Abstract
The nervous system can regulate the mechanical properties of the human ankle through feed-forward mechanisms such as co-contraction and rapid feedback mechanisms such as stretch reflexes. Though each of these strategies may contribute to joint stability, it is unclear how their relative contribution varies when ankle stability is threatened. We addressed this question by characterizing co-contraction and stretch reflexes during balance of an inverted pendulum simulated by a rotary motor configured as an admittance servo. The stability of this haptic environment was manipulated by varying the stiffness of a virtual spring supporting the pendulum. We hypothesized that co-contraction and stretch reflex amplitude would increase as the stability of the haptic load attached to the ankle was reduced. Electromyographic activity in soleus, medial and lateral gastrocnemius, and tibialis anterior was used to characterize co-contraction patterns and stretch reflex amplitude as subjects stabilized the haptic load. Our results revealed that co-contraction was heightened as stability was reduced, but that the resulting joint stiffness was not sufficient to fully counteract the imposed instability. Reflex amplitude, in comparison, was attenuated as load stability was reduced, contrary to results from upper limb studies using similar paradigms. Together these findings suggest that the nervous system utilizes feed-forward co-contraction rather than rapid involuntary feedback to increase ankle stability during simple balance tasks. Furthermore, since the stiffness generated through co-contraction was not sufficient to fully balance the haptic load, our results suggest an important role for slower, volitional feedback in the control of ankle stability during balancing tasks.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Maintaining a stable joint configuration requires the neuromuscular system to generate restoring forces through a combination of feed-forward and feedback mechanisms. Feed-forward commands can be executed in an open-loop manner in the absence of sensory input from the periphery. Co-contraction of antagonists represents the simplest example of a feed-forward strategy. Feed-forward strategies may also involve ballistic movements that are generated based on an estimate of the inverse dynamics of the body and environment (Hanneton et al. 1997). This type of predictive control is likely to be useful when disturbances are predictable. However, this strategy is unlikely to be relevant in the context of unexpected perturbations of posture where movement is not preplanned. Feedback commands involve a broader class of short and long latency responses driven by sensory information from numerous sources. The earliest of these responses is the short latency stretch reflex (Gottlieb and Agarwal 1979; Sinkjaer et al. 1988), followed sequentially by the medium latency or M2 response (Toft et al. 1991), triggered reactions (Gottlieb and Agarwal 1980), and volitional corrections. These include those often referred to as predictive control due to their anticipatory nature with respect to how the current state of the body will lead to future motions (Loram et al. 2005; Gawthrop et al. 2009). Although feed-forward and feedback strategies may each augment joint stability, their relative contributions and how these contributions vary when stability is compromised remain unknown.
In situations when the body is likely to experience unexpected perturbations, co-contraction represents an effective feed-forward strategy to counteract these disturbances. Co-contraction alters the impedance of the musculoskeletal system and provides instantaneous resistance to unexpected perturbations (Hirokawa et al. 1991; Carter et al. 1993; Milner et al. 1995). If the increase in joint stiffness resulting from co-contraction is of sufficient magnitude, co-contraction could be a strategy for completely counteracting destabilizing forces such as gravity. However, constant activation of antagonists can result in a high energetic cost, making this strategy sub-optimal. Additionally, using co-contraction to stabilize joints such as the ankle may not always be feasible due to limitations imposed by the limited stiffness of compliant tendons (de Zee and Voigt 2001; Loram and Lakie 2002). Though co-contraction is clearly useful for increasing joint impedance, it is likely to be insufficient for tasks when stability is highly compromised and must therefore be used in concert with feedback strategies.
Feedback commands are likely to be more energetically efficient than co-contraction and are associated with various advantages and limitations. The most rapid form of feedback-mediated control, the stretch reflex, has been shown to make significant contributions to joint impedance (Sinkjaer et al. 1988; Kearney et al. 1997). For example, the reflex contribution to ankle stiffness has been estimated to range from 28 to 85 Nm/rad for a single leg (Sinkjaer et al. 1988). These contributions are likely to be most relevant for rapid disturbances, as the muscle spindles contributing to the stretch reflex are largely sensitive to velocity (Gottlieb and Agarwal 1979). Longer latency reflexes compose the next, most rapid line of defense and may provide a more task-appropriate response resulting from integration with transcortical networks (Christensen et al. 2000). These responses, however, are not consistently generated in all muscles of the ankle (Toft et al. 1989, 1991). As the transmission delays associated with the choice of feedback increase, the likelihood of reducing stability may also increase if feedback gains are large. Therefore, it may be advantageous for the nervous system to utilize the most rapid form of feedback in concert with co-contraction in cases when joint stability is threatened.
There is substantial evidence from upper limb studies that stretch reflexes and co-contraction are both heightened when joint stability is compromised. For instance, stretch reflexes exhibit increased sensitivity during interactions with compliant loads (Doemges and Rack 1992a, b; Dietz et al. 1994; Perreault et al. 2008). These results suggest that reflexes are useful for increasing stability when sufficient postural stability is not provided by the environment. If joint stability is further compromised by having individuals interact with unstable environments, heightened co-contraction is commonly observed and may be accompanied by a further increase in stretch reflex sensitivity (De Serres and Milner 1991). This increased sensitivity can occur even without accompanying changes in muscle co-contraction (Krutky et al. 2010). Together these studies demonstrate that contributions of feedback and feed-forward mechanisms may be increased when limb stability is threatened. However, the utility of the stretch reflex at the ankle relative to that observed in the arm may be limited due to increased transmission delays, which can be approximately twice that observed for proximal arm muscles at the shoulder and elbow (Kearney et al. 1997; Krutky et al. 2010), slower muscle activation time constants (Buchthal and Schmalbruch 1970) and the high compliance of the Achilles tendon. Furthermore, the fact that the lower limbs are commonly used to stabilize the entire body, a large mass, long time-constant system, may reduce the need for feedback adaptation at the fastest time scales. Thus, it remains to be seen if lower limb stretch reflexes are modulated with load stability, as has been observed in the upper limb.
The purpose of this study was to examine how the nervous system balances the contributions from co-contraction and stretch reflexes to regulate stability in the lower limb. Though slower feedback responses mediated through supraspinal pathways are clearly involved in maintaining joint stability, it remains to be seen how the most rapid feedback responses are modulated when stability is compromised. We chose the ankle joint as a model system in the lower limb because of its relevance to common tasks that require postural stability such as stance and walking. In these experiments, ankle stability was challenged by instructing subjects to balance a haptic load designed to simulate the dynamics of an inverted pendulum. Based on the findings from upper limb studies (Doemges and Rack 1992a, b; Dietz et al. 1994; Perreault et al. 2008; Krutky et al. 2010), we hypothesized that both co-contraction magnitude and stretch reflex amplitude would increase as the stability of the haptic load was reduced and that these increases would lead to a corresponding increase in ankle stiffness. Identifying the interactions between volitional and reflex-mediated control during interaction with simulated loads may provide a basis for later studies investigating control of joint stability during more natural tasks like standing.
