Abstract
Primary gaze fixation is never perfectly stable but can be interrupted by involuntary, conjugate saccadic intrusions (SI). SI have a high prevalence in the normal population and are characterised by a horizontal fast eye movement away from the desired eye position, followed, after a variable duration, by a return saccade or drift. Amplitudes are usually below 1° and they often exhibit a directional bias. The aim of the present study was to investigate the aetiology of SI in relation to saccadic behaviour. It was hypothesised that if SI resulted from deficits in the saccadic system (i.e. reduced inhibitory mechanisms), changes in voluntary saccade behaviour may be apparent and related to SI frequency. To examine this, synchrony (no gap), gap, overlap and antisaccade tasks were conducted on ten normal subjects. No significant correlations were found between SI frequency and voluntary saccade latencies, the percentage of express saccades, or the percentage of antisaccade errors. In addition, no significant correlations were found between SI directional biases and saccade latency directional biases, express saccade biases or antisaccade error biases. These results suggest that an underlying alteration to saccadic behaviour is unlikely to be involved in SI production, and that the SI command signal may arise from the influence of attention on an intact saccadic system. Specifically, descending corticofugal signals relating to attention level and orientation may alter the balance between fixation and saccade generation, so determining SI characteristics.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Fixation to a stationary target is never perfectly stable, but is frequently interrupted by physiological, involuntary eye movements (Carpenter 1988; Ditchburn 1973). These take the form of disconjugate slow micro-drifts (1–3′ arc), small conjugate microsaccades (5–10′ arc) and disconjugate micro-tremors (15” arc). A further class of involuntary eye movements found during fixation are saccadic intrusions (SI). They are characterised by an initial fast eye movement away from the desired eye position, followed, after a variable duration, by a return saccade or drift.
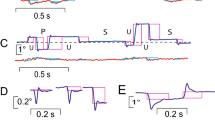
Physiological SI have a high prevalence in the normal population, and in their study Abadi and Gowen (2004) described four identifiable types: monophasic square wave intrusions (MSWI), biphasic square wave intrusions (BSWI), single saccadic pulses (SSP) and double saccadic pulses (DSP) (Fig. 1). They were found to be almost always conjugate and horizontal with typical amplitude and frequency means and ranges of 0.6±0.5°, 0.1–4.1° and 18.0±14.3 per minute, 0.6–54.8 per minute, respectively, and their characteristics changed little between binocular and monocular viewing (Abadi and Gowen 2004). SI often displayed a directional bias, defined as the side (right or left) to which the majority of SI are directed. The commonest bias was rightward (54%), with only 15% being leftward and the remainder showing no bias.
The aetiology of physiological SI still remains unclear. Possible suggestions include omnipause neuron dysfunction (Zee and Robinson 1979), a spurious supranuclear error signal (Doslak et al 1983) and enlarged microsaccades (Feldon and Langston 1977; Ohtsuka et al 1986). Abadi and Gowen (2004) have shown that, in regards to physiological SI, omnipause neuron dysfunction is unlikely, as SI displayed normal (not raised) peak velocities, indicating that they represent a planned rather than interrupted saccade. The contribution of other areas of the saccadic system towards SI production has not previously been tested.
The saccadic system comprises extensive cortical and subcortical networks, which function in a complementary manner to provide a balance between saccade generation and periods of fixation (for a review see Pierrot-Deseilligny et al 2004). The frontal eye fields are thought to be involved in fixation disengagement and triggering of intentional saccades to visual or remembered target locations (Dias and Bruce 1994; Friedman et al 1997; Gaymard et al 1999; Sommer and Wurtz 2000), while the dorsolateral prefrontal cortex is involved in saccade inhibition, short term spatial memory and prediction (Gaymard et al 2003; Guitton, Buchtel and Douglas 1985; Pierrot-Deseilligny et al 1991, 2003). The parietal eye fields control reflex saccade triggering made to the sudden appearance of a target (Braun et al 1992; Pierrot-Deseilligny et al 1991; Powell et al 1999), and the supplementary eye fields are important in triggering sequences of voluntary saccades to internally defined goals such as memory-guided saccades (Amador et al 2004; Gaymard et al 1993).
