Abstract
One outstanding question in the contemplative science literature relates to the direct impact of meditation experience on the monitoring of internal states and its respective correspondence with neural activity. In particular, to what extent does meditation influence the awareness, duration and frequency of the tendency of the mind to wander. To assess the relation between mind wandering and meditation, we tested 2 groups of meditators, one with a moderate level of experience (non-expert) and those who are well advanced in their practice (expert). We designed a novel paradigm using self-reports of internal mental states based on an experiential sampling probe paradigm presented during ~1 h of seated concentration meditation to gain insight into the dynamic measures of electroencephalography (EEG) during absorption in meditation as compared to reported mind wandering episodes. Our results show that expert meditation practitioners report a greater depth and frequency of sustained meditation, whereas non-expert practitioners report a greater depth and frequency of mind wandering episodes. This is one of the first direct behavioral indices of meditation expertise and its associated impact on the reduced frequency of mind wandering, with corresponding EEG activations showing increased frontal midline theta and somatosensory alpha rhythms during meditation as compared to mind wandering in expert practitioners. Frontal midline theta and somatosensory alpha rhythms are often observed during executive functioning, cognitive control and the active monitoring of sensory information. Our study thus provides additional new evidence to support the hypothesis that the maintenance of both internal and external orientations of attention may be maintained by similar neural mechanisms and that these mechanisms may be modulated by meditation training.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
One of the most unique human attributes is our capacity for a vastly complex inner landscape, and our ability to recall, generate and then manifest insight based on experience and predict out into the future. While an elaborate internal dialogue is fundamental to our human experience, this ongoing narrative can surface unknowingly and at inopportune points in time. The later phenomena are commonly referred to as mind wandering or task-unrelated thought and are the experience of thoughts involuntarily drifting to topics unrelated to the task at hand, often occurring under conditions where external demands on our attention are low (Smallwood et al. 2003). In a seminal study by Killingsworth and Gilbert (2010), participants reported being engaged in task-unrelated thought during almost half of their waking hours. While research has demonstrated that mind wandering is essential for creativity and memory consolidation (Baird et al. 2012), under less desirable circumstances excessive mind wandering is associated with problems in learning, rumination, anxiety and depression (Poerio et al. 2013; Smallwood et al. 2007). Given the pervasive and complex nature of mind wandering, exploring the neural dynamics underlying mind wandering is a crucial and necessary step toward understanding how the brain produces what William James first referred to as the “stream of consciousness” (James 1890).
Research in functional magnetic resonance imaging (fMRI) has begun to identify the neural networks largely contributing to mind wandering and the generation and maintenance of self-referential thought processes. The default mode network (DMN), comprised of the medial prefrontal cortex (mPFC), posterior cingulate cortex (PCC), superior and inferior frontal gyri, medial and lateral temporal lobes and the posterior portion of the inferior parietal lobule (Gusnard and Raichle 2001; Raichle 2015), shows consistent activations during both probe-caught and self-reported episodes of mind wandering. These episodes typically involve thinking about oneself, others, remembering the past and planning for the future (Buckner et al. 2008). Mind wandering has also been linked to activations in the frontal parietal control network (FPC), a network comprised mainly of the anterior cingulate cortex (ACC), mPFC, amygdala, PCC and the insula, and has been proposed to modulate top-down mechanisms involved in sustaining both endogenous and exogenous forms of attention allocation (Spreng et al. 2013). Spreng et al. (2013) suggest that goal-directed cognition is facilitated by the FPC, which functions as a gatekeeping system by moderating the dynamic balance between activations in the DMN and the dorsal attention network (DAN). It might also facilitate alternating or competing goal representations while maintaining directed attention to a given external task (i.e., driving, running; Spreng 2012). Concurrent activations in both the DMN and core regions of the executive functioning network (dorsolateral mPFC, ACC), networks that were traditionally considered independent, anti-correlated and thought to compete for cognitive resources, were also shown to co-activate during mind wandering episodes, and increasingly so when subjects reported being unaware of their mind wandering (Christoff et al. 2009). Interestingly, emphasis on the flexible monitoring of ongoing experience during meditation is thought to be responsible for the increased functional connectivity in DMN activations observed in expert meditation practitioners trained in internally guided forms of sustained attention (Tang et al. 2015; Jang et al. 2011). Taken together, these findings support the notion that both internally and externally directed forms of cognitive activity may recruit the DMN, as well as some overlapping regions of the executive networks.
An intriguing finding emerging from the field of contemplative neuroscience involves the mediating role of contemplative and meditative practices on the neural mechanisms underlying top-down regulation of sustained attention and sensory perception. An electroencephalography (EEG) study by Braboszcz and Delorme (2011) showed enhanced cortical processing of sensory stimuli during a sustained breath awareness task when compared to periods of time in which subjects reported mind wandering. Known as the perceptual decoupling hypothesis, the processing of sensory information during periods of mind wandering is more superficial (Schooler et al. 2011). Mrazek et al. (2013) found that after two weeks of mindfulness meditation training, participants who were initially most prone to distraction showed improved verbal GRE scores after meditation training in addition to enhanced working memory capacity as measured by the OSPAN working memory task. These changes were directly mediated by reduced mind wandering as measured by experience sampling using both probe-caught and self-reported mind wandering episodes during both the GRE and OSPAN. In another study by Mrazek et al. (2012), eight minutes of mindful breathing attenuated indirect performance markers of mind wandering during a sustained attention task. In a set of studies by Zanesco et al. (2016) exploring the effects of intense meditation training on mind wandering, two separate groups of participants who took part in either a one month insight meditation retreat, or a three month shamatha meditation retreat, showed a reduced tendency of the mind to wander as measured by reduced mindless reading and reduced probe-caught mind wandering measured during a reading task requiring ongoing error monitoring.