Materials and methods
Ethical approval
All experimental procedures were approved by the Institutional Review Board of Northwestern University (IRB protocol #0673-010) and complied with the principles of the Declaration of Helsinki. Written informed consent was obtained prior to testing.
Subjects
Fifteen subjects (8 men and 7 women, ages 25–33) who had no prior history of injury to the ankle participated in this study. Ten subjects participated in the primary protocol. Three of these subjects and five additional subjects were recruited for the control experiment.
Equipment
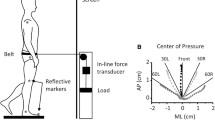
A computer-controlled brushless servomotor (BSM90N-3150AX; Baldor Electric Company, Fort Smith, AR) was used to apply angular rotations to the ankle joint. Angular feedback was provided by an encoder with an effective resolution of 6.3 × 10−5 rad. Forces and moments were measured using a six degree-of-freedom load cell (630N80; JR3, Inc, Woodland, CA), and the motor was controlled in real-time using Matlab xPC®. A number of safety mechanisms were implemented in the system including: software-based amplitude limits of ±0.26 rad, inductive proximity sensors (TL-W5E; Omron Industrial Automation, Schaumburg, IL), an emergency stop button to cut power to the motor, and hard stops at the end of the movement range. Subjects were seated in a Biodex chair, and movement of the trunk was minimized using a set of straps placed across the torso (Fig. 1a). Each subject’s right knee was flexed 0.52 rad (30°), and the foot was secured to an aluminum footplate using straps placed across the forefoot and heel. The ankle’s center of rotation was then aligned with the axis of the motor and locked into position at 0.17 rad (10°) of plantar flexion. This configuration was selected to be consistent with previous investigations of reflex function in the ankle (Gottlieb and Agarwal 1979; Kimura et al. 2003).
Experimental setup for study of ankle control. a Subjects were seated with their foot attached to a servomotor via a rigid foot plate. Individuals used visual feedback of the foot plate angle to maintain the target ankle position. b Subjects controlled an unstable haptic load which behaved like an inverted pendulum supported by a torsional spring of stiffness K env. The simulated pendulum had a mass equal to 25% of the subject’s mass and a height of 1 m
The purpose of this study was to quantify how volitional muscle activity and stretch reflex amplitude are regulated during interactions with stiff, compliant, and unstable environments. In the stiff (S) environment, the motor was controlled as a rigid position servo with a stiffness of 35 kNm/rad, and subjects generated an isometric torque guided by visual feedback. This task served as a baseline against which subjects’ performance in the remaining position control tasks could be compared. In the remaining compliant (C) and unstable environments (U1 and U2), subjects controlled the position of a second-order load guided by visual feedback of ankle position. For each of these remaining environments, an admittance control algorithm was used to simulate a haptic environment which included an inertial element, an elastic element, and gravity. The inertial element was designed to simulate the dynamics of a pendulum rotating about the primary axis of the ankle (Fig. 1b). For all subjects, this pendulum had a mass (m) equal to 25% of the subject’s mass and a height (l) of 1 m. The mass and height were selected so that subjects could control its position without fatigue. The simulated environment also included an elastic element of stiffness K env in parallel with the pendulum. In addition to these elements, a bias torque (τbias) equal to 5% of the subject’s maximum voluntary contraction was applied in dorsiflexion. Overall, the relationship between the torque generated by the ankle (τankle) and the ankle’s position was determined from the following equation,
where θ and θtarget represent the actual and desired ankle angle and K load is equal to −mgl. If this relationship is linearized about θtarget, the relationship between the motor torque and ankle position can be represented as the sum of the load stiffness, K load, and the stiffness of the environment, K env. Our simulated mechanical system was stable when K env ≥ |K load|. This stability implied that the system would return to its equilibrium point when perturbed. The overall stability of the system was manipulated by varying the value of K env relative to |K load|. For the compliant environment, K env was equal to 1.25*|K load|; K env was set to 0.75*|K load| and 0.5*|K load| for the unstable 1 (U1) and unstable 2 (U2) conditions, respectively. It should be noted that subjects would need to produce a torque that is of equal magnitude and opposite sign of the bias torque when each load is at its equilibrium point.
Electromyography
Bipolar, surface electrodes (model #272; Noraxon USA, Scottsdale, AZ) were used to record EMG activity in soleus (Sol), medial gastrocnemius (MG), lateral gastrocnemius (LG), and tibialis anterior (TA). Standard skin preparation techniques were used before applying each electrode to the skin. The resulting signals were amplified using a Bortec® AMT-16 system (Bortec Biomedical, Calgary, AB, Canada), which has a bandwidth of 10–1,000 Hz, an input impedance of 10 GΩ, and a common-mode rejection ratio of 115 dB at 60 Hz. Analog signals were anti-alias filtered using custom-built, differential input, 5th-order Bessel filters with a cutoff frequency of 500 Hz and then sampled at 2,500 Hz with a 16-bit data acquisition system (PCI-DAS1602/16; Measurement Computing Corporation, Norton, MA).
Protocol
Maximum voluntary contractions (MVCs) were recorded at the beginning of each experiment, and these values were used to normalize EMG activity across subjects. MVCs in plantar flexion and dorsiflexion were performed against the motor while the ankle angle was set at 0.17 rad (10°) of plantar flexion. Each MVC trial was conducted for 5 s, and two trials were performed in each direction. The MVC for each muscle was computed by taking the maximum average rectified EMG amplitude calculated over a 50-ms moving window, and the MVC torque was taken to be the peak torque measured across both trials for each direction.
Subjects then performed either a torque or position-matching task in each of the four environments imposed by the servomotor. The torque-matching task was always performed in the stiff environment with the ankle fixed at 0.17 rad (10°) of plantar flexion. Here, subjects were instructed to maintain a torque of 5 ± 2% of MVC using visual feedback of torque amplitude. Torque was displayed as the height of a vertical bar on an LCD display. The range of the feedback corresponded to 0–10% of MVC. The desired torque was represented by a horizontal bar spanning from 3 to 7% of MVC, which was found to be consistent with the torque variability observed during the position-matching tasks described below. Subjects’ control strategy during this torque-matching task served as the reference for the remaining position-matching tasks because previous studies have demonstrated large differences in reflex amplitude between force- and position-matching tasks (Doemges and Rack 1992a, b; Dietz et al. 1994; Perreault et al. 2008). The position-matching task was performed in the C, U1, and U2 environments. During this task, subjects were provided with a visual display of ankle position and instructed to maintain 0.17 ± 0.017 rad of plantar flexion, which was found to be consistently achievable for all tested environments. Ankle angle was displayed on an LCD monitor, scaled to represent a range from 0.087 to 0.26 rad of plantar flexion. The desired position target was placed at the middle of this range. Because of the bias torque applied during the position-matching tasks, subjects always exerted a plantar flexion torque equal to 5% of MVC when the cursor was at the center of the target. This resulted in a consistent ankle position and torque across the position- and torque-matching tasks. The order of each environment was randomized for all subjects.
