Abstract
Antiremodeling agents reduce bone loss in part through direct actions on osteoclasts. Their effects on osteoblasts and bone formation activity are less clear and may differ at sites undergoing modeling vs. remodeling. Skeletally mature intact beagles, 1–2 years old at the start of the study, were treated daily with clinically relevant doses of alendronate (0.10 or 0.20 mg/kg), risedronate (0.05 or 0.10 mg/kg), raloxifene (0.50 mg/kg), or vehicle (1 mL/kg). Dynamic bone formation parameters were histologically assessed on periosteal, endocortical/trabecular, and intracortical bone envelopes of the rib. Raloxifene significantly increased periosteal surface mineral apposition rate (MAR), a measure of osteoblast activity, compared to all other treatments (+108 to +175%, P < 0.02), while having no significant effect on MAR at either the endocortical/trabecular or intracortical envelope. Alendronate (both 0.10 and 0.20 doses) and risedronate (only the 0.10 dose) significantly (P ≤ 0.05) suppressed MAR on the endocortical/trabecular envelope, while none of the bisphosphonate doses significantly altered MAR at either the periosteal or intracortical envelopes compared to vehicle. Based on these results, we conclude that (1) at clinically relevant doses the two classes of antiremodeling agents, bisphosphonates and selective estrogen receptor modulators, exert differential effects on osteoblast activity in the canine rib and (2) this effect depends on whether modeling or remodeling is the predominant mechanism of bone formation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Antiremodeling agents significantly reduce vertebral fracture risk in part through their direct effects on osteoclasts. By inducing osteoclast apoptosis and suppressing activity/proliferation/differentiation [1–5], the number of newly initiated remodeling sites is reduced [6]. Due to the coupling of resorption and formation activities during remodeling, antiremodeling agents effect a systemic decrease in bone-formation activity [7, 8]. It is unclear whether these agents have separate effects on osteoblasts independent of their suppression of remodeling.
Numerous studies have attempted to determine the effects of antiremodeling agents on osteoblasts. In vitro data show positive effects of bisphosphonates on osteoblast proliferation and differentiation [9–11] as well as suppression of apoptosis [12, 13]. These data are contrasted by in vivo animal studies that have shown bisphosphonate suppression of mineral apposition rate (MAR), indicative of reduced osteoblast activity, at both modeling and remodeling sites [14–17]. Selective estrogen receptor modulators (SERMs) suppress osteoblast apoptosis [18] and increase osteoblast differentiation [19] and proliferation [20] in vitro. In vivo, SERMs have produced conflicting results with respect to effects on MAR [21–23].
Antiremodeling agents could exert differential effects on osteoblasts depending on whether the cells are involved in remodeling or modeling bone-formation activity. Remodeling-associated formation activity, the predominant form on endocortical, trabecular, and intracortical envelopes [24, 25], is coupled to osteoclasts. Antiremodeling agents may indirectly reduce the individual activity of osteoblasts through reductions in basic multicellular unit-level osteoclast activity (i.e., reduced erosion depth) [26]. These agents may also directly influence osteoblasts in vivo. This effect would be most evident at sites undergoing modeling, where formation occurs without previous resorption. Modeling is the predominant mechanism of formation activity on periosteal surfaces, although it can occur on other surfaces [24, 25].
The goal of this study was to determine the effect of bisphosphonates (alendronate and risedronate) and a SERM (raloxifene) on MAR, an indicator of osteoblast activity. Following 1-year treatment of intact beagle dogs with clinically relevant doses of these agents, ribs were histologically assessed for bone formation variables. MAR was used as an indicator of osteoblast activity and assessed separately at sites that predominantly undergo remodeling (endocortical/trabecular and intracortical envelopes) or modeling (periosteal) bone formation. We hypothesized that these three antiremodeling agents would suppress osteoblast activity (MAR) at both modeling and remodeling sites.
Materials and Methods
Animals
Seventy-two skeletally mature female beagles (1.3 ± 0.02 years old) were purchased from Marshall Farms (North Rose, NY). Upon arrival, lateral X-rays of all dogs were obtained to confirm skeletal maturity (closed proximal tibia and lumbar vertebra growth plates). Animals were housed two per cage in environmentally controlled rooms at Indiana University School of Medicine’s Association for Assessment and Accreditation of Laboratory Animal Care-accredited facility and provided standard dog chow and water. All procedures were approved prior to the study by the Indiana University School of Medicine Animal Care and Use Committee.
