Abstract
We investigated ontogenetic, temporal and spatial patterns in the composition and size of prey in the diet of crested terns, Sterna bergii. Diet analyses indicated that crested terns are a generalist predator on surface-schooling clupeids (Australian anchovy Engraulis australis, sardine Sardinops sagax and blue sprat Spratelloides robustus), Degens leatherjacket Thamnaconus degeni, southern sea garfish Hyporhamphus melanochir, Australian herring Arripis georgianus, slender bullseye Parapriacanthus elongatus and barracouta Thyrsites atun. Ontogenetic differences in prey size indicated that adults are constrained in their foraging behaviour during the early chick-provisioning period by the need to self feed and select smaller prey that can be ingested by their chicks. Chicks consumed significantly higher proportions of clupeids than adults, which consumed mainly Degens leatherjackets and barracouta, suggesting that adults may select higher quality prey for their chicks compared to what they consume themselves. Spatial differences in prey composition were driven by differing proportions of sardine, Australian anchovy and Degens leatherjacket and could reflect local differences in the abundances of these prey. The size of prey taxa consumed by adults also reflected a North–South gradient in prey size. The large component of juvenile sardine in the diet of crested terns suggests future dietary measures may inform fisheries managers about changes in local juvenile sardine abundance. These data could assist in highlighting any fishery-related decreases in sardine recruitment and help ensure commercial fishing practices address principals of Ecologically Sustainable Development developed for Australian fisheries.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Research on marine predators has the potential to underpin ecosystem-based management because being at the top of marine food webs, they reflect changes in marine system processes through measures of their diet, demography, behaviour or physiology. Particular focus has been placed on using seabirds as indicators because they are easily accessed, and several studies have linked seabird data to ecosystem health and changes in prey abundance (Nettleship et al. 1982; Cairns 1987; Montevecchi 1993; ICES 2000; Schreiber and Burger 2002). However, seabird responses to changes in ecosystem processes and prey availability are varied, and if species are to be useful as indicators of change a prior understanding is required of the factors that constrain seabird behaviour and influence diet and seabird responses (Crawford 2004; Hall and Mainprize 2004; Boyd et al. 2006).
Seabird foraging behaviour and reproduction are constrained during breeding by the availability of prey within their foraging range, their reproductive state, and innate characters of reproductive phenology and physiology (Cairns 1987; Burger and Piatt 1990; Montevecchi 1993). For instance, adults that are provisioning chicks face different energetic constraints to adults that are only self-feeding. During provisioning, adults must balance their own energetic requirements with that of their chicks by adjusting foraging behaviour. This dichotomy represents a trade-off between somatic and reproductive investment (Swihart and Johnson 1986). Prey favoured while self-feeding may be available at distances and travel times that exceed the fasting abilities of chicks during the breeding season (Weimerskirch 1998; Barrett et al. 2007). Also, single-prey loaders such as terns or guillemots, which must first provision themselves before locating and returning single-prey items of a suitable size to their chicks, have different foraging constraints to species such as penguins that regurgitate macerated prey to their chicks (Furness and Tasker 2000; Stienen et al. 2000). For single-prey loaders, central-place foraging theory predicts that adults should maximise their effort provisioning per unit time of foraging by providing as large or energetically rich prey as possible (Orians and Pearson 1979; Davoren and Burger 1999). However, adult foraging behaviour during chick provisioning is also regulated by what chicks can physically ingest and chick energetic requirements as they grow. Consequently, chick and adult diets may differ as a function of prey selection by adults at different stages of breeding (Hulsman et al. 1989; Ramos et al. 1998; Shealer 1998). Despite the different intrinsic constraints imposed on seabirds by species-specific foraging behaviour and reproduction over the course of the breeding season, only a few studies have simultaneously examined the diets of chicks and adults (Cairns 1984; Vermeer et al. 1987; Mahon et al. 1992; Leopold et al. 1996; Davoren and Burger 1999; Barrett et al. 2007).
Seabird dietary studies provide a profile of prey composition, size, mass, quality and availability, thereby highlighting the key trophic linkages within a species’ foraging range and differences between places and periods (Cairns 1987; Montevecchi 1993; Montevecchi and Myers 1995, 1996; Jaquemet et al. 2008). Diet data also provide a means of assessing links between seabird behavioural responses and the environment. Several studies have linked seabird diet information to oceanographic indices and estimates of prey abundance, and these data have sometimes been used to inform management decisions (Hislop and Harris 1985; Montevecchi et al. 1987; Monaghan et al. 1989; Montevecchi 1993; Montevecchi and Myers 1995; Lewis et al. 2001; ICES 2002; Frederiksen et al. 2004; Velarde et al. 2004). For instance, in the North Sea, the diet and breeding success of black-legged kittiwakes Rissa tridactyla is highly correlated with the abundance of sandeels Ammodytes marinus. If kittiwake production falls below 0.5 for 3 consecutive years, commercial sand eel fishing is ceased (ICES 2002).
