Abstract
This study investigates whether there is lateralized processing of emotional scenes in the visual periphery, in the absence of eye fixations; and whether this varies with emotional valence (pleasant vs. unpleasant), specific emotional scene content (babies, erotica, human attack, mutilation, etc.), and sex of the viewer. Pairs of emotional (positive or negative) and neutral photographs were presented for 150 ms peripherally (≥6.5° away from fixation). Observers judged on which side the emotional picture was located. Low-level image properties, scene visual saliency, and eye movements were controlled. Results showed that (a) correct identification of the emotional scene exceeded the chance level; (b) performance was more accurate and faster when the emotional scene appeared in the left than in the right visual field; (c) lateralization was equivalent for females and males for pleasant scenes, but was greater for females and unpleasant scenes; and (d) lateralization occurred similarly for different emotional scene categories. These findings reveal discrimination between emotional and neutral scenes, and right brain hemisphere dominance for emotional processing, which is modulated by sex of the viewer and scene valence, and suggest that coarse affective significance can be extracted in peripheral vision.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Probably due to their adaptive importance as appetitive and aversive cues, emotional visual stimuli receive heightened attention in the cognitive and neural systems, relative to neutral stimuli (see Bradley et al. 2012; Domínguez-Borrás and Vuilleumier 2013; Mohanty and Sussman 2013; Pessoa et al. 2013). In most prior research, emotional pictures have been presented in central vision, while fewer studies have investigated extrafoveal vision (see below).Footnote 1 We aim to extend this approach by investigating: (a) whether affective significance can be discriminated when emotional and neutral visual scenes appear in peripheral vision, in the absence of eye fixations; (b) whether there is any visual field advantage, and therefore brain lateralization or hemisphere dominance, in emotional scene processing; (c) whether lateralization depends on the scene affective valence (pleasant vs. unpleasant) and specific content (e.g., erotica, mutilation, etc.); and (d) whether sex of the observers interacts with visual field, valence, or specific scene content.
Eye-movement research has found that emotional scenes attract overt attention in extrafoveal vision (>2° away from current fixation). When an emotional and a neutral image are presented concurrently, the probability of the first fixation on the emotional picture is higher (Alpers 2008; Calvo and Lang 2004; Calvo et al. 2008; Nummenmaa et al. 2006), which reveals selective initial orienting. This occurs—albeit attenuated—even when participants are explicitly instructed to look first at the neutral picture (Nummenmaa et al. 2006). Saccade latencies have also been found to be shorter for emotional than for neutral scenes (Calvo et al. 2007). Relatedly, when viewers are asked to perform a saccade to an exogenous cue, saccade trajectories deviate toward the location of emotional scene distractors (McSorley and van Reekum 2013), and saccade latency is delayed when the image opposite to the instructed direction is emotional (Nummenmaa et al. 2009). Similarly, anti-saccade responses (i.e., to the opposite side) show more errors toward emotional than to neutral images (Kissler and Keil 2008). In the absence of low-level image differences, these findings suggest that emotional content was perceived and captured attention extrafoveally.
Relatedly, some electrocortical ERP (event-related potential) studies in which scenes were displayed in extrafoveal vision have also provided evidence of emotional processing, as shown by an affective modulation of both early and late ERP components. Negatively valenced emotional scenes (~2°) have been found to enhance activation in the temporo-occipital and parietal cortex, in comparison with neutral scenes (Keil et al. 2005). Also, emotional scenes in the near periphery (8.2°), but not in the far periphery (16.4°), elicited a negative ERP differential (relative to neutral scenes) over temporo-occipital scalp regions (early posterior negativity; EPN, 200–280 ms), and a later positivity over centro-parietal areas (late positive potential; LPP, 400–800 ms) (De Cesarei et al. 2009). Nevertheless, an affective modulation of early ERPs (P1, 145 ms) in lateral-occipital and infero-temporal cortices has been reported for emotional pictures even at a 30° eccentricity (Rigoulot et al. 2008). Altogether, the fact that these electrocortical potentials were enhanced by emotional relative to neutral scenes suggests that emotional content was encoded as different from non-emotional content.
The previous studies did not explicitly investigate lateralization effects, with the exception of Alpers (2008) and Keil et al. (2005). Alpers (2008) found that the initial saccade was more often spontaneously directed toward the emotional scene (both pleasant and unpleasant) than toward the paired neutral picture, when the former was presented in the left-visual hemifield. Keil et al. (2005) found right occipito-temporal and parietal-enhanced ERP activation when an unpleasant scene was presented in the left-visual field. This suggests, in agreement with a traditional ‘right-hemisphere hypothesis’, that emotional processing in the brain is mediated mainly by the right hemisphere (see Heller et al. 1998). Nevertheless, recent neurophysiological research has revealed some empirical complexities beyond this hypothesis, according to meta-analyses of neuroimaging studies (Duerden et al. 2013; Stevens and Hamann 2012) and also narrative reviews of studies using a variety of approaches (Kret and de Gelder 2012; Gianotti 2012; Whittle et al. 2011).
From the data reviewed by these studies, four hypotheses can be proposed (see Gianotti 2012): (a) perception of emotional stimuli involves a general dominance of the right hemisphere regardless of specific affective valence (‘right-hemisphere hypothesis’); (b) hemispheric specialization depends on the scene affective valence, with an opposite dominance of the left hemisphere for positive emotions and the right hemisphere for negative emotions (‘valence–specificity hypothesis’); (c) modulation of the right and the left hemisphere depends on specific types of emotional content (‘emotional-type hypothesis’); and (d) lateralization is modulated by sex of the viewer, with females and males recruiting different brain structures interactively with affective valence (‘sex–valence–specificity hypothesis’). The meta-analyses and narrative reviews have shown that some brain structures are typically activated, such as the amygdala, in the processing of emotional—relative to neutral—visual stimuli. However, a variable pattern has emerged regarding lateralization, depending on combinations of valence (pleasant vs. unpleasant), specific emotional content (e.g., erotica, mutilation, etc.), and sex of the viewer (Duerden et al. 2013; Kret and de Gelder 2012; Stevens and Hamann 2012; Whittle et al. 2011). In such integrative analyses and reviews, a wide range of emotional stimuli was included (faces, scenes, words, sounds, imagery, films, odors, etc.), which may have contributed to empirical complexities and discrepancies.
