Abstract
We obtained baseline and follow-up bone mineral density (BMD) values of the lumbar spine from sagittal reformations of routine abdominal contrast-enhanced multidetector computed tomography (MDCT) using a reference phantom and assessed their performance in differentiating patients with no, existing, and incidental osteoporotic fractures of the spine. A MDCT-to-QCT (quantitative computed tomography) conversion equation for lumbar BMD measurements was developed by using 15 postmenopausal women (63 ± 12 years), who underwent standard lumbar QCT (L1–L3) and afterward routine abdominal contrast-enhanced MDCT. Sagittal reformations were used for corresponding lumbar BMD measurements. The MDCT-to-QCT conversion equation was applied to baseline and follow-up routine abdominal contrast-enhanced MDCT scans of 149 postmenopausal women (63 ± 10 years). Their vertebral fracture status (no, existing, or incidental osteoporotic fracture) was assessed in the sagittal reformations. A correlation coefficient of r = 0.914 (p < 0.001) was calculated for the BMD values of MDCT and standard QCT with the conversion equation BMDQCT = 0.695 × BMDMDCT − 7.9 mg/mL. Mean follow-up time of the 149 patients was 20 ± 12 months. Fifteen patients (10.1 %) had an existing osteoporotic vertebral fracture at baseline. Incidental osteoporotic vertebral fractures were diagnosed in 13 patients (8.7 %). Patients with existing and incidental fractures showed significantly (p < 0.05) lower converted BMD values (averaged over L1–L3) than patients without fracture at baseline and at follow-up. In this longitudinal study, BMD values of the lumbar spine derived from sagittal reformations of routine abdominal contrast-enhanced MDCT predicted incidental osteoporotic vertebral fractures.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Osteoporosis is defined as a skeletal disorder characterized by compromised bone strength predisposing an individual to an increased risk of fracture [1]. The prevalence of osteoporosis is increasing with the aging population and prolonged life expectancy [2]. Due to its association with increased risk of fracture, osteoporosis is classified as a public health problem [3]. Osteoporotic fractures, in particular spine and hip fractures, have a high morbidity and mortality and generate immense financial cost [4–6]. Therefore, different methods have been established to identify patients at high risk of osteoporotic fractures and to initiate appropriate therapy. These include dual-energy X-ray absorptiometry (DXA) and quantitative computed tomography (QCT) of the spine measuring bone mineral density (BMD) [7–10]. Since routine abdominal multidetector computed tomography (MDCT) is one of the most frequently used radiologic examinations, it would be beneficial to use the obtained MDCT images to conduct additional BMD measurements of the spine in case clinical risk factors for osteoporosis are present in the examined patient. In particular, patients with cancer would benefit from these measurements since they routinely undergo MDCT examinations and are often at increased risk of osteoporosis due to the cancer-related treatment [11, 12]. Since many MDCT examinations are solely performed with intravenous contrast medium, correspondingly measured BMD values have to be converted to standard QCT equivalent BMD values, which are better for fracture risk prediction. Previous studies showed the potential of routine abdominal contrast-enhanced MDCT to determine the BMD of the lumbar spine using either axial images or sagittal reformations [13–15]. Since osteoporotic vertebral fractures can be substantially better detected in sagittal reformations of abdominal MDCT studies compared to axial MDCT images [16–18], it may be advantageous and time-saving to use only the sagittal reformations for both vertebral BMD measurements and assessment of vertebral fracture status.
It was demonstrated recently that lumbar BMD measurements derived from sagittal reformations of routine abdominal contrast-enhanced MDCT scans could differentiate postmenopausal women with versus without osteoporotic vertebral fracture [14]. However, it remains to be investigated if these BMD measurements are also able to predict incidental (i.e., newly occurred) osteoporotic vertebral fractures. If this ability were demonstrated, patients who routinely undergo abdominal MDCT examinations due to various medical indications and are at high risk for osteoporotic vertebral fractures could be identified and appropriate therapy could be initiated.
