Abstract
Self-recruitment rates are essential parameters in the estimation of connectivity among populations, having important consequences in marine conservation biology. Using ten highly polymorphic microsatellite loci, we estimate, over 3 years, the self-recruitment in a population of Tripterygion delaisi in the NW Mediterranean. Six previously described source populations were used for the assignment (Costa Brava, Columbretes, Formentera, Cabo de Palos, Cabo de Gata and Tarifa). Even though this species has a 16–21 day larval duration, a mean of 66.4 ± 1.4% of the recruits settled in their natal population. When refining in a more local scale the origin of individuals self-recruited to Costa Brava, using as source the three sampling localities that conform this population (Cap de Creus, Tossa and Blanes), the highest percentage (40.6 ± 8.9%) was self-assigned to the adult source locality (Blanes) where recruits were sampled each year. Our results suggest that a high proportion of the larvae of T. delaisi remained close to, or never leave, their natal spawning area. This observation can be extrapolated to other species with similar early life-history traits and low adult mobility and can have important implications for the conservation and management of Mediterranean littoral fishes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
One of the main objectives of research on fish populations is to identify the factors that determine the number of new individuals recruited into the adult population (Cushing 1996). The majority of shallow-water marine species have a two-phase life cycle, in which quite sedentary, demersal adults (no mobile phase) produce pelagic larvae (mobile phase) (Leis 1991; Leis and Carson-Ewart 2000). These larvae disperse and their settlement processes can be influenced by different environmental factors, e.g. currents, winds, that determine the settlement strength (Wilson and Meekan 2001; Cowen 2002; Raventós and Macpherson 2005). For many years, it was assumed that these larvae disperse away from the parental population operating as an open system (Sale 1991; Caley et al. 1996). These initial studies considered larvae as passive particles and focused on hydrodynamic features to explain their distribution, predicting that larvae are flushed away from their natal locality in the predominant current direction (Roberts 1997). However, larvae of many fishes have been found capable to maintain strong and sustained swimming activity, as well as to use their sensory abilities to regulate their distribution and dispersion (Cowen 2002; Kingsford et al. 2002; Leis and McCormick 2002). Accordingly, some recent studies (e.g. Jones et al. 1999, 2005; Swearer et al. 1999) have demonstrated that populations are not always open and that the proportion of larvae that may return to their natal population (self-recruitment) is very high. These studies, therefore, suggest that the extent of dispersal between populations is lower than currently assumed, affecting the connectivity among populations and having important implications in marine conservation policies.
Unfortunately, at present, the number of studies is still scarce. Otolith marking and trace-element concentrations in otoliths have been used to estimate the self-recruitment rate in different fish populations (Jones et al. 1999; Swearer et al. 1999; Thorrold et al. 2001; Miller and Shanks 2004; Patterson et al. 2005). Furthermore, Jones et al. (2005) using two different methods (larval marking and parentage analyses using microsatellites) in a population of Amphiprion polymnus concluded that most settled juveniles had returned to a 2-ha natal area. Knutsen et al. (2004) using microsatellites demonstrated an extensive but temporally variable drift of offshore cod larvae into coastal populations.
Microsatellites are highly polymorphic nuclear loci that have been successfully used to describe population structuring on a wide range of geographical levels (Appleyard et al. 2001; Rico and Turner 2002; Carlsson et al. 2004). Therefore, microsatellites seem to be a powerful tool to estimate population isolation and self-recruitment levels in fishes (Knutsen et al. 2004; Jones et al. 2005).
Tripterygion delaisi is a common littoral fish in the Mediterranean Sea, living always in rocky habitats, preferentially in biotopes of reduced light, between 6 and 12 m (Zander 1986). It is a short-lived species (maximum 3 years old) and maturity reaches at the first year of life. Adult individuals are highly territorial, showing high levels of homing behaviour (Heymer 1977), parental care of the benthic eggs (Wirtz 1978) and cannot swim even short distances (tens of metres) in open water or on sandy bottoms. Larvae of T. delaisi remain in plankton for 16–21 days (Raventós and Macpherson 2001), although they are present almost exclusively in coastal waters (Sabatés et al. 2003).