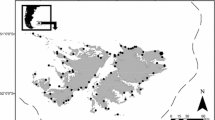
Crested terns Sterna bergii, are a small piscivorous plunge-diving seabird distributed throughout the Indo-Pacific and on all coasts of Australia. The total number of breeding colonies in South Australia is unknown but at least 12 colonies have been recorded in Gulf St Vincent and Spencer Gulf (Fig. 1). A previous study of the diet, demographic and morphological characteristics of a crested tern Sterna bergii population in South Australia indicated that this species may be sensitive to changes in sardine abundance (McLeay et al. 2009). Other studies have described the diets of crested tern chicks (Walter et al. 1987; Hulsman et al. 1989; Smith 1993; Chiaradia et al. 2002) or adults (Walter 1984; Blaber and Wassenberg 1989; Surman and Wooler 2003) but not the ontogenetic, temporal or spatial patterns in diet. These data are required to understand how species-specific intrinsic constraints influence the types and sizes of prey consumed, and mediate demographic, behavioural or physiological responses.
We investigated the diet of crested terns at four colonies in gulf waters of South Australia during their breeding seasons between 2003 and 2008. We hypothesise that the diet of crested terns will exhibit ontogenetic differences as a function of a trade-off between the physical ingestion capabilities of chicks, and adult foraging behaviour that has evolved to maximise the amount of energy provided to chicks per foraging trip. We specifically ask (1) What are the key prey in the diets of chick and adult crested terns? (2) Are there ontogenetic differences in prey composition and size? and (3) Are there seasonal and spatial patterns in prey composition and size? We address these questions by quantifying the level of ontogenetic, spatial, temporal and variation in the prey composition and size of crested tern diets, and discuss how future information collected from crested terns may inform marine resource management.
Methods
Study sites
This study was carried out at four locations in South Australia during the breeding seasons (between November and February) in the years 2003/2004 to 2007/2008. We refer to seasons as belonging to the year that sampling or breeding began, respectively (e.g. 2005/2006 = 2005) (Fig. 1). Troubridge Island is a sand island approximately 260 ha in southern Gulf St Vincent (35°4′S, 137°49′33″E). The crested tern colony at Troubridge Island is the largest in South Australia, having approximately 3,000 breeding pairs (McLeay unpublished data). Goose Island (34°27′S, 137°22′E), Lipson Island (34°15′S, 136°15′E) and Rocky Island (34°29′S, 37°25′E) are smaller islands (3–35 ha) in southern Spencer Gulf (Fig. 1). The colonies at these islands are generally smaller, numbering approximately 900 breeding pairs at Goose Island, 2,000 at Lipson Island and 1,300 at Rocky Island, respectively (McLeay, unpublished data).
Diet sampling
When adults and chicks were caught they regurgitated spontaneously. We collected regurgitates from crested tern chicks aged 0–5 weeks and adults that were incubating eggs or provisioning chicks. To minimise disturbance to the colony, adults and chicks were caught by moving from one end of the colony to the other which ensured no bird was caught more than once per visit. Samples were placed in a plastic bag and frozen before being transported to a laboratory and sorted, where individual prey were separated and identified to the lowest taxonomic group possible. Individual prey that were whole were weighed (wet) to ±0.01 g using a digital balance and length was measured to the nearest ±1.0 mm. Individual prey were sometimes too digested to enable accurate measurements of mass and size. Prey mass and size were estimated using regression equations obtained from published information and from regressions constructed in this study for the relationships between, caudal fin-ray, otolith or body/mantle length measurements, and fish/cephalopod mass. Data for regressions constructed in this study were obtained by measuring whole individual prey in diet samples and from fish collected during fishery research surveys (Table 1). Fish prey identified in regurgitates often only consisted of the posterior body and caudal fin. Consequently, species level identification was not possible for some individuals from the families Clupeidae and Engraulidae and so they were grouped into a distinct group. To determine the most important prey, we allocated prey from this group, which could have been either sardine Sardinops sagax or Australian anchovy Engraulis australis, proportionally to the groups identified specifically as sardine or Australian anchovy. For individual prey items that could be identified but not measured, the mean mass of individual cephalopod/fish species consumed by chicks/adults in the same location/year was used in biomass reconstructions. The unidentified prey biomass proportion was based on the minimum number of unidentified individuals, multiplied by the average mass of prey consumed by either chicks or adults in the same year at the same location.
Data analysis
We apportioned our diet data from each colony to one of four groups based on the time of the breeding season in which regurgitates were collected:
-
1.
Incubation: samples collected from adults during incubation.
-
2.
Early provisioning: samples collected from chicks and adults 1–10 days after hatching. Chicks weighed 30–108 g during this time (McLeay, unpublished data).
-
3.
Mid provisioning: samples collected 11 and 25 days post-hatching when chicks weighed 108–207 g.
-
4.
Late provisioning: samples collected >25 days post-hatching when chicks weighed >207 g.
The relative importance of the prey composition of chick and adult diets was assessed by estimating the percentage biomass contribution of each prey taxon. Ontogenetic, seasonal and spatial differences in the species composition of chick and adult diets were assessed using nonparametric analysis of similarity (ANOSIM), on a Bray–Curtis similarity matrix (PRIMER version 5.1.2, PRIMER-E Ltd., Plymouth, UK) (P ≤ 0.05). ANOSIM is a hypothesis-testing procedure that generates a probability value and a test statistic (R), which lies between 1 and −1. High positive R values indicate greater variation between groups than within groups, and negative values indicate high levels of within group variation compared to between groups. Values of R equal to zero represent the null hypothesis of no significant difference between groups. Similarity percentages (SIMPER, Plymouth Routines in Multivariate Ecological Research) were used to identify which prey taxa were responsible for the inter-group differences (Catalan et al. 2006).