Therefore, the purpose of the study was to obtain baseline and follow-up BMD values of the lumbar spine from sagittal reformations of routine abdominal contrast-enhanced MDCT in postmenopausal women and to assess their performance in differentiating postmenopausal women with no, existing, and incidental osteoporotic fractures of the spine.
Materials and Methods
This retrospective study was approved by the Institutional Committee on Human Research. The patients gave consent for scientific evaluation of material at the time of admission.
Patients
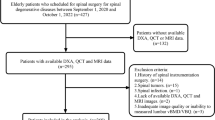
Postmenopausal women older than 50 years were retrospectively identified in our institution’s digital image archive picture archiving communication system (PACS), who underwent both standard lumbar QCT and routine abdominal contrast-enhanced MDCT within 3 months to develop a MDCT-to-QCT conversion equation for lumbar BMD. Patients with pathological bone changes like bone metastases, hematological disorders, or metabolic bone disorders aside from osteoporosis were excluded from the study. Fifteen postmenopausal women with a mean ± standard deviation (SD) age of 63 ± 12 years met the inclusion criteria and were included in the study.
The MDCT-to-QCT conversion equation for lumbar BMD was calculated as outlined below and applied to baseline and follow-up routine abdominal contrast-enhanced MDCT examinations of 149 postmenopausal women older than 50 years. These patients were retrospectively identified in our institution’s digital image archive (PACS). They had a history of cancer (such as esophageal, colorectal, or breast cancer) and chemotherapy but no pathological bone changes other than osteoporosis. They underwent MDCT examinations as long-term follow-up to rule out tumor recurrence. Only patients with a follow-up routine abdominal contrast-enhanced MDCT of at least 6 months were included in the study. Standard lumbar QCT was not performed for these patients. The mean ± SD age of the 149 postmenopausal women was 63 ± 10 years.
Standard QCT
All standard lumbar QCT examinations were performed with a 64-row MDCT scanner (Somatom Sensation Cardiac 64; Siemens Medical Solutions, Erlangen, Germany). A reference phantom (Osteo Phantom, Siemens Medical Solutions) was placed on the scanner mat beneath the patients. A standard protocol according to the manufacturer’s instructions was used. Tube voltage was 80 kVp, tube load was 140 mAs, and axial slice thickness was 10 mm. Based on lateral scout images of the lumbar spine of the patients, the implemented automated Osteo software (Siemens Medical Solutions) was applied to select the midvertebral sections of L1–L3 vertebrae. Axial image slices were acquired in these midvertebral sections. The orientation of each image slice was always parallel to the upper and lower endplates of the corresponding vertebra by tilting the gantry appropriately before each acquisition. The Osteo software automatically fitted a region of interest (ROI) in the trabecular part of each vertebra, and trabecular BMD of L1–L3 was determined. Fractured vertebrae were excluded. QCT analyses were performed by experienced radiology technologists who were supervised by a radiologist.
Routine Abdominal Contrast-Enhanced MDCT
All routine abdominal contrast-enhanced MDCT scans were performed with the same 64-row MDCT scanner and reference phantom as used for standard lumbar QCT examinations. The routine abdominal contrast-enhanced MDCT images were obtained with a standard protocol. Scanning parameters were 120 kVp tube voltage, adapted tube load of averaged 200 mAs, and minimum collimation (0.6 mm), creating 5-mm axial sections. Sagittal reformations of the spine were reconstructed with a slice thickness of 3 mm. Examinations were performed after standardized administration of intravenous contrast medium (Imeron 400; Bracco, Konstanz, Germany) using a high-pressure injector (Fresenius Pilot C; Fresenius Kabi, Bad Homburg, Germany). Intravenous contrast medium injection was performed with a delay of 70 s, a flow rate of 3 mL/s, and a body weight–dependent dose (80 mL for body weight up to 80 kg, 90 mL for body weight up to 100 kg, and 100 mL for body weight over 100 kg). Additionally, all patients were given 1,000 mL oral contrast medium (Barilux Scan; Sanochemia Diagnostics, Neuss, Germany).