During each task, ramp and hold perturbations were used to elicit stretch reflexes in the plantar flexors. A single trial consisted of a period where subjects maintained ankle torque or position followed by a randomly timed perturbation. The perturbation had a velocity and amplitude of 3.5 rad/s (200°/s) and 0.17 rad (10°), respectively. Subjects required 2–10 s to achieve the desired torque or position across all environments with trials in the U2 environment requiring approximately twice as much time as trials in the S and C environments. The total perturbation duration was only 300 ms, a small fraction of the total trial duration. A total of 20 trials were performed in each environment and at the conclusion of each trial, subjects were free to begin the next trial in their own time. These perturbation parameters were selected to be within the range of reported parameters used in prior studies of stretch reflexes at the ankle (Gottlieb and Agarwal 1979; Grey et al. 2001; Kimura et al. 2003). For the compliant and unstable environments, the motor was rapidly switched to a stiff position servo prior to perturbation onset as has been described previously (Shemmell et al. 2009). To minimize the probability of systematic muscle activation in preparation for the perturbation, the interval between perturbations was uniformly varied from 1 to 3 s. Subjects were instructed to not react to the perturbation, as it has been demonstrated that stretch reflexes are sensitive to the instructions provided to the subjects (Hammond 1956; Crago et al. 1976).
Control experiment
A control experiment was performed to quantify the influence of agonist–antagonist co-contraction on the amplitude of stretch reflexes elicited in the plantar flexors. Co-contraction is a means by which unstable loads can be controlled, and co-contraction was observed in our experiments. Because stretch reflexes are sensitive to changes in co-contraction (Nielsen et al. 1994), we tested whether the reflexes we observed during interactions with unstable loads could simply result from co-contraction. In this experiment, subjects initially performed the position-matching task in the U2 environment and we computed the mean activity in the plantar flexors and TA during the 100-ms prior to perturbation onset. In the subsequent task, subjects matched the mean background activity measured in the U2 environment while the ankle position was fixed (Cocon task). As it was not feasible to match EMG activity across all muscles, subjects were only provided with feedback of Sol and TA. Sol was selected because it was the primary plantar flexor used in each environment and also because the stretch reflexes in Sol were larger than those in the other plantar flexors. Once subjects maintained the desired level of muscle activity, 20 perturbations (velocity: 3.5 rad/s, amplitude: 0.17 rad) were applied to assess the influence of co-contraction on reflex amplitude. These reflexes were compared to those measured in the U2 environment and to those measured in the stiff environment when there was minimal co-contraction.
Data analysis
EMGs were processed by first removing the mean value, then rectifying and normalizing to the EMGs recorded during MVCs. For each trial in the stiff environment, the mean torque during the 100 ms prior to perturbation onset was computed. If the error between this torque and the target torque was more than 2% of MVC, the trial was then discarded. Similarly, for the remaining environments, the mean position during the 100 ms prior to the perturbation was computed. If the average position during this period was greater than 0.017 rad (1°) from the target angle, the trial was discarded. Of the 200 trials recorded for each environment, these criteria resulted in the removal of 7 (4%), 22 (11%), and 36 (18%) trials for the C, U1, and U2 environments, respectively; no trials were removed from the stiff environment protocols. Average responses for each environment were computed using all trials that were not excluded.
Feed-forward (co-contraction) and feedback (stretch reflex) control strategies were assessed using the average rectified EMG from each task. The feed-forward component in each muscle was estimated as the average rectified EMG during the 100 ms prior to perturbation onset, referred to as the background EMG. Onset of the stretch reflex was defined as the initial time following perturbation onset when the subsequent EMG exceeded three standard deviations above the average background activity for at least 5 ms. Reflex amplitude was quantified as the mean amplitude of the rectified EMG during the 20 ms following the onset of the response. We tested for potential learning and fatigue effects by comparing the average background EMG and stretch reflex amplitude during the first five trials and last five trials in each environment.
Changes in the mechanical properties of the ankle were quantified using estimates of joint stiffness. Quasi-static estimates of ankle stiffness were estimated by computing the ratio of the mean change in torque (Δτ) to the displacement (Δθ) during the hold portion of the perturbation (Fig. 2). Displacement and torque data were averaged within 100–200 ms following perturbation onset. This time period was selected to center our estimates over the period of peak reflex torque. During this period, the contributions from inertial or viscous transients would be minimal. Any observed changes in stiffness across environments would reflect the combined contributions from feed-forward and reflexive muscle activity. No efforts were made to separately identify contributions from these sources during this time period.
Representative data used to calculate ankle stiffness. Steady-state joint torque was averaged over a 100-ms window beginning 100 ms after perturbation onset (shaded area). Stiffness was quantified by normalizing the average change in torque (Δt) to the displacement amplitude (Δθ). The torque at steady state included contributions from both intrinsic joint mechanics and stretch reflex-mediated muscle contraction, but no effort was made to differentiate their relative contributions
Statistical analysis
Linear mixed effect models, with subject number as a random factor, were used to determine whether there was a significant influence of the environment on the background activity, reflex amplitude, and joint stiffness. Separate analyses were performed for each output measure. Analysis of variance (ANOVA) was used to identify significant effects. Post hoc analyses comparing environments were performed using the Tukey method. Significance was assessed at the 5% level, and all statistical analyses were performed in the R environment for statistical computing (The R Foundation for Statistical Computing, Vienna, Austria).
Results
Subjects successfully performed each task by staying within the desired torque bounds in the stiff environment and the desired position bounds in the remaining environments. For the stiff environment, subjects produced a mean plantar flexion torque of 5.2 ± 0.4% (95% CI) of MVC guided by visual feedback. In the remaining environments, the measured torque was generated by the motor based on the subject’s ankle position. Although the visual feedback differed between the stiff and remaining environments, both torque and position were matched across all conditions (torque: P = 0.48, position: P = 0.82).
There was no difference in the background activity of any muscle when comparing the average activity during the first five and last five trials for each environment (all P > 0.4). This suggests that fatigue was not a factor in these experiments and that the co-contraction strategy did not change from the beginning to the end of any trial set.