Arguably the cornerstones of most contemplative practices include the training and development of sustained attention (Slagter et al. 2007), the flexible monitoring of sensory experience (Kerr et al. 2011; Tang et al. 2015) and the cultivation of metacognitive awareness via active monitoring of mental states (Baird et al. 2014). Taken together, these faculties may facilitate increased efficiency in the distribution of our limited cognitive resources (Global workspace theory; Baars 2005). Increased functional connectivity in networks associated with both attention and executive functioning (Hasenkamp and Barsalou 2012; Teper and Inzlicht 2013) has been observed in advanced meditation practitioners, in addition to the aforementioned findings in the DMN (Jang et al. 2011). Given the large body of literature showing reduced DMN activations in advanced practitioners and that meditation leads to reduced DMN processing beyond that observed during other types of cognitive tasks (Garrison et al. 2015), together these findings could suggest that increased functional connectivity may reduce BOLD activity, reflecting an efficient use of cognitive resources. Contrary to these findings, a recent study by Berkovich-Ohana et al. (2016) found reductions in functional connectivity in experienced meditators; therefore, more research is needed to clarify the neurophysiological implications of functional connectivity in meditation practice and beyond.
In a study using magnetoencephalographic (MEG) recording of the SI finger representation, Kerr et al. (2011) found that experienced meditators showed an enhanced alpha power modulation in response to a cue, potentially reflecting an enhanced filtering of inputs to primary sensory cortex. They also found that experienced meditators demonstrated modified alpha rhythm properties and an increase in non-localized tonic alpha power when compared to controls. These findings can most likely be attributed to the emphasis on somatic attention training in mindfulness meditation techniques in which individuals train to develop metacognition, a process in which one directs their attention, moment-by-moment, to an overall somatosensory awareness of physical sensations, feelings and thoughts (Segal et al. 2004; Cahn and Polich 2006). Whitmarsh et al. (2014) investigated participants metacognitive ability to report on their attentional focus and found that contralateral somatosensory alpha depression correlated with higher reported attentional focus on either their left or right hand, respectively. Baird et al. (2014) found that a 2-week meditation program leads to significantly enhanced metacognitive ability for memory, but not for perceptual decisions, suggesting that while meditation training can enhance certain elements of introspective acuity, such improvements may not translate equally to all cognitive domains. Enhanced body awareness was also found to be associated with greater subjective emotional experience and awareness of the heart during exposure to emotionally provocative stimuli in vipassana meditators, when compared to expert dancers, and controls (Sze et al. 2010). Given that top-down attentional modulations of cortical excitability have been repeatedly shown to result in better discrimination and performance accuracy, the aforementioned findings provide support for both the enhancement of metacognitive accuracy via the direct monitoring of current mental states resulting from long-term meditation practice, and for potential changes in the supporting neural structures underlying sustained attention processes.
One outstanding question in the contemplative science literature relates to the direct impact of meditation experience on the monitoring of mind wandering and the degree to which practice influences the metacognitive awareness, duration and frequency of mind wandering events. In order to extend our scientific understanding of these temporally fluctuating mental states and phenomena in experimental settings, and given that subjects are generally unaware of mind wandering at the moment it occurs, the direct measurement challenge this poses for identifying the underlying mechanisms involved in attentional lapses requires nuanced neuroimaging methodology. Thus, we designed a novel paradigm based on experience-sampling probe presentations to gain insight into the dynamic measures of EEG by comparing the degree (subjects responded on a scale from 0 to 3) of self-reported absorption experienced during meditation with the self-reported absorption experienced during mind wandering. To assess the relation between mind wandering and meditation, we tested 2 groups of meditators, one with a moderate level of experience and one with an advanced practice level. The central question in this investigation is to test whether the level of meditation proficiency enhances the capacity for sustained attention, the awareness of and accuracy of self-report, and the metacognitive labeling of mental states. Our goal was also to contrast the neural dynamics of mind wandering and meditation, as well as an overall correlation between the EEG data and the first person behavioral data in this context.
Methods and materials
Participants
The study was conducted at the Meditation Research Institute (MRI) in Rishikesh, India. Twenty-five meditators from the Himalayan Yoga tradition participated in this study and were assigned to one of two groups based on experience and hours of daily practice. After data collection, one participant reported that they did not fully understand the task instructions and was excluded from the analyses; therefore, twenty-four participants were included in the analyses. Individuals who had engaged in a daily meditation practice for a minimum of 2 h daily for 1 year or longer were considered expert practitioners (3 females; mean hours weekly = 14.8, SD = 1.6 h; mean age = 39.3, SD = 12.0). Participants who were trained and familiar with the techniques, but who reported irregular practice (10 females; 3.2 mean weekly hours with SD of 3.1 h; mean age of 45.0 with SD of 14.8), were considered non-expert practitioners. All participants provided written consent to participate in the study and completed an extensive list of questions regarding their meditation background. Participants stated that they were not taking any medications that could potentially affect their concentration. The study received ethical approval from both the ethics committee of the Meditation Research Institute in India, and from the Comite de Protection des Personnes in France. Participants were all volunteers and were not compensated.