To assess differences in the size of prey in chick and adult diets, we calculated a ratio for the mass of individual prey collected from chicks and adults on the same day and at the same site (adult to chick prey mass ratio; ACPMR). We calculated the ACPMR for each prey species and for all prey, where:
For ACPMR = 1, the mean mass of individual prey taxa in chick and adult diets are the same. If ACPMR >1, the mass of individual prey consumed by adults was larger than consumed by chicks. For ACPMR <1, the mass of individual prey in chick diets is smaller than in adult diets. To assess differences between the early, middle and late stages of the chick-provisioning period, we used the ACPMR in ANOSIM on a Bray–Curtis similarity matrix (PRIMER version 5.1.2, PRIMER-E Ltd., Plymouth, UK) (P ≤ 0.05). Analyses were carried out between season stages that had >2 samples collected from chicks and adults on the same day.
To assess the relationship between prey mass/length and chick mass, we collected regurgitates from a sample of chicks that were weighed with a spring balance (1,000 g ± 5 g) after regurgitating. We assigned diet data to one of three groups, based on the mass of chicks, their approximate age (McLeay, unpublished data) and the different stages of the chick-provisioning period (see above) at the time of sampling.
To assess ontogenetic, seasonal and spatial differences in the mass of the main prey species, we used T tests, Mann–Whitney tests or ANOVA, for which respective t, Z and F values are reported (P ≤ 0.05 was used as the threshold of significance). All analyses were undertaken using SPSS® and data were tested for assumptions of normality and homoscedasticity using Shapiro–Wilks’ test and Box’s M test, respectively. The average sizes of prey are reported ± SD.
Results
Prey composition
Between 2003 and 2007, a total of 2,146 and 3,921 prey were identified from 1,400 and 1,561 regurgitates collected from chicks and adults, respectively. Samples represented a reconstructed biomass of 67.3 kg. In total, 47 fish, 5 cephalopod and three crustacean taxa were identified (Tables 2, 3, 4, 5). Insects were found in samples from Troubridge Island and Rocky Island, but contributed a low proportion of the total biomass (<0.5) (Tables 2, 5). Australian anchovy Engraulis australis, sardine Sardinops sagax, blue sprat Spratelloides robustus, barracouta Thyrsites atun, Degens leatherjacket Thamnaconus degeni, southern sea garfish Hyporhamphus melanochir, Australian herring Arripis georgianus and slender bullseye Parapriacanthus elongatus comprised >66% of the total prey biomass consumed by chicks or adults at all locations (Tables 2, 3, 4, 5).
Ontogenetic variation in prey composition
Chick and adult diets differed significantly during the early and middle stages of the chick-provisioning period (ANOSIM, P ≤ 0.05) (Tables 2, 3, 4, 5, 6). This pattern was consistent among all locations and years except at Goose Island and Lipson Island in 2005 where low numbers of adult samples were collected (N = 13, N = 9, samples respectively; Table 6). Ontogenetic differences were also apparent in late provisioning at Troubridge Island in 2005 and 2007 (Table 6). In the early provisioning period SIMPER results indicated that differences were attributed to higher relative proportions of clupeids (blue sprat, Australian anchovy and sardine) and Australian herring in chick diets, and sardine, barracouta and Degens leatherjacket in adult diets (Table 6). In the mid provisioning period, diet differences were attributed to higher relative proportions of Australian anchovy, barracouta or sardine in chick diets, and Degens leatherjacket and/or garfish in adult diets (Table 6).
Spatial and interannual patterns in prey composition
There were significant differences in the prey composition of chick and adult diets between colonies in Gulf St Vincent (Troubridge Island) and Spencer Gulf (Goose Island, Lipson Island and Rocky Island) (ANOSIM P ≤ 0.05). SIMPER analyses indicated that differences were a consequence of relatively high proportions of sardine in chick and adult diets from colonies in Spencer Gulf, and Australian anchovy at Troubridge Island (Tables 2, 3, 4, 5). High proportions of Degens leatherjacket in adult diets at Troubridge Island also contributed to spatial differences (Tables 2, 3, 4, 5).
There were no distinct interannual patterns in the prey composition of chick and adult diets at Troubridge Island between 2004 and 2007 (Tables 2, 3, 4, 5). Differences among years were typically driven by large variation in the relative abundance of a few main prey. In chicks the main prey that varied from year to year were Australian anchovy, blue sprat and sardine (ANOSIM, P ≤ 0.05). In adults the main prey contributing to interannual differences were sardine, Degens leatherjacket and barracouta (ANOSIM, P ≤ 0.05) (Tables 2, 3, 4, 5).
