Abstract
This study reports on ethnic differences in bone mass before and after adjusting for differences in body size and bone area (BA). Lumbar spine (LSBMC), proximal femur (PFBMC) and femoral neck (FNBMC) bone mineral contents were measured in black (‘black’; n = 263) and white (‘white’; n = 73) children from Johannesburg and children of mixed ancestral origin (‘mixed’; n = 64) from Cape Town, South Africa. Geometric estimates and the power coefficient from the regression analyses of BMC on BA were calculated. After adjusting for age, weight, and height, LSBMC in girls and FNBMC in girls and boys were greatest in mixed, followed by black and then white, groups. Mixed boys and girls also had greater PFBMC than their black and white peers, but only in the boys was PFBMC greater in the black than the white groups. When including BA in the adjustment, differences remained at the FN in boys and girls, and the LS and PF in girls, but disappeared at the PF in mixed and black boys. The difference in LSBMC between mixed and black boys became significant after adjustment for age, weight, height, and BA. Geometric estimates at the femoral neck were greater in the mixed group. Power coefficients were greater in the white group, suggesting differences in shape or bone distribution. In conclusion, this study suggests that, in addition to differences in BMC, differences in bone strength and geometry are present which might confer advantages to the bone of mixed-ancestry children.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Ethnic differences in bone mass have been extensively investigated in adults and children living in developed countries such as the United States (USA), the United Kingdom, and New Zealand [1, 2], with very few studies reporting on ethnic differences in developing countries. Previous cross-sectional South African data comparing black and white female nurses reported significantly higher proximal femur bone mineral density (BMD) in black women, however, no ethnic differences in BMD of the lumbar spine or radius were found after adjusting for differences in height [3]. Similarly, ethnic differences in bone mass between black and white South African pre- and early pubertal children have been reported by Vidulich et al. [4]. Their study showed that after adjusting for anthropometric and pubertal differences, black children had a greater femoral neck (FN), proximal femur (PF), and midradius bone mineral content (BMC), however, there were no differences at the whole body, lumbar spine (LS), or distal radius.
Most of the interethnic studies completed in South Africa have compared the black and white populations, with recent data from our laboratory reporting on differences in whole body BMC between black and white children, and children of mixed ancestral origin [5]. The mixed ancestral origin community has been historically disadvantaged socioeconomically and nutritionally, and has a high prevalence of maternal smoking and alcohol consumption during pregnancy [6]. We found that whole body BMC was lower in white children than black children, irrespective of whether they were residing in the USA or South Africa, and that South African children had significantly higher BMC compared to North American children of the same age. In addition, the children of mixed ancestral origin had the highest whole body BMC, a finding which was unexpected and of particular interest given the poor socioeconomic and adverse early life experiences in this community.
When comparing bone mass results of children derived from dual-energy X-ray absorptiometry (DXA), it is important to control for differences in body size [7, 8]. Size-related artifacts, as suggested by Prentice et al. [9], may result in differences between ethnic groups that are not real. It is for this reason that additional analyses that account for differences in body and bone size, such as calculation of the power coefficient describing the relationship between BMC and bone area (BA) [9], and estimation of the geometric properties of the bone such as section modulus and cross-sectional area proposed by Beck et al. [10], should be considered when comparing ethnic groups to more accurately describe bone health in these populations.
Thus the aims of this study are twofold: to describe the differences in site-specific BMC and strength among South African black, white, and mixed ancestry children and to investigate various methods of correcting for body size when interpreting BMC.
Materials and Methods
Subjects
Data from a sample of 400 children from three ethnic groups, aged 9 years, who participated in studies conducted at the Department of Human Biology, University of Cape Town, and Department of Paediatrics, University of Witwatersrand, Johannesburg, were examined. The local ethics committees approved both studies, and written informed consent was obtained from the mothers or primary caregivers prior to testing of the children.
The Johannesburg sample consisted of 336 participants (73 white subjects—38 male and 35 female; and 263 black subjects—142 male and 121 female) recruited from the Birth to Twenty cohort, a longitudinal study of child health and development of infants born within a 6-week period (April 23 to June 8, 1990) in the greater Johannesburg metropolitan area [11, 12]. These children were part of a subsample of 682 children stratified by ethnic group (black and white), gender, and socioeconomic status who, participating in the Birth to Twenty cohort study, were enrolled in a longitudinal study assessing factors influencing bone mass during childhood and adolescence (Bone Health Study) in 1999. Only prepubertal children were included in this study.