Accordingly, in the current study, we focused on visual scenes (depicting people doing things), to reduce the range of stimulus format variability. At the same time, we expanded on the specificity of emotional scene contents, to explore potential differences (or generalization) in lateralization and interactions with sex. Both factors (visual stimulus format and specific scene content) have proved to be important in emotional brain processing (Sabatinelli et al. 2011; Weinberg and Hajcak 2010). As an extension of prior studies, which were mainly concerned with stimuli in central vision, we presented them in peripheral vision (between 6.5° and 19.0° away from a central fixation point). A major reason of importance for the investigation of peripheral vision is that it facilitates exploration of laterality mechanisms in the brain, by means of presenting the stimuli in either one or the other hemifield. Given the contralateral neuroanatomical organization of the visual system—with stimuli presented to the right hemifield being initially projected to a greater extent to the left brain hemisphere, and viceversa, facilitated perception of stimuli in, for example, the left-visual field would reveal a right-hemisphere dominance.
To examine the four lateralization hypotheses, we orthogonally combined the following factors: visual field (left vs. right) of the stimuli, sex of the participant (females vs. males), and scene affective valence (pleasant vs. unpleasant). Such manipulations were relevant to test the ‘right-hemisphere’, the ‘valence–specificity’, and the ‘sex–valence–specificity’ hypotheses, respectively. In addition, to examine the ‘emotional-type hypothesis’, we distinguished specific content categories (e.g., erotica, attack, etc.) within each valence dimension. Pairs of emotional–neutral scenes appeared peripherally for 150 ms, while eye fixations were prevented. Participants responded on which side (left or right) the emotional scene was. The degree of emotional–neutral discrimination was determined by the probability and speed of accurate responding above chance level. Within this paradigm, support for the ‘right-hemisphere hypothesis’ involves more accurate and faster identification of both pleasant and unpleasant scenes in the left (LVF) than in the right (RVF) visual field. Support for the ‘valence–specificity hypothesis’ involves a recognition advantage (i.e., more accuracy and shorter RTs) for unpleasant scenes in the LVF but for pleasant scenes in the RVF. The other two hypotheses are less well developed (see above), and predictions are therefore exploratory. For the ‘sex–valence–specificity hypothesis’, the recognition advantage (on the LVF or RVF) will be different for females and males depending on pleasant or unpleasant content, i.e., a valence by sex interaction. For the ‘emotional-type hypothesis’, the recognition advantage will vary for the various specific scene contents.
Methods
Participants
In the main experiment, 48 undergraduates (24 females; between 20 and 30 years of age) received course credit or monetary incentive (5 €) for their participation. All were right-handed. They were informed that they would be presented with pleasant, unpleasant, and non-emotional photographs. Informed consent was obtained.
Stimuli
We used 70 pleasant scene images, 72 unpleasant images, and 142 neutral images. Most (91.5 %) of the emotional stimuli were photographs from the International Affective Picture System (IAPS; Lang et al. 2008). Within the pleasant dimension, the following seven scene categories were formed: babies, families, romance, erotica females, erotica males, erotica couples (heterosexual), and adventure/sports. Within the unpleasant dimension, there were six categories: accidents, loss/illness, human attack with or without weapons, mutilation, and male-to-female aggression.Footnote 2 All the scenes depicted people to keep some homogeneity across the variety of contents, and given that presence versus absence of people produce different effects on both behavioral and electrocortical measures of emotion perception, and interact with sex of the viewer (Colden et al. 2008; Groen et al. 2013).
The distinction of specific content categories was based on prior research in which pictures were presented to central vision (Bradley et al. 2001a, b; Calvo and Avero 2009; Weinberg and Hajcak 2010). Although the different categories may be seen as not strictly independent, significant differences have been found in subjective ratings of valence and arousal (see Bradley and Lang 2007; Gomez et al. 2013; Moltó et al. 2013), rating reaction times (Calvo and Avero 2009), psychophysiological measures (e.g., galvanic skin response; Bradley et al. 2001a, b), and brain activity (ERPs; Briggs and Martin 2009; Schupp et al. 2004; Weinberg and Hajcak 2010). In the current study, we aimed to extend such distinctions to extrafoveal vision.
For the pleasant valence dimension, 10 photographs were chosen for each of the seven categories; for the unpleasant dimension, 12 photographs for each of the six categories. The IAPS numbers of each photograph are shown in the ‘Appendix’. Nevertheless, to complete the number of images on all the categories, we had to add some new images freely available from the Internet. Then, we conducted a norming study to validate all the pictures with the same criterion and for participants with the same background as those in the main experiment. In the norming study, the photographs were shown to a new sample of undergraduates (34 females and 34 males), who provided valence ratings (in a 1—unpleasant—to 9—pleasant—scale), according to the SAM (self-assessment mannequin) procedure (Lang et al. 2008). Each participant saw one picture at a time, in random order and self-paced mode, on a computer screen. Results confirmed that the pleasant scenes were in fact rated as more pleasant (M = 7.15, females; 6.88, males), and the unpleasant scenes were more unpleasant (M = 1.55, females; 1.88, males), than the neutral scenes (M = 5.50, females; 5.35, males), both Fs(2, 281) ≥ 1,086.32, p < .0001 (all post hoc comparisons, ps < .0001, with Bonferroni correction). The respective reaction times were as follows: pleasant (M = 1,719 vs. 1,636 ms); unpleasant (M = 1,704 vs. 1,697 ms); neutral (M = 2,101 vs. 1,904 ms); both Fs(2, 281) ≥ 39.97, p < .0001, with shorter latencies for both the pleasant and the unpleasant relative to the neutral scenes (all ps < .0001).Footnote 3
Assessment of physical low-level image properties and visual saliency
Footnote 4On the assumption that luminance is a main factor attracting attention and facilitating perception in peripheral vision, we initially adjusted the mean luminance of all the scene images so that they were virtually identical, F(13, 270) = 0.017, p = 1, ns. Then, we computed the SD luminance, RMS or root-mean-square contrast, skewness, kurtosis, energy, and SNR or signal-to-noise ratio, by means of Matlab 7.0 (The Mathworks, Natick, MA, USA), and assessed red, green, and blue saturation by means of Adobe Photoshop 6.0. We also measured the image jpeg file size, as an index of perceptual complexity (see Peyk et al. 2009). Each of these measures was analyzed in a one-way (14: neutral scenes plus each of the 13 emotional categories: babies, accidents, etc.) ANOVA. No significant differences appeared for any of these variables (ps > .10), except for red saturation, F(13, 270) = 4.93, p < .0001, η 2 p = .19, with the erotica couples category having higher values than most of the other categories. Importantly, the neutral scenes did not differ from any of the other categories.
