Abstract
Many organisms survive stressful conditions through a tolerant life history stage. The life history known as the alternation of generations is typical of temperate kelps, producing diploid macroscopic stages, and both haploid and diploid microscopic stages, with the haploid stages thought to be stress tolerant. The survival of microscopic stages of the giant kelp Macrocystis pyrifera during El Niño has been suggested, yet has never been tested. This mechanism could be critical for population persistence, particularly at the southern limit of the range in the Northern Hemisphere, which is greatly impacted by El Niño conditions. The purpose of this study was to determine if microscopic stages of giant kelp could survive and recover from El Niño-type conditions and whether those from a population near its southern limit were more tolerant than a population at the center of its range. Microscopic stages were exposed to a laboratory simulation of potential El Niño conditions (high temperature, with and without light and nitrate) for 8 weeks and then allowed to recover at optimal conditions (low temperature and high nitrate) for 8 weeks, while controls were left at optimal conditions the entire 16 weeks test period. Haploid developmental stages from both populations survived and recovered from stressful conditions with no population level effect, suggesting haploid stress-tolerance may be widespread. The more advanced the developmental stage, and the presence of nitrate, resulted in significantly greater recovery for haploids. Yet, none of these stages were able to go on to produce sporophytes, whereas all controls did. There was a large population-level effect for diploids, however, with only microscopic diploid stages (embryonic sporophytes) from the southern-limit population recovering from El Niño simulated stress, suggesting ecotypic adaptation for microscopic sporophytes. Diploid recovery was significantly greater with light. We propose that the diploid stage is the most likely to survive and recover after El Niño conditions, as it would avoid obligate egg and sperm encounters after the stress period. The survivorship of the microscopic diploid in a seed bank analogue may be how the isolated southern-limit populations are able to recover after mass disappearance during El Niño.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The ability for certain life history stages to withstand ambient stress can be crucial to the survival of a population, particularly near an organism’s range limits, where it often encounters stressful physiological conditions (Hummel et al. 1996; Portner 2002; Clarke 2003). Temperature is a common stress along a latitudinal gradient (Vermeij 1980), and is a principal variable in determining distribution (Fields et al. 1993; Sagarin et al. 1999). Many factors covary with temperature, such as the inverse relationship of nitrogenous nutrients in temperate marine environments (Zimmerman and Kremer 1984; Ladah 2003). El Niño events are a good example, and are characterized by invasion of uncommon water masses, a reduction in upwelling of cool nutrient-replete waters, a depressed thermocline and nutricline, a warming of surface waters, and a reduction in primary productivity in the temperate Pacific Ocean (Huyer and Smith 1985; McGowan et al. 1998; Kahru and Mitchell 2000). El Niño’s cause significant physiological stress in temperate pelagic and benthic ecosystems along the Northeastern Pacific (Miller and Schneider 2000).
One ecosystem negatively affected by El Niño is the temperate kelp forest ecosystem (Dayton and Tegner 1984), dominated by large brown seaweeds, which are sensitive to warm temperatures and low nitrogenous nutrients (North 1994; North and Zimmerman 1984). These factors may be the most important in determining kelp forest distribution (Jackson 1977; Zimmerman and Robertson 1985; Foster and Schiel 1985), affecting the survival of all developmental life-history stages (Deysher and Dean 1986a, b; Gerard 1997; Martinez and Santelices 1998). Because kelps have a complex alternation of generations life history, with unique requirements at each stage, they respond to the warm temperature/low nutrient stress of El Niño in various ways. Mature kelps reduce reproductive output, fade in color, slough tissue and eventually dye (Gerard 1984). Recruitment is reduced and often entire macroscopic populations can disappear (Tegner and Dayton 1987; Ladah et al. 1999). Juveniles under stress survive for much less time than adults (Dean and Jacobsen 1984).
On the other hand, the microscopic stages of kelps seem to be stress tolerant (Bolton and Luning 1982; Laminaria spp., Lee and Brinkhuis 1986; Laminaria saccharina, and tom Dieck 1993; most Laminariales). The mechanism of “dormancy” for stress persistence in kelps and other large brown macroalgae has been suggested (Dayton 1973, 1985). The microscopic stages of kelps include haploid and diploid developmental stages. A pelagic haploid spore settles and produces a benthic gametophyte, which undergoes gametogenesis, and grows to an egg. This egg is fertilized by free swimming sperm (released from the benthic male gametophyte after it has undergone gametogenesis), producing a final diploid sporophyte, which grows into an adult (see North 1994 for a review). The ecological importance of the microscopic stages in the kelp life history is unclear, as they are difficult to study and identify in the field (Clayton 1990). They have been suggested to play a similar role to a seed bank (Santelices 1990; Ladah et al. 1999). Field research in California, however, has shown that microscopic stages have little capacity to survive long periods in nature (Reed et al. 1997). Surprisingly, the ability of microscopic stages to survive through adult plant extirpation during El Niño has not been tested, though empirical data suggests this may be a mechanism of recovery in the absence of adults (Ladah et al. 1999).