Experimental Design
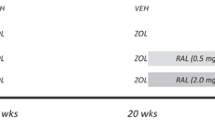
Following 2 weeks of acclimatization, animals were assigned to treatment groups (n = 12/group) by matching body weights. All dogs were treated daily for 1 year with oral doses of vehicle (saline, 1.0 mL/kg), risedronate sodium (0.05 or 0.10 mg/kg; Procter & Gamble Pharmaceuticals, Norwich, NY), alendronate sodium (0.10 or 0.20 mg/kg; Merck, RAHWAY, NJ), or raloxifene (0.50 mg/kg; Lilly Research Labs, Indianapolis, IN). The bisphosphonate doses (risedronate 0.10 and alendronate 0.20) are equivalent to those used for treatment of postmenopausal osteoporosis (on a mg/kg basis), and the lower doses of each drug correspond to approximately half the clinical treatment dose. The raloxifene dose was chosen to produce serum levels approximately equivalent to those documented in postmenopausal women. Risedronate and alendronate were dissolved in saline; raloxifene was diluted in 10% hydroxypropyl-β-cyclodextrin made with distilled water. All agents were administered orally with a syringe each morning after an overnight fast and at least 2 hours prior to feeding. Prior to necropsy, animals were injected with calcein (0.20 mL/kg, intravenous) using a 2-12-2-5 labeling schedule (n = 9 animals/group) or a 2-5-2-5 (n = 3 animals/group). The shorter interlabel duration was due to a scheduling error. Animals were killed after 1 year by intravenous administration of sodium pentobarbital (0.22 mg/kg). After death, the right ninth rib (∼20 mm) was dissected, placed in 10% neutral buffered formalin for 72 hours, and then transferred to 70% ethanol for processing.
Histology
Using an automatic tissue processor (Shandon/Lipshaw, Pittsburgh, PA), specimens were cycled through a graded series of ethanols, cleared using xylene, and infiltrated with methyl methacrylate (MMA; Aldrich, Milwaukee, WI) using routine embedding procedures. Transverse sections (80–100 μm) were cut using a diamond wire saw (Histosaw; Delaware Diamond Knives, Wilmington, DE). Histological measurements were made using a semiautomatic analysis system (Bioquant OSTEO 7.20.10; Bioquant Image Analysis, Nashville, TN) attached to a microscope equipped with an ultraviolet light source (Nikon Optiphot 2; Nikon, Tokyo, Japan). Measurements were made on one cross section per animal (Fig. 1). Periosteal, endocortical/trabecular (mineral surface/bone surface [MS/BS], MAR, bone formation rate [BFR]/BS), and intracortical (MAR, labeled osteon number, activation frequency) dynamic bone formation parameters were separately analyzed as previously described [16] and conformed to standard ASBMR nomenclature [27]. Endocortical and trabecular surfaces were analyzed as a single entity because it was not possible to consistently define the boundary between two surfaces across all tissue sections.
Statistics
Statistical tests were performed using SAS software (SAS Institute, Cary, NC). Differences among treatment groups were evaluated using one-way analysis of variance. When a significant overall F value (P ≤ 0.05) was present, differences between individual group means were tested using Fisher’s protected least-significant difference post-hoc test. For all tests, P ≤ 0.05 was considered statistically significant. Data are presented as mean ± standard error.
Results
The two classes of drugs, bisphosphonates (alendronate and risedronate) and SERMs (raloxifene), exerted envelope-specific effects on MAR of nonovariectomized dog ribs (Table 1, Fig. 2). Raloxifene-treated animals had significantly higher MAR (+108%, P = 0.019) on the periosteal surface compared to animals treated with vehicle (Fig. 2). Neither alendronate nor risedronate significantly altered periosteal MAR at either treatment dose compared to vehicle-treated animals. Periosteal surface MAR was significantly higher in raloxifene-treated animals compared to all bisphosphonate-treated groups (+112% to +175%, all P < 0.05). Endocortical/trabecular MAR was similar between vehicle- and raloxifene-treated animals but significantly higher in raloxifene-treated animals compared to all bisphosphonate-treated groups (+53% to +464%, all P < 0.05) (Fig. 2). MAR was significantly lower than vehicle in animals treated with alendronate at the 0.10 dose (−59%) and the 0.20 dose (−41%), as well as risedronate at the 0.10 dose (−80%). There was no difference in MAR between vehicle and the lower dose of risedronate (0.05) on the endocortical/trabecular surface. Intracortical MAR was not altered by any of the treatments.
