Abstract
According to the amyloid cascade hypothesis, sporadic Alzheimer’s disease (AD) is caused by the production and aggregation of β-amyloid (Aβ), and the production of Aβ has recently been linked to the metabolism of cholesterol. We have previously published clinical studies where the effect of statin treatment on Aβ production has been investigated. No effect on Aβ was found, which is in disagreement with cell and animal studies. In the present study we investigated the effect of statin treatment on a disease-specific pattern consisting of a C-terminally-truncated quintet of Aβ peptides. Nineteen patients with AD were treated with simvastatin for 12 months and the quintet of Aβ peptides were analysed in cerebrospinal fluid before and after treatment. Also included was a group of 15 untreated patients with AD. We found that the Aβ peptide pattern at baseline was in agreement with earlier findings; however, we did not find any change in the Aβ peptide pattern after statin treatment. We suggest that clinical studies with extended treatment periods are performed where higher dosages of statins are used. We also believe that the pleiotropic effects of statins should be investigated further in order to elucidate the connection between Alzheimer’s disease and statin treatment.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The processing of amyloid precursor protein (APP) results in the production of β-amyloid (Aβ) peptide, a major constituent of senile plaques (SP), a pathological hallmark in Alzheimer’s disease (AD). According to the amyloid cascade hypothesis, the production and aggregation of Aβ peptides are central events in the aetiology of sporadic AD. Evidence for this hypothesis comes from several lines of inquiry: (1) the disease-causing mutations found in APP, Presenilin 1 (PS1) and Presenilin 2 (PS2) genes all lead to an upregulation of Aβ40 and/or Aβ42 peptides (Scheuner et al 1996); (2) individuals with trisomy 21 (with three APP genes) develop AD pathology in their fourth or fifth decade of life (Wisniewski et al 1985; Iwatsubo et al 1995); (3) allelic variation of the apolipoprotein E (ApoE) gene is a risk factor for sporadic AD, and since ApoE binds to Aβ (Strittmatter et al 1993a, 1993b), it may be involved in the formation of SP.
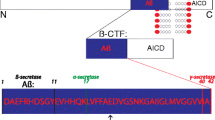
APP is ubiquitously expressed in all cells and is secreted by both brain and peripheral cells into extracellular fluids. The initial cleavage of APP is made by either α- or β-secretase, producing the soluble fragments αsAPP or βsAPP and the corresponding C-terminal fragment C83 or C99 (Andreasen and Blennow 2002). The C-terminal fragments remain membrane-bound and are substrates for the subsequent intramembranous cleavage by the γ-secretase complex, producing p3 or Aβ (Andreasen and Blennow 2002). The cleavage by α-secretase occurs after Lys16 within the Aβ sequence, thereby precluding the formation of Aβ. The identity of α-secretase is still not clear, but three candidates have been suggested: tumour necrosis factor-α-converting enzyme (TACE), also called ADAM17, ADAM10 (a disintegrin and metalloprotease) and MDC9 (metallo/disintegrin/cystein-rich proteins, also called ADAM9) (Black et al 1997; Moss et al 1997; Buxbaum et al 1998; Koike et al 1999; Lammich et al 1999). The β-secretase has been identified as BACE (β-site APP cleaving enzyme) (Sinha et al 1999; Vassar et al 1999; Yan et al 1999) and it cleaves APP at two different positions: at Met1 and after amino acid 10 (Aβ numbering). In addition, a BACE homologue, BACE 2, has been identified (Farzan et al 2000). The identity of the γ-secretase is not known, but accumulating evidence suggests that the γ-secretase is a high molecular weight complex formed by several proteins—APH1b, PEN2, Nicastrin and PS1 (Yu et al 2000)—and PS1 has been identified as an essential cofactor for γ-secretase activity (De Strooper et al 1998).
In recent years, several studies have indicated that elevated intracellular cholesterol levels might affect the processing of APP, promoting the production of Aβ. Changing the cholesterol and lipid compositions of cellular membranes have been shown to affect both α- and γ-secretase (Bodovitz and Klein 1996; Urmoneit et al 1998; Kojro et al 2001; Runz et al 2002; Wahrle et al 2002). Other cell studies and animal studies have revealed that increased levels of cholesterol increases the production of Aβ, while lowering cholesterol levels with statins (de novo cholesterol synthesis inhibitors) decreases the production of Aβ (Simons et al 1998; Fassbender et al 2001). Epidemiological studies suggest that statin treatment lowers the incidence of AD (Jick et al 2000; Wolozin et al 2000; Rockwood et al 2002; Yaffe et al 2002). The effect of statin treatment on APP processing and the production of Aβ has also been investigated in clinical studies (Friedhoff et al 2001; Tokuda et al 2001; Fassbender et al 2002; Simons et al 2002; Ishii et al 2003), but with contradictory results. Animal studies have also shown contradictory results (Park et al 2003). Our group has published two studies (Sjögren et al 2003; Höglund et al 2004) where patients treated with statins have shown no changes in cerebrospinal fluid (CSF) or plasma levels of Aβ1-40 or Aβ1-42.
Recently, a peptide pattern of a quintet of Aβ peptides truncated at the C-terminal (Aβ 1-37/38/39/40 and 42) has been found in AD (Wiltfang et al 2001, 2002), where the relative order of abundance differed in absolute and relative terms. This result has been further confirmed by SELDI-TOF (Lewczuk et al 2003). The pattern appears to be specific, indicating a tightly regulated pathway.