The visual saliency of an image has been proposed to influence shifts of covert and overt attention (Borji and Itti 2013). Accordingly, we assessed visual saliency to determine whether any emotional scenes could be more salient and, therefore, more easily perceivable than neutral scenes. By means of the iLab Neuromorphic Vision C++ Toolkit (iNVT; Itti 2006; Itti and Koch 2000; http://ilab.usc.edu/toolkit/) and Matlab 7.0 (the ‘pfmreadmatlab.m’ function was used to load the iNVT-generated saliency maps and perform the region analysis), we obtained the saliency values for each pair of emotional–neutral scenes when displayed in peripheral vision. The iNVT algorithm simulates which image attracts attention, by mimicking the response properties of retinal neurons, lateral geniculate nucleus, thalamus, and V1. The resulting saliency maps represent the conspicuity of an image as a function of contrast, color, and spatial orientation. The saliency values of our stimuli were analyzed in a 13 (specific emotional categories) × 2 (emotional vs. neutral) ANOVA. No significant effect emerged (all the Fs < 1, ps ≥ .58). Thus, visual saliency was equivalent for emotional and neutral scenes, with no differences among the emotional categories.
Procedure (main experiment)
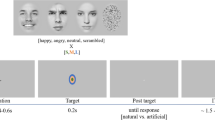
In the main experiment, each participant was presented with 284 experimental trials on a 22″ screen in four blocks. Stimulus presentation and response collection were controlled by E-Prime software. On each trial (see Fig. 1), following a 500-ms central fixation cross, two visual scenes, one emotional (either pleasant or unpleasant) and one neutral, were displayed simultaneously for 150 ms (while the central cross remained). At a constant 60-cm viewing distance, each picture was located 6.5° (inner edge of image) away (to the left or right) from a central cross, and subtended 10.5° (height) × 12.5° (width). The scenes were replaced by a pair of backward masks of the same luminance as the scenes. The masks remained until the participant responded whether the emotional scene was on the left or the right, by pressing one of two keys in a standard computer keyboard with the index fingers. Response accuracy and reaction times were collected. This was followed by a 1,500 ms inter-trial interval.
Instructions to participants indicated that pairs of pictures would be presented on each trial, on separate sides of the screen, with one image depicting an emotional (either positive or negative) scene and the other portraying a non-emotional scene. Participants should always look at a central fixation cross between the pictures and refrain from directly looking at the pictures (in fact, if this occurred, the experimenter could easily notice it online, and warn the participant). Participants were asked to respond ‘on which side the emotional scene had appeared’. For each participant, each emotional scene was presented twice, once in the LVF and another in the RVF, in different blocks, each time paired with a different neutral scene. All the participants were presented with all the images. Nevertheless, across participants, each emotional scene was paired with four different neutral scenes. On each block, there were 35 trials with pleasant-neutral pairs and 36 trials with unpleasant-neutral pairs. The order of blocks was counterbalanced, and trials were randomized.
Given that we aimed to investigate the processing of scenes in peripheral vision, it was critical that they were not foveally fixated. To this end, in addition to asking the participants to always look at the central fixation cross, they wore SensoMotoric Instruments GmbH (Teltow, Germany) Eye-Tracking Glasses (resolution: 1,280 × 960 pixels; accuracy: 1°; sampling rate: 30 Hz binocular; parallax compensation), which monitored oculomotor behavior on-line. Saccades were initiated toward one of the scenes on 7.8 % of trials, but on 90 % of them this occurred after the 150-ms scene display, and therefore, there were no fixations on the images. The remaining trials with anticipatory saccades (~2.2 times per participant, out of 284 trials) were removed.
Experimental design (main experiment)
The design involved an orthogonal combination of a between-subjects factor (Sex of the participant: female vs. male), and two within-subjects factors (scene emotional Valence: pleasant vs. unpleasant; and Visual field of emotional scene: left vs. right). Nevertheless, in an extended approach, we also took specific categories of scene content into account. To this end, we decomposed the pleasant and the unpleasant affective dimensions into seven and six levels, as described above. The scene categories could not be orthogonally combined with valence due to their being qualitatively different for each valence group. Accordingly, the scene category factor was analyzed separately for the pleasant and the unpleasant dimension, in a sex by visual field by category design. This was useful to determine whether lateralization varied for each type of scene content. Detection performance (‘on which side is the emotional scene?’) was indexed by response accuracy and latency, as the dependent variables.