Tripterygion delaisi from the Western Mediterranean shows genetic differentiation between most of its populations, although presenting small FST values (FST: 0–0.066) partly due to the high microsatellite loci polymorphism. Furthermore, significant isolation by distance was detected, suggesting the existence of a potential high degree of self-recruitment in each population (Carreras-Carbonell et al. 2006). The present study estimates, over 3 years, the self-recruitment in a population of T. delaisi in the NW Mediterranean. This could help to classify the population as genetically unconnected (closed) or connected (open), with a wide range of intermediate status depending on the percentage of recruits received from distant sources. Using ten highly polymorphic microsatellites (Carreras-Carbonell et al. 2004) we compare the new recruits of each year, with the adult reproductive specimens from the same locality and with adults from another seven adjacent localities separated by tens to hundreds of kilometres.
Materials and methods
Sampling and DNA extraction
We studied post-settlement individuals (<1 month old after settlement) of triplefin blenny (T. delaisi) from Blanes locality (North-western Mediterranean; Spain). A total of 112 specimens were collected during 2003 (n = 35; RBL03), 2004 (n = 47; RBL04) and 2005 (n = 30; RBL05) by SCUBA divers using hand nets. Each year, individuals were sampled from the same shallow rocky bay (St. Francesc—41°40.4′N, 2°48.2′E).
A small portion of the anal fin was removed from living fish, which were then measured and released into the same sample site. All fins were preserved individually in absolute ethanol at room temperature. Total genomic DNA was extracted from fin tissue using the Chelex 10% protocol (Estoup et al. 1996).
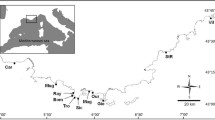
The triplefin recruits from Blanes were compared to adults from eight localities in the western Mediterranean collected in 2003 and previously analysed: Cap de Creus (CC), Tossa (TO), Blanes (BL), Columbretes Is. (CO), Formentera Is. (FO), Cabo de Palos (PA), Cabo de Gata (GA) and Tarifa (TA). Three of these localities (CC, TO and BL) presented no genetic differentiation and could not be considered isolated populations; furthermore, the existence of six populations was inferred using a Bayesian approach (Carreras-Carbonell et al. 2006). In accordance to this, we have grouped CC, TO and BL in a single population that in the present study we will refer to as Costa Brava (CB) (see Fig. 1).
Source localities of Tripterygion delaisi used in the assignment test and number of individuals analysed in each locality (n): Cap de Creus (CC, n = 20), Tossa (TO, n = 22), Blanes (BL, n = 34), Columbretes Is. (CO, n = 30), Formentera Is. (FO, n = 37), Cabo de Palos (PA, n = 30), Cabo de Gata (GA, n = 20) and Tarifa (TA, n = 33). Data obtained from Carreras-Carbonell et al. (2006). Costa Brava (CB) groups three sampling locations (CC, TO and BL), see text for details
PCR amplification and screening
We used the ten polymorphic microsatellite loci and polymerase chain reactions conditions described in Carreras-Carbonell et al. (2006). Amplified products were scored using an ABI 3700 automatic sequencer from the Scientific and Technical Services of the University of Barcelona. Alleles were sized by GENESCAN™ and GENOTYPER™ software, with an internal size marker CST Rox 70-500 (BioVentures Inc.).
Statistical analyses
Allele frequencies, mean allelic richness, expected (HE) and observed (HO) heterozygosity per locus, for each recruit year pools were calculated using GENECLASS2 program (Piry et al. 2004). The inbreeding coefficient (FIS) in each generation was computed with GENETIX version 4.05 (Belkhir et al. 2004) and its confidence interval was estimated with 10,000 bootstrapping values.
Linkage disequilibrium between pairs of loci were tested for each recruit year using GENEPOP version 3.4 (Raymond and Rousset 1995), which employs a Markov chain method, with 5,000 iterations, following the algorithm of Guo and Thompson (1992). Due to the multiple comparisons, these results were adjusted using the sequential Bonferroni procedure with ∝ = 0.05 (Rice 1989).
Genetic differentiation between the three samples of recruits of BL and the six adult source populations was estimated using the classical FST approach (Wright 1951; Weir and Cockerham 1984). Significance between each comparison pair was tested using the Fisher’s exact test implemented in GENEPOP program. These results were adjusted for multiple tests using the sequential Bonferroni procedure with ∝ = 0.05 (Rice 1989).