Intra-annual variation in prey composition
The prey composition of chick diets varied significantly within the breeding season (ANOSIM, P ≤ 0.05) typically due to the large variation in the consumption of clupeids (sardine, Australian anchovy and blue sprat) between early and subsequent provisioning stages (mid/late) (Tables 2, 3, 4, 5). There were no clear intra-annual trends in prey composition at any location (Tables 2, 3, 4, 5), but the abundance of blue sprat and Australian herring in chick diets typically decreased as the breeding season progressed (Tables 2, 4, 5). In contrast, there was typically little intra-annual variation in the prey composition of adult diets (ANOSIM P ≥ 0.05) (Tables 2, 3, 4, 5).
Prey size
The average mass of all prey taxa consumed by chicks ranged between 7.4 ± 4.5 g in early provisioning to 10.7 ± 9.3 g in late provisioning. Prey consumed by adults averaged between 10.4 ± 8.1 g in late provisioning to 11.6 ± 10.4 g in early provisioning. The average mass and length (all sites/years pooled) of Degens leatherjacket, southern sea garfish and sardine was significantly larger in adult diets compared to chick diets (all P ≤ 0.05) (Table 7). Conversely, the average mass and length of Australian anchovy, blue sprat and barracouta was significantly larger in the diets of chicks compared to adult diets (all P ≤ 0.05). Australian herring and slender bullseye were of similar size in chick and adult diets (all P ≥ 0.05) (Table 7).
Ontogenetic variation in prey size
The adult to chick prey mass ratios (ACPMRs) indicated that adults generally fed their chicks smaller prey than they ate themselves for the entire nestling period at Troubridge Island in 2007 (ACPMR > 1; Fig. 2). During the early provisioning period in 2007 at Troubridge Island, the mean mass of individual prey (all species pooled) in the diet of adults was more than double (mean ACPMR = 2.43 ± 0.15) that of prey delivered to chicks (Fig. 2). This trend in high ACPMR values for the early chick-provisioning period was also evident at Troubridge Island between 2004 and 2006, Lipson Island in 2006 and Rocky Island in 2007. After 10 days the ACPMR decreased (mean ACPMR = 1.26 ± 0.29), indicating that adults were providing chicks with prey more similar in size to what they ate themselves.
The high ACPMR in the early provisioning period could be attributed to the relatively smaller mass of common prey consumed by chicks (Fig. 2). Sardine in the diet of chicks (8.4 g ± 0.9) were significantly smaller than sardine found in the diets of adults on the same day (28.7 g ± 3.6) (ACPMR = 2.21 − 4.89; Z = 3.68, P ≤ 0.05; Fig. 2). High ACPMR values (>1) for barracouta, Degens leatherjacket and Australian herring indicated these taxa were of smaller mass in the diet of chicks compared to adults. In contrast, the ACPMR of Australian anchovy and blue sprat did not differ significantly between chick and adult diets in any chick-provisioning period indicating that adults were selecting these prey at a similar size for themselves and their chicks (P ≥ 0.05; Fig. 2).
Spatial differences in prey size
Chicks typically consumed larger prey at Troubridge Island than in Spencer Gulf colonies. Australian anchovy consumed by chicks were significantly larger in the mid provisioning period at Troubridge Island in 2005 (9.7 ± 3.2 g) and 2007 (8.2 ± 2.2 g), compared to Goose Island (6.7 ± 2.0 g) and Rocky Island (5.4 ± 1.4 g), respectively (all P ≤ 0.001). Barracouta consumed in the mid chick-provisioning period in 2005 were also larger at Troubridge Island compared to other sites (Troubridge vs. Goose 2005: 45.6 ± 34.9 vs. 12.7 ± 5.1 g, Z = 2.244, P = 0.025). Similarly, chicks at Troubridge Island consumed larger Degens leatherjacket (16.1 ± 5.6 g) than at Lipson Island in the late provisioning period of 2006 (11.3 ± 5.0 g) (Z = 2.160, P = 0.031).
Prey taxa consumed by adults were also typically larger at Troubridge Island than Spencer Gulf colonies. Degens leatherjacket consumed by adults at Troubridge Island were significantly larger than sampled from Lipson Island during the early provisioning period in 2006 (Troubridge Is. vs. Lipson Is.: 19.1 ± 7.4 vs. 15.3 ± 6.6 g) (t = 2.491, P = 0.015). Australian anchovy and sardine were also significantly larger at Troubridge Island between 2005 and 2007 compared to other sites (P ≤ 0.001).
Interannual differences in prey size
There were no distinct interannual patterns in the size of prey in chick or adult diets at Troubridge Island between 2004 and 2007. Australian anchovy and sardine consumed by chicks were significantly larger throughout the entire chick-provisioning period in 2005 compared to other years (P ≤ 0.05), but the size of most other key prey taxa found in chick diets did not typically vary between years (all P ≥ 0.05).
Intra-annual differences in prey size
In 2007, the average mass and length of prey (all taxa pooled) consumed by chicks at Troubridge Island and Rocky Island (locations pooled) increased throughout the provisioning period (Fig. 3). The larger sardine and Australian anchovy provisioned during the mid and late provisioning periods contributed to this result (Troubridge 2007, sardine: early 8.4 ± 3.1 vs. late 11.5 ± 4.2 g, Z = 2.520, P = 0.012; Australian anchovy: early 6.7 ± 2.8 vs. late 8.0 ± 2.1 g, Z = 2.461, P = 0.014) (Rocky 2007, sardine: mid 9.6 ± 3.9 g vs. late 13.3 ± 6.8 g, Z = 2.636 P = 0.008; Australian anchovy: mid 5.4 ± 1.4 vs. late 8.0 ± 2.7 g, t = 4.243, P ≤ 0.001).