We present an open biochemical study where patients with AD were treated with simvastatin (20 mg/day) for 12 months; a group of untreated patients with AD were also included. CSF samples were taken at baseline and after 12 months of treatment (endpoint) with simvastatin. The main aim of this study was to investigate whether treatment with simvastatin for 12 months affects the disease-specific peptide pattern. Our first step was to confirm previous findings (Wiltfang et al 2001, 2002; Lewczuk et al 2003). We also wanted to evaluate the two methods used for analysis of Aβ, the Luminex system and the western blot. We show that the peptide patterns in the untreated and treated groups at baseline are in agreement with earlier findings by Wiltfang et al (2002). However, we did not find any significant change in the peptide pattern after treatment with statins when comparing baseline with endpoint. A comparison of the absolute values of Aβ1-42 from the western blot analysis revealed a high correlation with the Aβ1-42 values quantified using xMAP technology on the Luminex system.
Materials and methods
Patients
Nineteen patients with AD were treated with 20 mg/day of simvastatin for 12 months, and CSF and plasma samples were collected at baseline and endpoint (12 months). One patient was excluded from the present study because western blot analysis failed. Fifteen untreated patients with AD were also included in the study, and only CSF samples, no plasma, were taken at baseline and endpoint from this group. Demographic data for both the untreated and treated groups are shown in Table 1. All patients underwent a thorough clinical investigation, which included medical history; physical, neurological and psychiatric examinations; screening laboratory tests; an electroencephalogram; and a computerized tomography scan of the brain. The diagnosis of probable AD was made according to the criteria of the National Institute of Neurological and Communicative Disorders and Stroke and Alzheimer’s Disease and Related Disorders Association (McKhann et al 1984). No patient with AD had a family history of dementia suggestive of autosomal dominant AD. The cognitive status was examined using the Mini Mental State Examination (MMSE) (Folstein et al 1975) and the Alzheimer Disease Assessment Scale-Cognition (ADAS-Cog) was used to examine cognitive function (Mohs and Cohen 1988).
Sample collection
Lumbar puncture was performed in the morning under standard conditions. A volume of 12 ml of CSF was collected, and gently mixed to avoid gradient effects. A blood sample was taken at the same time. All CSF samples with more than 500 erythrocytes/μl were excluded. The CSF/serum albumin ratio [CSF-albumin (mg/ml) divided by serum albumin (g/l)] was used as a measure of blood–brain barrier function. All samples were centrifuged to remove cells and debris, and the CSF was stored at −80 °C pending biochemical analyses. Due to previous analysis, all samples in the present study have been through two cycles of freezing/thawing, and a few samples have been through an additional third cycle. The local ethical committees approved the study. All the patients or their relatives gave informed consent to participate in the study, which was performed in accordance with the provisions of the Helsinki Declaration.
Lipoproteins, APOE genotype and Aβ1-42 on the Luminex
The plasma levels of total cholesterol (TC) and triglycerides (TG) were determined enzymatically with a Modular Hitachi analyzer (Roche Diagnostics Scandinavia AB, Stockholm, Sweden). Plasma high-density lipoprotein (HDL) cholesterol was determined enzymatically after low-density lipoprotein (LDL) and very low-density lipoprotein cholesterol was selectively removed by precipitation from the plasma by magnesium sulphate and dextran sulphate. LDL cholesterol concentration was estimated using the Friedewald formula (Friedewald et al 1972). ApoE genotyping was performed by polymerase chain reaction followed by mini sequencing, as described previously (Blennow et al 2000). The CSF levels of Aβ1-42 were measured using the xMAP technology on the Luminex system (Austin, Texas, USA) as described previously (Ohlsson et al 2005).
β-amyloid-sodium dodecyl sulphate-polyacrylamide gel electrophoresis
Following the protocol from Wiltfang et al (2002), the synthetic Aβ peptides were directly dissolved in sample buffer I containing 0.36 M BisTris, 0.16 M bicine, 1% (w/v) SDS, 15% (w/v) sucrose, 0.004% (w/v) bromophenol blue, and heated at 95 °C for 5 min. This latter buffer was also used for subsequent dilutions of the synthetic peptides. Sample buffer II was composed of 0.12 M BisTris, 0.053 M bicine, 5% sucrose, 0.5% SDS, and 0.0025% bromophenol blue containing one tablet of proteinase inhibitor cocktail Complete Mini per 10 ml. Sample buffer II was dried (SpeedVac, 40 °C) in polypropylene tubes (Eppendorf, Hamburg, Germany). The equivalent volume of CSF was added to the polypropylene tubes and vortexed until completely solubulized. After the addition of 2-mercaptoethanol to a final concentration of 2.5% v/v, the sample mix was heated at 95 °C for 5 min.
The urea version of the bicine/BisTris/Tris/sulphate SDS-PAGE of Wiltfang et al (1991) was used to separate the Aβ peptides . The composition of the separation gel was modified from 15%T%/5%C%8 M to 12%T%/5%C/8 M urea, and the gel thickness was 0.5 mm, as described in Wiltfang et al (2002). The gels were run at 25 mA/gel for 1 h, using the MiniProtean II electrophoresis unit (Bio-Rad Laboratories, Hercules, CA, USA). Ten microliters of CSF were loaded per lane. All samples were run in quadruplicate and each gel carried a five-step dilution series of the synthetic Aβ peptide mix. Mean values were used for subsequent calculations of Aβ1-37/38/39/40 and 42 peptides.