Lumbar BMD analysis in the routine abdominal contrast-enhanced MDCT scans were performed as previously described [14]: sagittal reformations were loaded into the institutional PACS (Sectra AB, Linköping, Sweden), and the most central slice of L1–L3 vertebrae was selected. The density measurement tools of the PACS software were used to place manually circular ROIs in the ventral halves of the trabecular compartment of the vertebral bodies of L1–L3, in each case equidistant to both endplates (Fig. 1). Thus, the impact of contrast medium accumulation in the vertebral venous plexus on BMD measurements was reduced. The ROI diameter of each vertebra was standardized to two-thirds vertebral height. ROIs were not placed in fractured vertebrae. The attenuation values measured in the ROIs in Hounsfield units had to be converted into milligrams of calcium hydroxylapatite per milliliter. For that purpose, attenuation values of the two phases of the reference phantom in Hounsfield units were measured in circular ROIs manually placed in the phantom. The placement of all ROIs was performed by a radiologist. The time effort for ROI placement and BMD calculation was less than 1 minute for one MDCT examination.
The vertebral fracture status of the 149 patients was assessed in the sagittal reformations of the MDCT studies at baseline and at follow-up. Osteoporotic vertebral fractures were classified by a radiologist according to the spinal fracture index described by Genant et al. [19]. Existing osteoporotic vertebral fractures were defined as fractures already present in the baseline images. Incidental osteoporotic vertebral fractures were defined as newly occurred fractures in the follow-up images (Fig. 2).
Statistical Analysis
The MDCT-to-QCT conversion equation for BMD measurements was computed with linear regression analysis. Using that conversion equation, a leave-one-out cross-validation was performed to evaluate the future prediction errors [20]. The calculated prediction errors were summarized as the root mean squared error (RMSE, mg/mL) and the coefficient of variation (CV) of the RMSE (%).
The 149 postmenopausal women were divided in three groups according to vertebral fracture status (no, existing, or incidental osteoporotic vertebral fracture). ANOVA was used to determine statistically significant differences in age and follow-up time between the three groups. Linear regression models, adjusting for age and follow-up time, were used to compare the converted BMD values of patients with no, existing, and incidental osteoporotic vertebral fracture at baseline and follow-up. Receiver operator characteristic (ROC) analysis was performed, and the area under the ROC curve (AUC) was used to evaluate the overall diagnostic performance of the acquired BMD data to differentiate patients with existing versus no vertebral fracture, respectively, with incidental versus no vertebral fracture. Felsenberg and Gowin [21] suggested that lumbar BMD values below 80 mg/mL are indicative of osteoporosis. Logistic regression models, adjusting for age, were used to calculate the odds ratio and 95 % confidence interval (CI) for patients with converted BMD values below 80 mg/mL at baseline to have an existing osteoporotic vertebral fracture at baseline to develop an incidental osteoporotic vertebral fracture during follow-up.
Statistical analyses were performed with SPSS (SPSS, Inc., Chicago, IL) and R (R Foundation for Statistical Computing, Vienna, Austria) and supervised by a statistician. All tests were done using a two-sided 0.05 level of significance.
Reproducibility
Reproducibility errors for lumbar BMD measurements in the sagittal reformations of routine abdominal contrast-enhanced MDCT studies were reported previously [14]. Short- and long-term reproducibility errors for BMD measurements amounted to 2.09 % (3.61 mg/mL) and 7.70 % (11.48 mg/mL), respectively. Short-term reproducibility error for the BMD measurements was assessed by placing ROIs three times in each vertebra (L1–L3) in the sagittal reformations of contrast-enhanced MDCT studies of 30 randomly selected patients. To determine the long-term reproducibility error, 30 patients with two consecutive contrast-enhanced MDCT examinations within 8 weeks were identified and BMD of L1–L3 was measured in the sagittal reformations in each scan one time.
Results
Forty-one lumbar vertebral bodies were selected for calculation of the MDCT-to-QCT conversion equation in 15 patients. A correlation coefficient of r = 0.914 (p < 0.001) was calculated for the BMD values of contrast-enhanced MDCT and standard QCT with the MDCT-to-QCT conversion equation BMDQCT = 0.695 × BMDMDCT − 7.9 mg/mL (Fig. 3). The leave-one-out cross-validation showed an RMSE of 16.1 mg/mL and CV of the RMSE of 16.7 %.