These cortical areas project to the superior colliculus (SC), which is central to both fixation control and saccade generation. Here, cells have been identified in the rostral SC (in the region corresponding to the foveal portion of the retinotopic map) that discharge during fixation, and pause or decrease their activity during saccades (Munoz and Wurtz 1992, 1993a). Together with the saccade-related burst and build-up cells of the caudal SC, these rostral fixation cells are thought to provide a mutually inhibitory network with the omnipause neurons and excitatory burst neurons of the brainstem (Gandhi and Keller 1999; Munoz and Wurtz 1993a, 1993b, 1995a, 1995b). During fixation, rostral fixation cells prevent saccade production by inhibiting the caudal SC saccade cells and the brainstem excitatory burst neurons and by exciting the omnipause neurons. When a saccade is required, the caudal SC saccade cells inhibit the rostral fixation cells and the omnipause neurons and excite the brainstem excitatory burst neurons. The basal ganglia also provide inhibitory modulation over the SC and can suppress reflexive and volitional saccades generated by the SC (Hikosaka and Wurtz 1985).
Variation in the activity of any of the aforementioned saccadic areas can present as latency changes and difficulty in initiating or suppressing saccades. Increased distractibility and the production of unwanted saccades are a feature of lesions to the dorsolateral prefrontal cortex (Gaymard et al 2003; Guitton et al 1985; Pierrot-Deseilligny et al 1991) and the basal ganglia (Hikosaka and Wurtz 1985; Lasker et al 1987). A reduction in saccade latency together with a higher number of express saccades and antisaccade errors accompanies increased frontal eye field activity due to an increase in its excitatory drive to the burst and build-up cells of the SC (Everling and Munoz 2000). In contrast, it has been observed that neurons in the supplementary eye field fire more vigorously before correct antisaccades than regular saccades, and that their activity is less if an error is made on the antisaccade task, suggesting that they may influence the SC fixation cells (Amador et al 2004; Everling et al 1998b; Everling and Munoz 2000; Schlag-Rey et al 1997). Inactivation of SC fixation cells causes a reduction in the ability to fixate and an inability to suppress unwanted saccades (Munoz and Wurtz 1992, 1993b). It is possible that SI could be the consequence of an increase or decrease in activity in areas such as the dorsolateral prefrontal cortex, frontal eye field, supplementary eye field, basal ganglia or SC, that upsets the balance between fixation and saccade generation.
It was hypothesised that if SI resulted from altered activity of these saccade-related areas, visually-guided saccades may display different latency characteristics that correlate with SI frequency. For example, if SI were caused by a reduction in the threshold required to produce a saccade, this may predispose the system towards saccade generation and shorter latency voluntary saccades. Subjects with higher frequencies of SI would be more likely to display shorter saccade latencies, a greater number of express saccades (particularly on the overlap task when the fixation system should be engaged) and a higher number of antisaccade errors. In addition, we investigated whether voluntary saccade biases correlated with SI directional biases. Asymmetry in the activity between the cortical hemispheres may lead to shorter voluntary saccade latencies (and a greater number of SI) in one direction than the other. Our results show no significant relationship between SI metrics and voluntary saccadic behaviour, suggesting that SI do not arise from modified properties of the saccadic system.
Materials and methods
Subjects
Ten normal control subjects participated in the experiments, prior to which informed consent was obtained. Out of the ten controls, five were naïve in respect to performing experiments and the nature of the tasks. Subject age range, mean and standard deviation were 21–54 years and 33±13 years respectively. The subjects had no previous or current history of ocular disease, general health problems or medication that had been linked to any ocular complications. Corrected visual acuity was 0.2 LogMAR or better in each eye and stereoacuity (TNO) was 60″ arc or better. All studies were approved by a university ethics committee and performed in accordance with the Declaration of Helsinki.
Eye movement recording and instrumentation
Binocular horizontal eye movements were recorded using an IRIS 6500 infrared limbal tracker (Skalar Medical, Delft, The Netherlands). The analogue output was filtered through a passive 100 Hz low-pass filter, digitised to 12-bit resolution and then sampled at intervals of 5 ms (200 Hz). The system was linear to ±20° and had a resolution of >5′ arc. Each subject’s head was restrained using chin and cheek rests. Head movements were calculated to be less than 6′ arc in amplitude.
The targets were back-projected onto a 210×82 cm screen and viewed from 114 cm, giving a field of 105° (horizontal) × 41° (vertical). Screen luminance was 0.01 cd/m2. Test targets consisted of two illuminated concentric circles containing an inner (0.3°) and outer cross (3.02°). Target luminance was 0.04 cd/m2, giving a contrast of 75%. Testing was carried out in a near-dark room unless stated otherwise. Subject’s eye movements were calibrated by moving the target sinusoidally at 0.32 Hz over a horizontal range of ±10°.