Western blot
Western blot and immunostaining was performed according to the the following protocol (Wiltfang et al 2002): proteins were transferred for 30 min at 1 mA/cm2 and room temperature under semi-dry conditions (Hoefer Semiphor) onto Immobilon-P PVDF membranes according to Wiltfang et al (1997). All of the following washing and incubation steps were performed at room temperature if not otherwise stated. For immunostaining, the Immobilon-P PVDF membranes were washed in deionized H20 and boiled for 3 min in PBS (phosphate buffer saline) using a microwave oven (Ida et al 1996; Wiltfang et al 1997). Blocking was performed for 1 h using Roti-Block (Carl Roth GmbH& Co. KG, Karlsruhe, Germany). Incubation with the primary antibody, which was diluted 1:200 in RotiBlock, was performed overnight at +4 °C. The membranes were washed for 30, 15 and 2×10 min in PBS–T (0.075% v/v Tween 20) and then incubated for 1 h with anti-mouse biotinylated IgG (H+L) antibody (Vector Laboratories, Burlingham, CA, USA) (1 mg/ml), which was diluted 1:3,000 in PBS–T. A second PBS–T wash was preformed for 3×10 min and then the membranes were incubated for 1 h with streptavidin-biotinylated horseradish peroxidase complex (Amersham Pharmacia Biotech, Buckinghamshire, UK) diluted 3,000-fold. Following 3×10 min washes, the membranes were developed for 5 min with ECL Advanced (Amersham Pharmacia Biotech) solution according to the manufacturer’s instructions. The emitted signal was detected by a CCD camera (Fluor-S Max MultiImager; BioRad), using a series of 5, 10, 15, 30, 45, 60, 75, 90, 105, 120, 135 and 150 s for data acquisition. Band intensities were quantified relative to an internal five-step dilution series of the Aβ peptide standard mix using Quantity One software.
Statistics
All statistical procedures were performed using the Statistical Package of the Social Sciences (SPSS). The Shapiro–Wilk test was performed to test the data distribution. The data was not normally distributed, so the groups were compared using the non-parametric Kruskal–Wallis test. The paired Wilcoxon test was used to investigate any significant changes between baseline and endpoint. The Spearman correlation coefficient was used for correlations. Individual Aβ peptides were expressed as absolute values (ng/ml) and as percentages of the total Aβ peptide concentration (Aβ1-X%, fractional Aβ peptide values). For multivariate analysis, the data was transformed and the ANOVA test was used. Values of p lower than 0.05 were considered significant.
Results
At baseline, the Aβ quintet showed the following order of abundance in absolute and relative terms: Aβ1-40>Aβ1-38>Aβ1-39>Aβ1-37>Aβ1-42, in both the untreated group (Table 2) and the treated group (Table 3).
In the untreated group there was a slight but significant decrease in the absolute values of Aβ1-39 (p=0.030) when comparing endpoint with baseline. However, this decrease was not significant when the levels of Aβ1-39 were expressed as percentages of the total Aβ peptide concentration.
In the treated group there were no changes in any of the absolute peptide levels when comparing endpoint with baseline (Fig. 1a–f). Neither were there any changes in the ratios between Aβ1-42 and Aβ1-40, Aβ1-42 and Aβ1-38 or the ratio between Aβ1-38 and Aβ1-40. However, there was a slight but significant decrease (p=0.032) in the levels of Aβ1-39 when the value was expressed as a percentage of the total Aβ peptide concentration.
We investigated the effect of confounding factors on changes between endpoint and baseline for the five different Aβ peptides using multivariate analysis. The following confounding factors were analysed; sex, age, APOE4 and both baseline values and difference between baseline and endpoint in MMSE, ADAS-Cog and cholesterol levels. We only found effects on the Aβ peptides 1–38 and 1–40. The ADAS-Cog score at baseline was significantly related to the change in Aβ1-38 (p=0.036) and the change in ADAS-Cog was significantly related to the change in Aβ1-40 (p=0.040). The MMSE score at baseline was significantly (p=0.043) related to the change in Aβ1-40 and the decrease in MMSE was significantly related to the change in Aβ1-38 (p=0.039). In addition, the decrease of cholesterol in plasma significantly influenced the change in Aβ1-40 (p=0.031).
The plasma levels of cholesterol (p=0.000) and LDL (p=0.000) were significantly decreased (Table 4) in the treated AD group when comparing endpoint with baseline, and the plasma levels of HDL were significantly increased (p=0.046) (Table 4). The CSF levels of Aβ1-42 measured by xMAP technology correlated with the levels of Aβ1-42 quantified by western blot, r=0.593 (p=0.000] (Fig. 2).
Discussion
Disease-specific Aβ peptide pattern
A disease-specific pattern of C-terminally-truncated Aβ peptides in CSF has been found in AD, with decreased absolute and relative quantities of Aβ1-42 and increased relative quantities of Aβ1-38 (Wiltfang et al 2002). In nondemented controls, the Aβ quintet showed the following order of abundance in absolute and relative terms: Aβ1-40>Aβ1-38>Aβ1-42>Aβ1-39≥Aβ1-37, while in patients with AD the following pattern was found: Aβ1-40>Aβ1-38>Aβ1-39> Aβ1-37>Aβ1-42. In the present study we found the same peptide pattern in both the treated and untreated groups of patients with AD at baseline, in other words Aβ1-40>Aβ1-38>Aβ1-39>Aβ1-37>Aβ1-42, suggesting that this pattern is highly regulated. In the untreated group we found a slight but significant reduction in the absolute levels of Aβ1-39. However, this did not change the relative order of abundance. The method shows the high reproducibility of the absolute and relative quantities of CSF Aβ peptides measured by Aβ SDS-PAGE/immunoblot when comparing endpoint with baseline.