Population assignment test
Assignment tests were carried out using GENECLASS2 program (Piry et al. 2004) under the Bayesian assignment method of Rannala and Mountain (1997) which, according to Cornuet et al. (1999), performed better in assigning/excluding individuals to their correct sampling populations than other likelihood-based and distance-based methods. To detect individuals that could not be assigned, the simulation algorithm of Paetkau et al. (2004) was used with 105 simulations. Individuals were considered unassigned when the probabilities of being assigned to any population were lower than 0.05 (Type error I) or when they showed high probabilities of assignment to more than one population.
The six previously differentiated populations described above (Costa Brava, Columbretes Is., Formentera Is., Cabo de Palos, Cabo de Gata and Tarifa) were used as the source populations of the recruits. Afterwards, in order to estimate the self-recruitment in a smaller geographical scale, only the recruits assigned to Costa Brava population were reassigned using as source the three localities (Cap de Creus, Tossa and Blanes) that were grouped in this population.
Results
Genetic variability
An extensive polymorphism per generation and locus was found among recruit samples with high mean number of alleles (17.6 ± 1.46) and high expected (0.855 ± 0.020) and observed (0.778 ± 0.030) heterozygosities. No differences were found between the three generations sampled in the mean number of alleles (Friedman ANOVA, χ2 = 1.81, P > 0.4) and the expected heterozygosity (χ2 = 0.97, P > 0.6) (Table 1). All loci were considered statistically independent since no linkage disequilibrium between loci pairs was observed, after Bonferroni correction, in any T. delaisi generation sampled. Alleles not previously detected in any adult locality were found in each generation: five within 2003 recruits, two within 2004 recruits and seven within 2005 recruits; all of them in very low frequency.
Global FIS values for the recruits of each year were statistically significant. We observed that these departures were mainly due to loci Td08 and Td09 for the three generations. Moreover, loci Td01 and Td02 in 2003 recruits and Td02 in 2004 recruits also showed significant FIS values (Table 1). Loci Td08 and Td09 also presented deviations in all source populations (Carreras-Carbonell et al. 2006) that could be explained by the presence of null-alleles in these loci. Null-alleles appear when one allele is unamplified due to mutations in the sequence where one of the primers was designed, and/or when technical problems associated with amplification and scoring arise (Hoarau et al. 2002). Technical issue could be ruled out since accurate scoring of alleles with poor amplification was carried out and all homozygous individuals and failed amplifications for loci Td01, Td02, Td08 and Td09 were re-amplified twice lowering the annealing temperature to 50°C.
Significant genetic differentiation was found between the three samples of recruits of Blanes and all adult source populations, with the exception of Costa Brava (Table 2). The distances of the recruits of the different years were always smaller when compared to Costa Brava; however, significant genetic differentiation was found when recruits of 2003 were compared. Nevertheless we observed strong correlations when we compared the genetic distances between the recruits of 2003 to each source population with the genetic distances between the recruits of 2004 and 2005 to each source population (r ≥ 0.95, P < 0.005). This would indicate that in spite of the differences found between years their relative distances to adult populations were maintained through time (Table 2). Similar results were obtained when locus Td08 and Td09, showing null alleles, were excluded from the analyses.
Self-recruitment estimation
The assignment method of Rannala and Mountain (1997) assigned most recruits to their expected source population in all 3 years. Across the 3 years most recruits (mean = 66.4 ± 1.4%) were assigned to Costa Brava while an average of 11.0 ± 3.2% were assigned to more distant populations, and an average of 22.6 ± 2.9% were unassigned (Fig. 2). A similar percentage of self-recruitment was obtained when loci having null alleles were excluded (mean percentage across 3 years = 64.4 ± 3.0%). Furthermore, the rest of the individuals were assigned to the other source population with similar frequencies as detected when all loci were used, with Formentera Is. being the greatest contributor of recruits (mean percentage across 3 years = 12.9 ± 1.9%). The average probability of recruits to be assigned to the first population was high (95.9 ± 0.7%), while the average probability to be assigned to the next most likely population was significantly lower (3.6 ± 0.7%) (Fig. 3). No significant differences were observed when comparing across the different years the probability of each individual to be assigned to the most likely population (Kruskal–Wallis test: H = 1.37, P = 0.50). Furthermore, in order to corroborate the results of the assignment test, we compared the presence of shared private alleles between recruits and source populations after correcting for the number of individuals per population. More shared private alleles were significantly found when the recruits were compared to Costa Brava than to all other populations (Wilcoxon matched pairs test, Z = 2.69, P = 0.007).