In the overlap paradigm, one target was back-projected and the other target was front-projected. Both fixation and saccade targets were of equal size and luminance. Liquid crystal shutters attached to the back-projector were used to obscure the target during the gap task.
Experimental tasks
Fixation task
Ten subjects performed at least 2 × 50 s sets of binocular primary fixation, randomly at different times during their visits. Instructions to subjects were “to keep their eyes as still as possible whilst looking at the centre of the cross”. SI frequency, defined as the frequency per minute of all four types of SI, was calculated. The resolution of the recording system limited the inclusion of SI to those above 0.09°.
Saccade tasks
Synchrony (no gap) paradigm
Following a 2 s period of primary fixation, the target jumped pseudo-randomly either to the right or left by 4°. At the end of each rightward or leftward jump, the target returned to the primary position within 1 s. In all cases, extinction of the central fixation target and presentation of the peripheral target were synchronous (no gap). Six blocks of saccades, each lasting 50 s, were repeated twice, resulting in a total of 192 saccades, 96 in each direction (rightward or leftward). Subjects were instructed to follow the target as quickly and accurately as possible, but not to attempt to predict the motion of the target.
Gap, overlap and synchrony antisaccade paradigms
The same six saccade sets used for the synchrony task were also employed for gap, antisaccade and overlap tasks. During the gap task, the central fixation target was extinguished 200 ms prior to the presentation of the peripheral target (Fig. 2a). During the overlap task the central fixation target remained illuminated while the peripheral target jumped to the right or left (Fig. 2b). In both the gap and overlap tasks, subjects were instructed to follow the target as quickly and as accurately as possible, but not to attempt to predict its direction. The synchrony antisaccade task was identical to the synchrony task, except the subject was instructed to look as quickly as possible and with similar amplitude in the direction opposite to the target movement. In all tasks the target returned to primary position after 1 second. A total of 192 saccades were performed, 96 in each direction.
Saccade tasks were performed in a random order. A target amplitude of 4° was chosen as it has been reported that a maximum number of express saccades are generated at this amplitude (Biscaldi et al 1996; Weber et al 1992). A 200 ms gap was used as a higher number of express saccades have been documented at this gap interval. This is hypothesised to be due to SC fixation cell activity being at a minimum after 200 ms (Dorris and Munoz 1995; Dorris et al 1997; Fischer et al 1995; Fischer and Weber 1997; Mayfrank et al 1986; Suzuki and Hirai 2000).
Data and statistical analysis
Data recorded from the right eye only was used for analysis. Modified software packages (Visual Basic and Microsoft Excel) were used to detect voluntary saccades. Saccade start and end points were determined by a 40°/s per cut-off criterion, and the calculated amplitude, latency, peak velocity and duration stored to disc. Parameters measured included saccade latency (from target presentation), percentage of express saccades, percentage of antisaccade errors and percentage of those errors corrected. Artifacts such as blinks and drift were discarded (<3% of dataset). In order to exclude non-visually driven saccades, anticipatory saccades (<80 ms) (Fischer et al 1993b; Wenban-Smith and Findlay 1991) and saccades that were two standard deviations above the mean latency were removed from the analysis. Express saccades were defined as having latencies between 80–134 ms (Fisher et al 1997).
SI were detected manually by scanning the position, velocity and acceleration data for peaks indicative of saccades. Start and end positions of saccades were detected by visual observation employing an amplitude-dependent velocity criterion (>10°/s). This was due to the amplitude of SI often being less than 1°, with peak velocities rarely above 40°/s.
The Student’s two-tailed t-test was used to test significance (P<0.05) of saccade latencies and biases. Z-scores (P<0.05) were used to calculate significant SI directional biases and significant difference between two proportions. Relationships were tested using Pearson correlation coefficient.