Kelp gametophytes may play an important ecological role similar to a seed bank in land plants (see Hoffman and Santelices 1991; Santelices et al. 1995). Yet kelps differ from terrestrial seeds because sexual reproduction occurs after spore settlement and gametophyte development. Fertilization of eggs by sperm requires an active female pheromone, and it is unclear how long this chemical remains active. After production of benthic embryonic diploids, there is reliance on compensation levels of light for photosynthesis, suggesting the analogy may be closer to that of germinated seedlings stunted under an adult canopy. Because of these differences between the traditional terms coined for terrestrial populations, we refer to the process of survivorship and recovery after stressful conditions by kelp microscopic stages as a “seed bank analogue”.
During El Niño episodes, conditions of high temperatures and low nitrogen concentrations occur throughout the distribution of giant kelp in the Northern Hemisphere [about 1,000 km from the Monterey area of California (about 36°N) to Southern Baja California (about 27°N) (Foster and Schiel 1985)]. Conditions are particularly extreme at the southern limit of the species located near Bahía Tortugas, Baja California Sur, Mexico (27°39.97 N, 114°54.18 W) (Ladah et al. 1999). The southern-limit populations are discontinuous from other populations (Dawson 1951, 1952; Lynn and Simpson 1987; Palacios-Hernandez et al. 1996) and can grow in very shallow or even intertidal areas. Because El Niño conditions negatively affect the distribution and abundance of macroscopic stages of giant kelp (Dayton 1985; Tegner and Dayton 1987, 1991; Tegner et al. 1996), and can cause wide spread extirpation of macroscopic plants (Hernandez-Carmona et al. 1991; Ladah et al. 1999; Edwards 2004), the survival of microscopic stages in a seed bank analogue may be very important, particularly in marginal or isolated areas.
At the southern limit of distribution, during El Niño 1982–83 (Hernandez-Carmona et al. 1991) and El Niño 1997–98 (Ladah et al. 1999; Edwards 2004), all macroscopic Macrocystis pyrifera plants disappeared in the southern limit region. During El Niño 1997–98, in shallow populations in Bahia Tortugas, water temperatures approached 25°C for approximately 2 months (unpublished data) throughout the entire water column, and much rocky substrate became covered with sand from subsequent storms due to hurricanes Linda and Nora. The nearest reproductive population was found over 400 km to the north, yet recruitment was documented less than a year later at the southern limit. A microscopic stage was proposed to survive at the southern limit on the benthos, potentially buried under sediment, in a seed bank analogue (Ladah et al. 1999).
The following microscopic stages of kelp might persist in a seed bank analogue: (1) newly settled haploid spores, (2) non-fecund gametophytes (males and females) (often filamentous under stressful conditions, pre-gametogenesis), (3) fecund female (eggs) and male (antheridia) gametophytes (post-gametogenesis), and (4) diploid embryonic microscopic sporophytes (after fertilization of egg by sperm). Diploid macroscopic sporophytes (<1 cm) may also survive undetected on the benthos awaiting more appropriate growing conditions. In order to resume growth and contribute to recruitment after stress exposure, the spores and the filamentous gametophytes would have to undergo gametogenesis and produce viable gametes. The fecund gametophytes, with eggs and antheridia already produced, would require release of sperm and release of the pheromone by the egg, both of which may only occur once. Ideally, if development could be arrested in the diploid microscopic stage that was already fertilized, the tiny blade would simply need to resume growth.
Recently, it has been shown during exposure to low light and nutrients in the laboratory, that diploid microscopic stages from one California population could arrest development for 1 month and then regrow (Kinlan et al. 2003). However, during El Niño conditions in Central Baja California, the microscopic stages would not only have to survive conditions of limiting resources (nutrients and light), they would also have to survive high temperatures (approaching 25°C for approximately 2 months) (Durazo and Baumgartner 2002, unpublished data). There is no information available on how different microscopic stages of giant kelp respond to combined thermal stress and limiting resources, as would occur during El Niño conditions.
Although a seed bank analogue is debated as a possible explanation for the recovery of distant populations after mass disappearance, particularly after El Niño, little is known, about which stage is the resistant stage in nature, and no work to our knowledge has combined the three major stressors for giant kelp (i.e., temperature, nutrients, and light). Considering the wide range of conditions inhabited along the coast of N. America, understanding if the ability to arrest development in response to limiting resources varies among populations in an ecotypic form would also be particularly interesting and relevant to the southern, more subtropical, populations. Physiological ecotypes for temperature tolerance, nutrient uptake and photosynthetic parameters have been shown for kelps from geographically isolated populations (Espinoza and Chapman 1983; Kopzcak et al. 1991; Gerard 1990, 1997; Martinez 1999; Ladah in prep.). Although the idea of a marine seed bank analogue for algae is not novel, the elucidation of which microscopic stage actually persists during periods of high temperatures, and whether or not different populations exhibit differences in ability to persist, has not been previously addressed.