Percentage of recruits of the year of T. delaisi assigned to each source population over 3 years (2003, 2004 and 2005). Population abbreviations as in Fig. 1. U unassigned individuals, not included in any previously defined source population
Probability of assignment (%) for each recruit to each population. Note: u unassigned individuals. Individuals were considered unassigned when the probabilities of being assigned to any population were lower than 0.05 (Type error I) or whenever they showed high probabilities of assignment to more than one population
In order to refine in a more local scale the origin of the recruits, the individuals that were assigned to Costa Brava were reassigned to the three sampling localities (Cap de Creus, Tossa and Blanes). Most individuals were strongly assigned to one of these three localities with an average probability to be assigned to the first population of 96.8 ± 0.7, and 3.0 ± 0.7% for the next most likely population. A small percentage of unassigned individuals was observed for the 2003 recruits, being the percentage of unassigned for the other two years much larger (Fig. 4). The highest percentage of the recruits was usually self-assigned to Blanes with mean percentage across three years of 40.6 ± 8.9%, while smaller percentages were found for Tossa (21.1 ± 4.4%) and Cap de Creus (14.2 ± 3.0%) (Fig. 4). When loci with null alleles were excluded, the mean percentage of assigned recruits became more similar among the three localities (BL = 29.8.7 ± 2.8%, TO = 21.9 ± 3.6%, CC = 22.5 ± 2.6%).
Discussion
Self-recruitment rates are essential parameters in the estimation of connectivity among populations, having important consequences in marine conservation biology (Swearer et al. 2002; Thorrold et al. 2002). Self-recruitment studies in marine fishes are scarce, and they have used different methodologies, e.g. chemical marking (Jones et al. 1999, 2005), otolith microstructure and/or microchemistry (Swearer et al. 1999; Thorrold et al. 2001; Miller and Shanks 2004; Patterson et al. 2005), and more recently adding molecular techniques (Jones et al. 2005). Molecular markers have demonstrated their utility in assigning the origin of colonizers invading new areas, mainly in continental habitats (Genton et al. 2005). Assignment tests and paternity analyses have been used to establish the origin of cod (Knutsen et al. 2004) and clownfish (Jones et al. 2005) recruits, respectively, demonstrating the utility of this methodology in the estimation of self-recruitment rates. In the present work, assignment tests were very robust, since similar results were obtained with and without loci having null alleles. Nevertheless, we used all loci to identify the origin of recruits since increasing the number of loci seems to yield higher statistical power when estimating the number of populations (Carreras-Carbonell et al. 2006). The validity of the assignment method is sensitive to the level of population differentiation. As suggested by Cornuet et al. (1999) a correct assignment rate can be achieved by scoring ten microsatellites on 30–50 individuals, as in the present case, when FST is near 0.1. The FST values among our populations are quite small since FST values are highly influenced by the polymorphism of the markers (Carreras-Carbonell et al. 2006). Nevertheless our T. delaisi populations are highly genetically differentiated; consequently it is not the FST value itself what is relevant to achieve accuracy of the recruits’ assignment but the genetic differentiation among populations.
In T. delaisi, self-recruitment was very high as revealed by the assignment tests. During the three years studied, the self-recruitment in the Costa Brava population ranged between 63.8 and 68.6%. However, among the three years there is a mean percentage of 11.0 ± 3.2% of recruits assigned to other populations, mainly belonging to the geographically nearest ones (Columbretes Is. and Formentera Is.). Furthermore, the percentage of assignment to the most likely population was generally high (Fig. 3). Given the life span of this species, the correct assignment test was that comparing the recruits of 2003 with the six adult genetically differentiated populations sampled in the same year (Fig. 2). Nevertheless, we observed a similar percentage of recruits assigned to each adult population over the three years indicating certain genetic temporal stability. Furthermore, we obtained similar FST values when we compared the recruits of each year to each adult population, indicating that the genetic relationships among populations did not change over time (Table 2).