Results
Saccadic intrusion characteristics
Four different SI types were identified. Each has been described previously (Abadi and Gowen 2004) (Fig 1). MSWI were the most common type in all subjects, except in subjects 3, 5 and 9 where DSP were predominant and subject 1 who exhibited a higher number of BSWI (Table 1, column 4). The frequencies and directional biases of the combined and predominant SI in the ten subjects are shown in the second, third, fourth and fifth columns of Table 1. The mean combined SI amplitude, standard deviation and range in each of the ten subjects was 0.5±0.4° and 0.1–4.1° respectively. SI mean frequency, standard deviation and range were 26.8±17.5 and 8.9–54.8 per minute respectively. Four subjects displayed a significant rightward SI bias, five no significant bias, and one a significant leftward bias (P<0.05).
Voluntary saccade characteristics
Table 2 displays the voluntary saccade latencies from the right eyes of the ten control subjects and group means for each of the four different saccade paradigms. Group mean latencies significantly (P<0.01) decreased and increased during the gap and overlap task respectively in comparison to the synchrony task, and were significantly (P<0.01) longer in the antisaccade task than in the overlap task. All subjects, except 8, showed a significant (P<0.05) decrease in latency in at least one direction with the gap paradigm (column 6) compared to the synchrony task (column 3), and all exhibited a significant (P<0.05) increase in latency with the overlap (column 9) when compared to the synchrony task and a significant (P<0.05) increase in latency during the antisaccade task (column 12) when compared to the overlap task.
Table 2 displays the percentage of express saccades produced for each subject and the group means for each saccade paradigm (synchrony/gap/overlap) (columns 4, 7, 10). As a group, express saccade percentage non-significantly increased during the gap paradigm and significantly (P<0.05) decreased during the overlap paradigm when compared to the synchrony task. All but one subject (5) exhibited express saccades. When contrasting the synchrony and gap paradigm, all subjects except 1, 5, 8 and 10 produced significantly more express saccades in the latter task (P<0.05) and all except 5, 7, 8, 9 and 10 produced significantly more express saccades during the synchrony in comparison with the overlap task (P<0.05). However, no subjects produced significantly more express saccades on the overlap task than the synchrony and gap tasks (P<0.05) but subject 1 (Z=8.89, P≥0.05) and 10 (Z=9.09, P≥0.05) demonstrated significantly more express saccades during the synchrony task as opposed to the gap task.
Errors (saccades directed opposite to the target direction) were minimal on synchrony, gap and overlap tasks, although significantly more were produced during the gap and overlap task than on the prosaccade task (P<0.05) (Table 2, columns 5, 8, 11). All subjects exhibited varying percentages of antisaccade errors (Table 2, column 13).
Most subjects displayed a voluntary saccade latency bias for all four tasks. A significant difference (P<0.05) between rightward and leftward directed saccades is indicated by the asterisk in Table 2 and documented in greater detail in Table 1 (columns 6–9). Subjects also showed an express saccade (Table 1, columns 10–12) and/or an antisaccade error bias (Table 1, column 13).
Saccade latency and saccadic intrusions
Average rightward and leftward saccade latencies for each of the four saccade tasks were plotted against frequency of SI for each subject (Fig. 3). No significant correlation between SI frequency and saccade latency was found for any of the saccade tasks (P>0.05).
Three subjects (1, 2 and 6) had consistently shorter saccade latencies across all tasks. From Table 1 (column 2) it can be seen that only subject 6 had a high SI frequency. Six subjects did not show a significant gap effect for one (subjects 1, 2, 4, 5, 10) or both (subject 8) directions (P<0.05). Subjects 1 (t=−7.89, P<0.001) and 8 (t=1.65, P<0.05) had significantly shorter saccade latencies in one direction for the synchrony task compared with the gap task. All these subjects exhibited different SI frequencies, and no consistent relationship between the direction of the absent gap effect and the SI directional bias was evident.
Express saccades and saccadic intrusions
The percentage of express saccades for each subject was compared to SI frequency for the synchrony, gap and overlap saccade tasks. No correlation was found (P>1.0).
Three subjects (2, 6 and 7) displayed express saccade percentages above 30% on the gap paradigm (Table 2, column 7) but no subjects exhibited more that 15% on the overlap task (Table 2, column 10). Coincidently, subjects 6 and 7 had a relatively high SI frequency of 39 and 33 per minute respectively, whereas subject 2 had a low frequency of 9 per minute (Table 1, column 2). However, subject 5 did not produce express saccades for any of the paradigms yet exhibited a high frequency of SI (55 per min). Subjects 1 (Z=8.89, P<0.05) and 10 (Z=9.09, P<0.05) produced more express saccades in the synchrony task than in the gap paradigm. This was to the same side as that of the absent gap effect. Subject 1 had a low SI frequency and the SI directional bias did not match the express saccade bias, whereas subject 10 demonstrated a high SI frequency and the SI directional bias corresponded with the express saccade bias.