Effect of statin treatment on Aβ peptide pattern
When comparing endpoint with baseline in the treated AD group, no significant change was seen when the absolute levels of Aβ peptides were expressed. However, upon expressing the levels of different peptides as percentages of the total Aβ level, a slight decrease in Aβ1-39 was seen in the treated group when comparing endpoint with baseline, but with no change of the peptide pattern. From these results we can also conclude that there is no change in the CSF levels of Aβ1-40 or Aβ1-42, and the level of Aβ1-42 was also shown to be unchanged by xMAP technology. It has previously been shown that the level of Aβ1-42 does not to decline during disease progression in untreated patients with AD (Andreasen et al 1999). The constant level of Aβ observed in this study correlates with two previously published studies from our group (Sjögren et al 2003; Höglund et al 2004) where the production of Aβ was studied and where statin treatment was shown to have no effect on the level of Aβ. In our first study (Sjögren et al 2003), the CSF and plasma levels of Aβ1-42 were investigated in patients with AD before and after 12 weeks of treatment with simvastatin. In the second study (Höglund et al 2004), plasma levels of Aβ1-40, 1-42 and total Aβ were investigated in patients with hypercholesterolemia, before and after 6, 12 and 24 weeks of treatment with either atorvastatin or simvastatin.
The lack of influence on the Aβ pattern observed in this study could perhaps be explained by the dosage of simvastatin being too low. However, in a study performed recently by our group (Höglund et al 2005), we found that treatment with 20 mg/day of simvastatin over 12 months is sufficient to affect cholesterol metabolism in both the peripheral body and the CNS, as indicated by reduced levels of lathosterol (a marker for cholesterol synthesis) and 24S-hydroxycholesterol (a marker for cholesterol homeostasis is in the CNS). In addition, doses of between 10 and 40 mg/day of statins have been shown to significantly reduce the CSF levels of 24S-hydroxycholesterol (Fassbender et al 2002), which is a marker for brain cholesterol homeostasis (Björkhem et al 1998). In cell and animal experiments where statins were found to affect Aβ production (Simons et al 1998; Fassbender et al 2001), the doses given exceeded clinically-relevant doses many times over. In epidemiological studies (Jick et al 2000; Wolozin et al 2000; Rockwood et al 2002; Yaffe et al 2002), the results were not sub-grouped by statin dosage. It has been shown that different statins have different effects (Kirsch et al 2003), which could also explain the lack of effect in the present study.
In vitro studies (Simons et al 1998; Frears et al 1999) have shown that lowering intracellular levels of cholesterol with statins affects the cholesterol content of cellular membranes. The γ-secretase complex believed to be responsible for the production of Aβ peptides would be expected to be sensitive to changes in the cholesterol content of cellular membranes since the cleavage occurs intramembraneously. Perhaps there are alternative proteolytical pathways. An ε-cleavage site has been identified (Weidemann et al 2002), located distal to the γ-cleavage site, and there are results indicating additional cleavages, with the Aβ peptide itself as substrate (Shi et al 2003). Together these findings could form a regulated pathway, producing the different peptides 1-37/38 and 39.
In epidemiological studies (Jick et al 2000; Wolozin et al 2000; Rockwood et al 2002; Yaffe et al 2002) where statin treatment was found to reduce the prevalence of AD, longer treatment periods (2–5 years) were investigated than explored here. Reducing the synthesis of cholesterol, which must be compensated for in the long run, may result in a new equilibrium, where the cholesterol content of the membrane is slightly affected. It should be noticed that the effect of statins on the cholesterol content of cellular membranes in humans is unknown and that the full effect might not be seen after only 12 months of treatment. Although statins work preventatively against the negative impacts that Aβ has on cholesterol and lipid homeostasis (Mason et al 1999; Chochina et al 2001; Michikawa et al 2001) and on the postulated deposition of Aβ on the plasma membrane (Yamaguchi et al 2000) it may be too late to see any change in Aβ production after statin treatment in patients who have already developed AD.
Mutually exclusive regulation of the APP cleavage pathways is suggested to occur, and enhanced α-cleavage may suppress β-cleavage at the same time (Checler 1995). However, this generally accepted hypothesis does not appear to be valid under all circumstances (Tomita et al 1998; Mills and Reiner 1999), and the processing of APP is probably much more complicated. There are findings suggesting that APP is processed differently, depending on the cell type (Hartmann et al 1997). That the Aβ peptides produced are heterogeneous in both the N-terminal and the C-terminal further indicates complicated processing. Together these findings suggest that several proteolytical products from the processing of APP should be analysed simultaneously.
The pathological significance of the Aβ peptide pattern relevant to AD is still not clear and must be further elucidated. However, considering that the CSF level of Aβ1-42 is a biochemical marker for AD (Blennow and Hampel 2003), we believe that changes in CSF are able to reflect those in the brain. We also believe that further investigations of Aβ peptides that are truncated in both the C-terminal and N-terminal are important if we are to elucidate the role of Aβ in AD.
Confounding factors
It is important to be cautious when interpreting the results from the multivariate analyses in the present study, since only 18 patients were included in the study and the multivariate analyses included several factors. We investigated the effects of the following confounding factors on changes in the different Aβ peptide levels between endpoint and baseline: sex, age, APOE4, baseline values of MMSE, ADAS-Cog and cholesterol, and finally changes in MMSE, ADAS-Cog and cholesterol. We did not find any effects of sex, age or APOE4 genotype on the change between baseline and endpoint for the five different peptides. For the Aβ peptide 1–40, a less pronounced decrease in plasma cholesterol was significantly related to a more pronounced decrease of Aβ1-40. We also found that both the change in ADAS-Cog and MMSE and the baseline values of ADAS-Cog and MMSE could be related to decreases in Aβ1-38 and 1–40. There were no correlations, but when looking at the plots we see that: (1) a more affected cognitive state at baseline (that is, low MMSE and high ADAS-Cog) is linked to a more pronounced decrease in Aβ1-38 and Aβ1-40, and (2) those patients who did not detoriate as much as others during the 12 months of treatment had a more pronounced decrease in the CSF levels of Aβ1-38 and Aβ1-40. These two groups of patients—the ones with a more affected cognitive state at baseline and the ones who did not detoriate as much—are more or less the same patients. This indicates that Aβ1-38 and Aβ1-40 may be related to severity of dementia. In agreement with this, Lue et al (1999 and McLean et al (1999) suggest that the soluble fraction of Aβ peptides is more closely correlated to the severity of dementia, compared to the insoluble Aβ peptides aggregated in plaques. Interestingly, in the study by Wiltfang et al (2002) there were also indications that Aβ1-38 and Aβ1-40 are connected to severity of dementia. In that study, the connection was found when the peptides were expressed as percentages of total Aβ. This may not be comparable to this study but is an interesting finding.