The size of the key prey taxa consumed by adults did not typically vary throughout the breeding season (P ≥ 0.05). Nonetheless, Australian anchovy consumed by adults in the late provisioning period at Troubridge Island and Rocky Island in 2007 were significantly larger than Australian anchovy in adult diets earlier in the season (Troubridge 2007: Australian anchovy early 7.6 ± 1.4 vs. late 8.7 ± 1.4 g, t = 1.951, P = 0.061; Rocky 2007: Australian anchovy mid 5.6 ± 1.5 vs. late 7.3 ± 1.6 g, t = 2.872, P = 0.008).
Discussion
This study profiles the diet of crested tern chicks and adults, and highlights some of their key prey in South Australia. Our research confirms findings of previous studies, which indicated that crested terns are generalist predators that feed predominantly on surface-schooling clupeids (Australian anchovy, sardine and blue sprat) (Walter et al. 1987; Hulsman et al. 1989; Smith 1993; Chiaradia et al. 2002). Degens leatherjacket, barracouta, southern sea garfish, Australian herring and slender bullseye were also commonly utilised. The importance of different prey varied spatially, temporally, between chicks and adults, and between chicks of different ages.
The observed decreases in the adult to chick prey mass ratio (ACPMR) over the course of the chick-provisioning period indicate that adults are constrained in their foraging behaviour by the need to select prey according to the physical ingestion capabilities of chicks during the early provisioning period (0–10 days). Crested tern chicks are small in comparison to many seabird species, weighing between 30 and 40 g when hatched (approximately 10% of adult body mass), and are limited immediately post-hatch in the size of prey they can ingest (McLeay, unpublished data). As chicks grow, energetic demands increase and adults may either increase the rate of fish delivered and/or deliver larger prey. Our research supports previous studies on other tern species, and indicates that adult crested terns adjust the size of prey provided to chicks as chicks grow throughout the nestling period (Hulsman et al. 1989; Ramos et al. 1998; Shealer 1998).
Seabirds provisioning chicks at a central place must adjust their foraging behaviour according to the distance of prey patches from their colony, their own prey requirements and the changing dietary requirements of their chicks. For single-prey loaders such as crested terns, central-place foraging models predict that adults should increase the amount of energy delivered to chicks in line with the amount of time they spend away from the colony (Orians and Pearson 1979; Leopold et al. 1996). As a result, a crested tern that had conducted a long foraging trip would be expected to provision its chick with relatively large prey. Adults consumed larger prey than chicks throughout the provisioning period, but differences in individual prey mass between chick and adult diets were greatest during early provisioning. These results indicate that small prey may not have been available in high enough densities near the colony to support self-feeding.
Chicks also consumed higher proportions of clupeids (Australian anchovy, sardine and blue sprat) than adults, which consumed mainly Degens leatherjackets and barracouta. Previous research on little penguin Eudyptula minor diets at Troubridge Island showed these clupeids were of higher calorific value and had higher amounts of lipid and protein than leatherjackets, southern sea garfish or slender bullseye (A. Wiebkin, unpublished data). This suggests that adult crested terns may select and provide higher quality prey to their chicks compared to what they consume themselves. Such a strategy has been demonstrated for other seabirds (Vermeer et al. 1987; Hodum and Hobsum 2000; Jaquemet et al. 2008). Clupeids have a high calorific value and may contain key nutritional elements required for the physiological processes that regulate chick growth and survival (Pichegru et al. 2007). Poorer quality “junk food” was the likely cause of breeding failure of common guillemots in the North Sea (Wanless et al. 2005). Future research should examine the relationship between breeding success, chick growth and diet quality for crested tern populations.
All diet studies have inherent biases which are associated with differences in prey-specific digestion rates and differential regurgitation of prey remains (Duffy and Jackson 1986; Gonzales-Solis et al. 1997; Barrett et al. 2007). The proportion of ingested prey in regurgitations varies according to when the prey were captured and the type of prey ingested. Small prey or otoliths from fish remains may be digested more quickly than larger items and therefore underestimated in diet analyses. Our study was not completely free of these biases. However, crested terns provisioned their chicks with an average of 3.1 ± 1.9SD single prey per day (McLeay, unpublished data) indicating that they forage in waters near to the colony. Also, prey remains collected were generally always in good condition and could be easily identified. This is supported by our identification of >97% of the prey collected in this study. Also, fish otoliths used in prey weight regressions were obtained via fish dissections and were consequently not degraded. These factors reduced the potential bias associated with differences in prey or size-specific digestion rates.
The ontogenetic and seasonal variation in prey composition and size indicated by this study highlights the potential biases associated with disproportionate, non-systematic sampling of seabird diets throughout the breeding season. Detection of fine-scale temporal patterns in seabird diets are often inhibited by the cost and effort of collecting samples. Nonetheless, if the factors constraining foraging behaviour and driving patterns of diet variation are to be elucidated, preliminary studies should attempt to stratify diet sampling from a range of life history stages throughout incubation and while adults are provisioning their chicks.