Sixty-four subjects of mixed ancestral origin (mixed group) from a working-class community in Cape Town were also recruited. This community is indigenous to South Africa and its members are descended from slaves brought from East and central Africa, the indigenous Khoisan who lived in the Cape at the time, indigenous Africans, and white settlers. They were part of a larger follow-up study investigating various determinants of growth and development in children. An original cohort of 253 mothers had participated in a nutrition and pregnancy study during which data on birth weight, maternal nutritional status, and anthropometry were collected. A follow-up to this study required subjects and their mothers to visit the university laboratory to complete further testing, including DXA measurements, which could not be done in the field. The majority of the children were not available for further testing for various reasons including relocation, unavailability, and other commitments, and for the purposes of this study, only 64 children were tested. Only prepubertal children were included in the study.
Body Composition
At both sites, height and weight were measured using similar methodologies, with participants wearing light clothing. Whole body fat mass and lean mass (fat-free soft tissue) and BMC and BA at the LS and the PF (including total PF and FN) were measured using Hologic QDR bone densitometers (QDR Discovery-W in Cape Town and QDR 4500A in Johannesburg).
Quality assurance checks were carried out on each machine prior to scanning. In Cape Town, a LS phantom was scanned at least three times a week to determine the intrinsic coefficient of variation of the machine, which, during the course of the study, was 0.32% for BMD and 0.53% for BMC. In Johannesburg, a spine phantom was scanned daily and the coefficients of variation for BMD and BMC were 0.35 and 0.48%, respectively. At each testing site a single trained DXA technician performed all scans. Intraobserver variation was 0.87% for Cape Town at the left hip and 0.98% at the LS and < 1% for Johannesburg at all skeletal sites. For analyses of the FN, the FN box size default setting was used in the majority of cases, however, if the technician felt it was necessary, the box size was adjusted to accommodate children of different sizes.
To ensure the comparability of data from both sites, we assessed the calibration difference between the two DXA instruments used. The same three-step calibration phantom obtained from Hologic (Bedford, MA, USA) was scanned 10 times on both densitometers. Pixel values were generated for the block phantom and the mean value of mass was calculated for each step of the phantom. The Cape Town values were regressed on the Johannesburg values to generate a linear calibration function. The regression formula predicting the Hologic QDR Discovery W (Cape Town) value from the Hologic QDR4500A (Johannesburg) readings indicates a strong association, with a slope of nearly 1: y = 0.9728x – 0.1951. The subject data obtained from Cape Town were appropriately adjusted using the above equation as recommended for multisite DXA studies [13, 14].
Power Coefficient
To determine the relationship between BMC and BA at each of the measured bone sites, BA and BMC were converted to their natural logarithms, and the regression coefficient obtained from the regression analysis of BMC on BA is known as the power coefficient [9]. This is the power to which BA should be raised to appropriately adjust for BA differences.
Structural Geometry of the Femoral Neck
Geometric parameters, including section modulus (Z; cm3) and cross-sectional area (cm2) of the FN, were estimated from the DXA output using various formulas (T. J. Beck, personal communication). The development of these equations to estimate geometry and its contribution to bone strength has been discussed extensively in the literature for adults and children [10, 15, 16]. The equations are included in the Appendix.
Statistical Analysis
The Statsoft (Statistica v8.0, 2007) package and the free programming environment, R (R Development Core team, 2006), were used for all statistical analyses. All analyses were completed separately for boys and girls. Data are summarized as means (standard deviations) or medians (interquartile range) depending on their distribution. We used linear models to assess the difference between ethnic groups for all numerical variables (age, weight, height, and bone parameters) with and without adjusting for covariates. Adjusted data are presented as mean (95% CI). Where significant group differences were found, we used Tukey’s HSD (honestly significant differences) procedure to compare pairs of groups. All effect estimates (β) and P-values are from Tukey’s HSD unless stated otherwise.