Lateralization bias: ancillary study
As a control condition, an ancillary study was conducted to explore the possibility that there was general lateralization toward the left (or the right) regardless of emotional content. Sixteen new undergraduates from the same pool as those in the main experiment served as participants. As the lateralization effects in the main experiment were stronger for female than for male participants, all those in this ancillary study were females, to maximize potential lateralization effects in the new task. Participants were presented with the same pairings of stimuli as in the main experiment, and the same procedure, with one important exception: The task was to judge ‘on which side the neutral scene was’ (instead of the emotional one). This way, presumably, emotional processing was downplayed in the discrimination criterion (i.e., the viewer had to ‘look for’ non-emotional scenes). If, as implied by the ‘right-hemisphere advantage hypothesis’, emotional processing should be facilitated for pictures presented to the LVF, when such processing is downplayed, the predicted lateralization will disappear for neutral scenes. In contrast, if there is a general leftward processing bias, also the discrimination of neutral scenes (not only emotional ones) will be more accurate and efficient in the LVF.
Results
Effects of emotional valence, visual field, and sex of participant (main experiment)
Emotional scene discrimination accuracy and latencies of correct responses were analyzed by a scene Valence (2) × Visual Field (2) × Sex (2) ANOVA. In this analysis, the performance for the seven pleasant categories, or for the six unpleasant categories, was averaged.
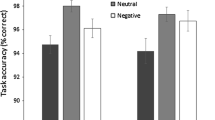
For response accuracy (i.e., the percentage of correct identification of emotional scenes), effects of valence, F(1, 46) = 4.87, p = .032, η 2 p = .10, and visual field, F(1, 46) = 8.94, p < .01, η 2 p = .16, emerged, but not of sex, (F < 1; females vs. males: 75.0 vs. 74.4 %), or the interactions (all Fs < 1). Discrimination accuracy was higher for pleasant than for unpleasant scenes (M = 76.9 vs. 72.5, respectively), and in the LVF than the RVF (M = 76.2 vs. 73.2). In Table 1, the mean scores between the LVF and the RVF are shown for valence and sex. One-sample t tests were conducted to determine whether hits exceeded the chance 0.5 level. For all the levels of valence, visual field, and sex, accuracy was above chance (all ts (23) > 7.5, p < .0001).
For response latencies, there were effects of valence, F(1, 46) = 23.95, p < .0001, η 2 p = .34, and visual field, F(1, 46) = 13.22, p < .001, η 2 p = .22. Discrimination was faster for pleasant than for unpleasant scenes (M = 694 vs. 754 ms), and in the LVF than the RVF (M = 708 vs. 740 ms), with no main effect of sex (F < 1; females = 747; males = 701). Table 1 shows the mean scores between the LVF and the RVF for valence and sex. These effects were qualified by a valence by visual field by sex interaction, F(1, 46) = 6.41, p = .016, η 2 p = .12. To decompose this interaction, visual field by sex ANOVAs was conducted separately for each valence level. For pleasant scenes, a visual field effect, F(1, 46) = 7.87, p < .01, η 2 p = .15, appeared (LVF: 677; RVF: 710), but not of sex or the interaction (Fs < 1; females: 668; males: 719). In contrast, for unpleasant scenes, in addition to a visual field effect, F(1, 46) = 7.01, p = .011, η 2 p = .13 (LVF: 739; RVF: 769), and the lack of a sex effect, (F < 1; females: 734; males: 775), the interaction was significant, F(1, 46) = 5.57, p = .023, η 2 p = .11. The comparison between the LVF and the RVF revealed significant differences for females, t(23) = 5.02, p < .0001, but not for males, t(23) = 0.17, p = .87, ns.
Analysis of specific scene categories (main experiment)
Footnote 5Visual field (2) × Sex (2) × Category (7 or 6, for pleasant or unpleasant scenes, respectively) Greenhouse-Geisser-corrected ANOVAs were conducted for each valence separately, to explore potential interactions. This would allow us to determine whether lateralization varies as a function of specific emotional scene content. Against this hypothesis, all the interactions involving category—for both response accuracy and reaction times—were statistically nonsignificant (all Fs ≤ 2.08, ps ≥ .08, ns). Importantly, this means that the previously described LVF advantage was consistent across all the scene categories.
In contrast, main effects of category emerged for accuracy, F(6, 276) = 26.11, p < .0001, η 2 p = .36, and latencies, F(6, 276) = 20.90, p < .0001, η 2 p = .31, for pleasant scenes; and F(5, 230) = 9.03, p < .0001, η 2 p = .16 (accuracy), and F(5, 230) = 8.26, p < .0001, η 2 p = .15 (latencies), for unpleasant scenes. In post hoc, Bonferroni-corrected multiple contrasts, the categories of babies, families, and erotica couples were discriminated more accurately and faster than the other pleasant categories. Also, the mutilation and the male-to-female aggression were discriminated more accurately and faster than the other unpleasant categories. In addition, and consistently with the results in the previous section, significant effects of visual field appeared for all the pleasant categories, F(1, 46) = 7.03, p = .011, η 2 p = .13 (accuracy); and F(1, 46) = 7.87, p < .01, η 2 p = .15 (latencies), and all the unpleasant categories, F(1, 46) = 6.06, p = .018, η 2 p = .12 (accuracy), and, F(1, 46) = 7.01, p = .011, η 2 p = .13 (latencies). One-sample t tests revealed that accuracy for all the categories was above the chance 0.5 level (all ts (23) > 2.10, p < .05), except for the same-sex erotica for male participants (ts < 1, ps > .36, ns, on both hemifields).