Stretch reflex modulation
Stretch reflex amplitude varied with the mechanical properties of the environment. Across all environments, only a single reflex response was observed in each of the plantar flexors. The average latency of this response was 37 ± 0.6, 36 ± 0.6, and 36 ± 0.6 ms for Sol, MG, and LG, respectively. Each of these latencies is consistent with the short latency reflex (Toft et al. 1991). There was no difference in reflex amplitude for any of the muscles when comparing the average responses in the first five and last five trials for each environment (all P > 0.6), suggesting that the feedback strategy was consistent from the beginning to the end of all trial sets. Reflex responses elicited during interactions with the stiff environment were larger than those elicited in the unstable environment (Fig. 3). For the subject shown in Fig. 3, the stretch reflex elicited in the U2 environment was smaller than the reflex in the stiff environment for all muscles. These findings held for the group as well (Fig. 4). Across all subjects, stretch reflex amplitude was significantly attenuated in all muscles during interaction with the most unstable (U2) environment relative to the stiff environment (all P < 0.05). This finding differs from similar tasks in the upper limb where prior studies report heightened stretch reflex sensitivity during interactions with compliant (Dietz et al. 1994; Perreault et al. 2008) or unstable loads (Akazawa et al. 1983; De Serres and Milner 1991; Krutky et al. 2010).
Perturbation trajectories and corresponding reflex responses for subject S4. Ankle perturbations with an amplitude of 0.17 rad (10°) were applied at 3.5 rad/s (200°/s). In each of the first two panels, positive values correspond to dorsiflexion. Mean, rectified EMG responses are shown for both the stiff environment (black line) and the most unstable environment, U2 (grey line). Consistent short latency stretch reflexes were elicited in each of the plantar flexors in both environments. However, the stretch reflex in each muscle was attenuated in the U2 environment relative to the stiff environment though the perturbation trajectories were matched in each condition
Average reflex amplitude for each environment. Reflexes were quantified as the mean amplitude of the rectified EMG over a 20-ms window following reflex onset. Data are group responses. Each row represents a different muscle. Within each plot, different bars correspond to different environments and the dashed horizontal lines indicate the average reflex amplitude in the stiff environment. Error bars represent 95% confidence intervals for the mean EMG amplitude across all subjects. Significant differences were assessed using a linear mixed effect model with subject as a random effect. This method removed subject variability before making comparisons across environments
Modulation of volitional activity
The changes in reflex amplitude across environments were accompanied by changes in background activity in all muscles. As the controlled load became more unstable, subjects increased the level of agonist–antagonist co-contraction. This increase in co-contraction was reflected by an increase in background activity in TA as the environment was changed from stiff to compliant and increased activity in MG, LG, and TA as the environment was changed from U1 to U2 (Fig. 5). Since the level of voluntary EMG co-varied with changes in the mechanical properties of the environment, it is possible that the observed changes in stretch reflex amplitude (Fig. 5) could simply reflect this change in voluntary EMG.
Average background activity for each environment. Background activity was quantified as the mean amplitude of the rectified EMG over a 100-ms window preceding movement onset. Data are group responses. Each row represents a different muscle. Within each plot, different bars correspond to different environments and the dashed horizontal lines indicate the average background activity in the stiff environment. Error bars represent 95% confidence intervals for the mean EMG amplitude across all subjects. Significant differences were assessed using a linear mixed effect model with subject as a random effect. This method removed subject variability before making comparisons across environments
Indeed, the observed changes in voluntary EMG produced reductions in stretch reflex amplitude independent of changes in the mechanical properties of the environment. This was determined by measuring reflex responses at matched levels of voluntary EMG in the stiff and U2 environments (Fig. 6). In the control condition, subjects interacted with the stiff environment and produced the same level of EMG in Sol and TA as was measured in the U2 environment (Sol: P = 0.60, TA: P = 0.43). The standard deviation of Sol and TA activity was similar in the control condition and the U2 environment (Sol: P = 0.11, TA: P = 0.38), which suggests that there was a comparable degree of reciprocal agonist–antagonist activity between these tasks. Analysis of the group data revealed that the amplitude of the Sol stretch reflex in the U2 and control conditions was comparable (P = 0.90), and each was significantly less than the stiff environment (both P < 0.01). Though subjects received feedback of only Sol and TA during the Control trials, background activity in LG was also matched between conditions (P = 0.54). Similar to Sol, reflex amplitude in LG in both the U2 and control conditions was significantly less than the stiff condition (both P < 0.001). In MG, reflex amplitude in the control condition was less than both the stiff condition (P < 0.001) and U2 condition (P < 0.001), but background activity was not matched across conditions (P = 0.027). Together, these data indicate that changes in co-contraction alone could cause changes in reflex amplitude comparable to those observed during interactions with unstable mechanical environments.
Influence of co-contraction of tibialis anterior (TA) on reflex amplitude in the plantar flexors. a Mean background activity in each muscle. The minimal activation of TA observed in the stiff condition indicates that there was limited co-contraction for this task. However, co-contraction was observed in the most unstable environment (U2). In the control condition, subjects produced the same level of Sol and TA activity observed in U2 while interacting with the stiff load. b Mean stretch reflex amplitude in each of the plantar flexors. Data are group responses. Each muscle is represented by a different set of bars. EMG from the stiff, U2, and control conditions is represented by the black, grey, and open bars, respectively. Error bars represent 95% confidence intervals for the mean EMG activity across subjects. Stars represent significant differences at the 5% level
Joint stiffness modulation
The observed agonist–antagonist co-contraction resulted in increased ankle stiffness in the most unstable (U2) environment (Fig. 7a). Although co-contraction varied across environments, the average joint stiffness did not differ significantly between the Stiff, Compliant, or U1 environments (all P > 0.05). The largest ankle stiffness was observed in the U2 environment, corresponding to the task where the highest level of background EMG was observed. Since subjects did not increase reflex sensitivity as a strategy to compensate for the unstable load dynamics, it is possible that co-contraction provided a sufficient increase in joint stiffness to offset the negative load stiffness in the unstable environments. This was investigated by calculating the net stiffness of the ankle, environment, and haptic load (K ank + K env + K load) for both the U1 and U2 environments. For the U1 environment, the net stiffness was indeed positive (Fig. 7b), indicating that ankle stiffness due to co-contraction and stretch reflexes was sufficient to offset the negative stiffness of the environment. However, this was not the case in the U2 environment. Although co-contraction did result in an increase in ankle stiffness in the U2 environment (Fig. 7a), this increase in stiffness was not sufficient to produce a net positive stiffness for the coupled system. This suggests that contributions from sources other than co-contraction and stretch reflexes were used to stabilize the load in this environment.