Results
Descriptive characteristics for the various ethnic groups are presented in Table 1. The mean age difference between the black and the white groups was not significant, however, both were slightly older than the mixed group, by 2–3 months (P < 0.001 for all four comparisons). White and black boys were significantly heavier than mixed boys (white β = 6.73, P < 0.001; black β = 3.56, P = 0.05), however, the weights were similar among the girls. White boys were significantly taller than black boys (β = 4.00, P = 0.001), who were significantly taller than mixed boys (β = 4.47, P < 0.001). White girls were significantly taller than both black (β = 3.37, P = 0.046) and mixed (β = 5.11, P = 0.014) girls. There was no difference in BMI among any of the groups.
Unadjusted and adjusted statistics, and indicators of significant differences in BA at the three sites among the various ethnic groups, are presented in Table 2. Unadjusted LS BA was significantly lower in the mixed boys and girls compared to the other two groups. After adjusting for height, these differences were no longer significant in the boys, however, LS BA was significantly higher in black girls compared to the other two groups (white β = 1.31, P = 0.021; mixed β = 1.53, P = 0.013). Unadjusted PF BA was higher in the white boys and girls compared to the other groups. After adjusting for height, there was no difference in PF BA between the girls, however, the mixed boys had a significantly greater PF BA than the other two groups (black β = 0.189, P < 0.001; white β = 2.095, P < 0.001). Unadjusted FN BA was significantly higher in the boys and girls of the mixed group, and after adjusting for height these differences remained significant, with the mixed boys and girls having a greater BA than the white (boys β = 0.440, P < 0.001; girls β = 0.501, P < 0.001) and black (boys β = 0.456, P < 0.001; girls β = 0.501, P < 0.001) groups.
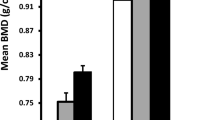
Unadjusted and adjusted statistics, and indicators of significant differences in BMC at the three sites among the various ethnic groups, are presented in Table 3 and graphically in Figs. 1, 2, 3. After adjusting for age, weight, and height, LSBMC was not different among the boys but was highly significantly different between the three groups of girls. The white girls had the lowest and the mixed girls the highest LSBMC (mixed-black β = 1.95, P < 0.001; black-white β = 2.09, P < 0.001; mixed-white β = 4.04, P < 0.001). When including BA in the adjustment, adjusted LSBMC was higher in the mixed group of boys compared to the other two groups, and differences remained significant in the girls. PFBMC (adjusted for age, weight, and height) was greater in the mixed boys and girls compared to the black (boys β = 1.77, P < 0.001; girls β = 1.93, P < 0.001) and white (boys β = 3.22, P < 0.001; girls β = 2.74, P < 0.001) groups. In addition, adjusted PFBMC was significantly higher in black boys and girls than white boys (β = 1.45, P < 0.001) and girls (β = 0.81, P = 0.026), respectively. When including BA in the adjustment, all groups remained significantly different from each other, except the difference was no longer significant between the black and the mixed boys. Adjusted for age, weight, and height, FNBMC was significantly higher in the mixed boys and girls compared to the black boys and girls (boys β = 0.479, P < 0.001; girls β = 0.571, P < 0.001), who, in turn, had a higher adjusted FNBMC than their white peers (boys β = 0.073, P < 0.001; girls β = 0.811, P < 0.001). These differences remained significant even when including BA in the adjustment.
Lumbar spine bone mineral content (LSBMC) for boys and girls: (a) unadjusted, (b) means adjusted for age, weight, and height, and (c) means adjusted for age, weight, height, and bone area, with simultaneous 95% confidence intervals. T-bars representing the confidence intervals overlap if the differences in means are not significant
Proximal femur bone mineral content (PFBMC) for boys and girls: (a) unadjusted, (b) means adjusted for age, weight, and height, and (c) means adjusted for age, weight, height, and bone area, with simultaneous 95% confidence intervals. T-bars representing the confidence intervals overlap if the differences in means are not significant
Femoral neck bone mineral content (FNBMC) for boys and girls: (a) unadjusted, (b) means adjusted for age, weight, and height, and (c) means adjusted for age, weight, height and bone area, with simultaneous 95% confidence intervals. T-bars representing the confidence intervals overlap if the differences in means are not significant
Power coefficients were calculated by linear-regression analysis of log(BMC) on log(BA) at the three skeletal sites, LS, PF, and FN, for the three ethnic groups, and are presented in Table 4. A power coefficient of 1 indicates that BMC is directly proportional to BA. The power coefficient was significantly > 1, but not different from 1.5, at all the anatomical sites for the white group (LS, 1.64 ± 0.10; PF,: 1.38 ± 0.12; FN, 1.59 ± 0.17), but only at the LS for the black group (LS, 1.27 ± 0.07) and at none of the sites for the mixed ancestry group. When adjusting BMC at the various sites for age and BA(power), there was no difference in the R 2 value compared to that adjusting for age and BA. R 2 values did, however, improve when including height and weight in the regression model, suggesting that adjusting BMC for BA(power) is not sufficient to account for differences in body size.