Analysis of the lateralization control study: ancillary study
Footnote 6The same analyses were conducted for data from the lateralization control experiment. No significant effects emerged in the ANOVA for accuracy or reaction times. Particularly, the effects of visual field (accuracy: LVF = 65.1 %; RVF = 65.5; RTs: LVF = 1,116 ms; RVF = 1,109; all ps > .80) and the interactions with valence (all ps > .40) were nonsignificant. One-sample t tests, nevertheless, revealed that hits exceeded the chance 0.5 level (all ps < .05) for all the scene categories and both hemifields. When data from the main experiment (‘emotionality’ task) and the control experiment (‘neutrality’ task) were compared, responses were more accurate, F(1, 38) = 9.31, p < .01, η 2 p = .20 (unpleasant scenes), F(1, 38) = 9.20, p < .01, η 2 p = .19 (pleasant scenes), and faster, F(1, 38) = 30.67, p < .0001, η 2 p = .45 (unpleasant scenes), F(1, 38) = 37.08, p < .0001, η 2 p = .49 (pleasant scenes), for the ‘emotionality’ than for the ‘neutrality’ task (accuracy: M unpleasant = 73.1 vs. 64.8 %; M pleasant = 76.9 vs. 65.6; RTs: M unpleasant = 775 vs. 1,142 ms; M pleasant = 719 vs. 1,084).
Discussion
The major results of this study were as follows. First, emotional scenes were reliably discriminated from simultaneously presented neutral scenes in peripheral vision. Second, there was facilitated discrimination when the emotional picture appeared in the left relative to the right visual field, and this occurred regardless of type of specific scene content. Third, lateralization was equivalent for females and males when processing pleasant images, but was greater for women when processing unpleasant images. These effects occurred in the absence of fixations on the pictures, and with no low-level image and visual saliency differences between emotional and neutral scenes, or among the various emotional scene categories.
Discrimination of emotional from neutral scenes in peripheral vision
Response accuracy was above chance level for both visual fields, all types of emotional scenes, and females and males. As there were no eye fixations on the otherwise briefly displayed scenes, this indicates that ‘something’ of them was perceived outside the focus of attention and allowed for an emotional vs. neutral discrimination. Given that emotional and neutral pictures were presented simultaneously and an emotionality criterion (i.e., ‘choose the emotional scene’) was used, correct responding implies that emotional content was extracted. Paradigms allowing for implicit encoding, as indexed by spontaneous overt attentional orienting (i.e., eye movements; Alpers 2008; Calvo et al. 2008; Kissler and Keil 2008; McSorley and Van Reekum 2013; Nummenmaa et al. 2006) and brain activity (De Cesarei et al. 2009; Keil et al. 2005; Rigoulot et al. 2008), have also found evidence of emotional processing in extrafoveal vision. In the current study, the use of an explicit measure adds to prior research by showing that affective significance is, in fact, encoded (see below, Affective encoding in peripheral vision).
Alternative interpretations can, nevertheless, be considered. First, it may be argued that, rather than using an affective or semantic criterion to select the emotional scene of the pair, participants could have simply relied on some physical cues distinguishing emotional from neutral images. If so, the neutral–emotional discrimination would be due to purely perceptual properties devoid of affective meaning. This hypothesis can, however, be ruled out on the grounds that emotional and neutral scenes did not differ in any of a wide range low-level image properties and visual saliency. Second, emotional scenes might have been more ‘perceptible’ on their own than neutral scenes. A way to address this issue has involved presenting the scenes alone rather than paired. Many of the same images in the current study were displayed by Calvo and Lang (2005) alone in parafoveal vision to assess perceptibility. Prime scenes were displayed for 150 or 450 ms, followed by a 500-ms mask, and a probe scene. Recognition of the people and actions depicted in the images was equivalent (hits, A’ sensitivity, and reaction times of correct responses) for pleasant, unpleasant, and neutral scenes. Similarly, Gutiérrez-García et al. (2009) presented the pictures alone in peripheral or foveal vision for 150 ms, followed by a 500-ms mask, and a recognition probe. A’ sensitivity and correct response RTs were also equivalent for the three types of pictures. Accordingly, the basic perceptibility of critical elements (people and actions) in the scenes was comparable, yet there was coarse affective processing.
Lateralization: left-visual field advantage modulated by sex and affective valence
Discrimination of emotional scenes was performed more accurately and faster in the left-visual field (with neutral scenes in the opposite side) than in the right. This result can be interpreted within the context of brain hemisphere specializations, supporting the ‘right-hemisphere hypothesis’ (Heller et al. 1998; see Gianotti 2012). Such a right-hemisphere dominance for emotional processing of extrafoveally presented scenes has received support from different paradigms (Alpers 2008; Calvo and Avero 2009; Calvo and Nummenmaa 2007; Keil et al. 2005). In contrast, the fact that the LVF advantage generally occurred for both pleasant and unpleasant scenes is not consistent with the ‘valence–specificity hypothesis’, according to which there should be left-hemisphere dominance for positive affect and right-hemisphere dominance for negative affect (see Gianotti 2012). Also, the fact that the LVF advantage occurred for all types of emotional scene contents (e.g., human attack, romance, etc.) is not consistent with the ‘emotional-type hypothesis’, according to which there should be a different modulation of the right and the left hemisphere depending on specific contents (see Gianotti 2012).
The observed LVF lateralization for emotional scenes does not merely reflect a general ‘left-to-right’ bias in processing all types of visual stimuli. Such a bias occurred when the driving discrimination criterion involved identification of emotionality, but not when processing priority involved identification neutral scenes.Footnote 7 This suggests that the LVF lateralization is specific to emotional encoding. This view is consistent with data from a study by Calvo and Nummenmaa (2007) in which affective and semantic processing were compared for the same scene stimuli. Pleasant or unpleasant scenes portraying people or animals were presented as primes (parafoveally) and probes (foveally), and participants made either an affective evaluation (pleasant or unpleasant) or a semantic categorization (people or animal) of the probe. Results revealed both affective and semantic priming, although they were affected by visual field in a different fashion. There was semantic priming only when primes were presented in the RVF, whereas affective priming more generally appeared in the LVF. These findings further support the hypothesis of right-hemisphere specialization for valence encoding of emotional pictures.