Experimental paradigm and procedure
All participants were asked to meditate continuously throughout the experiment in their usual seated meditation position (either seated on the floor, or in a chair). Meditators were all practitioners of the Himalayan Yoga tradition. Once subjects were comfortably seated in their meditative posture, they were instructed to begin their meditation. All practitioners began with an initial body scan as they relaxed into their seated posture and then started to mentally recite their mantra. Mantras are traditionally a word or sentence assigned to them by their meditation teacher. When deeper levels of meditation or stillness are obtained, mantra repetitions gradually cease. Mantras are derived from Sanskrit root words and syllables, whose resonance is thought to induce stability of the mind without the need for an overly intense focus.
Experience-sampling probes were presented at random intervals ranging from 30 to 90 s throughout the duration of the experiment. Probes, in the form of pre-recorded vocalized questions, were presented on two freestanding speakers and were reported as clearly audible by all subjects. Each experience-sampling probe series consisted of three questions, which were presented in the same order throughout the experiment and are described in detail below. Subjects responded on a small customized numeric USB keypad resting on their right thigh, to enable their right hand to comfortably rest without having to move or open their eyes. The time range of the experiment lasted from 45 min to 1 h 30 min, as some subjects were willing and able to sit comfortably for longer periods of time. The minimum number of probes that participants received was 30. The entire experiment was programmed and automated using the MATLAB psychophysics toolbox. All participants completed a 5-min training block prior to performing the experiment.
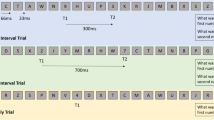
Experience-sampling probes consisted of three questions that followed sequentially (Fig. 1); the first question was Q1: “Please rate the depth of your meditation,” for which participants evaluated the subjective depth of their “meditative state” for the moments immediately preceding the first probe, on a scale from 0 (not meditating at all) to 3 (deep meditative state) by pressing the corresponding key on the keypad. After their response was registered, the second question Q2: “Please rate the depth of your mind wandering” automatically followed. Participants evaluated the subjective depth of their “mind wandering” for the period of time which immediately preceded the first probe, on a scale from 0 (not mind wandering at all) to 3 (immersed in their thoughts). The last question was Q3: “Please rate how tired you are,” where participants were asked to rate the subjective depth of their drowsiness at the time of the first question, from 0 (not drowsy at all) to 3 (very drowsy). All responses pertained to the evaluations of the same time period immediately preceding the first probe. Participants were then instructed to resume their meditation with the prompt: “You may now resume your meditation.”
Timeline of experimental design. Pseudorandom probes (randomly interspaced between 30 and 90 s) prompted subjects to respond via key press by subjectively evaluating the depth of their experience on a scale from 0 to 3 to three questions: Q1 for “Please rate the depth of your meditation”; Q2 for “Please rate the depth of your mind wandering”; Q3 for “Please rate how tired you are.” The letter R on the timeline corresponds to the instruction “You may now resume your meditation”
Data acquisition
We collected data using a 64-channel Biosemi system and a Biosemi 10–20 head cap montage at 2048 Hz sampling rate. All electrodes were kept within an offset of 15 using the Biosemi ActiView data acquisition system for measuring impedance. Respiration, heart rate (ECG/HRV) and galvanic skin response (GSR) were also recorded, but results from these data will not be reported here.
Data processing was done using MATLAB (The MathWorks, Inc.) and EEGLAB software (Delorme and Makeig 2004). The raw EEG data were average referenced and down-sampled from 2048 to 256 Hz. A high-pass filter at 2 Hz using a infinite impulse response filter (IIR; transition bandwidth of 0.7 Hz and order of 6) was applied, and the data were then average referenced again. The high-pass filter was necessary to obtain high-quality ICA decompositions on some subjects (see below) and, even though it was not necessary for all subjects, we opted to use the same high-pass filter settings for all subjects to ensure that all data were processed uniformly. Data were then segmented into 10 s-epochs, ranging from −10.05 to −0.05 s prior to the onset of question Q1 in the experience-sampling probe series. Bad electrodes (0–20 per subject, average of 6 per subject) and bad epochs containing paroxysmal activity were manually removed. Extended Infomax Independent Component Analysis (ICA) was then used to identify ocular and muscle artifacts (Delorme et al. 2007). ICA components for eye blink, lateral eye movements and temporal muscle noise were identified and subtracted from the data by the visual inspection of both the component scalp topographies and power spectrum distributions. Between 1 and 5 artifactual components were removed for each subject. After artifact rejection, between 21 and 64 clean data epochs (mean of 38.1; SD of 12.6) were included in subsequent analyses for each subject.