When we used the criterion algorithm of Rannala and Mountain (1997) and the simulation algorithm of Peatkau et al. (2004) to detect first-generation migrants among the source populations used in the present work, we detected 14 individuals with a probability below 0.01 that were assigned to a different population. This is in agreement with the isolation by distance observed among these western Mediterranean populations (Carreras-Carbonell et al. 2006), indicating that, although the populations were genetically differentiated, a small connexion between them could exist, allowing the interchange of individuals (via larvae) between populations (FST: 0.009–0.066).
When the recruits assigned to Costa Brava population were reassigned to a finer scale using as source the three localities that are part of this population separately (Cap de Creus, Tossa and Blanes), we observed that, on average, the highest proportion of the recruits settled in their natal locality (mean percentage across three years of 40.6 ± 8.9%, Blanes) (Fig. 4). However, the recruit contribution of the other two localities was also high, reinforcing the idea that these three localities conformed a homogeneous population (Costa Brava) as suggested in Carreras-Carbonell et al. (2006). In spite of the absence of genetic differentiation among the three adult populations, 57% of the assignment test of the recruits of the year 2003 pointed to Blanes as the most likely source population. This result suggests the presence of high self-recruitment even at smaller scales and with low genetic differentiation. Given the life span of the species and the low genetic differentiation between the Costa Brava localities, these assignments should be done within the same year and could not be correct among years due to annual variability. Therefore, the percentage of recruits of the year 2004 and 2005 assigned within the Costa Brava population should be considered with caution in agreement with the increase of unassigned recruits in these two years (Fig. 4).
We can conclude that the vast majority of larvae remain close to, or never leave, their population. Our results are in agreement with the high self-recruitment levels obtained in other studies (Jones et al. 1999, 2005; Swearer et al. 1999; Thorrold et al. 2001; Miller and Shanks 2004; Patterson et al. 2005), suggesting that the extent of dispersal between populations is lower than currently assumed. However, extensive inter-ocean scale dispersal has been revealed in the reef fish Naso vlamingii, indicating that dispersion may be highly influenced by the nature of the life cycles of species (Klanten et al. 2006).
Self-recruitment rate and, in general, gene flow among populations can be related with spawning characteristics and larval and adult strategies, e.g. Riginos and Victor 2001; Planes 2002 (however, see Shulman and Bermingham 1995). Eggs of T. delaisi are demersal and larvae remain in plankton for 16–21 days (Raventós and Macpherson 2001); however, these larvae are present almost exclusively in waters close to adult habitats during the spawning season (Sabatés et al. 2003). Thus, some retention mechanisms must be acting in these larvae during the mobile phase, since self-recruitment results imply that a significant percentage of spawned larvae come back to, or never leave, their natal population.
The inshore larval distribution of T. delaisi would determine that these species have lower dispersal possibilities than species with larvae situated offshore (Shanks and Eckert 2005). These differential dispersal capabilities could be due to stronger transport currents offshore than inshore (Tintoré et al. 1995; Largier 2003). Furthermore, larvae from benthic eggs, as those of T. delaisi, are larger, better swimmers, and have more developed sensory systems than larvae from pelagic eggs (Blaxter 1986). The combination of these characteristics may make retention more likely for larvae from benthic spawners than for larvae from pelagic spawners. Additionally, T. delaisi have planktonic larvae in spring-summer, when the wind regime (inshore winds) (Lloret et al. 2004) prevents dispersal of larvae promoting high self-recruitment rate. As Shanks and Eckert (2005) pointed out, the early life traits of many species may show an adaptation to the local oceanography, to avoid the alongshore loss of larvae. This promotes the settlement of larvae into their parental habitats. Nevertheless, self-recruitment could be also influenced by local adaptation and not only by retention, since larvae from distant populations may fail to settle due to habitat selection and overwhelm the homogenizing effect of dispersal (Rocha et al. 2005).