Envelope-specific effects of antiremodeling agents on MAR of dog ribs following 1-year treatment with clinically relevant doses of raloxifene, alendronate, or risedronate. (A) Periosteal MAR. (B) Endocortical/trabecular envelope MAR. Data presented as mean ± standard error. P < 0.05 vs. aall other treatments, ball bisphosphonate-treated groups, cvehicle, drisedronate 0.05, erisedronate 0.10.
Periosteal mineralizing surface (MS/BS) was not significantly different (P = 0.22) among the treatment groups (Table 1). On the endocortical/trabecular surface, there was no significant effect of raloxifene on MS/BS compared to vehicle. However, raloxifene-treated animals had significantly higher MS/BS on this surface compared to all bisphosphonate-treated groups (+60% to +254%, all P < 0.05). Endocortical/trabecular MS/BS was significantly lower than vehicle in groups treated with alendronate (both 0.10 and 0.20 doses) or risedronate (only the 0.10 dose). Labeled osteon number, the surrogate to MS/BS within the intracortical envelope, was not significantly altered by any of the treatments (Table 1).
Periosteal surface BFR was significantly higher in raloxifene-treated animals compared to both vehicle (+285%, P < 0.003) and all bisphosphonate-treated groups (Table 1). There was no significant difference between vehicle and any of the bisphosphonate-treated groups. Endocortical/trabecular surface BFR was unchanged with raloxifene compared to vehicle; bisphosphonate-treated animals (alendronate 0.10 and 0.20, risedronate 0.10) had significantly lower BFR compared to vehicle. Intracortical activation frequency was not different among the groups.
Discussion
In addition to direct effects on osteoclasts, antiremodeling agents may influence osteoblasts. It is well accepted that these agents reduce bone formation, but whether this is due to a decrease in the number of formation sites, a decrease in formation at each individual site, or both is unclear. In vitro studies have shown that both bisphosphonates and SERMs have direct positive effects on osteoblasts [9–13, 18–20]; however, in vivo data have produced conflicting results. Our data show that following 1-year treatment of intact dogs with clinically relevant doses, raloxifene significantly stimulates osteoblast activity on periosteal surfaces without an effect on endocortical/trabecular surfaces. In contrast to raloxifene, bisphosphonates do not alter periosteal osteoblast activity while significantly suppressing MAR on endocortical/trabecular surfaces.
In the mature skeleton, periosteal bone formation occurs predominantly through formation without prior resorption [24]. Our data show that raloxifene significantly increases osteoblast activity (assessed by MAR) on the rib periosteal surface compared to vehicle-treated animals. This is consistent with in vitro data showing that raloxifene can positively affect osteoblast activity. In culture, raloxifene stimulates type 1 collagen secretion and alkaline phosphatase activity [19, 20] and suppresses osteoblast apoptosis [18]. In our study, there was no significant effect of raloxifene (positive or negative) on MAR within either the endocortical/trabecular or intracortical envelopes of the rib, both sites that predominantly undergo coupled formation and resorption in the adult skeleton. These data suggest that raloxifene has a direct stimulatory effect on osteoblasts associated with formation on a surface that primarily undergoes modeling, while this effect is negated on surfaces that primarily undergo remodeling-associated formation activity.
The mechanism through which raloxifene stimulates osteoblast activity on the periosteal surface is unclear. Estrogen receptor-α (ERα) is more highly expressed in cortical bone [28] and appears to be a major regulator of bone modeling on the periosteal surface. Mice lacking ERα receptors exhibit reduced periosteal diameter [29, 30] and an attenuation of loading-induced periosteal formation [31]. Animals and cells lacking ERβ are minimally affected with respect to periosteal geometry or cellular activity [32]; this receptor appears to play a greater role in endocortical and trabecular sites. Our findings may be explained by the selective modulation of ERα or ERβ by raloxifene, which may differentially regulate osteoblast-related genes [33] at the various bone envelopes depending on the ER population expressed. It is also possible that factors associated with osteoclasts and/or resorption offset the positive effect of raloxifene at remodeling sites.