Cross-sectional area of the FN was significantly different among all the groups for boys (white, 1.55 ± 0.20 cm2, vs. black, 1.62 ± 0.21 cm2, vs. mixed, 1.83 ± 0.24 cm2; P < 0.001) and girls (white, 1.28 ± 0.21 cm2, vs. black, 1.39 ± 0.20 cm2, vs. mixed, 1.72 ± 0.25 cm2; P < 0.001). Section modulus (Z) was also significantly greater (P < 0.001) in the mixed groups (boys, 0.95 ± 0.25; girls, 0.91 ± 0.36) compared to the black (boys, 0.67 ± 0.18; girls, 0.54 ± 0.16) and white (boys, 0.70 ± 0.17; girls, 0.55 ± 0.19) groups. Adjusting for height and whole body lean mass did not alter these differences.
Discussion
This is the first study to compare site-specific (LS, PF, and FN) BA and BMC, and geometric measures of bone strength, between mixed ancestry, black, and white South African children. The significantly greater size-adjusted BA and BMC in the mixed group compared to the other two ethnic groups is of interest, as this is a population that has been characterized by a higher overall prevalence of socioeconomic and nutritional deprivation, as well as stunting and underweight for age [17]. A previous study comparing the bone mass of 10-year-old black and white South African children found that black children had a greater BMC at the FN and total hip than white children but a LSBMC similar to that of white children [4]. In the present study of 9 year olds, although the difference in LSBMC between the black and the white children was less than at the two hip sites, it was still significant. Possible reasons for this discrepancy in results between the two studies may be the difference in pubertal status, as all members of the present 9-year-old cohort were prepubertal, while some (± 1%) of the 10-year-old cohort were classified as midpubertal (Tanner stages 3–4). As differences in body size and other significant predictors were adjusted for in the present study, it seems likely that genetic factors may play a role in the ethnic differences, and must be investigated further. In addition, although environmental influences such as physical activity are important for the development of bone mass at this age [18], South African data suggest that this cannot explain the higher BMC in black children compared to white children, as physical activity is generally lower in black children [19].
Many researchers who adjust DXA-derived bone mineral data in children for body size do so assuming that BMC and BA, or height, are directly proportional to each other. This assumption is generally not true, as it does not take into account the associated changes in the third dimension of bone (depth) during growth. To take into account the influence of depth or height, a power coefficient of 1.5 would be expected if the dimensions of the shape are assumed to approximate those of a cube [7]. In the present study, the power coefficients at the LS, the PF, and the FN in the white group were not significantly different from 1.5. However, in the black group the power coefficients for the PF and the FN, and in the mixed group the power coefficients for all three sites, were not significantly different from 1. This finding suggests that factors other than the three-dimensional changes in size are important in determining the relationship between BA and BMC. These other factors may include differences in the distribution of trabecular and cortical bone at the various sites during growth, but verification of this requires further research. Support for this is provided by the finding of a higher cortical cross-sectional area at the FN in both black and mixed children compared to white children. Although the mixed children had a greater BA than black and white children at that site, black children had a BA similar to that of the white children, thus excluding bone size being responsible for the difference in cross-sectional area between black and white children.
The differences in adjusted bone mass, power coefficients, and geometric analyses between the ethnic groups may also have implications for fracture prevalence. Recent findings from the Birth to Twenty cohort in South Africa have shown a significant difference in fracture rates between the ethnic groups over the first 9 years of life, with ±53, ±18, and ±14 fractures per 1000 children/year in the white, black, and mixed groups respectively [20]. The significantly lower adjusted BMC of white children in our sample and the higher fracture prevalence in a comparable sample of white children support previous research that confirms bone mass to be an important predictor of fracture risk in children [21]. Whether the significantly higher power coefficient between BA and BMC in the white children in our study, which may be indicative of different bone dimensions or bone distribution, is also associated with a higher fracture prevalence needs to be investigated more closely.
