractile dysfunction. Circulation 100:346–353
Lopez BL, Christopher TA, Yue TL, Ruffolo R, Feuerstein GZ, Ma XL (1995) Carvedilol, a new beta-adrenoreceptor blocker antihypertensive drug, protects against free-radical induced endothelial dysfunction. Pharmacology 51:165–173
Louis WJ, McNeil JJ, Workman BS (1987) A pharmacokinetic study of carvedilol in elderly subjects: preliminary report. J Cardiovasc Pharmacol 10:89–93
Cargnoni A, Ceconi C, Bernocchi P, Boraso A, Parrinello G, Curello S, Ferrari R (2000) Reduction of oxidative stress by carvedilol: role in maintenance of ischaemic myocardium viability. Cardiovasc Res 47:556–566
Kukin ML, Kalman J, Charney RH, Levy DK, Buchholz-Varley C, Ocampo O, Eng C (1999) Prospective, randomized comparison of effect of long-term treatment with metoprolol or carvedilol on symptoms, exercise, ejection fraction, and oxidative stress in heart failure. Circulation 99:2645–2651
The Heart Outcomes Prevention Evaluation Study Investigators (2000) Vitamin E supplementation and cardiovascular events in high-risk patients. N Engl J Med 342:154–160
Minuz P, Patrignani P, Gaino S, Degan M, Menapace L, Tommasoli R, Seta F, Capone ML, Tacconelli S, Palatresi S, Bencini C, Del Vecchio C, Mansueto G, Arosio E, Santonastaso CL, Lechi A, Morganti A, Patrono C (2002) Increased oxidative stress and platelet activation in patients with hypertension and renovascular disease. Circulation 106:2800–2805
Tsikas D, Schwedhelm E, Suchy MT, Niemann J, Gutzki FM, Erpenbeck VJ, Hohlfeld JM, Surdacki A, Frölich JC (2003) Divergence in urinary 8-iso-PGF2α (iPF2α-III, 15-F2 t-IsoP) levels from gas chromatography-tandem mass spectrometry quantification after thin-layer chromatography and immunoaffinity column chromatography reveals heterogeneity of 8-iso-PGF2α. Possible methodological, mechanistic and clinical implications. J Chromatogr B 794:237–255
Troost R, Schwedhelm E, Rojczyk S, Tsikas D, Frölich JC (2000) Nebivolol decreases systemic oxidative stress in healthy volunteers. Br J Clin Pharmacol 50:377–379
Omenn GS, Goodman GE, Thornquist MD, Balmes J, Cullen MR, Glass A, Keogh JP, Meyskens FL, Valanis B, Williams JH, Barnhart S, Hammar S (1996) Effects of a combination of beta carotene and vitamin A on lung cancer and cardiovascular disease. N Engl J Med 334:1150–1155
Rapola JM, Virtamo J, Ripatti S, Huttunen JK, Albanes D, Taylor PR, Heinonen OP (1997) Randomised trial of alpha-tocopherol and beta-carotene supplements on incidence of major coronary events in men with previous myocardial infarction. Lancet 349: 1715–1720
Serruys PW, Foley DP, Höfling B, Puel J, Glogar HD, Seabra-Gomes R, Goicolea J, Coste P, Rutsch W, Katus H, Bonnier H, Wijns W, Betriu A, Hauf-Zachariou U, van Swijndregt EM, Melkert R, Simon R (2000) Carvedilol for prevention of restenosis after directional coronary atherectomy: final results of the European carvedilol atherectomy restenosis (EUROCARE) trial. Circulation 101:1512–1522
Acknowledgements
We thank Mrs. B. Schubert and Mrs. I. Fuchs for their skillful technical assistance. The authors are indebted to L. Hoy, Ph.D., Department of Biometrics, Medizinische Hochschule Hannover, for statistical advice. This study was supported by an institutional grant by Hoffmann-La Roche, Grenzach-Wyhlen, Germany. The experiments in the study complied with the current laws of Germany.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fahlbusch, S.A., Tsikas, D., Mehls, C. et al. Effects of carvedilol on oxidative stress in human endothelial cells and healthy volunteers. Eur J Clin Pharmacol 60, 83–88 (2004). https://doi.org/10.1007/s00228-004-0729-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-004-0729-0