The diets of crested terns at Troubridge Island, Gulf St Vincent contained larger prey (Australian Australian anchovy, sardine, Degens leatherjacket and barracouta) than colonies in Spencer Gulf and support the hypothesis proposed for other finfish species (Australian anchovy and King George whiting Sillaginodes punctata) of the occurrence of a spatial gradient in prey size from north of Spencer Gulf and Gulf St Vincent to the south (i.e. smaller in north vs. larger in south) (Fowler et al. 2000; Dimmlich and Ward 2006). The diets of crested terns at Spencer Gulf colonies had typically more sardine compared to diets sampled from Troubridge Island, Gulf St Vincent and may reflect differences in the abundance of sardine between the two gulfs. This finding is consistent with the lower estimates of sardine egg production calculated via annual fisheries assessment for Gulf St Vincent (Ward et al. 2008). A longer time series of diet data and continued sardine fishery assessment are required to assess whether the proportion of sardine in crested terns diets is related to sardine stock recruitment or adult sardine abundance.
Use of data in fisheries management
Small pelagic fish (‘forage fish’) are increasingly the target of commercial fisheries and form a key link between apex predators and lower trophic levels in many marine ecosystems. However their distribution and abundance is highly variable. The South Australian Sardine Fishery (SASF), which targets sardine and other small pelagic fishes, has grown in recent years and fisheries managers have raised concerns about the potential ecological impacts on apex predators such as crested terns (Shanks 2004). Previous research indicated that crested terns may be sensitive to large scale variations in sardine abundance (McLeay et al. 2009). However, before data from crested terns are applied in management, it is first necessary to understand the intrinsic factors that underpin diet variation and mediate demographic, behavioural or physiological responses.
The large component of sardine, which were predominantly juveniles, in the diet of crested terns from Spencer Gulf colonies suggests that future dietary measures may provide additional information about changes in juvenile sardine abundance. Most of the SASFs annual total allowable catch (TAC) of sardine is taken in close proximity to at least 11 islands in Spencer Gulf where breeding of crested terns has been recorded (Fig. 1) (McLeay et al. 2009). The fishery does not currently target juvenile sardine, however, localised depletion of adult sardine could reduce subsequent rates of sardine egg production and juvenile recruitment. Measures of crested tern diet, coupled with estimates of breeding success, foraging behaviour (provisioning), or physiology (chick growth) and fishery data (catch information: location, sardine length) may assist in highlighting whether fishing is indirectly impacting on the abundance of juvenile sardine. These data could help to ensure commercial fishing practices address principals of ecologically sustainable development developed for Australian fisheries (Fletcher et al. 2002).
References
Barrett RT, Camphuysen CJ, Anker-Nilssen T, Chardine JW, Furness RW, Garthe S, Huppop O, Leopold MF, Montevecchi WA, Veit RR (2007) Diet studies of seabirds: a review and recommendations. ICES J Mar Sci 64:1675–1691. doi:https://doi.org/10.1093/icesjms/fsm152
Blaber SJM, Wassenberg TJ (1989) Feeding ecology of the piscivorous birds Phalocrocorax varius, Phalocrocorax melanoleucos and Sterna bergii in Moreton Bay, Australia: diets and dependence on trawler discards. Mar Biol (Berl) 101:1–10. doi:https://doi.org/10.1007/BF00393473
Boyd IL, Wanless S, Camphuysen CJ (2006) Introduction. In: Boyd IL, Wanless S, Camphuysen CJ (eds) Top predators in marine ecosystems. Cambridge University Press, pp 378
Burger AE, Piatt JF (1990) Flexible time budgets in breeding common murres: buffers against variable prey abundance. Stud Avian Biol 14:71–83
Cairns DK (1984) The foraging ecology of the black guillemot (Cepphus grylle) Ph.D. thesis, Ontario
Cairns DK (1987) Seabirds as indicators of marine food supplies. Biol Oceanogr 5:261–271
Catalan IA, Jimenez MT, Alconchel JI (2006) Spatial and temporal changes of coastal demersal assemblages in the Gulf of Cadiz (Spain) in relation to environmental conditions. Deep-sea research paper II, topical studies in oceanography 53:1402–1419
Chiaradia A, Dann P, Jessop R, Collins P (2002) The diet of crested tern (Sterna bergii) chicks on Phillip Island, Victoria, Australia. Emu 102:367–371. doi:https://doi.org/10.1071/MU02004
Crawford RJM (2004) Accounting for food requirements of seabirds in fisheries management—the case of the South African purse-seine fishery. Ecosystem Approaches to fisheries in the southern Benguela. Afr J Mar Sci 26:197–203