Estimated ankle stiffness. a Average ankle stiffness for each environment. There was no significant difference in ankle stiffness between the S, C, and U1 environments. Ankle stiffness increased from the U1 to U2 environments, reflecting the increased co-contraction observed in the U2 environment. b Net stiffness of the ankle and simulated load. The net stiffness includes the linearized load stiffness, the stiffness of the simulated supporting spring, and the stiffness of the ankle. The positive net stiffness in the U1 environment indicates that the coupled system has a stable equilibrium position. Here, stability implies that the system would return to its equilibrium point when perturbed, Despite the co-contraction observed in the U2 environment, the net system remains passively unstable. Error bars represent the 95% confidence intervals and significance is indicated at the 5% level
Evidence for volitional contribution to joint stabilization
Though the net stiffness of the coupled system remained negative in the most unstable environment, subjects were able to actively maintain the target position prior to the perturbation, providing evidence to active control beyond the regulation of joint stiffness. For each environment, three trajectories of ankle angle prior to perturbation onset are shown for a single subject (Fig. 8a). In the compliant and U1 environments, the variability in ankle position remains low and the subject was able to hold his ankle near the center of the target throughout the 1-s window. In the U2 environment, however, the variability in ankle position was significantly greater than the corresponding trajectories in the compliant and U1 environments. Though the peak-to-peak displacements were largest in this environment, subjects were able to maintain their ankle position within the target window even though the net stiffness of the coupled system was negative (Fig. 7b). This trend held across subjects (Fig. 8b), as the standard deviation of ankle position, was larger in the U2 environment relative to the compliant or U1 conditions (P < 0.05).
Variability in ankle position during stabilization. a Trajectories of ankle position prior to perturbation onset. For each environment, three trajectories are shown for the same subject. As the load becomes less stable, larger ankle excursions are observed, but they remain with the target window. b Average standard deviation of ankle position over a 1-s period immediately prior to the perturbation. Comparable variability was observed in the C and U1 environments, but variability was increased in the most unstable environment (U2). Error bars represent 95% confidence intervals
Discussion
The present study was performed to assess how changes in load stability influence both feed-forward muscle activation and reflex excitability in the ankle musculature. We hypothesized that subjects would compensate for unstable loads by increasing the magnitude of agonist–antagonist co-contraction and also by increasing stretch reflex excitability. Our results demonstrated that individuals did indeed increase the magnitude of co-contraction to stabilize the ankle as stiffness was reduced. This increase in co-contraction was reflected by an increase in ankle joint stiffness, consistent with the idea that feed-forward stiffness control is used in part to compensate for reduced stability. However, this co-contraction led to a reduction in stretch reflex sensitivity. To our knowledge, this is the first study to demonstrate a reduction in stretch reflex amplitude during tasks that require enhanced stability. This finding is in contrast to similar studies in the upper limb, each reporting increased reflex sensitivity as stability is compromised (De Serres and Milner 1991; Doemges and Rack 1992a, b; Dietz et al. 1994; Perreault et al. 2008; Krutky et al. 2010). Though our results do not support the hypothesis that rapid, involuntary reflexes are used to compensate for compromised stability in the lower limbs; our findings suggest that joint stability is actively controlled through a combination of feed-forward co-activation and longer latency, volitional intervention.
Potential mechanisms
A number of central and peripheral mechanisms could contribute to the reduction in reflex excitability observed in the current study. Increases in the level of tonic activity in the plantar flexors during the most unstable condition could reduce the fraction of the motor neuron pool available to fire in response to afferent input. However, within the torque range used in this study, reflex amplitude typically becomes greater with increasing levels of background activity (Gottlieb and Agarwal 1979; Mirbagheri et al. 2000; Cronin et al. 2008), making this an unlikely explanation for our results. Alternatively, TA activity also was increased in these tasks and disynaptic reciprocal inhibition from TA spindle afferents onto the plantar flexors could have influenced our results. In the cat, these projections extend to the MG, LG and SOL (Nichols 1994). Similar projections in humans could contribute to our observed modulation of reflexes in all measured plantar flexor muscles, even though changes in co-contraction were observed only in the MG and LG muscles, but not the SOL. A confounding report is that this source of inhibition is typically reduced during voluntary activation of the soleus (Nielsen and Kagamihara 1992), making it unclear how much reciprocal activation can contribute to our results.
Another explanation for the observed reflex attenuation could be presynaptic inhibition of Ia terminals from descending sources (Nielsen and Kagamihara 1993; Nielsen et al. 1995). The co-contraction of TA and the plantar flexors in the most unstable environment may have contributed to a change in “central set” during this task. The resulting command to TA could contribute to the smaller reflexes observed in the plantar flexors through presynaptic inhibition (Nielsen et al. 1995). Though estimates of presynaptic inhibition were not performed in our study, our observation of reduced reflex excitability during co-contraction alone is consistent with this mechanism. It is also possible that the reflexes were attenuated because of the small sways which occurred while subjects were within the target. Passive movement of the ankle has been shown to reduce the amplitude of the H-reflex (Brooke et al. 1995), possibly due to a depression in Ia transmission (Hultborn et al. 1996). While we cannot rule out this possibility, the reduction in reflex amplitude during the isometric control studies strongly suggests that a change in the central set contributed to at least a portion of the reflex behavior observed during the balancing of the unstable loads.
The influence of co-contraction on the stretch reflex may depend on the strength of the descending co-contraction command. Nielsen et al. (1994) characterized the influence of co-contraction on stretch reflexes in the ankle over a wide range of activation levels. They observed a reduction in stretch reflex amplitude in four of seven subjects, but only during weak co-contraction. As the level of co-contraction was increased, stretch reflex amplitude became larger relative to responses elicited during isolated plantar flexion, suggesting that sufficiently high levels of co-contraction can facilitate the stretch reflex. The contraction levels used in our study correspond to the weak levels of activation described by Nielsen and these levels were set by the magnitude of the unstable load that subjects were able to successfully balance. Hence, even though increased levels of co-contraction may lead to larger stretch reflexes, these increases are unlikely to be present during most tasks which challenge ankle stability.
It should be noted that our measure of co-contraction is unable to differentiate between a sustained increase in tonic activity and an attempted rapid alternation of agonist–antagonist activity. However, if this rapid alternation was present in the U2 environment, we would have expected to see more variability in muscle activity in this task relative to the isometric control task and this was not the case. Furthermore, during our isometric control study, we observed a reduction in reflex amplitude comparable to what was found during the stabilization task. Since it is unlikely that significant alternating activity would be observed in this task, our data suggest that the smaller reflex responses were most likely due to a change in tonic activity.
Comparison with task-dependent modulation in the upper limb
The influence of the mechanical properties of the environment on co-contraction and reflex excitability appears to differ between the upper and lower limbs. Both single and multi-joint studies in the upper limb report larger stretch reflexes during control of compliant loads relative to the responses measured during interaction with stiff loads (Doemges and Rack 1992a, b; Dietz et al. 1994; Perreault et al. 2008). Although a torque-matching task is used with the stiff load, whereas a position-matching task is used with the compliant load, the added inertial dynamics and reduced stiffness associated with the compliant load is sufficient to elicit larger stretch reflexes. In contrast to these findings, we observed no difference in reflex amplitude in the compliant environment when compared to the stiff environment. As load stability was further reduced, we did observe an increase in co-contraction similar to previous observations of control of the upper limb during interactions with unstable loads (Akazawa et al. 1983; De Serres and Milner 1991; Krutky et al. 2010). Despite this similarity, the resulting modulation of stretch reflex amplitude opposed previous reports as responses increased in amplitude in previous studies (Akazawa et al. 1983; De Serres and Milner 1991; Krutky et al. 2010), but grew smaller in our study.