There is a significant difference in the number of carriers of the E4 allele of ApoE between the treated and untreated group. Even though ApoE is a risk factor for AD, there seems to be no connection between presence of the ApoE4 allele and cognitive decline (Bunce et al 2004).
Correlation between Luminex and western blot
One potential drawback when measuring Aβ in the Luminex system is that the measurement can be affected if other proteins bind to Aβ. In the present study we measured the CSF levels of Aβ1-42 using both western blot analysis and xMAP technology on the Luminex system. Using the western blot method, potential protein complexes are dissolved upon subjecting the proteins to sodium dodecyl sulphate and heating. The levels of Aβ1-42 measured by the Luminex system and the levels of Aβ1-42 quantified by western blot showed a high correlation, and the absolute values of Aβ1-42 also corresponded when comparing the two methods, so the methods seem to be comparable.
In conclusion, the disease-specific peptide pattern of Aβ1-37/38/39/40 and 42 was confirmed in the present study. However, despite an effect on both peripheral and CNS cholesterol synthesis after 12 months of treatment with simvastatin (20 mg/day), we found no effect on the Aβ peptide pattern. We suggest that clinical studies with extended treatment periods are performed, where different statins in higher dosages are used. We also believe that the pleiotropic effects of statins should be investigated in order to further elucidate the connection between AD and statin treatment.
References
Andreasen N, Blennow K (2002) Beta-amyloid (Abeta) protein in cerebrospinal fluid as a biomarker for Alzheimer’s disease. Peptides 23:1205–1214
Andreasen N, Hesse C, Davidsson P, Minthon L, Wallin A, Winblad B, Vanderstichele H, Vanmechelen E, Blennow K (1999) Cerebrospinal fluid beta-amyloid(1–42) in Alzheimer disease: differences between early- and late-onset Alzheimer disease and stability during the course of disease. Arch Neurol 56:673–680
Björkhem I, Lütjohann D, Diczfalusy U, Stahle L, Ahlborg G, Wahren J (1998) Cholesterol homeostasis in human brain: turnover of 24S-hydroxycholesterol and evidence for a cerebral origin of most of this oxysterol in the circulation. J Lipid Res 39:1594–1600
Black RA, Rauch CT, Kozlosky CJ, Peschon JJ, Slack JL, Wolfson MF, Castner BJ, Stocking KL, Reddy P, Srinivasan S, Nelson N, Boiani N, Schooley KA, Gerhart M, Davis R, Fitzner JN, Johnson RS, Paxton RJ, March CJ, Cerretti DP (1997) A metalloproteinase disintegrin that releases tumour-necrosis factor-alpha from cells. Nature 385:729–733
Blennow K, Hampel H (2003) CSF markers for incipient Alzheimer’s disease. Lancet Neurol 2:605–613
Blennow K, Ricksten A, Prince JA, Brookes AJ, Emahazion T, Wasslavik C, Bogdanovic N, Andreasen N, Batsman S, Marcusson J, Nagga K, Wallin A, Regland B, Olofsson H, Hesse C, Davidsson P, Minthon L, Jansson A, Palmqvist L, Rymo L (2000) No association between the alpha2-macroglobulin (A2M) deletion and Alzheimer’s disease, and no change in A2M mRNA, protein, or protein expression. J Neural Transm 107:1065–1079
Bodovitz S, Klein WL (1996) Cholesterol modulates alpha-secretase cleavage of amyloid precursor protein. J Biol Chem 271:4436–4440
Bunce D, Fratiglioni L, Small BJ, Winblad B, Backman L (2004) APOE and cognitive decline in preclinical Alzheimer disease and non-demented aging. Neurology 63:816–821
Buxbaum JD, Liu KN, Luo Y, Slack JL, Stocking KL, Peschon JJ, Johnson RS, Castner BJ, Cerretti DP, Black RA (1998) Evidence that tumor necrosis factor alpha converting enzyme is involved in regulated alpha-secretase cleavage of the Alzheimer amyloid protein precursor. J Biol Chem 273:27765–27767
Checler F (1995) Processing of the beta-amyloid precursor protein and its regulation in Alzheimer’s disease. J Neurochem 65:1431–1444
Chochina SV, Avdulov NA, Igbavboa U, Cleary JP, O’Hare EO, Wood WG (2001) Amyloid beta-peptide(1–40) increases neuronal membrane fluidity. Role of cholesterol and brain region. J Lipid Res 42:1292–1297
De Strooper B, Saftig P, Craessaerts K, Vanderstichele H, Guhde G, Annaert W, Von Figura K, Van Leuven F (1998) Deficiency of presenilin-1 inhibits the normal cleavage of amyloid precursor protein. Nature 391:387–390
Farzan M, Schnitzler CE, Vasilieva N, Leung D, Choe H (2000) BACE2, a beta-secretase homolog, cleaves at the beta site and within the amyloid-beta region of the amyloid-beta precursor protein. Proc Natl Acad Sci USA 97:9712–9717