The purpose of the present study was to determine if microscopic stages could survive and recover from a laboratory re-creation of an El Niño stress representative of the 1997–98 El Niño in Central Baja California. We wanted to identify which stage was most probable to persist in the field, and if there were any differences between geographically separated populations. Each developmental microscopic stage of giant kelp was cultured from spores from two populations, one at the center of distribution (temperate) and one at the southern limit (subtropical) of the M. pyrifera range in Baja California (separated by approx. 500 km) in the Northern Hemisphere. Each stage was then exposed to laboratory simulated El Niño conditions representative of the El Niño 1997–98 in shallow populations in Central Baja California (note these conditions are very different from what may be experienced in California, USA). Simulations entailed a high temperature stress. Treatments considered the presence and absence of both nitrate and light, as some stages may have been buried below the sediment shading them from light, and because both sediment and different water masses (see Ladah et al. 2003 for an example of nutrient enrichment at high temperatures during El Niño in Central Baja California) may provide some level of nutrients in the field, even at high temperatures. Treatments did not consider temperature, since El Niño temperatures are always warm in this region and because colder temperatures never occur without nutrients in these areas. Temperature was intentionally not orthogonal as a low temperature, low nutrient treatment is not representative of any natural condition in these sites to our knowledge.
Materials and methods
Fertile sporophylls were collected from five different adult canopy-forming individuals at 15 m depth separated by 20 m each in a population from the center of the range (Campo Kennedy, 31°41.96 N, 116°40.90 W) and a population from the southern-limit (Bahia Tortugas, 27°39.97 N, 114°54.18 W) of M. pyrifera (Fig. 1), wrapped in damp paper towel, and stored on ice overnight. Spores from each population were then released in the laboratory by submerging sporophylls in an artificial seawater solution (Ott 1965) (without nitrate added) containing all Provasoli’s reagents (PES) except nitrogen (PES-N) (Provasoli 1968) at 16°C. Spore density was quantified using a hemocytometer. Spores were seeded at a density of ten spores mm−2 in all treatments. It can, of course, be argued that sporophylls from five plants do not represent a large population and that neither does a single spore release. However, we believe that spores were healthy as all controls developed to blade stage plants. Also, because spores were mixed from all five plants from each population we believe our results are representative of each population.
Map of Baja California showing sites from which spores were collected in N. Baja California (at the center of the giant kelp range in the Northern Hemisphere) and in Baja California Sur (near its southern limit in the Northern Hemisphere), about 500 km to the south. The northern limit is located about 500 km to the north in Monterey, California (not shown)
Haploid stress-recovery experiment
The ability of haploids to survive and recover from stress exposure was tested by seeding spore solutions from each population into petri dishes that contained 20 ml of PES-N with 10 μM of nitrate. These were cultured in an incubator at standard conditions (16°C, 50 μE m−2 s−1, 12:12 photoperiod). A scuffed cover slip was placed on the bottom of each petri dish to serve as a removable substrate for evaluation. As the spores developed, petri dishes were randomly chosen from each population at each developmental stage of the haploid life cycle. Settled spores were approx. 2 days old, gametophytes were approx. 1 week old, and eggs (gametophytes that had undergone gametogenesis and had produced an egg, so only females were evaluated) were approx. 3 weeks old. The number of haploid individuals was evaluated for each developmental stage in four permanent microscopic quadrants on the cover slip (1 mm2) per petri dish, and then moved to a 24°C (±1°C) environment, at 50 μE m−2 s−1 irradiance, in Ott’s artificial seawater, with PES-N. Each stage was exposed to the following four treatments at 24°C: (1) nitrate (10 μM NO3 added) and light (no shade), (2) nitrate and dark (petri dishes covered with a black cardboard hat), (3) no nitrate and light, and (4) no nitrate and dark, representing potential El Niño conditions encountered at the southern limit (see Introduction for explanation). Three replicates for each nitrate/light combination were tested for each population for each haploid stage. Each stage was exposed to the above treatments for 8 weeks and then evaluated for survivorship in the quadrants.
Any individual that was still recognizable after the eight weeks of stress exposure was considered a survivor. Unpigmented individuals were still counted as survivors if their outline could be distinguished. After survivorship was evaluated, the cultures were returned to standard culture conditions (16°C, 10 μM NO3, 50 μEm−2 s−1) for 8 weeks (termed the recovery period) and then monitored for recovery.
Any individual that had maintained or regained pigment or begun to grow during the period of recovery (a total of 16 weeks in culture, 8 weeks under stress and 8 weeks under recovery conditions) was considered to have recovered. If any haploids had become diploids (detected by cell division), this was also noted. Culture media was changed weekly.
Data is expressed in two ways. Frequency surviving refers to the frequency of initial (pre-stress) individuals that survived the 8 weeks El Niño simulated stress exposure. Frequency recovering refers to the frequency of initial (pre-stress) individuals that survived the 8 weeks El Niño simulated stress exposure and had recovered (regaining or maintaining pigment or resuming development) after further 8 weeks of recovery conditions. Frequency recovering was considered the ecologically relevant measure in this study because in the field, individuals would have to survive the original El Niño stress as well as recover and continue development to contribute to population restoration. We limited this recovery time to 2 months of optimum conditions. Of course, recovery could have still occurred after the 2 months cut off point in this study, however, 2 months was chosen as the limit of the recovery period as that was what occurred in nature during the El Niño of 1997–1998.