The results of this study suggest that adjustment for bone size, and not just body size, is important when comparing bone parameters between groups of children. Our results show that height differences may account for BA differences at the LS, however, differences in BA at the PF and the FN become either significant or more significant between the mixed group and the other two groups after adjusting for height. Calculating the relationship between BMC and BA using the actual data, rather than the assumed relationship, will result in a BMC value that is independent of bone size and therefore easier to compare among groups of children of differing ethnic backgrounds and varying weights and heights. The literature suggests that this relationship changes during growth, particularly during puberty [22]. In a 5-year longitudinal study of Chinese girls, it was found that the relationship between BA and BMC changed in the years prior to, during, and after menarche [22]. In a similar study of Canadian girls, the size-corrected BMD declined prior to age at peak height velocity and then showed a rapid increase [23].
It is clear that there are a number of limitations when measuring BMC in children using DXA, due to the constantly changing bone and body size. In this study we have investigated possible differences in bone mass among various South African ethnic groups, independent of size, by adjusting for differences in bone and body size, using power coefficients, as well as differences in geometric calculations of cross-sectional area and section modulus. We have shown ethnic differences in bone mass at the LS and PF similar to those shown previously for the whole body after adjusting for body size [5], which suggest that these site differences may be due to differing bone shapes and/or internal structural differences. In addition, the significant ethnic differences in the geometric estimates, section modulus, and cross-sectional area suggest that measuring or estimating the geometric properties of bone is also essential when comparing groups and assessing fracture risk. Our findings now need to be investigated further by more direct measures of geometric properties such as pQCT and MRI.
Conclusion
We have shown a significantly higher BMC at the LS, PF, and FN of a mixed-ancestral group of 9-year-old children compared to black and white children of a similar age. These differences are not due to differences in body size, as size differences were adjusted for. The results are concordant with our previous work showing similar patterns in the whole body skeleton. These differences are remarkable in that, despite significant social inequalities in South Africa, there are potentially strong genetic factors protecting bone mass in the mixed and black ethnic groups. Additional research is necessary to investigate whether there is a significant association between higher size-adjusted bone mass and decreased fracture risk in mixed-ancestry children and adolescents. Furthermore, research is also needed to investigate changes in the geometric properties of the bone during growth in these groups and the effect that these might have on bone mass and strength.
References
Weaver CM, McCabe LD, McCabe GP et al (2007) Bone mineral and predictors of bone mass in white, Hispanic, and Asian early pubertal girls. Calcif Tissue Int 81:352–363
Grant AM, Gordon FK, Ferguson EL et al (2005) Do young New Zealand Pacific Island and European children differ in bone size or bone mineral? Calcif Tissue Int 76:397–403
Daniels ED, Pettifor JM, Schnitzler CM, Russell SW, Patel DN (1995) Ethnic differences in bone density in female South African nurses. J Bone Miner Res 10:359–367
Vidulich L, Norris SA, Cameron N, Pettifor JM (2006) Differences in bone size and bone mass between black and white 10-year-old South African children. Osteoporos Int 17:433–440
Micklesfield LK, Norris SA, Nelson DA, Lambert EV, van der ML, Pettifor JM (2007) Comparisons of body size, composition, and whole body bone mass between North American and South African children. J Bone Miner Res 22:1869–1877
Croxford J, Viljoen D (1999) Alcohol consumption by pregnant women in the Western Cape. S Afr Med J 89:962–965
Carter DR, Bouxsein ML, Marcus R (1992) New approaches for interpreting projected bone densitometry data. J Bone Miner Res 7:137–145
Webber CE, Sala A, Barr RD (2009) Accounting for body size deviations when reporting bone mineral density variables in children. Osteoporos Int 20:113–121
Prentice A, Parsons TJ, Cole TJ (1994) Uncritical use of bone mineral density in absorptiometry may lead to size-related artifacts in the identification of bone mineral determinants. Am J Clin Nutr 60:837–842