The high self-recruitment rate observed in T. delaisi can be extrapolated to other species with short planktonic larval duration, larvae situated inshore and low adult mobility, e.g. Gobiesocidae, Syngnathidae (Macpherson and Raventós 2006) and can have important implications for the conservation and management of Mediterranean littoral fishes. Furthermore, T. delaisi populations are genetically isolated when large discontinuities of sand or deep-water channels (>30 km) are present among them, preventing larval and adult exchange (Carreras-Carbonell et al. 2006). The results observed in the present paper and works from other authors (see references cited above) suggest that larval retention and current population isolation can be more elevated than presently assumed. These parameters are essential in the estimation of the population connectivity among areas, and are critical for sizing and spacing marine protected areas (Sala et al. 2002; Cowen et al. 2006). Therefore, in order to maintain the connectivity among marine reserves in the western Mediterranean, the location of these protected zones may consider the degree of genetic isolation among populations, and the existence of geographic and ecological discontinuities that prevent gene flow among areas.
References
Appleyard SA, Grewe PM, Innes BH, Ward RD (2001) Population structure of yellowfin tuna (Thunnus albacares) in the western Pacific Ocean, inferred from microsatellite loci. Mar Biol DOI 10.1007/s002270100578
Belkhir K, Borsa P, Chikhi L, Raufaste N, Bonhomme F (2004) GENETIX 4.05, logiciel sous Windows TM pour la génétique des populations. Laboratoire Génome, Populations, Interactions, CNRS UMR 5171, Université de Montpellier II, Montpellier (France)
Blaxter JHS (1986) Development of sense organs and behaviour of teleost larvae with special reference to feeding and predator avoidance. Trans Am Fish Soc 115:98–114
Caley MJ, Carr MH, Hixon MA, Hughes TP, Jones GP, Menge BA (1996) Recruitment and the local dynamics of open marine populations. Annu Rev Ecol Syst 27:477–500
Carlsson J, McDowell JR, Díaz-James P (2004) Microsatellite and mitochondrial DNA analyses of Atlantic bluefin tuna (Thunnus thynnus thynnus) population structure in the Mediterranean Sea. Mol Ecol DOI 10.1111/j.1365–294X.2004.02336.x
Carreras-Carbonell J, Macpherson E, Pascual M (2004) Isolation and characterization of microsatellite loci in Tripterygion delaisi. Mol Ecol Notes DOI 10.1111/j.1471-8286.2004.00688.x
Carreras-Carbonell J, Macpherson E, Pascual M (2006) Population structure within and between subspecies of the Mediterranean triplefin fish Tripterygion delaisi revealed by highly polymorphic microsatellite loci. Mol Ecol DOI 10.1111/j.1365-294X.2006.03003.x
Cornuet JM, Piry S, Luikart G, Estoup A, Solignac M (1999) New methods employing multilocus genotypes to select or exclude populations as origins of individuals. Genetics 153:1989–2000
Cowen RK (2002) Larval dispersal and retention and consequences for population connectivity. In: Sale P (ed) Coral reef fishes; diversity and dynamics in a complex ecosystem. Academic, San Diego, pp 149–170
Cowen RK, Paris CB, Srinivasan A (2006) Scaling of connectivity in marine populations. Science DOI 10.1126/science.1122039
Cushing DH (1996) Towards a science of recruitment in fish populations. Ecology Institute, Oldendorf
Estoup A, Largiadèr CR, Perrot E, Chourrout D (1996) Rapid one–tube DNA extraction for reliable pcr detection of fish polymorphic markers and transgenes. Mol Mar Biol Biotechnol 5:295–298
Genton BJ, Shykoff JA, Giraud T (2005) High genetic diversity in French invasive populations of common ragweed, Ambrosia artemisiifolia, as a result of multiple sources of introduction. Mol Ecol DOI 10.1111/j.1365-294X.2005.02750.x
Guo SW, Thompson EA (1992) Performing the exact test for Hardy-Weinberg proportions for multiple alleles. Biometrics 48:361–372