The current data are also relevant to a ‘sex–valence–specificity hypothesis’, with lateralization varying for females and males, although this takes place in rather complex ways (see Duerden et al. 2013; Kret and de Gelder 2012; Stevens and Hamann 2012; Whittle et al. 2011). Our valence by sex interaction can be related to this hypothesis: While the LVF advantage was comparable for females and males when viewing pleasant scenes, lateralization disappeared for males, but not for females, when viewing unpleasant scenes. It must, nevertheless, be noted that this happened for—presumably more discriminative—reaction times, albeit not for response accuracy. Thus, the right-hemisphere dominance is to some extent modulated by valence and sex. Consistently, in some prior studies, women but not men showed laterality effects depending on emotional valence (Rodway et al. 2003; van Strien and van Beek 2000). We can add further information to account for the lateralization effect in females—but not in males—for unpleasant scenes: A reanalysis of the affective valence data in our norming study revealed that both females and males rated pleasant scenes as similarly positive, whereas males rated unpleasant scenes as less negative than females did (see also Bradley et al. 2001b; Calvo and Avero 2009; Moltó et al. 2013).Footnote 8 This result, in fact, matches with the observed lateralization patterns: Equivalent positive valence is associated with equivalent lateralization for females and males; less negative valence is associated with reduced or no lateralization for males. This suggests that the strength of lateralization depends on how much pleasant or unpleasant an emotional scene is judged to be.
Affective encoding in peripheral vision
What is perceived of emotional scenes in peripheral vision that allows for discrimination from neutral scenes and leads to lateralized dominance? We have suggested that affective valence is processed. The fact, just described, that lateralization differences between males and females are related to the amount of perceived pleasantness or unpleasantness is consistent with this view. Additional support comes from studies showing emotional scene gist encoding, i.e., a coarse impression about whether something good or bad is depicted in the scene, in the absence of recognition of object details in peripheral vision (Calvo 2006; Calvo et al. 2008). Such a coarse affective gist would lead to confusions between scenes that share the same affective valence and to delayed correct rejection times (i.e., stimulus identification) (Calvo et al. 2008). At the same time, the emotional gist would be sufficient to attract overt attention, as shown by selective eye-movement orienting (McSorley and van Reekum 2013; Nummenmaa et al. 2006).
A demonstration that affect is extracted through lateralized mechanisms has been provided by affective priming paradigms in which a prime scene is presented extrafoveally, followed by a probe (e.g., another scene) in central vision. The prime and the probe are physically different but congruent or incongruent in affect. The viewer ignores the prime and judges the probe as pleasant or unpleasant. If the prime activates affective information, probe reaction times will be faster when the prime and the probe are congruent vs. incongruent. Shorter latencies have been found for affectively congruent probes following parafoveal (2.5°), foveally masked primes (Calvo and Avero 2008; Calvo and Nummenmaa 2007). This reveals that emotional information was extracted from the prime scenes. Importantly, although not in truly peripheral vision (≥5°), these effects occurred when the prime scenes appeared in the LVF, but not in the RVF. This lends support to the affective encoding interpretation that we have assigned to the lateralized effects in the current study.
Conclusions
The neuro-cognitive system can discriminate between emotional and neutral scenes in the visual periphery, without overt attention. The detection advantage when emotional scenes appear in the left hemifield is consistent with a brain right-hemisphere dominance hypothesis. Nevertheless, lateralization is modulated by valence and sex, with enhanced effects for females and unpleasant scenes. Lateralization generalizes to a variety of specific emotional scene contents and is related to greater-rated scene valence. This suggests that affective significance per se is indeed extracted from the scenes in peripheral vision. Alternative explanations based on the contribution of perceptual (low-level image properties, visual saliency, and perceptibility) rather than affective factors can be ruled out.
Notes
Apart from emotional visual scenes, facial expressions of emotion have been investigated in extrafoveal vision, with behavioral (Bayle et al. 2011; Calvo et al. 2014b; Goren and Wilson 2006) and neurophysiological (Bayle et al. 2009; Calvo et al. 2014a; Rigoulot et al. 2012) measures. In a meta-analysis of 157 fMRI (functional magnetic resonance imaging) studies, Sabatinelli et al. (2011) found that, although there is extensive overlap of brain regions activated by expressive faces and natural scenes, each type of stimulus also recruits specific regions and neural networks. In addition, emotional scenes have been found to produce stronger subjective and ERP responses than facial expressions (Thom et al. 2014). Accordingly, we will focus on visual scenes involving people, rather than single face stimuli.
Bernat et al. (2006) have underscored the importance of classifying emotional pictures of different types into meaningful content categories, to determine the content specificity of emotional processing.
See the mean scores for each category and sex group in a supplementary data file.
See the mean scores for each category in a supplementary data file.
See the mean scores for each category, visual field, and sex group in a supplementary data file.
See the mean scores for each category and visual field in a supplementary data file.
The fact that “neutrality” discrimination was more difficult (less accurate and longer responses, yet above chance level) than “emotionality” discrimination is probably due to the “neutral” scenes being perceived as emotionally ambiguous, at least at initial processing stages (e.g., 150-ms display).
A 2 (Valence: pleasant vs. unpleasant scenes) × 2 (Sex of participant: female vs. male) ANOVA on valence ratings in the norming study yielded a significant interaction, F(1, 140) = 11.90, p < .001, η 2 p = .08. Females rated unpleasant scenes as more unpleasant than males did (M = 1.55 vs. 1.88, respectively, in a 1- to 9-point scale), t(71) = 9.90, p < .0001, while there were no sex differences for the pleasant scenes (M = 7.15 vs. 6.88), t(71) = 1.59, p = .12, ns.