Beck T (2003) Measuring the structural strength of bones with dual-energy X-ray absorptiometry: principles, technical limitations, and future possibilities. Osteoporos Int 14(Suppl 5):S81–S88
Richter LM, Yach D, Cameron N, Griesel RD, de Wet T (1995) Enrolment into Birth to Ten (BTT): population and sample characteristics. Paediatr Perinat Epidemiol 9:109–120
Richter LM, Norris SA, de Wet T (2004) Transition from Birth to Ten to Birth to Twenty: the South African cohort reaches 13 years of age. Paediatr Perinat Epidemiol 18:290–301
Pearson J, Dequeker J, Reeve J et al (1995) Dual X-ray absorptiometry of the proximal femur: normal European values standardized with the European Spine Phantom. J Bone Miner Res 10:315–324
Pearson J, Dequeker J, Henley M et al (1995) European semi-anthropomorphic spine phantom for the calibration of bone densitometers: assessment of precision, stability and accuracy. The European Quantitation of Osteoporosis Study Group. Osteoporos Int 5:174–184
Bonnick SL (2007) HSA: beyond BMD with DXA. Bone 41:S9–S12
Macdonald HM, Kontulainen SA, Petit MA, Beck TJ, Khan KM, McKay HA (2008) Does a novel school-based physical activity model benefit femoral neck bone strength in pre- and early pubertal children? Osteoporos Int 19:1445–1456
Zere E, McIntyre D (2003) Inequities in under-five child malnutrition in South Africa. Int J Equity Health 2:7
Mackelvie KJ, Khan KM, McKay HA (2002) Is there a critical period for bone response to weight-bearing exercise in children and adolescents? A systematic review. Br J Sports Med 36:250–257
McVeigh JA, Norris SA, Cameron N, Pettifor JM (2004) Associations between physical activity and bone mass in black and white South African children at age 9 yr. J Appl Physiol 97:1006–1012
Thandrayen K, Norris SA, Pettifor JM (2008) Fracture rates in urban South African children of different ethnic origins: The Birth to Twenty Cohort. Osteoporos Int 20:47–52
Clark EM, Ness AR, Bishop NJ, Tobias JH (2006) Association between bone mass and fractures in children: a prospective cohort study. J Bone Miner Res 21:1489–1495
Zhu K, Greenfield H, Zhang Q et al (2008) Growth and bone mineral accretion during puberty in Chinese girls: a five-year longitudinal study. J Bone Miner Res 23:167–172
Faulkner RA, Davison KS, Bailey DA, Mirwald RL, Baxter-Jones AD (2006) Size-corrected BMD decreases during peak linear growth: implications for fracture incidence during adolescence. J Bone Miner Res 21:1864–1870
Acknowledgments
Appreciation is extended to all funders and research teams of the respective studies presented in this paper. The Cape Town study was funded by the International Atomic Energy Agency. The Johannesburg study was funded by the Wellcome Trust (UK), South African Medical Research Council, and University of the Witwatersrand, Johannesburg. Dr. Micklesfield holds the Hillel Friedland Postdoctoral Fellowship at the University of the Witwatersrand and Dr. Norris holds a Wellcome Trust Postdoctoral Research Fellowship.
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
The method for estimating geometry in the femoral neck (FN; T. J. Beck, Sc.D.) is as follows. Structural geometry can be calculated directly from DXA mass image data, but in cases where the image data are not available it is possible to estimate the geometry from conventional outputs, although the method is meaningful only in DXA regions that traverse the long axis of a long bone (FN). It should be realized that the methods are necessarily crude and assume that the bone within the region is a uniform right circular cylinder. This is not a very good assumption in children and persons with smaller bones since cross sections of the FN change greatly in shape along the length of the neck. How much cross sections change in a fixed region length varies with the size of the bone.
FN width (W) is estimated by dividing the neck region area by its fixed length in Hologic scanners, i.e., 1.5 cm. The cross-sectional area (CSA) is computed as
where ρm is the effective density of bone mineral in fully mineralized bone tissue (~1.05 g/cm3).
The section modulus (Z) is computed as
The cross-sectional moment of inertia (CSMI) is computed as
where p is the trabecular porosity and ED is the estimated endosteal diameter.
where f c is the assumed proportion of cortical mass in the FN (0.6).
An estimate of mean cortical thickness is given by
Buckling ratio can then be estimated as
Rights and permissions
About this article
Cite this article
Micklesfield, L.K., Norris, S.A., van der Merwe, L. et al. Comparison of Site-Specific Bone Mass Indices in South African Children of Different Ethnic Groups. Calcif Tissue Int 85, 317–325 (2009). https://doi.org/10.1007/s00223-009-9286-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-009-9286-y