Antisaccade errors and saccadic intrusions
No correlation was found (r=−0.52; P>1.0) between antisaccade errors and the frequency of SI. All subjects corrected the majority of their errors by making a further saccade towards the side opposite to the target (Table 2, column 14).
Whilst subjects 1, 2 and 9 produced the highest percentage of errors (Table 2, column 13), none of the three exhibited a high frequency of SI (Table 1, column 2). Subjects 1 and 2 were the youngest members of the study.
Saccade latency directional bias and saccadic intrusion directional bias
The SI directional bias was compared to voluntary saccade latency bias, with the view that the SI directional bias may correspond to the direction of the shorter latency saccades. No subject showed a consistent relationship between SI directional bias and voluntary saccade latency bias for all four saccade paradigms (Table 1, columns 3, 6–9). Indeed, latency bias was not consistent across the four tasks.
Express saccade bias was also compared to SI bias by separately plotting the percentage of rightward express saccades from the synchrony and gap tasks against the percentage of rightward SI for each subject (Fig. 4a,b). The percentage of rightward express saccades from combined synchrony, gap and overlap tasks was also plotted against the percentage of rightward SI for each subject (Fig. 4c). No correlations were found, the r values being −0.46, −0.26 and −0.27 (P>0.05) for the synchrony, gap and combined synchrony, gap and overlap tasks respectively.
Express saccade biases of all subjects are shown in Table 1 (columns 10–12). As with voluntary saccade latency biases, express saccade bias was not consistent for the synchrony, gap or overlap tasks. Furthermore, biases were more apparent in the gap and synchrony tasks where 8/10 and 4/10 subjects respectively displayed a bias, in contrast to the overlap task where 1/10 subjects demonstrated a bias.
Table 1 compares the SI directional bias (column 3) with the antisaccade error bias (column 13). Rightward errors indicate that the target jumped right, and instead of making a saccade to the left the subject made a saccade to the right. SI and antisaccade error bias corresponded in two subjects only.
Comparisons with predominant SI type
The same comparisons described above were repeated but using the predominant SI type (Table 1, columns 4–5). The predominant SI type was defined as the most common SI type present for each subject. Once again, no consistent relationships were apparent.
Discussion
Voluntary saccades and SI characteristics
The present experiments tested the hypothesis that SI may be a consequence of altered activity of the saccadic system, specifically an imbalance that favours saccade initiation as opposed to saccade inhibition. Four saccade tasks—synchrony, gap, overlap and antisaccades—were used to explore the relationship between volitional saccade latencies, express saccades and antisaccade errors with SI characteristics.
SI frequency was not found to be correlated with voluntary saccade latencies, express saccade frequency or antisaccade error frequency, suggesting that the baseline activity of the saccadic system was not predisposed towards SI generation. Furthermore, no relationship was observed between SI directional bias and voluntary saccade latency, express saccade and antisaccade directional biases. Indeed the intra-subject voluntary saccade latency biases were not the same across all four tasks and did not match the express saccade or antisaccade error bias.
The examination of individual subject differences further discourages any link between altered saccadic activity and SI. Of the three subjects (1, 2 and 6) who displayed the shortest saccade latencies across all tasks, high numbers of SI were recorded only in one of the three subjects (6). These results do not support the hypothesis that shorter saccade latencies are associated with the production of SI. It has been previously suggested that less then 30% and 15% express saccades on the gap and overlap paradigms respectively is within normal limits (Fischer et al 1993a). Three subjects (2, 6, 7) produced greater than 30% express saccades on the gap task; subjects 6 and 7 had a relatively high SI frequency of 39 and 33 per minute, respectively, whereas, subject 2 had a low frequency of 9 per minute (Table 1). However, two subjects (5 and 10) exhibited exceptionally high frequency rates of SI, but few express saccades on any of the tasks. Furthermore, none of the ten subjects exhibited greater than 8% express saccades on the overlap task, implying that the SC rostral fixation cells and their afferent and efferent projections were essentially functioning within normal limits as the presence of a fixation target prevented disengagement.