Microscopic diploid stress-recovery experiment
The ability of diploids to survive and recover from stress exposure was tested by removing diploid blades of each population after 1.5 months in standard culture (16°C, 10 μM NO3, 50 μE m−2 s−1, 12:12 photoperiod) and placing them in petri dishes kept at 24°C. For each population, three petri dishes with ten blades each were placed under the same four treatments as in the haploid treatments and exposed to the same stress treatments for eight weeks. They were then evaluated for survivorship. A diploid survivor was considered any individual still recognizable, even if completely bleached. After exposure to stress, the cultures were returned to standard conditions as above and then monitored for recovery after eight more weeks. Criterion for recovery was considered any blade that maintained or regained pigment or growth. If after 8 weeks of recovery, individuals were still bleached or sloughing, they were marked as dead. Data is expressed as explained above.
Control treatments
More petri dishes were seeded than were needed for the experimental treatments and those not used remained at the original treatment in the incubator at standard conditions. Culture media was replaced in these controls at the same time as the treatments. All control cultures successfully became diploid during the experiment. Density could not be determined as when the haploid cultures produced diploids, the blades formed overlapping canopy resulting in a percent cover of 100% on the cover slip. Diploid controls had 100% survivorship.
Statistical analyses
A fixed factor ANOVA (Statistica, version 6.0) was used to compare the effects of population (two levels), developmental stage (three levels tested for haploid experiments, not tested for diploid experiments), light (two levels) and their interactions on percent recovery. Percent recovery was arc-sine transformed and homocedasticity was confirmed using the Cochran test.
Results
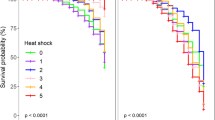
In all stages, many individuals that appeared to be alive after exposure to stress for 8 weeks had completely disappeared (tissue sloughed and disintegrated) during the recovery period (Fig. 2) (i.e., individuals continued to perish, rather than recover, under the recovery conditions).
Frequency of initial (pre-stress) individuals that survived the 8 weeks El Niño simulated stress exposure (in black) and frequency of initial (pre-stress) individuals that had recovered (regaining or maintaining pigment or resuming development) after further 8 weeks of recovery conditions (in white) for different microscopic life history stages of M. pyrifera (all treatments and populations combined to show that organisms continued dying even in recovery conditions) (n = 24, mean ± 1 SE shown
Recovery of haploid life history stages
More than 90% of treatments with haploid stages showed at least some recovery according to our criterion. Only three replicates did not show any survival or recovery. All three were from the northern population and were from the spore stage. The more advanced the haploid stage, the greater the survival and recovery (Figs. 2, 3), with the egg stage having the highest recovery of the haploid stages tested for both populations (Fig. 3).
Frequency of initial (pre-stress) individuals that survived the 8 weeks El Niño simulated stress exposure and that had recovered (regaining or maintaining pigment or resuming development) after further 8 weeks of recovery conditions for different microscopic life history stages of M. pyrifera from two different populations: Campo Kennedy, N. Baja population, and Bahia Tortugas, S. Baja population (n = 3, mean ± 1 SE shown). N nitrate, L light
There was a significant effect on recovery of developmental stage, the presence of nitrate during stress exposure, and the interaction between the population of origin and the presence of light during stress exposure (see Table 1). The egg stage had significantly greater recovery than the gametophyte stage, which in turn was significantly greater than the spore stage (Post hoc tukey honest significant differences test, p < 0.05).
In general, the presence of nitrate resulted in significantly greater recovery. Similar levels of recovery were detected for individuals from both populations in the presence of light. In the absence of light, the southern population recovered just as well as with light, while the northern population showed significantly lower recovery (Table 1). Despite some individuals regaining pigmentation, none of the haploid stages that had survived the stress exposure went on to produce sporophytes during the 8 weeks recovery period.
Recovery of diploid sporophytes
Less than 30% of the diploid cultures showed recovery at the end of the experiment and all those that did were from the southern-limit population (Fig. 3). Diploid sporophytes from the northern population showed no survival according to our criterion at the end of the experiment and were necrotic, whereas some of the southern-limit sporophytes did regain pigment and grow, however only from the basal portion of the microscopic blade closest to the haptera and not over the entire blade. The southern limit population had significantly greater recovery in the presence of light during stress exposure (see Table 2).
Discussion
The recovery of haploid stages of M. pyrifera after exposure to El Niño simulated stress was not affected by population, while for diploid stages only those from the southern limit population showed recovery. These results support two main ideas. One, that in organisms with a heteromorphic alternation of generations, such as kelps, the haploid microscopic stages have a widespread ability to tolerate and recover from stressful conditions, regardless of population. And two, that M. pyrifera from the southern limit population has developed ecotypic adaptation to thermal stress. It is important to consider, however, that the haploid stage capability to recover after exposure to El Niño stress would not necessarily be “ecologically relevant” in nature, since no diploids were produced during the two-month recovery period allowed for in this study. The diploid recovery detected in this study for the southern limit population would, on the other hand, be highly relevant ecologically, if they could go on and produce reproductive adults. A stress survival adaptation that is specific to the southern-limit diploids may provide an adaptive advantage, allowing populations at the southern limit to persist in a seed bank analogue during marginal conditions despite mass disappearance of adults across large geographic areas. Diploid physiological ecotypes may have come about due to recurrent selection for high temperature tolerance at the southern limit during warm summers and/or increased warming during El Niño events.