Fassbender K, Simons M, Bergmann C, Stroick M, Lütjohann D, Keller P, Runz H, Kühl S, Bertsch T, von Bergmann K, Hennerici M, Beyreuther K, Hartmann T (2001) Simvastatin strongly reduces levels of Alzheimer’s disease beta-amyloid peptides Abeta 42 and Abeta 40 in vitro and in vivo. Proc Natl Acad Sci USA 98:5856–5861
Fassbender K, Stroick M, Bertsch T, Ragoschke A, Kuehl S, Walter S, Walter J, Brechtel K, Muehlhauser F, Von Bergmann K, Lütjohann D (2002) Effects of statins on human cerebral cholesterol metabolism and secretion of Alzheimer amyloid peptide. Neurology 59:1257–1258
Folstein MF, Folstein SE, McHugh PR (1975) “Mini-mental state”: a practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 12:189–198
Frears ER, Stephens DJ, Walters CE, Davies H, Austen BM (1999) The role of cholesterol in the biosynthesis of beta-amyloid. Neuroreport 10:1699–1705
Friedewald WT, Levy RI, Fredrickson DS (1972) Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem 18:499–502
Friedhoff LT, Cullen EI, Geoghagen NS, Buxbaum JD (2001) Treatment with controlled-release lovastatin decreases serum concentrations of human beta-amyloid (A beta) peptide. Int J Neuropsychopharmacol 4:127–130
Hartmann T, Bieger SC, Bruhl B, Tienari PJ, Ida N, Allsop D, Roberts GW, Masters CL, Dotti CG, Unsicker K, Beyreuther K (1997) Distinct sites of intracellular production for Alzheimer’s disease A beta40/42 amyloid peptides. Nat Med 3:1016–1020
Höglund K, Wiklund O, Vanderstichele H, Eikenberg O, Vanmechelen E, Blennow K (2004) Plasma levels of beta-amyloid(1–40), beta-amyloid(1–42), and total beta-amyloid remain unaffected in adult patients with hypercholesterolemia after treatment with statins. Arch Neurol 61:333–337
Höglund K, Thelen K, Syversen S et al (2005) The effect of simvastatin treatment on the amyloid precursor protein in patients with Alzheimer’s disease. Dement Geriatr Cogn Disord 19:256–265
Ida N, Hartmann T, Pantel J, Schroder J, Zerfass R, Forstl H, Sandbrink R, Masters CL, Beyreuther K (1996) Analysis of heterogeneous A4 peptides in human cerebrospinal fluid and blood by a newly developed sensitive western blot assay. J Biol Chem 271:22908–22914
Ishii K, Tokuda T, Matsushima T, Miya F, Shoji S, Ikeda S, Tamaoka A (2003) Pravastatin at 10 mg/day does not decrease plasma levels of either amyloid-beta (Abeta) 40 or Abeta 42 in humans. Neurosci Lett 350:161–164
Iwatsubo T, Mann DM, Odaka A, Suzuki N, Ihara Y (1995) Amyloid beta protein (A beta) deposition: A beta 42(43) precedes A beta 40 in Down syndrome. Ann Neurol 37:294–299
Jick H, Zornberg GL, Jick SS, Seshadri S, Drachman DA (2000) Statins and the risk of dementia. Lancet 356:1627–1631
Kirsch C, Eckert GP, Mueller WE (2003) Statin effects on cholesterol micro-domains in brain plasma membranes. Biochem Pharmacol 65:843–856
Koike H, Tomioka S, Sorimachi H, Saido TC, Maruyama K, Okuyama A, Fujisawa-Sehara A, Ohno S, Suzuki K, Ishiura S (1999) Membrane-anchored metalloprotease MDC9 has an alpha-secretase activity responsible for processing the amyloid precursor protein. Biochem J 343(Pt 2):371–375
Kojro E, Gimpl G, Lammich S, Marz W, Fahrenholz F (2001) Low cholesterol stimulates the nonamyloidogenic pathway by its effect on the alpha-secretase ADAM 10. Proc Natl Acad Sci USA 98:5815–5820
Lammich S, Kojro E, Postina R, Gilbert S, Pfeiffer R, Jasionowski M, Haass C, Fahrenholz F (1999) Constitutive and regulated alpha-secretase cleavage of Alzheimer’s amyloid precursor protein by a disintegrin metalloprotease. Proc Natl Acad Sci USA 96:3922–3927
Lewczuk P, Esselmann H, Meyer M, Wollscheid V, Neumann M, Otto M, Maler JM, Ruther E, Kornhuber J, Wiltfang J (2003) The amyloid-beta (Abeta) peptide pattern in cerebrospinal fluid in Alzheimer’s disease: evidence of a novel carboxyterminally elongated Abeta peptide. Rapid Commun Mass Spectrom 17:1291–1296
Lue LF, Kuo YM, Roher AE, Brachova L, Shen Y, Sue L, Beach T, Kurth JH, Rydel RE, Rogers J (1999) Soluble amyloid beta peptide concentration as a predictor of synaptic change in Alzheimer’s disease. Am J Pathol 155:853–862
Mason RP, Jacob RF, Walter MF, Mason PE, Avdulov NA, Chochina SV, Igbavboa U, Wood WG (1999) Distribution and fluidizing action of soluble and aggregated amyloid beta-peptide in rat synaptic plasma membranes. J Biol Chem 274:18801–18807
McKhann G, Drachman D, Folstein M, Katzman R, Price D, Stadlan EM (1984) Clinical diagnosis of Alzheimer’s disease: report of the NINCDS-ADRDA Work Group under the auspices of Department of Health and Human Services Task Force on Alzheimer’s Disease. Neurology 34:939–944
McLean CA, Cherny RA, Fraser FW, Fuller SJ, Smith MJ, Beyreuther K, Bush AI, Masters CL (1999) Soluble pool of Abeta amyloid as a determinant of severity of neurodegeneration in Alzheimer’s disease. Ann Neurol 46:860–866