The mean ± SD follow-up time of the 149 postmenopausal women was 20 ± 12 months. Fifteen patients (10.1 %) had an existing osteoporotic vertebral fracture at baseline. Incidental osteoporotic vertebral fractures were diagnosed in 13 patients (8.7 %). The remaining 121 patients (81.2 %) had no osteoporotic vertebral fracture at baseline as well as at follow-up. While the age of patients with no, existing, and incidental osteoporotic vertebral fractures was significantly different (62 ± 10 vs. 69 ± 11 vs. 68 ± 8 years, respectively; p = 0.004), the three groups showed similar follow-up times (20 ± 13 vs. 21 ± 12 vs. 17 ± 9 months, respectively; p = 0.744).
Patients with existing and incidental osteoporotic vertebral fracture showed lower converted BMD values (averaged over L1–L3) than patients without fracture at baseline (73.9 ± 18.3 vs. 76.6 ± 28.8 vs. 103.7 ± 30.7 mg/mL, respectively) (Fig. 4). After adjustment for age and follow-up time, converted BMD values at baseline were still significantly different between patients without fracture and with existing fracture (Table 1, p = 0.010) and between patients without fracture and with incidental fracture (Table 1, p = 0.019). Converted BMD values decreased over time in all three groups. The BMD decrease amounted 4.0 ± 18.9 versus 1.3 ± 10.9 versus 7.1 ± 24.3 mg/mL in patients with no, existing, and incidental osteoporotic vertebral fracture, respectively. Differences in BMD decrease between the three groups were not statistically significant (p = 0.730). Similar to baseline, higher converted BMD values were observed at follow-up in patients without osteoporotic vertebral fracture compared to patients with existing and incidental osteoporotic vertebral fracture (99.7 ± 30.4 vs. 72.6 ± 21.9 vs. 69.5 ± 24.0 mg/mL, respectively) (Fig. 4). BMD differences between patients without fracture and with existing fracture and between patients without fracture and with incidental fracture were still statistically significant after adjustment for age and follow-up time (p < 0.05, Table 1).
Box plots for baseline and follow-up mean (L1–L3) converted BMD values (mg/mL) in patients with no, existing, and incidental osteoporotic vertebral fracture. Converted BMD values were significantly (p < 0.05, Table 1) lower in patients with existing and incidental fracture compared to patients without fracture
An AUC value of 0.791 (p < 0.001) was computed in the ROC analysis for converted BMD values at baseline to differentiate patients with existing versus no vertebral fracture. AUC = 0.782 (p = 0.001) was obtained for converted BMD values at baseline to differentiate patients with incidental versus no vertebral fracture.
The odds ratio (95 % CI, adjusted p value) for patients with converted BMD values below 80 mg/mL at baseline to have an existing osteoporotic vertebral fracture at baseline amounted to 3.65 (1.12–12.04, p = 0.033). An odds ratio (95 % CI, adjusted p value) of 9.00 (2.20–36.87, p = 0.002) was calculated for patients with converted BMD values below 80 mg/mL at baseline to develop an incidental osteoporotic vertebral fracture during follow-up.
Discussion
In this longitudinal study, we obtained BMD values of the lumbar spine from sagittal reformations of routine abdominal contrast-enhanced MDCT in postmenopausal women using a reference phantom placed on the scanner mat beneath the patients. A reliable MDCT-to-QCT conversion equation was developed for these lumbar BMD measurements. The converted BMD values at baseline were able not only to differentiate adequately between patients with existing versus no osteoporotic vertebral fracture but also to predict incidental osteoporotic vertebral fractures.