Periosteal expansion significantly increases bone strength [34]. The relationship between periosteal dimensions and bone strength is exponential; increases of periosteal radii enhance section modulus (an estimator of bone strength) by the fourth power [35]. Thus, small increases in periosteal apposition are mechanically advantageous as limited amounts of new bone can substantially increase fracture resistance and can mechanically offset loss of endocortical/trabecular bone. The increases in MAR (and BFR) with raloxifene would be expected to significantly increase nonvertebral bone cross-sectional area and reduce nonvertebral fracture risk, yet neither of these effects has been confirmed in clinical trials. Raloxifene did not enhance periosteal expansion beyond that of placebo-treated postmenopausal women after 3 years [36], although the technique used to assess bone size (dual-energy X-ray absorptiometry) may not have sufficient resolution to detect small differences. Additionally, clinical trials with raloxifene have not shown a significant reduction in nonvertebral fractures [37] (in contrast to both risedronate [38] and alendronate [39]). Increased trabecular BMD with bisphosphonates is largely responsible for the reduced fracture risk. Because raloxifene is a less potent antiremodeling agent, it has less effect on trabecular BMD, which may explain its failure to significantly reduce nonvertebral fracture risk. Still, it is entirely possible that periosteal expansion, although known to increase bone strength, is not sufficient with raloxifene treatment to decrease fracture risk in the absence of a more significant trabecular bone mineral density response.
Bisphosphonate treatment (either alendronate or risedronate) did not significantly alter periosteal surface MAR but did significantly suppress MAR on endocortical/trabecular surfaces. These effects on different bone surfaces are consistent with previous studies in dogs [16, 17, 40, 41] and nonhuman primates [42]. In growing [14] and skeletally mature [15] rats, however, treatment with either alendronate or risedronate significantly suppresses osteoblast activity on both periosteal and endocortical surfaces. Our data suggest that bisphosphonates negatively influence the work rate of osteoblasts associated with remodeling-associated formation. High concentrations of bisphosphonates are known to be liberated from the bone matrix during resorption, and osteoblasts have been shown to internalize bisphosphonates [43]. Since bisphosphonates cannot be metabolized, sufficient uptake in osteoblasts would compromise their function via inhibition of protein prenylation [44]. We therefore hypothesize that decreases in formation with bisphosphonates are due to both a decrease in the number of forming sites and the osteoblast activity at each site. However, human data quantifying MAR with bisphosphonate treatment generally show no effect at any bone envelope [6, 26, 45, 46]. Although these human data are limited by relatively small sample sizes and confined to the iliac crest, until similar drug effects on osteoblast activity can be confirmed in humans our interpretations remain hypothetical.
Periosteal bone formation can be a compensatory mechanism to maintain bone strength in situations where bone loss occurs from trabecular and endocortical surfaces [47]. At their respective clinical doses, raloxifene is known to suppress remodeling less than the bisphosphoantes [48]. In these same dogs, vertebral bone turnover was suppressed ∼70% with alendronate (0.20) and risedronate (0.10) and only ∼20% with raloxifene compared to vehicle [49]. It is therefore possible that bisphosphonates did not increase periosteal formation in our study because the suppression of remodeling on endocortical/trabecular surfaces was sufficient to increase bone mass to some critical level. If this were the case, one might expect that a bisphosphonate dose that had less suppressive effect on the endocortical/trabecular surface would have a more significant anabolic effect on the periosteal surface. However, we did not find this to be the case as the lower dose of risedronate (0.05) suppressed endocortical/trabecular bone formation significantly less than the higher dose (0.10), yet the two had similar periosteal bone formation parameters. These data suggest that the level of turnover suppression on the endocortical/trabecular surface is not a main determinant of periosteal osteoblast activity.
Our data describing bisphosphonate suppression of MAR at remodeling sites may provide some explanation for results from clinical trials using combination treatments. Treatment with alendronate, either concurrently or sequentially with teriparatide, blunts teriparatide-induced increases in bone formation biomarkers and bone mineral density of postmenopausal women [50, 51] and men with low bone mass [52]. This blunting was hypothesized to be related to the reduction in the number of remodeling sites by alendronate, leaving few active formation sites for teriparatide to stimulate. Based on our data, an alternative hypothesis may be that bisphosphonate suppression of osteoblast activity directly offsets the osteoblast stimulatory effect of teriparatide. Additionally, a suppressive effect of bisphosphonates on remodeling-associated bone-formation activity could help explain the failure to consistently find increased trabecular bone volume in bisphosphonate clinical trials [6, 53].