Haploid tolerance of extreme temperatures, light, and nutrient limitation has been previously shown for marine algae (Bolton and Luning 1982; Lee and Brinkhuis 1986; Gerard 1990; tom Dieck 1993; Santelices et al. 1995, Martinez 1999). In this study, the egg stage was shown to have the greatest recovery of the haploid stages, which might be due to the rich protoplasm produced for prolonged survival until fertilization. Resistance of eggs to adverse conditions has been demonstrated for other marine species (Trowbride 1994; Cohen and Strathmann 1996). In theory it would be more ecologically efficient if the surviving mature female gametophytes (i.e., eggs) were more stress tolerant than males. A single male [if within the 1 mm distance required (Reed et al. 1991)] could fertilize numerous surviving females if the pheromone was still active and antheridia could still release sperm. Tolerance at the haploid stage may be universal due to the reduced complexity (Lewis 1985). Haploid resistance suggests there exists large populations of microscopic stages, which are undetectable (Hoffman and Santelices 1991; Santelices et al. 1995; Ladah et al. 1999). However, studies have shown that M. pyrifera gametophytes do not survive long in nature (Deysher and Dean 1986a, b; Reed et al. 1997), and we showed they did not produce sporophytes after laboratory exposure to limiting resources. We are unable to speculate whether they could have done so had they been given more recovery time in our study.
For the diploid stage, the presence of light during stress exposure significantly increased recovery, though it was surprising to find that individuals from the southern limit population did recover even after exposure to reduced levels of nitrate and light. Recovery after exposure to reduced resources suggests that at high temperatures, limiting resources may not play such an important role as previously expected or shown at lower temperatures (e.g., 10°C, Kinlan et al. 2003). This indicates that temperature is an additional factor partially independent of light and nutrients that could control emergence of “resting” embryonic sporophytes, and that its role exhibits ecotypic variation. For the diploid microscopic stage from the southern limit, tolerance of high temperatures seems to occur regardless of other factors. Ecotypic thermal tolerance occurs in other kelps at the microscopic stages and appears to be related with their distribution (Martinez 1999).
The diploid recovery after stress exposure at high temperature detected in this study for the southern population may be explained by ecotypic adaptation to a warmer, more nutrient depleted, environment at the southern limit. However, it is important to recognize that the stressful temperature used in this study to simulate El Niño is not typical of more northern populations and generally only occurs at the southern limit in shallow kelp populations during extreme El Niño conditions. The fact that diploids recovered better if they were grown under light during the stress exposure suggests some dependence on photosynthetic processes for surviving and recovering from stress. Gerard (1990) showed similar results among light-related ecotypes in the kelp, Laminaria, where microscopic haploid gametophytes grew at the same rate regardless, of which population they originated from. The ecotypic trait did not confer any advantage in the haploid stage, whereas the diploid stage manifested the light-related ecotype trait and was more likely to survive stress when originating from the stressful environment. Ecotypic adaptation has also been shown for kelps in temperature tolerance, photosynthetic parameters, nutrient physiology, and growth (Espinoza and Chapman 1983; Kopczak et al. 1991; Gerard 1988, 1990, 1997; Gerard and Dubois 1988; Martinez 1999).
The recovery of the southern-limit diploids after exposure to stress detected in this study is highly relevant from the ecological point of view. Recovery of diploids after stress would eliminate the problems associated with egg and sperm encounters at low densities, which would most probably be the situation after a stressful El Niño event. Thus, if diploids were the persistent stage, a high density of post-event survivors in the field would not be necessary to renew an extirpated population (e.g., Dayton 1985; Martinez 1999; Kinlan et al. 2003). Diploids have been suggested to be more tolerant in some species due to advanced cellular regulation and metabolism (Maynard-Smith 1978), having the advantage of dominance as a protection against deleterious mutations (Crow and Kimura 1978; Bernstein et al. 1985; Perrot et al. 1991).
Stage-specific adaptations would be expected in a complex heteromorphic life cycle such as in the kelps, and may show the adaptive advantage of such a life history spanning a wide range of environmental conditions. In the northern population, individuals may rarely be exposed to stressful conditions (certainly rarely exposed to the treatments in this study in deeper populations), particularly if diploid production is cued by environmental factors, which allow for coordination with adequate environmental conditions (John 1994). At the southern limit, this coordination may not exist as conditions for growth and reproduction may be more sporadic, or unpredictable. Reproductive adults are more frequently wiped out over large areas at the southern limit, and rarely in the north (Ladah and Zertuche 2004), another factor that could select for resistance of the microscopic stages at the southern limit. The adaptive advantage of a resistant diploid in the southern-limit populations would be persistence of the population even in an unstable environment with a limited growth season.