This apparent discrepancy in the role of the stretch reflex might be explained by differences in the mechanics underlying the control of ankle stability during standing balance, for which the lower limb muscles are typically used, and the control of upper limb posture. Due to the mass of the body supported by the ankles, the time constant of the system they must stabilize is much longer than that seen by the arm, when supporting typical loads that can be lifted above horizontal. Thus, any perturbation from a given posture will grow much faster in the upper limb relative to the lower limb. Because of this, volitional reactions may be too slow to restore arm posture following unexpected disturbances, leaving stretch reflexes as the most effective means of maintaining stability. In contrast, the slower movements associated with standing balance are not likely to necessitate the rapid corrections necessary in the upper limb. Although our experiments were not conducted during standing and did not replicate many critical features of standing balance control such as body-weight support and trunk motion, they did utilize ankle loads with time constants similar to those observed during standing balance. It would be interesting to observe how the neuromotor systems controlling upper and lower limb posture adapt to loads with time constants different than those encountered during typical functional tasks.
Contribution of co-contraction to ankle stability
The observed increases in co-contraction with reduced load stability are consistent with regulation of joint posture via stiffness control. However, although subjects were able to stabilize the most unstable load, the stiffness generated via co-contraction was not sufficient to stabilize this load without feedback contributions from other sources. There has been controversy for some time regarding the contribution of intrinsic mechanics to ankle stability, most notably in inverted pendulum models of stance. The debate primarily revolves around whether intrinsic ankle stiffness is sufficient to maintain balance, as suggested by some (Kelton and Wright 1949; Fitzpatrick et al. 1992; Winter et al. 1998, 2001), or whether active modulation of muscle activity is required (Morasso and Schieppati 1999; Loram and Lakie 2002; Morasso and Sanguineti 2002; Peterka 2002). Our results support the latter proposition, demonstrating that co-contraction is used to increase ankle stiffness during the interaction with unstable loads, but that these increases alone are likely insufficient to provide stability. If co-contraction was a sufficient strategy for stance, we would expect that the stiffness generated in our task would be sufficient for the much smaller load which we simulated, but this was not observed. It is important to note that although the gastrocnemius was shortened in our experiments relative to its length during stance, this is not likely to explain the insufficient joint stiffness we observed. Prior estimates of ankle stiffness measured with the knee in a more flexed position (80° from neutral) demonstrated that high levels of co-contraction could result in an ankle stiffness of up to 230 Nm/rad which is more than twice the stiffness that was measured in the current study (Nielsen et al. 1994). Furthermore, the EMGs we recorded indicated that subjects never exceeded more than 17% of the activation in their SOL, MG, LG and TA. Together, these results suggest that subjects may have been capable of producing a larger stiffness, but chose a lower level of muscle activity.
Though insufficient to guarantee stability, moderate levels of co-contraction may simplify the task of feedback control. To use delayed feedback to stabilize a system about an unstable equilibrium point, it is generally necessary for the time constant (τ) of the system to be larger than the corresponding feedback delay (Milton et al. 2009). This time constant is a measure of how fast the system deviates away from equilibrium following perturbations or changes in initial conditions. For the simulated pendulum in our study with a height l = 1 m, the effective time constant \( \tau = \sqrt {l/g} \approx 320\,{\text{ms}} \). A lower bound for the feedback delay corresponding to volitional intervention can be estimated based on volitional reaction times and the time constants for muscle activation. For instance, at the ankle, reaction times during simple choice tasks typically extend beyond 120 ms (Gottlieb and Agarwal 1980). These delays are compounded by the twitch times of the ankle musculature which, at least in the plantar flexors, are ~75 ms (Buchthal and Schmalbruch 1970). Hence, a reasonable estimate of the fastest time between registration of disturbances at the joint and subsequent changes in torque would be ~200 ms. This provides a safety margin of only ~120 ms, and this margin would likely be much smaller due to the processing time associated with choosing the appropriate intervention strategy. Co-contraction could greatly increase the available time to react. For example, the stiffness of the ankle in our most unstable environment increases the time constant \( \tau = \sqrt {{{ml^{2} } \mathord{\left/ {\vphantom {{ml^{2} } {\left( {mgl - K_{\text{ank}} } \right)}}} \right. \kern-\nulldelimiterspace} {\left( {mgl - K_{\text{ank}} } \right)}}} \approx 480\,{\text{ms}} \) on average, providing an additional 160 ms. Therefore, moderate augmentation of stiffness via co-contraction, which alone is insufficient for stability, may still play an important role in the maintenance of balance. Such co-contraction is not commonly observed during quiet stance (Carpenter et al. 2001), but it is used for in more difficult tasks such as standing on unstable surfaces (Gantchev and Dimitrova 1996) or walking along balance beams (Llewellyn et al. 1990).
Influence of series elasticity on the contributions of co-contraction and stretch reflexes
A discussion of the role of co-contraction and stretch reflexes during control of ankle stability must also address the limitations imposed by the mechanics of the Achilles tendon. It is known that the maximum stiffness achievable through co-contraction is limited by the stiffness of the Achilles tendon (Hof 1998; Loram and Lakie 2002; Morasso and Sanguineti 2002). Previous measurements of ankle stiffness during co-contraction have reported values near 280 Nm/rad (Nielsen et al. 1994) and up to 400 Nm/rad for contractions near 50% of MVC (Kearney and Hunter 1990). While these values are not sufficient to maintain postural stability during upright stance (Morasso and Schieppati 1999), they would be sufficient to maintain ankle stability during our experiments. For the most unstable load in our task, a stiffness of 140 Nm/rad would be sufficient for a 70-kg subject, and this is well within the range of values reported in the literature. Thus, it does not appear that the control strategy subjects used was constrained by the limited stiffness of the Achilles tendon.
Relevance to previous studies on the control of 2nd-order loads
The results of our study extend the current body of knowledge regarding how the nervous system controls unstable loads with long time constants. Our observation of increased ankle sway as load stiffness was reduced is consistent with the larger sways reported when changes in the inertia (Loram et al. 2006) or stiffness of a manually controlled second-order load have been used to alter stability (Lakie et al. 2003; Chew et al. 2008). Using a spectral analysis of wrist movements during manual pendulum control, Chew et al. (2008) concluded that there was little or no change in the contribution of reflexes when load stability was reduced. This conclusion was based on invariance of the high-frequency components of wrist movements when stiffness was reduced. However, because this was a closed-loop task, the role of stretch reflexes cannot be easily inferred from analysis of the sway power spectrum, as acknowledged in the original study. Nevertheless, our more direct measures of reflex sensitivity are consistent with these previous conclusions and further provide evidence of a limited role for reflex activity when posture is perturbed.