One aim of this study was to compare the effects of raloxifene and bisphosphonates at doses corresponding to those used for treatment of postmenopausal women. Poststudy analyses of serum from three raloxifene-treated dogs, for reasons unrelated to this study, revealed that the serum concentration of raloxifene was approximately half that predicted from the original dosing calculations. These serum levels were still within the range quantified in postmenopausal women receiving the 60 mg/day dose of raloxifene (Lilly, unpublished data). We therefore compared the raloxifene dose to both the clinical and half-clinical dose of each bisphosphonate. Based on the similarity among the four different bisphosphonate-treated groups with respect to periosteal MAR, the significantly greater MAR with raloxifene appears to represent a true difference in the biological activity of the drug administered at clinically relevant doses. We cannot discount the possibility, however, that bisphosphonate doses lower than those used in this study could potentially stimulate periosteal formation to levels comparable with raloxifene.
The current study has various limitations that should be noted. We analyzed only one bone site of intact, nonovariectomized beagle dogs and therefore cannot be certain whether similar changes would occur in the absence of estrogen (i.e., postmenopausal women) or at other bone sites. The endocortical/trabecular surfaces were not separately assessed as the boundaries are difficult to differentiate and in our experience the surfaces respond in similar ways. Although raloxifene dosing levels provided serum values within the range quantified in postmenopausal women receiving the 60 mg/day dose of raloxifene, we cannot discount the possibility that administering the drug at a higher dose would produce different results. Although there are no data to suggest the raloxifene carrier has any effect on bone formation (10% hydroxypropyl-β-cyclodextrin), we cannot exclude this possibility. Finally, although all growth plates are generally closed in the dog by 12 months [54], we did not assess the rib growth plates prior to treatment and therefore do not know whether continued growth occurred in the ribs of these animals during the course of the study.
In conclusion, we have shown that 1 year of treatment with clinically relevant doses of antiremodeling agents significantly influences osteoblast activity in the rib of beagle dogs. Raloxifene stimulates modeling-associated osteoblast activity on the periosteal surface while maintaining remodeling-associated activity on endocortical/trabecular surfaces at levels similar to untreated controls. In contrast, bisphosphonates suppress remodeling-associated osteoblast activity on the endocortical/trabecular surface while having no significant effect on periosteal osteoblast activity.
References
Fisher JE, Rodan GA, Reszka AA (2000) In vivo effects of bisphosphonates on the osteoclast mevalonate pathway. Endocrinology 141:4793–4796
Fisher JE, Rogers MJ, Halasy JM, Luckman SP, Hughes DE, Masarachia PJ, Wesolowski G, Russell RG, Rodan GA, Reszka AA (1999) Alendronate mechanism of action: geranylgeraniol, an intermediate in the mevalonate pathway, prevents inhibition of osteoclast formation, bone resorption, and kinase activation in vitro. Proc Natl Acad Sci USA 96:133–138
Hughes DE, Wright KR, Uy HL, Sasaki A, Yoneda T, Roodman GD, Mundy GR, Boyce BF (1995) Bisphosphonates promote apoptosis in murine osteoclasts in vitro and in vivo. J Bone Miner Res 10:1478–1487
Luckman SP, Hughes DE, Coxon FP, Graham R, Russell G, Rogers MJ (1998) Nitrogen-containing bisphosphonates inhibit the mevalonate pathway and prevent post-translational prenylation of GTP-binding proteins, including Ras. J Bone Miner Res 13:581–589