EEG time frequency analysis and statistics
We applied a Welch-like analysis on the 10-s-long epochs (Welch 1967). The difference with the Welch method is in its implementation of wavelets instead of the fast Fourier transform (FFT). We used a Morlet wavelet decomposition with 200 linearly spaced time windows and a series of 100 log-spaced frequencies that range from 1 to 128 Hz. The wavelet used to measure the amount of data in each successive, and overlapping time window has a 3-cycle wavelet at the lowest frequency. The number of cycles in the wavelets used for higher frequencies increases linearly, reaching 60 cycles at its highest frequency of 128 Hz.
Parametric statistics for behavioral results were performed in Excel, Statistica and MATLAB using paired t test, unpaired t test with unpooled variance estimates, and linear regression. For EEG data, statistics were conducted on topographic and time–frequency maps using two-tailed paired or unpaired statistics. For the EEG data, correction for multiple comparisons was performed using the cluster method developed by Maris and Oostenveld (2007), which is based on a nonparametric Monte Carlo permutation method. Channel topographies for this clustering method were established by setting the number of channel neighbors to 4.5 (Maris and Oostenveld 2007). We also used false discovery rate (Benjamini and Yekutieli 2001) to correct for multiple comparisons and obtained similar results as compared to the cluster method.
Results
Behavioral data
Expert practitioners reported a significantly lower depth of mind wandering than non-expert practitioners (mean 1.14 vs. 1.59; with parametric unpaired two-tailed t test with correction for non-homogeneous variance p = 0.03; with permutation statistics and 20,000 permutations p = 0.03). Expert practitioners also reported a greater depth of meditation than non-experts, although this effect failed to reach significance (mean 1.85 vs. 1.39; degree of freedom (df) of 11; with parametric statistics p = 0.06; with permutation statistics p = 0.06). When taking the rating difference between the two questions (Q1 minus Q2), the difference between expert and non-expert practitioners was even larger (mean −0.20 vs. 0.71; with parametric statistics p = 0.0084; with permutation statistics p = 0.0088). There was no difference in terms of alertness between expert and non-expert participants, and all participants were relatively alert as reflected by the low tiredness ratings (mean 0.69 vs. 0.73; ns with both parametric and permutation statistics). No significant correlations were observed between meditation and mind wandering (r 2 = 0.05; ns) or between meditation depth and drowsiness (r 2 = 0.02; ns) when all participants were pooled together. When considering the two groups of participants, such correlation was again low and not significant. However, a strong positive correlation was observed between mind wandering and drowsiness across all participants (r 2 = 0.31; p = 0.004), with positive correlations observed for both groups of subjects, and reaching significance for expert practitioners (r 2 = 0.66; p = 0.0013) but not for non-expert practitioners (r 2 = 0.2; ns). Average subject responses are summarized in Table 1.
To test whether responses to probes changed over time, we compared the behavioral responses for the first 20 probes, comprising at least 75% of the subjects (probes were delivered at random intervals; thus, subjects received a varying number of probes—3 subjects in each group had less than 20 probes). As shown in Fig. 2, although both groups start at approximately the same meditation depth, the group of expert practitioners showed a highly significant increase from 0.034 to 0.058 points in the depth of their meditation over time, p = 0.0000002; r 2 = 0.8 (the ranges provided here indicate a 95% confidence (CI) interval, with all statistics using parametric methods), as well as a significant increase for non-expert practitioners, p = 0.02; r 2 = 0.27 (increase from 0.003 to 0.026 points in meditation depth per probe). No significant difference was observed in the intercept between the two groups, with a 95% confidence interval for a non-overlapping slope indicating a significant difference between the two groups. A regression analysis revealed a significant reduction over time for the mind wandering scale for the expert practitioners (p = 0.02; r 2 = 0.27 for experts, and p = ns; r 2 = 0.02 for non-experts). The decrease in mind wandering for experts was significantly negative, with a slope CI of −0.033 to −0.003. The intercept between the two groups did not differ significantly. Interestingly, our sleep-scale measures indicate that the non-expert group experienced increased drowsiness compared to the expert group at the onset of the experiment (CI of 0.62–0.86 at the first probe for the expert group and 0.37–0.61 for non-expert group), suggesting that meditation experience may potentially affect overall subjective alertness level. Additionally, the expert group reported a significantly reduced depth of drowsiness as the experiment progressed, while the non-expert group report significantly increasing drowsiness (slope CI of −0.034 to −0.012 for experts and 0.008–0.029 for non-experts). Both regressions were highly significant (experts p = 0.0004; r 2 = 0.51 vs. non-experts p = 0.002; r 2 = 0.43).
Evolution of the responses to experiential probes throughout the experiment for the three questions pertaining to meditation depth, mind wandering and sleepiness. Only the first 20 probes for each subject are shown. Thick lines indicate mean response on each probe for the two groups of participants. Dashed lines indicate linear regressions. Significative results are reported in the text
While significant behavioral differences were observed between experts and non-experts when considering mind wandering, we observed large inter-subject variations. The relatively large variance across participants was most likely due the subjective nature of the task. Participants were likely to have rated the questions differently—with some participants being biased toward providing high ratings, as compared to others being biased toward providing low ratings. This is consistent with the fact that we observed larger differences between expert and non-expert practitioners when we considered the normalized individual ratings for both the meditation and mind wandering responses, than when we considered absolute ratings. Calculating the differences between the ratings of these two questions effectively minimized the absolute response bias participants may have had. Since a comparison of ratings across participants is subject to a large amount of noise, we adopted the strategy of splitting trials in two categories: trials for which ratings on the meditation scale were larger (considered meditation trials) than mind wandering and vice versa (mind wandering trials). Trials when the two ratings for the two conditions were equal were ignored. Two subjects were excluded from these analyses because they reported no behavioral trials for one of the two conditions.