Heymer A (1977) Expériences subaquatiques sur les performances d’orientation et de retour au gite chez Tripterygion tripteronotus et Tripterygion xanthosoma (Blennioidei, Tripterygiidae). Vie et Milieu 3e sér 27:425–435
Hoarau G, Rijnsdorp AD, Van der Veer HW, Stam WT, Olsen JL (2002) Population structure of plaice (Pleuronectes platessa l.) in northern Europe: microsatellites revealed large-scale spatial and temporal homogeneity. Mol Ecol DOI 10.1046/j.1365-294X.2002.01515.x
Jones GP, Milicich MJ, Emslie MJ, Lunow C (1999) Self-recruitment in a coral reef fish population. Nature DOI 10.1038/45538
Jones GP, Planes S, Thorrold SR (2005) Coral reef fish larvae settle close to home. Curr Biol DOI 10.1016/j.cub.2005.06.061
Kingsford MJ, Leis JM, Shanks A, Lindeman KC, Morgan SG, Pineda J (2002) Sensory environments, larval abilities and local self-recruitment. Bull Mar Sci 70:309–340
Klanten OS, Choat JH, van Herwerden L (2006) Extreme genetic diversity and temporal rather than spatial partitioning in a widely distributed coral reef fish. Mar Biol DOI 10.1007/s00227-006-0372-7
Knutsen H, André C, Jorde PE, Skogen MD, Thuróczy E, Stenseth NC (2004) Transport of North Sea cod larvae into the Skagerrak coastal populations. Proc R Soc Lond B DOI 10.1098/rspb.2004.2721
Largier JL (2003) Considerations in estimating larval dispersal distances from oceanographic data. Ecol Appl 13:S71–S89
Leis J, McCormick M (2002) The biology, behaviour and ecology of the pelagic, larval stage of coral reef fishes. In: Sale P (ed) Coral reef fishes; diversity and dynamics in a complex ecosystem. Academic, San Diego, pp 171–200
Leis JM (1991) The pelagic stages of reef fishes: the larval biology of coral reef fishes. In: Sale PF (ed) The ecology of fishes on coral reefs. Academic, San Diego, pp 183–230
Leis JM, Carson-Ewart BM (eds) (2000) The larvae of Indo-Pacific coastal fishes: an identification guide to marine fish larvae, 1st edn. Brill, Boston
Lloret J, Palomera I, Salat J, Sole I (2004) Impact of freshwater input and wind on landings of anchovy (Engraulis encrasicolus) and sardine (Sardina pilchardus) in shelf waters surrounding the Ebre (Ebro) River delta (north-western Mediterranean). Fish Oceanogr DOI 10.1046/j.1365-2419.2003.00279.x
Macpherson E, Raventós N (2006) Relationships between pelagic larval duration and geographic distribution in Mediterranean littoral fishes. Mar Ecol Prog Ser (in press)
Miller JA, Shanks AL (2004) Evidence for limited larval dispersal in black rockfish (Sebastes melanops): implications for population structure and marine-reserve design. Can J Fish Aquat Sci DOI 10.1139/F04-111
Paetkau D, Slade R, Burden M, Estoup A (2004) Genetic assignment methods for the direct, real-time estimation of migration rate: a simulation-based exploration of accuracy and power. Mol Ecol DOI 10.1046/j.1365-294X.2004.02008.x
Patterson HM, Kingsford MJ, McCulloch MT (2005) Resolution of the early life history of a reef fish using otolith chemistry. Coral Reefs DOI 10.1007/s00338-004-0469–8
Piry S, Alapetite A, Cornuet JM, Paetkau D, Baudouin L, Estoup A (2004) GENECLASS2: a software for genetic assignment and first-generation migrant detection. J Hered DOI 10.1093/jhered/esh074
Planes S (2002) In: Sale PF (ed) The ecology of fishes on coral reefs. Academic, San Diego, pp 201–220
Rannala B, Mountain JL (1997) Detecting immigration by using multilocus genotypes. Proc Natl Acad Sci USA 94:9197–9201
Raventós N, Macpherson E (2001) Planktonic larval duration and settlement marks on the otoliths of Mediterranean littoral fishes. Mar Biol DOI 10.1007/s002270000535
Raventós N, Macpherson E (2005) Environmental influences on temporal patterns of settlement in two littoral labrid fishes in the Mediterranean Sea. Estuar Coast Shelf Sci DOI 10.1016/j.ecss.2004.11.018
Raymond M, Rousset F (1995) GENEPOP: Population genetics software for exact tests and ecumenism, version 1.2. J Hered 86:248–249