Davoren GK, Burger AE (1999) Differences in prey selection and behaviour during self-feeding and chick provisioning in rhinoceros auklets. Anim Behav 58:853–863. doi:https://doi.org/10.1006/anbe.1999.1209
Dimmlich WF, Ward TM (2006) Ontogenetic shifts in the distribution and reproductive patterns of Australian anchovy (Engraulis australis) determined by otolith microstructure analysis. Mar Freshw Res 57:373–381. doi:https://doi.org/10.1071/MF05184
Duffy DC, Jackson S (1986) Diet studies of seabirds: a review of methods. Colon Waterbirds 9:1–17. doi:https://doi.org/10.2307/1521138
Fletcher WJ, Chesson J, Fisher M, Sainsbury KJ, Hundloe T, Smith ADM, Whitworth B (2002) National ESD reporting framework for Australian fisheries: The how to guide for wild capture fisheries. FRDC Project 2000/145, Canberra, Australia
Fowler AJ, McLeay LJ, Short DA (2000) Spatial variation in size and age structures and reproductive characteristics of the King George whiting (Percoidei: Sillaginidae) in South Australian waters. Mar Freshw Res 51:11–22. doi:https://doi.org/10.1071/MF99032
Frederiksen M, Wanless S, Harris MP, Rothery P, Wilson NJ (2004) The role of industrial fisheries and environmental change in the decline of North Sea black-legged kittiwakes. J Appl Ecol 41:1129–1139. doi:https://doi.org/10.1111/j.0021-8901.2004.00966.x
Furlani D, Gales R, Pemberton D (2007) Otoliths of common Australian temperate fish: a photographic guide. CSIRO, Victoria, pp 208
Furness RW, Tasker ML (2000) Seabird fishery interactions: quantifying the sensitivity of seabirds to reductions in sand eel abundance, and identification of key areas for sensitive seabirds in the North Sea. Mar Ecol Prog Ser 202:253–264. doi:https://doi.org/10.3354/meps202253
Gonzales-Solis J, Oro D, Pedrocchi V (1997) Bias associated with diet samples in Audouin’s gulls. Condor 99:773–779. doi:https://doi.org/10.2307/1370488
Hall S, Mainprize B (2004) Towards ecosystem-based fishery management. Fish Fish 5:1–20. doi:https://doi.org/10.1111/j.1467-2960.2004.00133.x
Hislop JRG, Harris MP (1985) Recent changes in the food of young puffins Fratercula arctica on the Isle of May in relation to fish stocks. Ibis 127:234–239. doi:https://doi.org/10.1111/j.1474-919X.1985.tb05057.x
Hodum PJ, Hobsum KA (2000) Trophic relationships among Antarctic fulmarine petrels: insights into dietary overlap and chick provisioning strategies inferred from stable isotope (ä15 and ä13C) analyses. Mar Ecol Prog Ser 198:273–281. doi:https://doi.org/10.3354/meps198273
Hulsman K, Langham NPE, Bluhdom D (1989) Factors affecting the diet of crested terns (Sterna bergii). Aust Wildl Res 16:478–489. doi:https://doi.org/10.1071/WR9890475
ICES (2000) Report of the working group on seabird ecology. International Council for the Exploration of the Sea, Wilhelmshaven, Germany
ICES (2002) International Council for the Exploration of the Sea Cooperative Research Report 254. International Council for the Exploration of the Sea, Copenhagen, Denmark
Jaquemet S, Potier M, Cherel Y, Kojadinovic J, Bustamante P, Richard P, Catry T, Ramos JA, Le Corre M (2008) Comparative feeding ecology and ecological niche of a superabundant tropical seabird: the sooty tern Sterna fuscata in the southwest Indian Ocean. Mar Biol (Berl) 155:505–520. doi:https://doi.org/10.1007/s00227-008-1049-1
Leopold MF, VanElk JF, VanHeezik YM (1996) Central place foraging in oystercatchers Haematopus ostralegus: can parents that transport mussels Mytilus edulis to their young profit from size selection? Ardea 84A:311–325
Lewis S, Wanless S, Wright PJ, Harris MP, Bull J, Elston DA (2001) Diet and breeding performance of black legged kittiwakes Rissa tridactyla at a North Sea colony. Mar Ecol Prog Ser 221:277–284. doi:https://doi.org/10.3354/meps221277
Mahon T, Kaiser G, Burger AE (1992) The role of marbled murrelets in mixed-species feeding flocks in British Columbia. Wilson Bull 104:738–743
McLeay LJ, Page B, Goldsworthy SD, Ward TM, Paton DC, Waterman M, Murray MD (2009) Demographic and morphological responses to prey depletion in a crested tern Sterna bergii population: can fish mortality events highlight performance indicators for fisheries management? ICES J Mar Sci 66:237–247. doi:https://doi.org/10.1093/icesjms/fsn195
Monaghan P, Uttley JD, Okill JD (1989) Terns and sand eels: seabirds as indicators of changes in marine fish populations. J Fish Biol 35:339–340
Montevecchi WA (1993) Birds as indicators of change in marine prey stocks. In: Furness RW, Greenwood JJD (eds) Birds as monitors of environmental change. Chapman and Hall, London, pp 217–266
Montevecchi WA, Myers RA (1995) Prey harvests of seabirds reflect pelagic fish and squid abundance on multiple spatial and temporal scales. Mar Ecol Prog Ser 117:1–9. doi:https://doi.org/10.3354/meps117001