References
Alpers GW (2008) Eye-catching: right hemisphere attentional bias for emotional pictures. Laterality 13:158–178
Bayle D, Henaff MA, Krolak-Salmon P (2009) Unconsciously perceived fear in peripheral vision alerts the limbic system: a MEG study. PLoS One 4(12):e8207
Bayle D, Schoendorff B, Henaff MA, Krolak-Salmon P (2011) Emotional facial expression detection in the peripheral visual field. PLoS One 6(6):e21584
Bernat E, Patrick CJ, Benning SD, Tellegen A (2006) Effects of picture content and intensity on affective physiological response. Psychophysiology 43:93–103
Borji A, Itti L (2013) State-of-the-art in visual attention modeling. IEEE Trans Pattern Anal Mach Intell 35:185–207
Bradley MM, Lang PJ (2007) The International Affective Picture System (IAPS) in the study of emotion and attention. In: Coan JA, Allen JJB (eds) Handbook of emotion elicitation and assessment. Oxford University Press, New York, pp 29–46
Bradley MM, Codispoti M, Cuthbert BN, Lang PJ (2001a) Emotion and motivation I: defensive and appetitive reactions in picture processing. Emotion 1:276–298
Bradley MM, Codispoti M, Sabatinelli D, Lang PJ (2001b) Emotion and motivation II: sex differences in picture processing. Emotion 1:300–319
Bradley MM, Keil A, Lang PJ (2012) Orienting and emotional perception: facilitation, attenuation, and interference. Front Psychol 3:493
Briggs K, Martin F (2009) Affective picture processing and motivational relevance: arousal and valence effects on ERPs in an oddball task. Int J Psychophysiol 72:299–306
Calvo MG (2006) Processing of emotional visual scenes outside the focus of spatial attention: the role of eccentricity. Vis Cogn 13:666–676
Calvo MG, Avero P (2008) Affective priming of emotional pictures in parafoveal vision: left visual field advantage. Cogn Affect Behav Neurosci 8:41–53
Calvo MG, Avero P (2009) Reaction time normative data for the IAPS as a function of display time, gender, and picture content. Behav Res Methods 41:184–191
Calvo MG, Lang PJ (2004) Gaze patterns when looking at emotional pictures: motivationally biased attention. Motiv Emot 28:221–243
Calvo MG, Lang PJ (2005) Parafoveal semantic processing of emotional scenes. J Exp Psychol Hum Percept Perform 31:502–519
Calvo MG, Nummenmaa L (2007) Processing of unattended emotional visual scenes. J Exp Psychol Gen 136:347–369
Calvo MG, Nummenmaa L, Hyönä J (2007) Emotional and neutral scenes in competition: orienting, efficiency, and identification. Q J Exp Psychol 60:1585–1593
Calvo MG, Nummenmaa L, Hyönä J (2008) Emotional scenes in peripheral vision: selective orienting and gist processing, but not content identification. Emotion 8:68–80
Calvo MG, Beltrán D, Fernández-Martín A (2014a) Early processing of happy facial expressions in peripheral vision: neurophysiological evidence. Biol Psychol 100:60–70
Calvo MG, Fernández-Martín A, Nummenmaa L (2014b) Recognition of facial expressions in peripheral vs. central vision: role of the eyes and the mouth. Psychol Res 78:180–195
Colden A, Bruder M, Manstead ASR (2008) Human content in affect-inducing stimuli: a secondary analysis of the international affective picture system. Motiv Emot 32:260–269
De Cesarei A, Codispoti M, Schupp HT (2009) Peripheral vision and preferential emotion processing. NeuroReport 20:1439–1443
Domínguez-Borrás J, Vuilleumier P (2013) Affective biases in attention and perception. In: Armony J, Vuilleumier P (eds) The Cambridge handbook of human affective neuroscience. Cambridge University Press, New York, pp 331–356
Duerden EG, Arsalidou M, Lee M, Taylor MJ (2013) Lateralization of affective processing in the insula. NeuroImage 78:159–175
Gianotti G (2012) Unconscious processing of emotions and the right hemisphere. Neuropsychologia 50:205–218
Gomez P, von Gunten A, Danuser B (2013) Content-specific gender differences in emotion ratings from early to late adulthood. Scand J Psychol 54:451–458
Goren D, Wilson HR (2006) Quantifying facial expression recognition across viewing conditions. Vis Res 46:1253–1262
Groen Y, Wijers AA, Tucha O, Althaus M (2013) Are there sex differences in ERPs related to processing empathy-evoking pictures? Neuropsychologia 51:142–155
Gutiérrez-García A, Nummenmaa L, Calvo MG (2009) Enhanced processing of emotional gist in peripheral vision. Span J Psychol 12:414–423
Heller W, Nitschke JN, Miller GA (1998) Lateralization in emotion and emotional disorders. Curr Dir Psychol Sci 7:26–32
Itti L (2006) Quantitative modeling of perceptual salience at human eye position. Vis Cogn 14:959–984
Itti L, Koch C (2000) A saliency-based search mechanism for overt and covert shifts of visual attention. Vis Res 40:1489–1506
Keil A, Moratti S, Sabatinelli D, Bradley MM, Lang PJ (2005) Additive effects of emotional content and spatial selective attention on electrocortical facilitation. Cereb Cortex 15:1187–1197
Kissler J, Keil A (2008) Look—don’t look! How emotional pictures affect pro- and antisaccades. Exp Brain Res 188:215–222
Kret ME, de Gelder B (2012) A review on sex differences in processing emotional signals. Neuropsychologia 50:1212–1221
Lang PJ, Bradley MM, Cuthbert BN (2008) International affective picture system (IAPS): affective ratings of pictures and instruction manual. Technical report A-8. University of Florida, Gainesville, FL, USA
McSorley E, van Reekum CM (2013) The time course of implicit affective picture processing: an eye movement study. Emotion 13:769–773