Rice WR (1989) Analysing tables of statistical tests. Evolution 43:223–225
Rico C, Turner GF (2002) Extrem microallopatric divergence in a cichlid species from Lake Malawi. Mol Ecol DOI 10.1046/j.1365-294X.2002.01537.x
Riginos C, Victor BC (2001) Larval spatial distributions and other early life-history characteristics predict genetic differentiation in eastern Pacific blennioid fishes. Proc R Soc Lond B 268:1931–1936
Roberts CM (1997) Connectivity and management of Caribbean coral reefs. Science DOI 10.1126/science.278.5342.1454
Rocha LA, Robertson DR, Roman J, Bowen BW (2005) Ecological speciation in tropical reef fishes. Proc R Soc Lond B 272:573–579
Sabatés A, Zabala M, Garcia-Rubies A (2003) Larval fish communities in the Medes Islands marine reserve (north-west Mediterranean). J Plankton Res 25:1035–1046
Sala E, Aburto-Oropeza O, Paredes G, Parra I, Barrera JC, Dayton PK (2002) A general model for designing networks of marine reserves. Science DOI 10.1126/science.1075284
Sale PF (1991) Reef fish communities: open nonequilibrial systems. In: Sale PF (ed) The ecology of fishes on coral reefs. Academic, San Diego, pp 564–598
Shanks AL, Eckert G (2005) Population persistence of California Current fishes and benthic crustaceans: a marine drift paradox. Ecol Monogr 75:505–524
Shulman MJ, Bermingham E (1995) Early life histories, ocean currents, and the population genetics of Caribbean reef fishes. Evolution 49:897–910
Swearer SE, Caselle JE, Lea DW, Warner RR (1999) Larval retention and recruitment in an island population of a coral-reef fish. Nature DOI 10.1038/45533
Swearer SE, Shima JS, Hellberg ME, Thorrold SR, Jones GP, Robertson DR, Morgan SG, Selkoe KA, Ruiz GM, Warner RR (2002) Evidence of self-recruitment in demersal marine populations. Bull Mar Sci 70:251–271
Thorrold SR, Latkoczy C, Swart PK, Jones CM (2001) Natal homing in a marine fish metapopulation. Science DOI 10.1126/science.291.5502.297
Thorrold SR, Jones GP, Hellberg ME, Burton RS, Swearer SE, Neigel JE, Morgan SG, Warner RR (2002) Quantifying larval retention and connectivity in marine populations with artificial and natural markers. Bull Mar Sci 70:291–308
Tintoré J, Wang DP, García E, Viúdez A (1995) Near inertial motions in the coastal ocean. J Mar Sys 6:301–312
Weir BS, Cockerham CC (1984) Estimating F-statistics for the analysis of population structure. Evolution 38:1358–1370
Wilson DT, Meekan MG (2001) Environmental influences on patterns of larval replenishment in coral reef fishes. Mar Ecol Prog Ser 222:197–208
Wirtz P (1978) The behaviour of the Mediterranean Tripterygion species (Pisces, Blennioidei). Zeitschrift für Tierpsychologie 48:142–174
Wright S (1951) The genetical structure of populations. Ann Hum Genet 15:323–354
Zander CD (1986) Tripterygiidae. In: Whitehead PJP, Bauchot ML, Hureau JC, Nielsen J, Tortonese E (eds) Fishes of the North-Eastern Atlantic and the Mediterranean, vol 3. Unesco, Paris, pp 1118–1121
Acknowledgments
We thank S. Planes his helpful comments. This research was supported by a Predoctoral fellowship from the Ministerio de Educación, Cultura y Deporte to J.C. (AP2001-0225). Research was funded by projects CTM2004-05265 and BOS2003-05904 of the MCYT and MMA 119/2003. Researchers are part of the SGR 2005SGR-00995 and 2005SGR-00277 of the Generalitat de Catalunya. All the experiments made comply with the current Spanish laws.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by S.A. Poulet, Roscoff.
Rights and permissions
About this article
Cite this article
Carreras-Carbonell, J., Macpherson, E. & Pascual, M. High self-recruitment levels in a Mediterranean littoral fish population revealed by microsatellite markers. Mar Biol 151, 719–727 (2007). https://doi.org/10.1007/s00227-006-0513-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00227-006-0513-z