DXA and QCT are the standard methods to assess BMD of the lumbar spine [7–9]. However, routine abdominal MDCT studies are one of the most frequently used radiologic examinations. If the examined patient has clinical risk factors for osteoporosis, it may be beneficial to use MDCT images to conduct additional BMD measurements of the spine. Previous studies demonstrated that routine abdominal non-contrast-enhanced and contrast-enhanced MDCT as well as CT examinations of the heart can be used to determine BMD of the spine [13–15, 22, 23]. Similar to these studies, we developed a MDCT-to-QCT conversion equation for lumbar BMD measurements. Due to the application of intravenous contrast medium, BMD values derived from the sagittal reformations of contrast-enhanced MDCT were considerably higher than those obtained with standard QCT. This finding is consistent with previous studies [13–15]. BMD values measured with contrast-enhanced MDCT and standard QCT showed a high correlation coefficient (r = 0.914) and an acceptable prediction error (16.7 %), which are comparable to previous studies [13–15]. The conversion equation is limited to the CT scanner and the protocols that we used. However, the equation can easily be adapted to different CT scanners and protocols by performing the described calibration procedure.
Similar to a previous study, we used the sagittal reformations and not the axial images of routine abdominal contrast-enhanced MDCT to perform lumbar BMD measurements [14]. It is advantageous to measure BMD of the lumbar spine in the same reformations that are known for a substantially better detection of vertebral fractures [16–18]. Furthermore, lumbar BMD was measured with low technical effort and time by using simple PACS measurement tools. Thus, assessment of vertebral fracture status and measurement of lumbar BMD can be performed in the same image plane in a reliable and time-saving way, which is required for application in everyday clinical practice.
This study demonstrated that lumbar BMD measurements from sagittal reformations of routine abdominal contrast-enhanced MDCT could predict incidental (i.e., newly occurred) osteoporotic vertebral fractures. This finding has an important clinical impact. Patients who routinely undergo abdominal MDCT examinations due to various medical indications and are at high risk for osteoporotic vertebral fractures could be identified and appropriate therapy could be initiated. In particular, elderly patients and patients undergoing chemotherapy due to cancer treatment are at increased risk of osteoporosis and may benefit from these additional BMD quantifications [11, 12, 24, 25]. The MDCT scans used for this study were clinically indicated for tumor follow-up care. Without further radiation exposure, lumbar BMD measurements could be performed in this study population and incidence of osteoporotic vertebral fractures could be predicted by converted lumbar BMD values.
Reproducibility of the obtained lumbar BMD measurements is critical in this longitudinal study. Long-term reproducibility errors of 1–4 % were reported for standard QCT measurements with automated software [26, 27]. The manual, but time-saving, BMD measurements in the sagittal reformations had a short-term reproducibility error of 2.09 % and a long-term reproducibility error of 7.70 % as reported previously [14]. The major limitation of the lumbar BMD measurements in the sagittal reformations of routine abdominal contrast-enhanced MDCT is the imponderability of the intravenous contrast medium. Contrast medium administration was standardized in this study. However, variations in the blood supply of the vertebral bodies can induce an inhomogeneous contrast enhancement. Therefore, the administration of intravenous contrast medium may decrease reproducibility.
This study had further limitations. Firstly, by using the MDCT-to-QCT conversion equation, BMD values derived from contrast-enhanced MDCT can be compared with defined BMD thresholds of standard QCT to diagnose osteopenia/osteoporosis [21]. However, MDCT-derived BMD cannot be used for the determination of World Health Organization T scores and application in FRAX (Fracture Risk Assessment in Osteoporosis) [28, 29]. Secondly, the sample size for the calculation of the MDCT-to-QCT conversion equation was relatively small due to the required simultaneous clinical indication of standard QCT and routine abdominal contrast-enhanced MDCT. Therefore, we did not calculate conversion equations for each L1–L3 vertebra. Thirdly, mean follow-up time was 20 ± 12 months, and a rather small percentage of the study population (8.7 %) had an incidental osteoporotic vertebral fracture. A higher percentage of incidental osteoporotic vertebral fractures may be diagnosed by extended follow-up times, thus strengthening the statistical analysis. Lastly, vertebral bone marrow changes were found in specific patient populations. Lower bone marrow perfusion and increased bone marrow fat content were observed in osteoporotic compared to healthy subjects [30–32]. Therefore, the developed BMD conversion equation may be limited to the investigated postmenopausal women. However, the lumbar BMD measurements in the sagittal reformations of routine abdominal contrast-enhanced MDCT may also be important in different patient populations such as men in an oncological setting. Therefore, further studies are required in the future to evaluate the utility of these BMD measurements in patient populations other than postmenopausal women.