Our behavioral analyses found that expert practitioners reported significantly more meditation trials as compared to mind wandering trials [mean of 75.4 trials for Med; mean of 24.6 trials for mind wandering; standard error (SE) of 4.4 in both cases, p = 0.00014], while non-expert practitioners showed no such effect (mean of 42.7 trials for Med; mean of 57.3 trials for mind wandering; SE of 6.2, ns) as shown in Fig. 3. Group-level statistics showed that non-expert practitioners reported a significantly greater number of mind wandering trials as compared to expert practitioners (difference of 32.7; p = 0.00038) and that expert practitioners reported a significantly greater number of meditation trials as compared to non-expert meditators (difference of 32.7; p = 0.00052).
Expert practitioners reported a greater number of probed trials in which they were actively engaged in their meditation as compared to mind wandering. They also reported a greater number of probed trials in which they were actively engaged in their meditation than non-expert practitioners. Error bars indicate 95% confidence intervals which was calculated by multiplying the standard error by 1.96
EEG activity time-locked to experience-sampling probes
Event-related spectral perturbation (ERSP) of the EEG signal was assessed during the 10-s immediately preceding probe onset (see “Methods and materials” section). Expert practitioners showed significantly increased modulation of theta activity (4–7 Hz) across the frontal cortex (p < 0.02 after correction for multiple comparisons), as well as alpha activity (9–11 Hz) primarily concentrated over the somatosensory cortex (p < 0.02), during meditation trials as compared to mind wandering trials. When the same analysis was conducted on the non-expert meditation group, no significant differences were observed (see discussion). No interaction was observed between trial type and meditation expertise.
For each expert subject, we averaged the theta and alpha power for the electrodes which showed significant differences in both frequency bands (Fig. 4). We observed a positive correlation between theta difference between meditation and mind wandering state and alpha power difference between meditation and mind wandering state (r 2 = 0.42; p = 0.02), indicating that subject that had a larger theta difference between conditions also had a larger alpha difference. We did not observe correlations between behavioral responses (responses averages for expert subjects from Q1 to Q3 in Table 1) and theta or alpha power differences.
Event-related spectral perturbation (ERSP) plots, and differential plots of significance in theta (4–7 Hz) and alpha (9–11 Hz) band activity for expert meditation practitioners. Power scale is expressed in μV2/Hz. Black dots on the difference plots indicate electrodes significant at p < 0.02 after cluster correction for multiple comparisons
Discussion
Our results provide some of the first evidence that meditation expertise is associated with an attenuated frequency of mind wandering. We observed that meditation expertise was associated with a significantly greater depth of meditative absorption and a significantly reduced number of mind wandering episodes meausred throughout our experience-sampling paradigm. These findings suggest that meditation training reduces the susceptibility of the mind to wander, subsequently leading to longer periods of meditative absorption (discussed below). Our findings provide supporting evidence that increased theta activity over mid-frontal theta regions and alpha activity primarily focused over the somatosensory cortex are markers of sustained and internally directed attentional states of awareness cultivated via long-term meditation practice. Additionally, the modulations seen in mid-frontal theta and somatosensory alpha, both traditionally markers of various types of executive functions, add to an expanding body of literature suggesting that meditation training may modulate some of the neural mechanisms involved in cognitive control and attention (Hölzel et al. 2011; Mrazek et al. 2013; Slagter et al. 2007). Given that our limited sample size (n = 24) was made of individuals who practiced exclusively in the Himalayan meditation tradition, further studies are necessary to validate our findings in other meditation traditions.
Our EEG findings indicate that the mid-frontal theta and somatosensory alpha rhythms, often observed during executive functioning tasks and cognitive control (Cavanagh and Shackman 2015; Cavanagh and Frank 2014; Enriquez-Geppert et al. 2014; Bollimunta et al. 2011), can also be seen during internally guided states of focus such as meditation and are consistent with previous research (Aftanas and Golocheikine 2001; Kerr et al. 2013). It may also suggest a functional relationship between the sources contributing to our observed mid-frontal theta activity and the broader frontoparietal control network involved in maintaining top-down representations of goal states, learning and directed attention (deBettencourt et al. 2015; Spreng 2012; Cavanagh and Frank 2014). The role of theta in meditation practice and the cultivation of top-down control via the enhancement of monitoring and possibly enhanced conflict detection falls in line with the established literature regarding its specific role in learning (Swick and Turken 2002; Haegens et al. 2010). Cavanagh and Frank (2014) have suggested that cortical theta-band oscillations may serve as a candidate mechanism by which neurons communicate top-down control over long-range and broad networks. Mid-frontal theta has been proposed to function as a temporal template for organizing mid-frontal neuronal processes, and theta-band phase dynamics may entrain disparate neural systems when cognitive control is needed (Cavanagh and Frank 2014). This is supported by findings suggesting that cortical and subcortical areas are interconnected via the cingulate cortex (Morecraft and Tanji 2009; Bollimunta et al. 2009). Our study thus provides new evidence to support the claims posited by Spreng (2012) that the maintenance of both internal and external orientations of focus may be maintained by similar neural mechanisms. Our findings suggest that meditation training may target the neural substrates underlying these oscillations.