Michikawa M, Gong JS, Fan QW, Sawamura N, Yanagisawa K (2001) A novel action of Alzheimer’s amyloid beta-protein (Abeta): oligomeric Abeta promotes lipid release. J Neurosci 21:7226–7235
Mills J, Reiner PB (1999) Regulation of amyloid precursor protein cleavage. J Neurochem 72:443–460
Mohs RC, Cohen L (1988) Alzheimer’s Disease Assessment Scale (ADAS). Psychopharmacol Bull 24:627–628
Moss ML, Jin SL, Milla ME, Bickett DM, Burkhart W, Carter HL, Chen WJ, Clay WC, Didsbury JR, Hassler D, Hoffman CR, Kost TA, Lambert MH, Leesnitzer MA, McCauley P, McGeehan G, Mitchell J, Moyer M, Pahel G, Rocque W, Overton LK, Schoenen F, Seaton T, Su JL, Becherer JD et al (1997) Cloning of a disintegrin metalloproteinase that processes precursor tumour-necrosis factor-alpha. Nature 385:733–736
Ohlsson A, Vanderstichele H, Andreasen N, Demeyer G, Holmberg G, Rosengren L, Vanmechelen E, Blennow K (2005) Simultaneous measurement of beta-amyloid(1–42), total tau and phosphorylated tau (Thr181) in cerebrospinal fluid using the xMAP technology. Clin Chem 51(2):336–345
Park IH, Hwang EM, Hong HS, Boo JH, Oh SS, Lee J, Jung MW, Bang OY, Kim SU, Mook-Jung I (2003) Lovastatin enhances Abeta production and senile plaque deposition in female Tg2576 mice. Neurobiol Aging 24:637–643
Rockwood K, Kirkland S, Hogan DB, MacKnight C, Merry H, Verreault R, Wolfson C, McDowell I (2002) Use of lipid-lowering agents, indication bias, and the risk of dementia in community-dwelling elderly people. Arch Neurol 59:223–227
Runz H, Rietdorf J, Tomic I, de Bernard M, Beyreuther K, Pepperkok R, Hartmann T (2002) Inhibition of intracellular cholesterol transport alters presenilin localization and amyloid precursor protein processing in neuronal cells. J Neurosci 22:1679–1689
Scheuner D, Eckman C, Jensen M, Song X, Citron M, Suzuki N, Bird TD, Hardy J, Hutton M, Kukull W, Larson E, Levy-Lahad E, Viitanen M, Peskind E, Poorkaj P, Schellenberg G, Tanzi R, Wasco W, Lannfelt L, Selkoe D, Younkin S (1996) Secreted amyloid beta-protein similar to that in the senile plaques of Alzheimer’s disease is increased in vivo by the presenilin 1 and 2 and APP mutations linked to familial Alzheimer’s disease. Nat Med 2:864–870
Shi XP, Tugusheva K, Bruce JE, Lucka A, Wu GX, Chen-Dodson E, Price E, Li Y, Xu M, Huang Q, Sardana MK, Hazuda DJ (2003) Beta-secretase cleavage at amino acid residue 34 in the amyloid beta peptide is dependent upon gamma-secretase activity. J Biol Chem 278:21286–21294
Simons M, Keller P, De Strooper B, Beyreuther K, Dotti CG, Simons K (1998) Cholesterol depletion inhibits the generation of beta-amyloid in hippocampal neurons. Proc Natl Acad Sci USA 95:6460–6464
Simons M, Schwarzler F, Lütjohann D, von Bergmann K, Beyreuther K, Dichgans J, Wormstall H, Hartmann T, Schulz JB (2002) Treatment with simvastatin in normocholesterolemic patients with Alzheimer’s disease: a 26-week randomized, placebo-controlled, double-blind trial. Ann Neurol 52:346–350
Sinha S, Anderson JP, Barbour R, Basi GS, Caccavello R, Davis D, Doan M, Dovey HF, Frigon N, Hong J, Jacobson-Croak K, Jewett N, Keim P, Knops J, Lieberburg I, Power M, Tan H, Tatsuno G, Tung J, Schenk D, Seubert P, Suomensaari SM, Wang S, Walker D, John V et al (1999) Purification and cloning of amyloid precursor protein beta-secretase from human brain. Nature 402:537–540
Sjögren M, Gustafsson K, Syversen S, Olsson A, Edman A, Davidsson P, Wallin A, Blennow K (2003) Treatment with Simvastatin in patients with Alzheimer’s disease lowers both alpha- and beta-cleaved amyloid precursor protein. Dement Geriatr Cogn Disord 16:25–30
Strittmatter WJ, Saunders AM, Schmechel D, Pericak-Vance M, Enghild J, Salvesen GS, Roses AD (1993a) Apolipoprotein E: high-avidity binding to beta-amyloid and increased frequency of type 4 allele in late-onset familial Alzheimer disease. Proc Natl Acad Sci USA 90:1977–1981
Strittmatter WJ, Weisgraber KH, Huang DY, Dong LM, Salvesen GS, Pericak-Vance M, Schmechel D, Saunders AM, Goldgaber D, Roses AD (1993b) Binding of human apolipoprotein E to synthetic amyloid beta peptide: isoform-specific effects and implications for late-onset Alzheimer disease. Proc Natl Acad Sci USA 90:8098–8102
Tokuda T, Tamaoka A, Matsuno S, Sakurai S, Shimada H, Morita H, Ikeda S (2001) Plasma levels of amyloid beta proteins did not differ between subjects taking statins and those not taking statins. Ann Neurol 49:546–547
Tomita S, Kirino Y, Suzuki T (1998) A basic amino acid in the cytoplasmic domain of Alzheimer’s beta-amyloid precursor protein (APP) is essential for cleavage of APP at the alpha-site. J Biol Chem 273:19304–19310