Rogers MJ, Frith JC, Luckman SP, Coxon FP, Benford HL, Monkkonen J, Auriola S, Chilton KM, Russell RG (1999) Molecular mechanisms of action of bisphosphonates. Bone 24:73S–79S
Chavassieux PM, Arlot ME, Reda C, Wei L, Yates AJ, Meunier PJ (1997) Histomorphometric assessment of the long-term effects of alendronate on bone quality and remodeling in patients with osteoporosis. J Clin Invest 100:1475–1480
Rosen CJ, Hochberg MC, Bonnick SL, McClung M, Miller P, Broy S, Kagan R, Chen E, Petruschke RA, Thompson DE, de Papp AE (2005) Treatment with once-weekly alendronate 70 mg compared with once-weekly risedronate 35 mg in women with postmenopausal osteoporosis: a randomized double-blind study. J Bone Miner Res 20:141–151
Delmas PD (2000) How does antiresorptive therapy decrease the risk of fracture in women with osteoporosis? Bone 27:1–3
Giuliani N, Pedrazzoni M, Negri G, Passeri G, Impicciatore M, Girasole G (1998) Bisphosphonates stimulate formation of osteoblast precursors and mineralized nodules in murine and human bone marrow cultures in vitro and promote early osteoblastogenesis in young and aged mice in vivo. Bone 22:455–461
Im GI, Qureshi SA, Kenney J, Rubash HE, Shanbhag AS (2004) Osteoblast proliferation and maturation by bisphosphonates. Biomaterials 25:4105–4115
Schindeler A, Little DG (2005) Osteoclasts but not osteoblasts are affected by a calcified surface treated with zoledronic acid in vitro. Biochem Biophys Res Commun 338:710–716
Plotkin LI, Weinstein RS, Parfitt AM, Roberson PK, Manolagas SC, Bellido T (1999) Prevention of osteocyte and osteoblast apoptosis by bisphosphonates and calcitonin. J Clin Invest 104:1363–1374
Rodan GA, Fleisch HA (1996) Bisphosphonates: mechanisms of action. J Clin Invest 97:2692–2696
Bikle D, Morey-Holton E, Doty S, Currier P, Tanner S, Halloran B (1994) Alendronate increases skeletal mass of growing rats during unloading by inhibiting resorption of calcified cartilage. J Bone Miner Res 9:1777–1787
Iwata K, Li J, Follet H, Phipps RJ, Burr DB (2006) Bisphosphonates suppress periosteal osteoblast activity independent of resorption in rat femur and tibia. Bone (Published online June 7, 2006)
Mashiba T, Hirano T, Turner CH, Forwood MR, Johnston CC, Burr DB (2000) Suppressed bone turnover by bisphosphonates increases microdamage accumulation and reduces some biomechanical properties in dog rib. J Bone Miner Res 15:613–620
Mashiba T, Turner CH, Hirano T, Forwood MR, Johnston CC, Burr DB (2001) Effects of suppressed bone turnover by bisphosphonates on microdamage accumulation and biomechanical properties in clinically relevant skeletal sites in beagles. Bone 28:524–531
Olivier S, Fillet M, Malaise M, Piette J, Bours V, Merville MP, Franchimont N (2005) Sodium nitroprusside-induced osteoblast apoptosis is mediated by long chain ceramide and is decreased by raloxifene. Biochem Pharmacol 69:891–901
Viereck V, Grundker C, Blaschke S, Niederkleine B, Siggelkow H, Frosch KH, Raddatz D, Emons G, Hofbauer LC (2003) Raloxifene concurrently stimulates osteoprotegerin and inhibits interleukin-6 production by human trabecular osteoblasts. J Clin Endocrinol Metab 88:4206–4213
Taranta A, Brama M, Teti A, De Luca V, Scandurra R, Spera G, Agnusdei D, Termine JD, Migliaccio S (2002) The selective estrogen receptor modulator raloxifene regulates osteoclast and osteoblast activity in vitro. Bone 30:368–376
Lees CJ, Register TC, Turner CH, Wang T, Stancill M, Jerome CP (2002) Effects of raloxifene on bone density, biomarkers, and histomorphometric and biomechanical measures in ovariectomized cynomolgus monkeys. Menopause 9:320–328
Iwamoto J, Yeh JK, Schmidt A, Rowley E, Stanfield L, Takeda T, Sato M (2005) Raloxifene and vitamin K2 combine to improve the femoral neck strength of ovariectomized rats. Calcif Tissue Int 77:119–126
Ott SM, Oleksik A, Lu Y, Harper K, Lips P (2002) Bone histomorphometric and biochemical marker results of a 2-year placebo-controlled trial of raloxifene in postmenopausal women. J Bone Miner Res 17:341–348