The observed increase in alpha activity in our expert meditation group supports it putative role in the processing and integration of somatosensory information (Kerr et al. 2013; Whitmarsh et al. 2014), and cognitive entrainment. Somatosensory alpha modulation has been established in the facilitation of working memory performance, in addition to mindfulness meditation practitioners showing an enhanced ability to modulate alpha power in sensory neocortex in response to a cue (Kerr et al. 2013). Kerr et al. (2013) suggest that mindfulness meditation enhances top-down modulation of alpha by facilitating precise alterations in timing and efficacy of the somatosensory cortex thalamocortical inputs. Thus, our findings of enhanced alpha activity support these respective findings and are consistent with the hypothesis that cortical mechanisms underlying somatosensory perception may be modulated by meditation training. Furthermore, our findings provide further support for theories that an enhanced integration of sensory information and attention can be learned and modulated via top-down mechanisms.
Our implementation of a probe-caught mind wandering paradigm was based on previous findings which suggest that this method is thought to reflect the actual frequency of mind wandering episodes, whereas mind wandering that is self-reported may reflect an individual’s metacognitive awareness of mind wandering (Smallwood and Schooler 2006). While meditation practice may increase the number of self-caught mind wandering episodes over time by enhancing metacognitive awareness of internal experience, it may also be due to a reduced number of lapses of task-related attention following extensive training, limiting the opportunities for practitioners to subsequently identify and report mind wandering episodes. Thus, variations in reports from both self- and probe-caught mind wandering paradigms should be mutually considered in future studies. While our research findings do suggest that overtime meditation practice may fundamentally reduce the frequency of spontaneous thought, this may occur alongside the ability to actively identify and disengage from mind wandering and subsequently reorient attention. It may also be the case that meditation practice facilitates the unification of various attentional mechanisms so as to moderate mind wandering. Future avenues of research on mind wandering and meditation training should focus on disentangling whether meditation increases the metacognitive awareness of mind wandering and the subsequent reorientation of attention, if meditation enhances a fundamental capacity of allocating attentional resources, or if meditation facilitates an overall reduction in the occurrence of mind wandering events as our findings suggest.
It remains possible that what distinguishes experts from novices is not necessarily their attentional capacity for internal focus per se, but rather their metacognitive capacity to accurately label mental states (Baird et al. 2014). It is also important to note that due to the nature of our experimental paradigm in which participants were being auditorily probed about every 3 min, exit interviews indicated that the majority of participants were unable to experience particularly “deep” meditative states. As indicated in our behavioral data, both groups experienced a progressive increase in the depth of their meditation over time, suggesting that both groups progressed in their ability to engage in their meditation practice and perform the task simultaneously. Given that meditative experience enhances individuals’ ability to monitor internal states, it would follow that expert meditators would also be better at labeling these states. Thus, we cannot rule out the possibility that our novice meditators were engaged in focused meditative practices in a way that would be similar to experts and that the differences in EEG we observed were due to a decreased capacity of novices to accurately label mental states. Our corresponding EEG and behavioral findings in expert practitioners may therefore provide supporting evidence for an enhanced metacognitive accuracy in reporting as a result of long-term meditation practice.
Cognitive control is one of the most essential sets of cognitive functions for our interactions with the external world, with individual differences in these cognitive functions predicting success across academic and professional domains (Hirsh and Inzlicht 2010). Research is beginning to confirm that impaired cognitive control in the hallmark of clinical disorders such as ADHD, obsessive compulsive disorder and schizophrenia (Kaser et al. 2013; Yordanova et al. 2013; Mazaheri et al. 2014). Our findings suggest that contemplative practices and techniques may be useful in treating an increasingly wide array of medical and clinical disorders through training and exercising the neural circuitry underlying the top-down regulation of executive functions, somatosensory processing and metacognition. Furthermore, they provide support for the development of cognitive protocols and brain computer interfaces that aim to modulate these neural networks and their underlying cortical and subcortical structures.
Our behavioral results are one of the first to show that an attenuated frequency of mind wandering can be considered a direct marker of meditation expertise. Furthermore, the corresponding behavioral and EEG findings in our expert practitioner group provide evidence for enhanced metacognitive accuracy in reporting as a result of long-term meditation practice. Finally, the increased mid-frontal theta and alpha rhythms observed during meditative absorption provide direct evidence to support the hypothesis that the maintenance of both internal and external orientations of focus may be maintained by similar neural mechanisms.