Life-history characteristics in a particular population or species are often referred to as “strategies” in the sense that they confer selective advantages, which have been favored by natural selection. The ecological and evolutionary importance of the development of certain life history characteristics or strategies in brown macroalgae have been reviewed by Clayton (1990). The obligate heteromorphic alternation of generations in Macrocystis results in two very different macroscopic and microscopic stages. The ecological niche occupied by each stage in kelps is unique due to their different sizes, complexity, and physiological requirements and provides an advantage depending on conditions (e.g., Martinez and Santelices 1998). The microscopic stages live floating in the plankton or within the boundary layer on the benthos, and are often unaffected by wave action or water motion, though they are severely affected by sediment scour (Devinny and Volse 1978) and herbivory (Lubchenco and Cubit 1980; Chapman 1984; Martinez and Santelices 1998). On the other hand, the large macroscopic plant stretches throughout the water column, producing a canopy, which dominates its environment by absorbing light from the upper layers of the water column and by assimilating nutrients from its lower blades near the benthos below the thermocline. By translocation of photosynthates and amino acids, nutrients and fixed carbon are moved to growing parts of the plant, increasing productivity. Yet, larger plants run the risk of being damaged by wave action, particularly during storms or El Niño events (Seymour et al. 1989).
The strategy of a heteromorphic life history, such as in kelps, may be at an evolutionary advantage if the principal selective agent is variable rather than constant (Scagel et al. 1982; John 1994). In this sense the physical and chemical environments are divided into alternate niches for the different life history stages both temporally and spatially. In temperate and polar areas, heteromorphic algae dominate (Clayton 1987), supporting the hypothesis that a heteromorphic life history provides an adaptive advantage to changing environments. Yet, it has been argued that the possession of long-lived perennial sporophytes in kelps like M. pyrifera eliminates the need for such alternative survival strategies of tolerant microscopic stages (Clayton 1990). This would be the case in an environment where the adult population is rarely extirpated, such as in the northern site where surface canopy disappears and plants slough down to 10 m, but still remain on the bottom during El Niño (Ladah and Zertuche 2004). This, however, is clearly not the case at the southern limit of distribution, which has seen great disturbance and recurring extirpation in the past three decades (see Ladah et al. 1999).
At the southern-limit, a transition zone between temperate and subtropical conditions, temperatures can vary between 10 and 26°C. During warm summers and/or El Niño events, microscopic diploid stages may have the potential to persist in a seed bank analogue, remaining on the benthos until conditions are favorable to resume growth. This could explain the rapid recovery after the massive disappearance at the southern limit during the El Niño of 1997–1998 (Ladah et al. 1999). At the southern limit, the persistence of diploid stages during El Niño warming may be the only chance for population persistence since the southern-limit populations are fairly isolated from other reproductive populations by the Vizcaino disjuncture (400 km, see Ladah et al. 1999) and may not receive spores from such distant populations. The selective agent of temperature in the scenario of warm summers and El Niño events would theoretically place the alternation of generations of M. pyrifera at an adaptive advantage at the southern limit of its distribution in the Northern Hemipshere. This would be particularly true in light of a warming ocean, increasing frequency of El Niño events, and global climate change.
References
Bernstein H, Byerly H, Hopf F, Michod R (1985) Genetic damage, mutation, and the evolution of sex. Science 229:1277–1281
Bolton J, Luning K (1982) Optimal growth and maximal survival temperatures of Atlantic Laminaria species in culture. Mar Biol 87:131–135
Chapman ARO (1984) Reproduction, recruitment, and mortality of two species of Laminaria in southwest Nova Scotia. J Exp Mar Biol Ecol 78:99–109
Clarke A (2003). Costs and consequences of evolutionary temperature adaptation. Trends Ecol Evol 18:573–581
Clayton M (1987) Isogamy and a fucalean type of life history in the antarctic brown algae, Ascoseira mirabilis. Botanica Marina 30:447–54
Clayton M (1990) The adaptive significance of life history characters in selected orders of marine brown macroalgae. Aust J Ecol 15:439–452
Cohen CS, Strathman R (1996) Embryos at the edge of tolerance: effects of environmental and structure of egg masses on supply of oxygen to embryos. Biol Bull 190:8–15
Crow JF, Kimura M (1978) Evolution in sexual and asexual populations. Am Nat 909:439–450
Dawson Y (1951) A further study of upwelling and associated vegetation along Pacific Baja California, Mexico. J Mar Res 10:39–58
Dawson Y (1952) Circulation within Bahía Vizcaino, Baja California, and its effects on marine vegetation. Am J Bot 39:425–432
Dayton P (1973) Dispersion and persistence of the annual intertidal alga Postelsia palmaeformis. Ecology 54:433–438
Dayton P (1985) Ecology of kelp communities. Annu Rev Ecol Syst 16:215–45
Dayton P, Tegner M (1984) Catastrophic storms, El Niño, and patch stability in a southern California kelp community. Science 224:283–85
Dean T, Jacobsen FR (1984) Growth of juvenile Macrocystis pyrifera (Laminariales) in relation to environmental factors. Mar Biol 83:301–11