Frost HM (2004) The Utah Paradigm of Skeletal Physiology. Chapters 3 and 4. International Society of Musculoskeletal and Neuronal Interactions, Greece
Erben RG (1996) Trabecular and endocortical bone surfaces in the rat: modeling or remodeling? Anat Rec 246:39–46
Eriksen EF, Melsen F, Sod E, Barton I, Chines A (2002) Effects of long-term risedronate on bone quality and bone turnover in women with postmenopausal osteoporosis. Bone 31:620–625
Parfitt A, Drezner M, Glorieux F, Kanis J, Malluche H, Meunier P, Ott S, Recker R (1987) Bone histomorphometry: standardization of nomenclature, symbols, and units. J Bone Miner Res 2:595–610
Bord S, Horner A, Beavan S, Compston J (2001) Estrogen receptors alpha and beta are differentially expressed in developing human bone. J Clin Endocrinol Metab 86:2309–2314
Vidal O, Lindberg MK, Hollberg K, Baylink DJ, Andersson G, Lubahn DB, Mohan S, Gustafsson J-A, Ohlsson C (2000) Estrogen receptor specificity in the regulation of skeletal growth and maturation in male mice. Proc Natl Acad Sci USA 97:5474–5479
Sims NA, Dupont S, Krust A, Clement-Lacroix P, Minet D, Resche-Rigon M, Gaillard-Kelly M, Baron R (2002) Deletion of estrogen receptors reveals a regulatory role for estrogen receptor-β in bone remodeling in females but not in males. Bone 30:18–25
Lee K, Jessop H, Suswillo R, Zaman G, Lanyon L (2003) Endocrinology: bone adaptation requires oestrogen receptor-alpha. Nature 424:389
Saxon LK, Turner CH (2005) Estrogen receptor beta: the antimechanostat? Bone 36:185–192
Kian Tee M, Rogatsky I, Tzagarakis-Foster C, Cvoro A, An J, Christy RJ, Yamamoto KR, Leitman DC (2004) Estradiol and selective estrogen receptor modulators differentially regulate target genes with estrogen receptors alpha and beta. Mol Biol Cell 15:1262–1272
Allen MR, Hock JM, Burr DB (2004) Periosteum: biology, regulation, and response to osteoporosis therapies. Bone 35:1003–1012
Orwoll E (2003) Toward an expanded understanding of the role of the periosteum in skeletal health. J Bone Miner Res 18:949–954
Uusi-Rasi K, Beck TJ, Semanick LM, Daphtary MM, Crans GG, Desaiah D, Harper KD (2006) Structural effects of raloxifene on the proximal femur: results from the multiple outcomes of raloxifene evaluation trial. Osteoporos Int 17:575–586
Siris ES, Harris ST, Eastell R, Zanchetta JR, Goemaere S, Diez-Perez A, Stock JL, Song J, Qu Y, Kulkarni PM, Siddhanti SR, Wong M, Cummings SR (2005) Skeletal effects of raloxifene after 8 years: results from the Continuing Outcomes Relevant to Evista (CORE) study. J Bone Miner Res 20:1514–1524
Harris ST, Watts NB, Genant HK, McKeever CD, Hangartner T, Keller M, Chesnut CH 3rd, Brown J, Eriksen EF, Hoseyni MS, Axelrod DW, Miller PD (1999) Effects of risedronate treatment on vertebral and nonvertebral fractures in women with postmenopausal osteoporosis: a randomized controlled trial. Vertebral Efficacy with Risedronate Therapy (VERT) Study Group. JAMA 282:1344–1352
Karpf DB, Shapiro DR, Seeman E, Ensrud KE, Johnston CC Jr, Adami S, Harris ST, Santora AC 2nd, Hirsch LJ, Oppenheimer L, Thompson D (1997) Prevention of nonvertebral fractures by alendronate. A meta-analysis. Alendronate Osteoporosis Treatment Study Groups. JAMA 277:1159–1164
Komatsubara S, Mori S, Mashiba T, Li J, Nonaka K, Kaji Y, Akiyama T, Miyamoto K, Cao Y, Kawanishi J, Norimatsu H (2004) Suppressed bone turnover by long-term bisphosphonate treatment accumulates microdamage but maintains intrinsic material properties in cortical bone of dog rib. J Bone Miner Res 19:999–1005
Balena R, Markatos A, Seedor JG, Gentile M, Stark C, Peter CP, Rodan GA (1996) Long-term safety of the aminobisphosphonate alendronate in adult dogs. II. Histomorphometric analysis of the L5 vertebrae. J Pharmacol Exp Ther 276:277–283