References
Aftanas LI, Golocheikine SA (2001) Human anterior and frontal midline theta and lower alpha reflect emotionally positive state and internalized attention: high-resolution EEG investigation of meditation. Neurosci Lett 310(1):57–60
Baars BJ (2005) Global workspace theory of consciousness: toward a cognitive neuroscience of human experience. Prog Brain Res 150:45–53
Baird B, Smallwood J, Mrazek MD, Kam JW, Franklin MS, Schooler JW (2012) Inspired by distraction mind wandering facilitates creative incubation. Psychol Sci 0956797612446024
Baird B, Mrazek M, Phillips DT, Schooler JW (2014) Domain-specific enhancement of metacognitive ability following meditation training. J Exp Psychol Gen 143(5):1972–1979
Benjamini Y, Yekutieli D (2001) The control of the false discovery rate in multiple testing under dependency. Ann Stat 29:1165–1188
Berkovich-Ohana A, Harel M, Hahamy A, Arieli A, Malach R (2016) Data for default network reduced functional connectivity in meditators, negatively correlated with meditation expertise. Data Brief 8:910–914
Bollimunta A, Chen Y, Schroeder CE, Ding M (2009) Characterizing oscillatory cortical networks with Granger causality. In: Rubin J, Josic K, Matias M, Romo R (eds). Coherent behavior in neuronal networks. Springer, New York, pp 169–189
Bollimunta A, Mo J, Schroeder CE, Ding M (2011) Neuronal mechanisms and attentional modulation of corticothalamic alpha oscillations. J Neurosci 31:4935–4943
Braboszcz C, Delorme A (2011) Lost in thoughts: neural markers of low alertness during mind wandering. Neuroimage 54(4):3040–3047
Buckner RL, Andrews-Hanna JR, Schacter DL (2008) The brain’s default network. Ann NY Acad Sci 1124(1):1–38
Cahn BR, Polich J (2006) Meditation states and traits: EEG, ERP, and neuroimaging studies. Psychol Bull 132(2):180
Cavanagh J, Frank M (2014) Frontal theta as a mechanism for cognitive control. Trends Cogn Sci 18(8):414–421
Cavanagh JF, Shackman AJ (2015) Frontal midline theta reflects anxiety and cognitive control: meta-analytic evidence. J Physiol Paris 109(1):3–15
Christoff K, Gordon AM, Smallwood J, Smith R, Schooler JW (2009) Experience sampling during fMRI reveals default network and executive system contributions to mind wandering. Proc Natl Acad Sci 106(21):8719–8724
deBettencourt MT, Cohen JD, Lee RF, Norman KA, Turk-Browne NB (2015) Closed-loop training of attention with real-time brain imaging. Nat Neurosci 18(3):470–475
Delorme A, Makeig S (2004) EEGLAB: an open source toolbox for analysis of single-trial EEG dynamics including independent component analysis. J Neurosci Methods 134(1):9–21
Delorme A, Sejnowski T, Makeig S (2007) Enhanced detection of artifacts in EEG data using higher-order statistics and independent component analysis. Neuroimage 34(4):1443–1449
Desbordes G, Gard T, Hoge EA, Hölzel BK, Kerr C, Lazar SW, Olendzki A, Vago DR (2015) Moving beyond mindfulness: defining equanimity as an outcome measure in meditation and contemplative research. Mindfulness 6(2):356–372
Enriquez-Geppert S, Huster RJ, Scharfenort R, Mokom ZN, Zimmermann J, Herrmann CS (2014) Modulation of frontal-midline theta by neurofeedback. Biol Psychol 95:59–69
Garrison KA, Zeffiro TA, Scheinost D, Constable RT, Brewer JA (2015) Meditation leads to reduced default mode network activity beyond an active task. Cogn Affect Behav Neurosci 15(3):712–720
Gusnard DA, Raichle ME (2001) Searching for a baseline: functional imaging and the resting human brain. Nat Rev Neurosci 2(10):685–694
Haegens S, Osipova D, Oostenveld R, Jensen O (2010) Somatosensory working memory performance in humans depends on both engagement and disengagement of regions in a distributed network. Hum Brain Mapp 31(1):26–35
Hasenkamp W, Barsalou LW (2012) Effects of meditation experience on functional connectivity of distributed brain networks. Front Hum Neurosci 6:38
Hirsh JB, Inzlicht M (2010) Error-related negativity predicts academic performance. Psychophysiology 47(1):192–196
Hölzel BK, Lazar SW, Gard T, Schuman-Olivier Z, Vago DR, Ott U (2011) How does mindfulness meditation work? Proposing mechanisms of action from a conceptual and neural perspective. Perspect Psychol Sci 6(6):537–559
James W (1890) The principles of psychology. Holt, New York
Jang JH, Jung WH, Kang DH, Byun MS, Kwon SJ, Choi CH, Kwon JS (2011) Increased default mode network connectivity associated with meditation. Neurosci Lett 487(3):358–362
Kaser M, Soltesz F, Lawrence P, Miller S, Dodds C, Croft R et al (2013) Oscillatory underpinnings of mismatch negativity and their relationship with cognitive function in patients with schizophrenia. PLoS ONE 8(12):e83255
Kerr CE, Jones SR, Wan Q, Pritchett DL, Wasserman RH, Wexler A et al (2011) Effects of mindfulness meditation training on anticipatory alpha modulation in primary somatosensory cortex. Brain Res Bull 85(3):96–103