Montevecchi WA, Myers RA (1996) Dietary changes of seabirds indicate shifts in pelagic food webs. Sarsia 80:313–322
Montevecchi WA, Birt VL, Cairns DK (1987) Dietary shifts of seabirds associated with local fisheries failures. Biol Oceanogr 5:153–159
Nettleship DN, Sanger GA, Springer PF (eds) (1982) Marine birds: their feeding ecology and commercial fisheries relationships. In: Proceedings of the Pacific seabird group symposium. Canadian wildlife service, Seattle, Washington, p 212
Orians GH, Pearson NE (1979) On the theory of central place foraging. In: Horn OJ, Stairs BR, Mitchell RD (eds) Analysis of ecological systems. Ohio Sate University Press, Ohio, pp 155–177
Pichegru L, Ryan PG, Van der Lingen CD, Coetzee J, Ropert-Coudert Y, Gremillet D (2007) Foraging behaviour of Cape gannets Morus capensis feeding on live prey and fishery discards in the Benguela upwelling system. Mar Ecol Prog Ser 350:127–136. doi:https://doi.org/10.3354/meps07128
Ramos JA, Encarnacion S, Monteiro LR (1998) Prey delivered to roseate tern chicks in the Azores. J Field Ornithol 69:419–429
Schreiber EA, Burger J (2002) Biology of marine birds. CRC Press, London
Shanks S (2004) Ecological assessment of the South Australian pilchard fishery. PIRSA, Adelaide
Shealer DA (1998) Size-selective predation by a specialist forager, the roseate tern. Auk 115:519–525
Smith GC (1993) Feeding and breeding of crested terns at a tropical locality. Comparison with sympatric black-naped terns. Emu 93:65–70
Stienen EWM, Van Beers PWM, Brenninkmeijer A, Habrakan JM, Maaike PM, Raaijmakers E, Van Tienen PGM (2000) Reflections of a specialist: patterns in food provisioning and foraging conditions in sandwich terns Sterna sandvicensis. Ardea 88:33–49
Surman CA, Wooler RD (2003) Comparative feeding ecology of five sympatric terns at a sub-tropical island in the eastern Indian Ocean. J Zool 259:219–230. doi:https://doi.org/10.1017/S0952836902003047
Swihart RK, Johnson SG (1986) Foraging decisions of American robins: somatic and reproductive tradeoffs. Behav Ecol Sociobiol 19:275–282. doi:https://doi.org/10.1007/BF00300642
Velarde E, Ezcurra E, Cisneros-Mata MA, Lavin MF (2004) Seabird ecology, El Nino anomalies, and prediction of sardine fisheries in the Gulf of California. Ecol Appl 14:607–615. doi:https://doi.org/10.1890/02-5320
Vermeer K, Sealy SG, Sanger GA (1987) Feeding ecology of the Alcidae in the eastern north Pacific Ocean. In: Croxall JP (ed) Seabirds: feeding ecology and their role in marine ecosystems. Cambridge University Press, New York, pp 189–228
Walter CB (1984) Fish prey remains in swift tern and Hartlaub’s gull pellets at Possession Island, off Namibia. Ostrich 58:49–53
Walter CB, Cooper J, Suter W (1987) Diet of swift tern chicks in the Saldanha Bay Region, South Africa. Ostrich 58:49–53
Wanless S, Harris MP, Redman P, Speakman JR (2005) Low energy values of fish as a probable cause of breeding failure in the North Sea. Mar Ecol Prog Ser 294:1–8. doi:https://doi.org/10.3354/meps294001
Ward TM, Ferguson G, Rogers PJ (2008) Australian Sardine (Sardinops sagax) Fishery. SARDI Aquatic Sciences, F2007/000765-2, Adelaide
Weimerskirch H (1998) How can a pelagic seabird provision its chick when relying on a distant food resource? Cyclic attendance at the colony, foraging decision and body condition in sooty shearwaters. J Anim Ecol 67:99–109. doi:https://doi.org/10.1046/j.1365-2656.1998.00180.x
Acknowledgments
This study was principally supported through the Australian Government’s Fisheries Research and Development Corporation (FRDC) Grants Scheme (PN 2005/031), co-funded by the South Australian Sardine Fishery. We also thank the South Australian Department for Environment and Heritage for financial assistance through the Wildlife Conservation Fund. All diet sampling and bird handling procedures were carried out under South Australian scientific research permit A24684. We thank volunteers A. Baylis, G. French, A. Ivey, T. Kemper, J. McKenzie, R. Mayo, G. McLeay, H. McLeay, L. K. McLeay, M. McLeay, A. Newman, J. Nichols, C. Platt, P. Rogers, and A. Wiebkin for their assistance during field operations, and C. and J. Johnson for providing logistical support and accommodation on Troubridge Island. We are grateful to A. Wiebkin, M. Steer and W. Dimmlich for providing data relating to prey regressions and P. Rogers and anonymous referees for useful comments that greatly improved the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by S. Garthe.
Rights and permissions
About this article
Cite this article
McLeay, L.J., Page, B., Goldsworthy, S.D. et al. Size matters: variation in the diet of chick and adult crested terns. Mar Biol 156, 1765–1780 (2009). https://doi.org/10.1007/s00227-009-1211-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00227-009-1211-4