Devinny J, Volse L (1978) Effects of sediments on the development of Macrocystis pyrifera gametophytes. Mar Biol 48:343–348
Deysher L, Dean T (1986a). In situ recruitment of sporophytes of the giant kelp, Macrocystis pyrifera: effects of physical factors. J Exp Mar Biol Ecol 103:41–63
Deysher L, Dean T (1986b) Interactive effect of light and temperature on sporophyte production in the giant kelp, Macrocystis pyrifera. Mar Biol 93:17–20
Durazo R, Baumgartner T (2002) Evolution of oceanographic conditions off Baja California: 1997–1999. Prog Oceanogr 54:7–31
Edwards M (2004) Estimating scale-dependency in disturbance impacts: El Niños and giant kelp forests in the northeast Pacific. Oecologia 138(3):436–447
Espinoza J, Chapman ARO (1983) Ecotypic differentiation of Laminaria longicruris in relation to seawater nitrate concentration. Mar Biol 74:213–218
Fields P, Graham J, Rosenblat H, Somero G (1993) Effects of expected global climate change on marine faunas. Trends Ecol Evol 8:361–3671
Foster M, Schiel D (1985) The ecology of giant kelp forests in California: a community profile. U.S. Fish and wildlife service, Biological Report 85(7.2)
Gerard V (1984) Physiological effects of El Niño on giant kelp in southern California. Mar Biol Letts 5:317–322
Gerard V (1988) Ecotypic differentiation in light related traits of the kelp Laminaria saccharina. Mar Biol 97:25–36
Gerard V (1990) Ecotypic differentiation in the kelp Laminaria saccharina:phase-specific adaptation in a complex life cycle. Mar Biol 107:519–28
Gerard V (1997) The role of nitrogen nutrition in high-temperature tolerance of the kelp Laminaria saccharina (Chromophyta). J Phycol 33:800–810
Gerard V, Dubois K (1988) Temperature ecotypes near the southern boundary of the kelp Laminaria saccharina. Mar Biol 97:575–580
Hernández-Carmona G, Rodríguez-Montesinos Y, Casas-Valdez M, Vilchis M, Sanchez-Rodríguez I (1991) Evaluation of the beds of Macrocystis pyrifera in the Baja California Peninsula, Mexico III. Summer 1986 and seasonal variation. Ciencias Marinas 17:121–145
Hoffman A, Santelices B (1991) Banks of algal microscopic forms: hypotheses on their functioning and comparisons with seed banks. Mar Ecol Prog Ser 79:185–194
Hummel H, Amiard-Triquet C, Bachelet G, Desprez M, Marchand J, Sylvand B, Amiard J, Rybarczyk R, Boogards H, Sinke J, de Wolf L (1996) Sensitivity to stress of the estuarine bivalve Macoma balthica from areas between the Netherlands and its southern limits. J Sea Res 35:315–321
Huyer A, Smith RL (1985) The signature of El Niño off Oregon in 1982–83. J Geophys Res 90:7133–7142
Jackson GA (1977) Nutrients and production of giant kelp, Macrocystis pyrifera, off southern California. Limnol Oceanogr 22:979–995
John D (1994) Alternation of generation in algae: its complexity, maintenance and evolution. Biol Rev 69:275–91
Kahru M, Mitchell G (2000) Influence of the 1997–98 El Niño on the surface chlorophyll in the California current. J Geophys Lett 26:2937–2940
Kinlan B, Graham M, Sala E, Dayton P (2003) Arrested development of giant kelp (Macrocystis pyrifera, Phaeophyceae) embryonic sporophytes: a mechanism for delayed recruitment in perennial kelps. J Phycol 39:47–57
Kopczak C, Zimmerman R, Kremer J (1991) Variation in nitrogen physiology and growth among geographically isolated populations of the giant kelp Macrocystis pyrifera (Phaeophyta). J Phycol 27:149–158
Ladah L (2003) The shoaling of nutrient-enriched subsurface waters as a mechanism to sustain primary productivity off Central Baja California during El Niño winters. J Mar Systems 42:145–152
Ladah L, Zertuche-Gonzalez JA (2004) Giant kelp (Macrocystis pyrifera) survival in deep water (25–40 m) during El Niño of 1997–1998 in Baja California, Mexico. Botanica Marina 47:367–372
Ladah L, Zertuche-Gonzalez JA, Hernandez-Carmona G (1999) Rapid recovery giant kelp (Macrocystis pyrifera, Phaeophyceae) recruitment near its southern limit in Baja California after mass disappearance during ENSO 1997–1998. J Phycol 35:1106–1112
Lee JA, Brinkuis BH (1986) Reproductive phenology of Laminaria saccharina (L.) Lamour (Phaeophyta) at the southern limit of its distribution in the northwestern Atlantic Ocean. J Phycol 22:276–285
Lewis WM (1985) Nutrient scarcity as an evolutionary cause of haploidy. Am Nat 125:692–701
Lubchenco J, Cubit J (1980) Heteromorphic life histories of certain marine algae as adaptations to variations in herbivory. Ecology 64:1116–23
Lynn R, Simpson J (1987) The California current system: the seasonal variability of its physical characteristics. J Geophys Res 92:12947–966
Martinez E (1999) Latitudinal differences in thermal tolerance among microscopic sporophytes of the kelp Lessonia nigrescens (Phaeophyta: Laminariales). Pac Sci 53(1):74–81
Martinez E, Santelices B (1998) Selective mortality on haploid and diploid microscopic stages of Lessonia nigrescens Bory (Phaeophyta, Laminariales). J Exp Mar Biol Ecol 229:219–239
Maynard-Smith J (1978) The evolution of sex. Cambridge University Press, Cambridge, pp 222
McGowan J, Cayan D, Dorman L (1998) Climate–ocean variability and ecosystem response in the Northeast Pacific. Science 281:210–217
Miller A, Schneider N (2000) Interdecadal climate regime dynamics in the North Pacific Ocean: theories, observations and ecosystem impacts. Prog Oceanogr 47:355–379
North W (1994) Review of Macrocystis biology. In: Akatsuka I (ed) Biology of economic algae. Academic Publishing, Netherlands, pp 447–527
North W, Zimmerman R (1984) Influences of macronutrients and water temperatures on summertime survival of Macrocystis canopies. Hydrobiología 116/117:419–24
Ott F (1965) Synthetic media and techniques for the xenic culture of marine algae and flagellates. VA J Sci (N.S.)