Balena R, Toolan BC, Shea M, Markatos A, Myers ER, Lee SC, Opas EE, Seedor JG, Klein H, Frankenfield D (1993) The effects of 2-year treatment with the aminobisphosphonate alendronate on bone metabolism, bone histomorphometry, and bone strength in ovariectomized nonhuman primates. J Clin Invest 92:2577–2586
Coxon F, Thompson K, Ebetino H, Rogers M (2006) Resorbing osteoclasts increase the availability of mineral-bound bisphosphonates to non-resorbing cells. Bone 38:S45
Gasser J, Ingold P, Rebmann A, Susa M, Green J (2006) Inhibition of FPP-synthase in osteoblasts may explain the blunting of bone anabolic response to PTH observed after chronic exposure of rats to bisphosphonates. Bone 38:S50
Bare S, Recker S, Recker R, Kimmel D (2005) Influence of alendronate on periosteal and endocortical bone formation in the ilium of osteoporotic women. J Bone Miner Res 20:SA414
Recker RR, Weinstein RS, Chesnut CH 3rd, Schimmer RC, Mahoney P, Hughes C, Bonvoisin B, Meunier PJ (2004) Histomorphometric evaluation of daily and intermittent oral ibandronate in women with postmenopausal osteoporosis: results from the BONE study. Osteoporos Int 15:231–237
Alhlborg H, Johnell O, Turner C, Rannevik G, Karlsson M (2003) Bone loss and bone size after menopause. N Engl J Med 349:327–334
Sambrook PN, Geusens P, Ribot C, Solimano JA, Ferrer-Barriendos J, Gaines K, Verbruggen N, Melton ME (2004) Alendronate produces greater effects than raloxifene on bone density and bone turnover in postmenopausal women with low bone density: results of EFFECT (Efficacy of Fosamax versus Evista Comparison Trial). Int J Intern Med 255:503–511
Allen MR, Iwata K, Sato M, Burr DB (2006) Raloxifene enhances vertebral mechanical properties independent of bone density. Bone (Published online June 30, 2006)
Black DM, Bilezikian JP, Ensrud KE, Greenspan SL, Palermo L, Hue T, Lang TF, McGowan JA, Rosen CJ (2005) One year of alendronate after one year of parathyroid hormone (1–84) for osteoporosis. N Engl J Med 353:555–565
Black DM, Greenspan SL, Ensrud KE, Palermo L, McGowan JA, Lang TF, Garnero P, Bouxsein ML, Bilezikian JP, Rosen CJ (2003) The effects of parathyroid hormone and alendronate alone or in combination in postmenopausal osteoporosis. N Engl J Med 349:1207–1215
Finkelstein JS, Hayes A, Hunzelman JL, Wyland JJ, Lee H, Neer RM (2003) The effects of parathyroid hormone, alendronate, or both in men with osteoporosis. N Engl J Med 349:1216–1226
Recker R, Masarachia P, Santora A, Howard T, Chavassieux P, Arlot M, Rodan G, Wehren L, Kimmel D (2005) Trabecular bone microarchitecture after alendronate treatment of osteoporotic women. Curr Med Res Opin 21:185–194
Wolvekamp P (2002) The many faces of elbow dysplasia [abstract]. Proceedings from the 27th World Small Animal Veterinary Association Congress, Granada, Spain, October 2002
Acknowledgments
The authors thank Dr. Keith Condon, Diana Jacob, and Lauren Waugh for histological preparation. This work was supported by National Institutes of Health grants R01-AR047838 and T32-AR007581 and research grants from The Alliance for Better Bone Health (Procter & Gamble Pharmaceuticals and Sanofi-Aventis) and Lilly Research Laboratories. Merck kindly provided the alendronate. This investigation utilized an animal facility constructed with support from Research Facilities Improvement Program grant C06 RR10601-01 from the National Center for Research Resources, National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Additional information
M. R. Allen and H. Follet contributed equally to this work.
Rights and permissions
About this article
Cite this article
Allen, M.R., Follet, H., Khurana, M. et al. Antiremodeling Agents Influence Osteoblast Activity Differently in Modeling and Remodeling Sites of Canine Rib. Calcif Tissue Int 79, 255–261 (2006). https://doi.org/10.1007/s00223-006-0031-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00223-006-0031-5