Modulation of stretch reflexes in the lower limb during balance control
The observed reduction in stretch reflex amplitude during control of unstable loads is consistent with reported observations of H-reflex modulation during challenging postural tasks. Such results have been reported during tandem stance (Trimble and Koceja 2001; Taube et al. 2008), stance on unstable surfaces (Solopova et al. 2003; Taube et al. 2008), beam walking (Llewellyn et al. 1990), and ballistic leg extension (Holl and Zschorlich 2011). Although each of these studies revealed a reduced influence of sensory feedback when postural stability was challenged, H-reflex studies do not account for possible changes in fusimotor drive. If present, increased fusimotor drive could counteract the reduction in the efficacy of afferent transmission assessed by the H-reflex. By quantifying the mechanically induced stretch reflex, we have demonstrated that any potential changes in fusimotor drive are not sufficient to offset the reduced transmission of afferent input to motor neurons. These results suggest that the nervous system is organized in such a way as to limit the reflex contributions to ankle stability in conditions when postural stability is compromised.
There appear to be certain situations when challenges to postural stability result in increased reflex amplitude in the lower limbs. Krauss and Misiaszek (2007) demonstrated that H-reflex amplitude is enhanced at heel-strike when fore-aft trunk perturbations are applied during locomotion. The apparent difference between these findings and those described above may reflect differences in the biomechanical demands of stance and locomotion. During stance, balance is least stable in the saggital plane (Winter et al. 1998), whereas stability is compromised primarily in the frontal plane during locomotion (Bauby and Kuo 2000). If the reduced reflex gains reported above are indeed associated with challenges to postural stability, they would be expected to be greatest for perturbations along the least stable directions. This would predict a reduction in reflex sensitivity when perturbations are applied in the medio-lateral direction during locomotion, as has been observed during beam walking (Llewellyn et al. 1990).
It is important to note that subjects were seated in our experiments and that conclusions regarding how our results pertain to standing balance must be made with caution. Subjects were not in a weight-bearing position and the semi-flexed position of the knee may have particularly limited the contributions of the gastrocnemius. However, our conditions were selected so that load stability could be controlled precisely and to focus on the contributions of peripheral afferents in a manner consistent with previous studies (Gottlieb and Agarwal 1979; Grey et al. 2001; Kimura et al. 2003). Nevertheless, the agreement between our results and those from the standing tasks described above suggests that the role of afferent feedback during balance can be assessed during simpler tasks in which subjects control a load mimicking that mechanics of their own bodies. Our results may also be directly relevant to perturbations encountered during stumbling when large and rapid perturbations, such as those used in this study, are often encountered.
Conclusions
In summary, our results suggest that the influence of feed-forward strategies must be accounted for to fully understand the task-dependent contribution of feedback mechanisms in postural control. We found that reducing the level of support provided by the environment leads to altered feed-forward control as evidenced by increased co-contraction. This co-contraction is accompanied by a reduction in reflex amplitude leaving slower, potentially volitional commands as the likely source of the necessary feedback control. Though the role of stretch reflexes in tasks that compromise stability differs between the upper and lower limbs, this may simply reflect an optimal tuning of feedback commands to the mechanics of the musculoskeletal system. Use of higher level feedback during balance tasks is likely ideal because it enables the nervous system to integrate information from multiple sensory modalities and generate the appropriate commands necessary to maintain balance.
References
Akazawa K, Milner TE, Stein RB (1983) Modulation of reflex EMG and stiffness in response to stretch of human finger muscle. J Neurophysiol 49:16–27
Bauby CE, Kuo AD (2000) Active control of lateral balance in human walking. J Biomech 33:1433–1440
Brooke JD, Cheng J, Misiaszek JE, Lafferty K (1995) Amplitude modulation of the soleus H reflex in the human during active and passive stepping movements. J Neurophysiol 73:102–111
Buchthal F, Schmalbruch H (1970) Contraction times and fibre types in intact human muscle. Acta Physiol Scand 79:435–452
Carpenter M, Frank J, Silcher C, Peysar G (2001) The influence of postural threat on the control of upright stance. Exp Brain Res 138:210–218
Carter RR, Crago PE, Gorman PH (1993) Nonlinear stretch reflex interaction during cocontraction. J Neurophysiol 69:943–952
Chew JZ, Gandevia SC, Fitzpatrick RC (2008) Postural control at the human wrist. J Physiol 586:1265–1275
Christensen LO, Petersen N, Andersen JB, Sinkjaer T, Nielsen JB (2000) Evidence for transcortical reflex pathways in the lower limb of man. Prog Neurobiol 62:251–272
Crago PE, Houk JC, Hasan Z (1976) Regulatory actions of human stretch reflex. J Neurophysiol 39:925–935
Cronin NJ, Peltonen J, Ishikawa M, Komi PV, Avela J, Sinkjaer T, Voigt M (2008) Effects of contraction intensity on muscle fascicle and stretch reflex behavior in the human triceps surae. J Appl Physiol 105:226–232
De Serres SJ, Milner TE (1991) Wrist muscle activation patterns and stiffness associated with stable and unstable mechanical loads. Exp Brain Res 86:451–458
de Zee M, Voigt M (2001) Moment dependency of the series elastic stiffness in the human plantar flexors measured in vivo. J Biomech 34:1399–1406
Dietz V, Discher M, Trippel M (1994) Task-dependent modulation of short- and long-latency electromyographic responses in upper limb muscles. Electroencephalogr Clin Neurophysiol 93:49–56
Doemges F, Rack PM (1992a) Task-dependent changes in the response of human wrist joints to mechanical disturbance. J Physiol 447:575–585
Doemges F, Rack PM (1992b) Changes in the stretch reflex of the human first dorsal interosseous muscle during different tasks. J Physiol 447:563–573
Fitzpatrick RC, Taylor JL, McCloskey DI (1992) Ankle stiffness of standing humans in response to imperceptible perturbation: reflex and task-dependent components. J Physiol 454:533–547
Gantchev GN, Dimitrova DM (1996) Anticipatory postural adjustments associated with arm movements during balancing on unstable support surface. Int J Psychophysiol 22:117–122
Gawthrop P, Loram I, Lakie M (2009) Predictive feedback in human simulated pendulum balancing. Biol Cybern 101:131–146