Palacios-Hernandez E, Argote-Espinosa M, Amador-Buenrostro A, Mancilla-Peraza M (1996) Simulación de la circulación barotropica inducida por viento en Bahía Sebastián Vizcaíno, B.C. Atmosfera 9:171–188
Perrot V, Richerd S, Valero M (1991) Transition from haploidy to diploidy. Nature 351:315–317
Portner H (2002) Climate variations and the physiological basis of temperature dependant biogeography: systemic to molecular hierarchy of thermal tolerance in animals. Comp Biochem Physiol A Mol Integr Physiol 133:303–321
Provasoli L (1968) Media and prospects for the cultivation of marine algae. In: Watanabe A, A. Hattori (eds) Cultures and collections of algae. Proc US–Japan Conf. Hakone. Sept 1966. Japanese Society of Plant Physiology 63–75
Reed D, Neushul M, Ebeling A (1991) Role of density on gametophyte growth and reproduction in the kelps Macrocystis pyrifera and Pterygophora californica. J Phycol 27:361–366
Reed D, Anderson T, Ebeling A, Anghera M (1997) The role of reproductive synchrony in the colonization potential of kelp. Ecology 78(8):2443–2457
Sagarin R, Barry J, Gilman S, Baxter C (1999) Climate-related change in an intertidal community over short and long time scales. Ecol Monogr 69:465–490
Santelices B (1990) Patterns of reproduction, dispersal, and recruitment in seaweeds. Oceanogr Mar Biol Annu Rev 28:177–276
Santelices B, Hoffman A, Aedo D, Bobadilla M, Otaiza R (1995) A bank of microscopic forms on disturbed boulders and stones in tide pools. Mar Ecol Prog Ser 129:215–228
Scagel R, Bandoni R, Maze J, Rouse R, Scholfield G, Stein J (1982) Nonvascular plants. An evolutionary survey. Wadsworth publishing Company, Belmont
Seymour R, Tegner M, Dayton P, Parnell P (1989) Storm wave induced mortality of giant kelp, Macrocystis pyrifera, in southern California. Estuar Coast Shelf Sci 28:277–292
Tegner M, Dayton P (1987) El Niño effects on southern California kelp communities. Adv Ecol Res 17:243–279
Tegner M, Dayton P (1991) Sea urchins, El Niño’s, and long term stability of southern California kelp forest communities. Mar Ecol Prog Ser 77:49–63
Tegner M, Dayton P, Edwards P, Riser K (1996) Is there evidence for long-term climatic change in Southern California kelp forests? Calcofi Reports 37:111–126
tom Dieck I (1993) Temperature tolerance and survival in darkness of kelp gametophytes (Laminariales, Phaeophyta): ecological and biogeographic implications. Mar Ecol Prog Ser 100:253–264
Trowbridge CD (1994) Life at the edge: population dynamics and salinity tolerance of a high intertidal, pool-dwelling ascoglossan opisthobranch on New Zealand rocky shores. J Exp Mar Biol Ecol 182:65–84
Vermeij GJ (1980) Biogeography and adaptation––patterns of marine life. Harvard University Press, Cambridge, p 332
Zimmerman R, Kremer J (1984) Episodic nutrient supply to a kelp forest ecosystem in Southern California. J Mar Res 42:591–604
Zimmerman R, Robertson DL (1985) Effects of El Niño on local hydrography and growth of giant kelp Macrocystis pyrifera, at Santa Catalina Island, California. Limnol Oceanogr 30:1298–1302
Acknowledgements
The companies of Productos del Pacifico and Abulones Cultivados, the Fishing Cooperative in Bahia Tortugas, and the Fishing Cooperative Estaban Cantu assisted with field research logistics and permits. Diving would not have been possible without assistance from J. Guzman and R. Bermudez. Marco Aurelio provided laboratory assistance. The authors acknowledge funding and support from the UABC (4078-22) and SIMACCONACYT (970106007) grants to J.A.Z., and UC MEXUSCONACYT awards, IAI-EPCOR (CRN-062), AMELIS-CONACYT (J37689/V), and SEP-CONACYT (J50046) research grants to L.B.L. Special thanks to Dr. C. Barilloti, Dr. A. Cabello, and P. Chagoya.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by P. W. Sammarco.
Rights and permissions
About this article
Cite this article
Ladah, L.B., Zertuche-González, J.A. Survival of microscopic stages of a perennial kelp (Macrocystis pyrifera) from the center and the southern extreme of its range in the Northern Hemisphere after exposure to simulated El Niño stress. Mar Biol 152, 677–686 (2007). https://doi.org/10.1007/s00227-007-0723-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00227-007-0723-z