Urmoneit B, Turner J, Dyrks T (1998) Cationic lipids (lipofectamine) and disturbance of cellular cholesterol and sphingomyelin distribution modulates gamma-secretase activity within amyloid precursor protein in vitro. Prostaglandins Other Lipid Mediat 55:331–343
Vassar R, Bennett BD, Babu-Khan S, Kahn S, Mendiaz EA, Denis P, Teplow DB, Ross S, Amarante P, Loeloff R, Luo Y, Fisher S, Fuller J, Edenson S, Lile J, Jarosinski MA, Biere AL, Curran E, Burgess T, Louis JC, Collins F, Treanor J, Rogers G, Citron M (1999) Beta-secretase cleavage of Alzheimer’s amyloid precursor protein by the transmembrane aspartic protease BACE. Science 286:735–741
Wahrle S, Das P, Nyborg AC, McLendon C, Shoji M, Kawarabayashi T, Younkin LH, Younkin SG, Golde TE (2002) Cholesterol-dependent gamma-secretase activity in buoyant cholesterol-rich membrane microdomains. Neurobiol Dis 9:11–23
Weidemann A, Eggert S, Reinhard FB, Vogel M, Paliga K, Baier G, Masters CL, Beyreuther K, Evin G (2002) A novel epsilon-cleavage within the transmembrane domain of the Alzheimer amyloid precursor protein demonstrates homology with Notch processing. Biochemistry 41:2825–2835
Wiltfang J, Arold N, Neuhoff V (1991) A new multiphasic buffer system for sodium dodecyl sulfate-polyacrylamide gel electrophoresis of proteins and peptides with molecular masses 100,000–1,000, and their detection with picomolar sensitivity. Electrophoresis 12:352–366
Wiltfang J, Smirnov A, Schnierstein B, Kelemen G, Matthies U, Klafki HW, Staufenbiel M, Huther G, Ruther E, Kornhuber J (1997) Improved electrophoretic separation and immunoblotting of beta-amyloid (A beta) peptides 1–40, 1–42, and 1–43. Electrophoresis 18:527–532
Wiltfang J, Esselmann H, Cupers P, Neumann M, Kretzschmar H, Beyermann M, Schleuder D, Jahn H, Ruther E, Kornhuber J, Annaert W, De Strooper B, Saftig P (2001) Elevation of beta-amyloid peptide 2–42 in sporadic and familial Alzheimer’s disease and its generation in PS1 knockout cells. J Biol Chem 276:42645–42657
Wiltfang J, Esselmann H, Bibl M, Smirnov A, Otto M, Paul S, Schmidt B, Klafki HW, Maler M, Dyrks T, Bienert M, Beyermann M, Ruther E, Kornhuber J (2002) Highly conserved and disease-specific patterns of carboxyterminally truncated Abeta peptides 1–37/38/39 in addition to 1–40/42 in Alzheimer’s disease and in patients with chronic neuroinflammation. J Neurochem 81:481–496
Wisniewski KE, Dalton AJ, McLachlan C, Wen GY, Wisniewski HM (1985) Alzheimer’s disease in Down’s syndrome: clinicopathologic studies. Neurology 35:957–961
Wolozin B, Kellman W, Ruosseau P, Celesia GG, Siegel G (2000) Decreased prevalence of Alzheimer disease associated with 3-hydroxy-3-methyglutaryl coenzyme A reductase inhibitors. Arch Neurol 57:1439–1443
Yaffe K, Barrett-Connor E, Lin F, Grady D (2002) Serum lipoprotein levels, statin use, and cognitive function in older women. Arch Neurol 59:378–384
Yamaguchi H, Maat-Schieman ML, van Duinen SG, Prins FA, Neeskens P, Natte R, Roos RA (2000) Amyloid beta protein (Abeta) starts to deposit as plasma membrane-bound form in diffuse plaques of brains from hereditary cerebral hemorrhage with amyloidosis-Dutch type, Alzheimer disease and nondemented aged subjects. J Neuropathol Exp Neurol 59:723–732
Yan R, Bienkowski MJ, Shuck ME, Miao H, Tory MC, Pauley AM, Brashier JR, Stratman NC, Mathews WR, Buhl AE, Carter DB, Tomasselli AG, Parodi LA, Heinrikson RL, Gurney ME (1999) Membrane-anchored aspartyl protease with Alzheimer’s disease beta-secretase activity. Nature 402:533–537
Yu G, Nishimura M, Arawaka S, Levitan D, Zhang L, Tandon A, Song YQ, Rogaeva E, Chen F, Kawarai T, Supala A, Levesque L, Yu H, Yang DS, Holmes E, Milman P, Liang Y, Zhang DM, Xu DH, Sato C, Rogaev E, Smith M, Janus C, Zhang Y, Aebersold R, Farrer LS, Sorbi S, Bruni A, Fraser P, St George-Hyslop P (2000) Nicastrin modulates presenilin-mediated notch/glp-1 signal transduction and betaAPP processing. Nature 407:48–54
Acknowledgements
This study has been supported by the Swedish Medical Research Council (projects # 12103,09946, 14981 and 14002); Alzheimerfonden, Lund, Sweden; Åhlèns Stiftelse, Stockholm, Sweden; Stohnes Stiftelse, Stockholm, Sweden; Pfannenstills Stiftelse, Sweden, Stiftelsen för Gamla Tjänarinnor, Stockholm, Sweden and BMBF Funded Competence Net Dementia, Germany. We are very grateful to Sabine Paul for skilful technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Höglund, K., Syversen, S., Lewczuk, P. et al. Statin treatment and a disease-specific pattern of β-amyloid peptides in Alzheimer’s disease. Exp Brain Res 164, 205–214 (2005). https://doi.org/10.1007/s00221-005-2243-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-005-2243-8