Gottlieb GL, Agarwal GC (1979) Response to sudden torques about ankle in man: myotatic reflex. J Neurophysiol 42:91–106
Gottlieb GL, Agarwal GC (1980) Response to sudden torques about ankle in man. II. Postmyotatic reactions. J Neurophysiol 43:86–101
Grey MJ, Ladouceur M, Andersen JB, Nielsen JB, Sinkjaer T (2001) Group II muscle afferents probably contribute to the medium latency soleus stretch reflex during walking in humans. J Physiol 534:925–933
Hammond PH (1956) The influence of prior instruction to the subject on an apparently involuntary neuro-muscular response. J Physiol 132:17–18P
Hanneton S, Berthoz A, Droulez J, Slotine JJ (1997) Does the brain use sliding variables for the control of movements? Biol Cybern 77:381–393
Hirokawa S, Solomonow M, Luo Z, Lu Y, Dambrosia R (1991) Muscular cocontraction and control of knee stability. J Electromyogr Kinesiol 1:199–208
Hof AL (1998) In vivo measurement of the series elasticity release curve of human triceps surae muscle. J Biomech 31:793–800
Holl N, Zschorlich V (2011) Neural control of joint stability during a ballistic force production task. Exp Brain Res 210:229–242
Hultborn H, Illert M, Nielsen J, Paul A, Ballegaard M, Wiese H (1996) On the mechanism of the post-activation depression of the H-reflex in human subjects. Exp Brain Res 108:450–462
Kearney RE, Hunter IW (1990) System identification of human joint dynamics. Crit Rev Biomed Eng 18:55–87
Kearney RE, Stein RB, Parameswaran L (1997) Identification of intrinsic and reflex contributions to human ankle stiffness dynamics. IEEE Trans Biomed Eng 44:493–504
Kelton IW, Wright RD (1949) The mechanism of easy standing by man. Aust J Exp Biol Med Sci 27:505–515
Kimura T, Nozaki D, Nakazawa K, Akai M, Ohtsuki T (2003) Gradual increment/decrement of isometric force modulates soleus stretch reflex response in humans. Neurosci Lett 347:25–28
Krauss EM, Misiaszek JE (2007) Phase-specific modulation of the soleus H-reflex as a function of threat to stability during walking. Exp Brain Res 181:665–672
Krutky MA, Ravichandran VJ, Trumbower RD, Perreault EJ (2010) Interactions between limb and environmental mechanics influence stretch reflex sensitivity in the human arm. J Neurophysiol 103:429–440
Lakie M, Caplan N, Loram ID (2003) Human balancing of an inverted pendulum with a compliant linkage: neural control by anticipatory intermittent bias. J Physiol 551:357–370
Llewellyn M, Yang JF, Prochazka A (1990) Human H-reflexes are smaller in difficult beam walking than in normal treadmill walking. Exp Brain Res 83:22–28
Loram ID, Lakie M (2002) Direct measurement of human ankle stiffness during quiet standing: the intrinsic mechanical stiffness is insufficient for stability. J Physiol 545:1041–1053
Loram ID, Maganaris CN, Lakie M (2005) Active, non-spring-like muscle movements in human postural sway: how might paradoxical changes in muscle length be produced? J Physiol 564:281–293
Loram ID, Gawthrop PJ, Lakie M (2006) The frequency of human, manual adjustments in balancing an inverted pendulum is constrained by intrinsic physiological factors. J Physiol 577:417–432
Milner TE, Cloutier C, Leger AB, Franklin DW (1995) Inability to activate muscles maximally during cocontraction and the effect on joint stiffness. Exp Brain Res 107:293–305
Milton J, Cabrera JL, Ohira T, Tajima S, Tonosaki Y, Eurich CW, Campbell SA (2009) The time-delayed inverted pendulum: implications for human balance control. Chaos 19:026110
Mirbagheri MM, Barbeau H, Kearney RE (2000) Intrinsic and reflex contributions to human ankle stiffness: variation with activation level and position. Exp Brain Res 135:423–436
Morasso PG, Sanguineti V (2002) Ankle muscle stiffness alone cannot stabilize balance during quiet standing. J Neurophysiol 88:2157–2162
Morasso PG, Schieppati M (1999) Can muscle stiffness alone stabilize upright standing? J Neurophysiol 82:1622–1626
Nichols TR (1994) A biomechanical perspective on spinal mechanisms of coordinated muscular action: an architecture principle. Acta Anat (Basel) 151:1–13
Nielsen J, Kagamihara Y (1992) The regulation of disynaptic reciprocal Ia inhibition during co-contraction of antagonistic muscles in man. J Physiol 456:373–391
Nielsen J, Kagamihara Y (1993) The regulation of presynaptic inhibition during co-contraction of antagonistic muscles in man. J Physiol 464:575–593
Nielsen J, Sinkjaer T, Toft E, Kagamihara Y (1994) Segmental reflexes and ankle joint stiffness during co-contraction of antagonistic ankle muscles in man. Exp Brain Res 102:350–358
Nielsen J, Crone C, Sinkjaer T, Toft E, Hultborn H (1995) Central control of reciprocal inhibition during fictive dorsiflexion in man. Exp Brain Res 104:99–106
Perreault EJ, Chen K, Trumbower RD, Lewis G (2008) Interactions with compliant loads alter stretch reflex gains but not intermuscular coordination. J Neurophysiol 99:2101–2113
Peterka RJ (2002) Sensorimotor integration in human postural control. J Neurophysiol 88:1097–1118
Shemmell J, An JH, Perreault EJ (2009) The differential role of motor cortex in stretch reflex modulation induced by changes in environmental mechanics and verbal instruction. J Neurosci 29:13255–13263
Sinkjaer T, Toft E, Andreassen S, Hornemann BC (1988) Muscle stiffness in human ankle dorsiflexors: intrinsic and reflex components. J Neurophysiol 60:1110–1121
Solopova IA, Kazennikov OV, Deniskina NB, Levik YS, Ivanenko YP (2003) Postural instability enhances motor responses to transcranial magnetic stimulation in humans. Neurosci Lett 337:25–28
Taube W, Leukel C, Gollhofer A (2008) Influence of enhanced visual feedback on postural control and spinal reflex modulation during stance. Exp Brain Res 188:353–361
Toft E, Sinkjaer T, Andreassen S (1989) Mechanical and electromyographic responses to stretch of the human anterior tibial muscle at different levels of contraction. Exp Brain Res 74:213–219
Toft E, Sinkjaer T, Andreassen S, Larsen K (1991) Mechanical and electromyographic responses to stretch of the human ankle extensors. J Neurophysiol 65:1402–1410
Trimble MH, Koceja DM (2001) Effect of a reduced base of support in standing and balance training on the soleus H-reflex. Int J Neurosci 106:1–20
Winter DA, Patla AE, Prince F, Ishac M, Gielo-Perczak K (1998) Stiffness control of balance in quiet standing. J Neurophysiol 80:1211–1221
Winter DA, Patla AE, Rietdyk S, Ishac MG (2001) Ankle muscle stiffness in the control of balance during quiet standing. J Neurophysiol 85:2630–2633
Acknowledgments
This work was supported by NIH grants T32 HD007418-17, R01 NS053813, and an NSF Graduate Research Fellowship. The authors would like to thank the subjects for their participation and Tim Haswell for technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Finley, J.M., Dhaher, Y.Y. & Perreault, E.J. Contributions of feed-forward and feedback strategies at the human ankle during control of unstable loads. Exp Brain Res 217, 53–66 (2012). https://doi.org/10.1007/s00221-011-2972-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-011-2972-9