Mohanty A, Sussman TJ (2013) Top-down modulation of attention by emotion. Front Hum Neurosci 7:102
Moltó J, Segarra P, López R, Esteller A, Fonfría A, Pastor MC, Poy R (2013) Adaptación española del International affective picture system (IAPS). Tercera parte [Spanish adaptation of the International Affective Picture System. Third part]. Anales de Psicología 29:965–984
Nummenmaa L, Hyönä J, Calvo MG (2006) Eye movement assessment of selective attentional capture by emotional pictures. Emotion 6:257–268
Nummenmaa L, Hyönä J, Calvo MG (2009) Emotional scene content drives the saccade generation system reflexively. J Exp Psychol Hum Percept Perform 35:305–323
Pessoa L, Oliveira L, Pereira M (2013) Top-down attention to the processing of emotional stimuli. In: Armony J, Vuilleumier P (eds) The Cambridge handbook of human affective neuroscience. Cambridge University Press, New York, pp 357–374
Peyk P, Schupp HT, Keil A, Elbert T, Junghöfer M (2009) Parallel processing of affective visual stimuli. Psychophysiology 46:200–208
Rigoulot S, Delplanque S, Despretz P, Defoort-Dhellemmes S, Honore J, Sequeira H (2008) Peripherally presented emotional scenes: a spatiotemporal analysis of early ERP responses. Brain Topogr 20:216–223
Rigoulot S, D’Hondt F, Honoré J, Sequeira H (2012) Implicit emotional processing in peripheral vision: behavioral and neural evidence. Neuropsychologia 50:2887–2896
Rodway P, Wright L, Hardie S (2003) The valence-specific laterality effect in free-viewing conditions: the influence of sex, handedness, and response bias. Brain Cogn 53:452–463
Sabatinelli D, Fortune EE, Li O, Siddiqui A, Krafft C, Oliver WT, Beck S, Jeffries J (2011) Emotional perception: meta-analyses of face and natural scene processing. Neuroimage 54:2524–2533
Schupp H, Cuthbert B, Bradley M, Hillman C, Hamm A, Lang P (2004) Brain processes in emotional perception: motivated attention. Cogn Emot 18:593–611
Stevens JS, Hamann S (2012) Sex differences in brain activation to emotional stimuli: a meta-analysis of neuroimaging studies. Neuropsychologia 50:1578–1593
Thom N, Knight J, Dishman R, Sabatinelli D, Johnson DC, Clementz B (2014) Emotional scenes elicit more pronounced self-reported emotional experience and greater EPN and LPP modulation when compared to emotional faces. Cogn Affect Behav Neurosci 14:849–860
van Strien JW, van Beek S (2000) Ratings of emotion in laterally presented faces: sex and handedness effects. Brain Cogn 44:645–652
Weinberg A, Hajcak G (2010) Beyond good and evil: the time-course of neural activity elicited by specific picture content. Emotion 10:767–782
Whittle S, Yücel M, Yap MB, Allen NB (2011) Sex differences in the neural correlates of emotion: evidence from neuroimaging. Biol Psychol 87:319–333
Acknowledgments
This research was supported by Grant PSI2014-54720-P from the Spanish Ministerio de Economía y Competitividad, and the Agencia Canaria de Investigación, Innovación, y Sociedad de la Información (Neurocog Project).
Ethical standard
The study was conducted in accordance with the WMA Declaration of Helsinki 2008, and approved by the Local Ethics Committee of the University of La Laguna.
Conflict of interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
221_2014_4174_MOESM1_ESM.xlsx
Supplementary material 1: A supplementary data file contains the mean scores and standard deviations for each scene category regarding: (1) norming study valence ratings and reaction times (sheet 1); (2) low-level image properties (sheet 2); (3) visual saliency values (sheet 3); (4) discrimination performance accuracy and latencies in the main experiment (sheet 4); and (5) discrimination performance accuracy and latencies in the ancillary study (sheet 5). (XLSX 25 kb)
Appendix
Appendix
IAPS stimuli used in the current experiment.
Neutral 2025, 2026, 2036, 2037, 2038, 2039, 2102, 2104, 2107, 2190, 2191, 2221, 2235, 2272, 2273, 2305, 2308, 2342, 2357, 2372, 2374, 2377, 2382, 2383, 2384, 2389, 2390, 2393, 2394, 2396, 2397, 2400, 2411, 2480, 2484, 2488, 2489, 2491, 2512, 2513, 2515, 2560, 2575, 2579, 2593, 2594, 2595, 2597, 2635, 2745, 2749, 2840, 2850, 2870, 5410, 5455, 5875, 7493, 7496, 7497, 7506, 7550, 7620, 7632, 9210.
Pleasant Sport/adventure (8021, 8032, 8040, 8161, 8200, 8205, 8370, 8420, 8490, 8499); Families (2151, 2152, 2155, 2160, 2165, 2340, 2360); Babies (2040, 2057, 2058, 2070, 2260, 2655, 2660); Romance (4597, 4598, 4599, 4612, 4619, 4624, 4626, 4628, 4641, 4700); Erotica men (4470, 4490, 4505, 4520, 4525, 4531, 4534, 4542, 4561, 4572); Erotica women (4002, 4003, 4008, 4071, 4085, 4130, 4141, 4150, 4232, 4235); Erotica couples (4604, 4611, 4647, 4658, 4659, 4668, 4669, 4680, 4687, 4690).
Unpleasant Accidents (3216, 6838, 8480, 8485, 9050, 9230, 9435, 9900, 9903, 9921, 9925, 9926); Attack without weapon (2691, 2694, 6821, 6825, 6836, 6840, 8065, 8232, 9402, 9413, 9414, 9428); Attack with weapon (2683, 3500, 3530, 6212, 6231, 6242, 6510, 6520, 6571, 6832, 9414, 9425); Male-to-female aggression (6312, 6313, 6315, 6530, 6560, 6561); Grief/illness (2141, 2205, 2700, 2703, 2710, 2799, 2900, 3300, 3350, 9040, 9332, 9421); Mutilation (3001, 3030, 3064, 3180, 3181, 3225, 6022, 9250, 9253, 9410, 9433, 9490).
Rights and permissions
About this article
Cite this article
Calvo, M.G., Rodríguez-Chinea, S. & Fernández-Martín, A. Lateralized discrimination of emotional scenes in peripheral vision. Exp Brain Res 233, 997–1006 (2015). https://doi.org/10.1007/s00221-014-4174-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-014-4174-8