In this longitudinal study, BMD values of the lumbar spine derived from sagittal reformations of routine abdominal contrast-enhanced MDCT predicted incidental osteoporotic vertebral fractures and differentiated postmenopausal women with and without osteoporotic vertebral fractures. Based on these findings, BMD measurements from routine MDCT studies have the potential to assess fracture risk for osteoporotic vertebral fractures without further radiation exposure in patients who routinely undergo MDCT examinations and are often at increased risk of osteoporosis, e.g., due to cancer-related treatment.
References
NIH Consensus Development Panel on Osteoporosis Prevention, Diagnosis, and Therapy (2001) March 7–29, 2000: highlights of the conference. South Med J 94(6):569–573
Cole ZA, Dennison EM, Cooper C (2008) Osteoporosis epidemiology update. Curr Rheumatol Rep 10(2):92–96
Holroyd C, Cooper C, Dennison E (2008) Epidemiology of osteoporosis. Best Pract Res Clin Endocrinol Metab 22(5):671–685
Ioannidis G, Papaioannou A, Hopman WM, Akhtar-Danesh N, Anastassiades T, Pickard L, Kennedy CC, Prior JC, Olszynski WP, Davison KS, Goltzman D, Thabane L, Gafni A, Papadimitropoulos EA, Brown JP, Josse RG, Hanley DA, Adachi JD (2009) Relation between fractures and mortality: results from the Canadian Multicentre Osteoporosis Study. CMAJ 181(5):265–271
Leboime A, Confavreux CB, Mehsen N, Paccou J, David C, Roux C (2010) Osteoporosis and mortality. Joint Bone Spine 77(Suppl 2):S107–S112
Johnell O, Kanis J (2005) Epidemiology of osteoporotic fractures. Osteoporos Int 16(Suppl 2):S3–S7
Adams JE (2009) Quantitative computed tomography. Eur J Radiol 71(3):415–424
Bergot C, Laval-Jeantet AM, Hutchinson K, Dautraix I, Caulin F, Genant HK (2001) A comparison of spinal quantitative computed tomography with dual energy X-ray absorptiometry in European women with vertebral and nonvertebral fractures. Calcif Tissue Int 68(2):74–82
Blake GM, Fogelman I (2010) An update on dual-energy X-ray absorptiometry. Semin Nucl Med 40(1):62–73
Damilakis J, Maris TG, Karantanas AH (2007) An update on the assessment of osteoporosis using radiologic techniques. Eur Radiol 17(6):1591–1602
Gralow JR, Biermann JS, Farooki A, Fornier MN, Gagel RF, Kumar RN, Shapiro CL, Shields A, Smith MR, Srinivas S, Van Poznak CH (2009) NCCN Task Force report. Bone health in cancer care. J Natl Compr Canc Netw 7(Suppl 3):S1–S32
Guise TA (2006) Bone loss and fracture risk associated with cancer therapy. Oncologist 11(10):1121–1131
Bauer JS, Henning TD, Mueller D, Lu Y, Majumdar S, Link TM (2007) Volumetric quantitative CT of the spine and hip derived from contrast-enhanced MDCT: conversion factors. AJR Am J Roentgenol 188(5):1294–1301
Baum T, Muller D, Dobritz M, Rummeny EJ, Link TM, Bauer JS (2011) BMD measurements of the spine derived from sagittal reformations of contrast-enhanced MDCT without dedicated software. Eur J Radiol 80(2):e140–e145
Link TM, Koppers BB, Licht T, Bauer J, Lu Y, Rummeny EJ (2004) In vitro and in vivo spiral CT to determine bone mineral density: initial experience in patients at risk for osteoporosis. Radiology 231(3):805–811
Muller D, Bauer JS, Zeile M, Rummeny EJ, Link TM (2008) Significance of sagittal reformations in routine thoracic and abdominal multislice CT studies for detecting osteoporotic fractures and other spine abnormalities. Eur Radiol 18(8):1696–1702