Kerr CE, Sacchet MD, Lazar SW, Moore CI, Jones SR (2013) Mindfulness starts with the body: somatosensory attention and top-down modulation of cortical alpha rhythms in mindfulness meditation. Front Hum Neurosci 7:12
Killingsworth MA, Gilbert DT (2010) A wandering mind is an unhappy mind. Science 330(6006):932
Maris E, Oostenveld R (2007) Nonparametric statistical testing of EEG-and MEG-data. J Neurosci Methods 164(1):177–190
Mazaheri A, Fassbender C, Coffey-Corina S, Hartanto TA, Schweitzer JB, Mangun GR (2014) Differential oscillatory electroencephalogram between attention-deficit/hyperactivity disorder subtypes and typically developing adolescents. Biol Psychiatry 76(5):422–429
Morecraft RJ, Tanji J (2009) Cingulofrontal interactions and the cingulate motor areas. In: Vogt BA (ed) Cingulate neurobiology and disease. Oxford University Press, Oxford, pp 113–144
Mrazek MD, Smallwood J, Schooler JW (2012) Mindfulness & mind-wandering: finding convergence through opposing constructs. Emotion. doi:10.1037/a0026678
Mrazek MD, Franklin MS, Phillips DT, Baird B, Schooler JW (2013) Mindfulness training improves working memory capacity & GRE performance while reducing mind wandering. Psychol Sci 24(5):776–781
Poerio G, Totterdell P, Miles E (2013) Mind wandering and negative mood: does one thing really lead to another? Conscious Cogn 22(4):1412–1421
Raichle ME (2015) The brain’s default mode network. Annu Rev Neurosci 38:433–447
Schooler JW, Smallwood J, Christoff K, Handy TC, Reichle ED, Sayette MA (2011) Meta-awareness, perceptual decoupling and the wandering mind. Trends Cogn Sci 15(7):319–326
Segal ZV, Teasdale JD, Williams JMG (2004) Mindfulness-based cognitive therapy: theoretical rationale and empirical status. In: Hayes SC, Follette VM, Linehan M (eds) Mindfulness and acceptance: expanding the cognitive-behavioral tradition. Guilford Press, New York, NY, pp 45–65
Slagter HA, Lutz A, Greischar LL, Francis AD, Nieuwenhuis S, Davis JM, Davidson RJ (2007) Mental training affects distribution of limited brain resources. PLoS Biol 5(6):e138
Smallwood J, Schooler JW (2006) The restless mind. Psychol Bull 132(6):946
Smallwood J, Baracaia S, Lowe M, Obonsawin M (2003) Task unrelated thought whilst encoding information. Conscious Cogn 12(3):452–484
Smallwood J, Fishman DJ, Schooler JW (2007) Counting the cost of an absent mind: mind wandering as an underrecognized influence on educational performance. Psychon Bull Rev 14(2):230–236
Spreng RN (2012) The fallacy of a “task-negative” network. Front Psychol 3:145
Spreng RN, Sepulcre J, Turner GR, Stevens WD, Schacter DL (2013) Intrinsic architecture underlying the relations among the default, dorsal attention, and frontoparietal control networks of the human brain. J Cogn Neurosci 25(1):74–86
Swick D, Turken U (2002) Dissociation between conflict detection and error monitoring in the human anterior cingulate cortex. Proc Natl Acad Sci 99(25):6354–16359
Sze JA, Gyurak A, Yuan JW, Levenson RW (2010) Coherence between emotional experience and physiology: does body awareness training have an impact? Emotion 10:803–814
Tang YY, Hölzel BK, Posner MI (2015) The neuroscience of mindfulness meditation. Nat Rev Neurosci 16(4):213–225
Teper R, Inzlicht M (2013) Meditation, mindfulness and executive control: the importance of emotional acceptance and brain-based performance monitoring. Soc Cogn Affect Neurosci 8(1):85–92
Welch PD (1967) The use of fast Fourier transform for the estimation of power spectra: a method based on time averaging over short, modified periodograms. IEEE Trans Audio Electroacoust 15(2):70–73
Whitmarsh S, Barendregt H, Schoffelen JM, Jensen O (2014) Metacognitive awareness of covert somatosensory attention corresponds to contralateral alpha power. Neuroimage 85:803–809
Yordanova J, Kolev V, Rothenberger A (2013) Event-related oscillations reflect functional asymmetry in children with attention deficit/hyperactivity disorder. Clin Neurophysiol 62:289–301
Zanesco AP, King BG, MacLean KA, Jacobs TL, Aichele SR, Wallace BA, Saron CD (2016) Meditation training influences mind wandering and mindless reading. Psychol Conscious Theory Res Pract 3(1):12
Acknowledgements
This work was supported by Grants from the ANR (Agence Nationale pour la Recherche) ANR-12-JSH2-0009 and the BIAL foundation BIAL-08-162. The authors wish to thank the late Swami Veda, Gopalkrishna Prabhu, Pravin Kumar Soni, John Sellinger and all the members of the Swami Rama Sadhaka Grama ashram where the data were collected and the volunteers who participated in this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Brandmeyer, T., Delorme, A. Reduced mind wandering in experienced meditators and associated EEG correlates. Exp Brain Res 236, 2519–2528 (2018). https://doi.org/10.1007/s00221-016-4811-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-016-4811-5