Bauer JS, Muller D, Ambekar A, Dobritz M, Matsuura M, Eckstein F, Rummeny EJ, Link TM (2006) Detection of osteoporotic vertebral fractures using multidetector CT. Osteoporos Int 17(4):608–615
Williams AL, Al Busaidi A, Sparrow PJ, Adams JE, Whitehouse RW (2009) Under-reporting of osteoporotic vertebral fractures on computed tomography. Eur J Radiol 69(1):179–183
Genant HK, Wu CY, van Kuijk C, Nevitt MC (1993) Vertebral fracture assessment using a semiquantitative technique. J Bone Miner Res 8(9):1137–1148
Barnhart HX, Haber MJ, Lin LI (2007) An overview on assessing agreement with continuous measurements. J Biopharm Stat 17(4):529–569
Felsenberg D, Gowin W (1999) Bone densitometry by dual energy methods [in German]. Radiologe 39(3):186–193
Lenchik L, Shi R, Register TC, Beck SR, Langefeld CD, Carr JJ (2004) Measurement of trabecular bone mineral density in the thoracic spine using cardiac gated quantitative computed tomography. J Comput Assist Tomogr 28(1):134–139
Papadakis AE, Karantanas AH, Papadokostakis G, Petinellis E, Damilakis J (2009) Can abdominal multi-detector CT diagnose spinal osteoporosis? Eur Radiol 19(1):172–176
Smith MR (2006) Treatment-related osteoporosis in men with prostate cancer. Clin Cancer Res 12(20 Pt 2):6315s–6319s
Yamamoto DS, Viale PH (2009) Update on identifying and managing osteoporosis in women with breast cancer. Clin J Oncol Nurs 13(5):E18–E29
Bligh M, Bidaut L, White RA, Murphy WA Jr, Stevens DM, Cody DD (2009) Helical multidetector row quantitative computed tomography (QCT) precision. Acad Radiol 16(2):150–159
Braillon PM (2002) Quantitative computed tomography precision and accuracy for long-term follow-up of bone mineral density measurements: a five year in vitro assessment. J Clin Densitom 5(3):259–266
WHO Study Group (1994) Assessment of fracture risk and its application to screening for postmenopausal osteoporosis. Report of a WHO Study Group. World Health Organ Tech Rep Ser 843:1–129
Kanis JA, Oden A, Johansson H, Borgstrom F, Strom O, McCloskey E (2009) FRAX and its applications to clinical practice. Bone 44(5):734–743
Griffith JF, Yeung DK, Antonio GE, Lee FK, Hong AW, Wong SY, Lau EM, Leung PC (2005) Vertebral bone mineral density, marrow perfusion, and fat content in healthy men and men with osteoporosis: dynamic contrast-enhanced MR imaging and MR spectroscopy. Radiology 236(3):945–951
Griffith JF, Yeung DK, Antonio GE, Wong SY, Kwok TC, Woo J, Leung PC (2006) Vertebral marrow fat content and diffusion and perfusion indexes in women with varying bone density: MR evaluation. Radiology 241(3):831–838
Li X, Kuo D, Schafer AL, Porzig A, Link TM, Black D, Schwartz AV (2011) Quantification of vertebral bone marrow fat content using 3 Tesla MR spectroscopy: reproducibility, vertebral variation, and applications in osteoporosis. J Magn Reson Imaging 33(4):974–979
Acknowledgments
This work was supported by grants of the Deutsche Forschungsgemeinschaft (DFG BA 4085/1-2).
Author information
Authors and Affiliations
Corresponding author
Additional information
The authors have stated that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Baum, T., Müller, D., Dobritz, M. et al. Converted Lumbar BMD Values Derived from Sagittal Reformations of Contrast-Enhanced MDCT Predict Incidental Osteoporotic Vertebral Fractures. Calcif Tissue Int 90, 481–487 (2012). https://doi.org/10.1007/s00223-012-9596-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-012-9596-3