Ten subjects (1, 2, 4, 5, 8 and 10) did not exhibit a gap effect, suggesting that either the onset of the gap had not promoted fixation disengagement or that the fixation system was already in a state of disengagement. In the latter case, one would also expect shorter saccade latencies, a higher number of express saccades in synchrony, gap and particularly overlap tasks and a higher number of antisaccade errors. Of the six subjects, only two subjects (1 and 2) demonstrated a higher number of express and antisaccade errors and shorter overlap latency values (Table 2). An increased level of pre-stimulus (preparatory) activity in the SC build-up neurons has been associated with the generation of shorter latency saccades (Dorris et al 1997; Gomez et al 1994; Suzuki and Hirai 2000) and antisaccade errors (Everling et al 1998a). Express saccades result if the visual burst created by the appearance of the target coincides with the region of increased excitement in the build-up cells (Dorris et al 1997). An increase in saccade preparation would also reduce the activity of the SC fixation cells without the need for a gap due to mutually inhibitory connections between the fixation and saccade-related cells. That subjects 1 and 2 produced relatively few SI (11 and 9 per minute, respectively) argues against the view that reduced fixation cell activity and increased levels of preparatory activity give rise to SI. The observations that SI frequency did not increase during the saccade tasks in subjects 1 and 2 and that there did not appear to be an increase in SI during the gap period for any subjects provides additional support for this conclusion.
Three of the subjects without a gap effect (5, 8 and 10) had moderate to high SI frequencies but did not produce increased numbers of express saccades or antisaccade errors, suggesting that the gap did not disengage fixation in these subjects. It is possible that higher frequencies of SI interfere with voluntary saccade production, preventing faster latency saccade preparation. Although this cannot be conclusively ruled out, Fig. 3 shows that some subjects with lower SI frequencies have longer latencies than subjects with higher SI frequencies.
All 10 subjects displayed minimal antisaccade errors, and when they did occur were able to correct them. An error rate of approximately 14% has been reported in gap antisaccade tasks (Fisher and Weber 1992). As the current antisaccade task did not involve a gap, one would expect lower error rates. Only two subjects (1 and 9) exceeded this value, although one subject (2) also demonstrated a higher percentage of antisaccade errors than the other seven subjects. All three displayed low SI frequencies, challenging the theory that SI are caused by deficient inhibitory mechanisms. Two subjects (1 and 2) exhibited shorter saccade latencies and frequent express saccades, which, as discussed earlier, may be attributed to increased SC preparatory activity. One subject (9) who had few express saccades on any of the tasks, was one of the oldest of the group. Increased numbers of antisaccade errors have been reported in older age groups (>20% as opposed to 13% in younger subjects) (Fischer et al 1997).
Saccade latencies using the gap and overlap task have been shown to increase (by approximately 20–50 ms) between the ages of 21 and 55 years (Fischer et al 1997; Munoz et al 1998). To examine whether this may have masked any relationship between the SI frequency and saccade latency we compared subjects of similar ages who had different SI frequencies (subjects 9, 10 and 5, 4) and found their saccade latencies to be similar. Two subjects (6 and 7) with moderate frequencies of SI do have significantly (P<0.01) shorter latencies than an age-matched subject 8 who had fewer SI. These two subjects also have comparable latencies with younger subjects (1, 2 and 3). However, in a previous study it was found that SI frequency does not appear to be age-related (Abadi and Gowen 2004). This reinforces the view that there is no relationship between saccade latencies and SI frequency, since a greater number of SI would be expected in early adulthood (when saccade latencies are shorter) and fewer SI in the older population.
Directional biases
Directional right-left asymmetries in voluntary saccade tasks have been observed previously (Fischer et al 1997; Honda 2002; Munoz et al 1998; Weber and Fischer 1995). Weber and Fischer (1995) reported that asymmetries in express saccade frequency and saccadic latencies were more apparent when attention was disengaged (gap 200 ms). Results of the present study also indicate that latency asymmetries are more apparent during the gap task, although in agreement with Honda (2002), asymmetries in the synchrony and overlap tasks were also observed. The subjects used by Weber and Fischer (1995) were trained and this may have contributed to a reduction in the asymmetries. Weber and Fischer (1995) suggested that the process of attentional disengagement might have a different strength/time course for the right and left hemispheres, so when attention is disengaged, asymmetries become apparent. However, the fact that a gap effect can be produced by offset of an unattended target implies that the gap is more akin to warning effects or fixation cell offset then attention disengagement (Kingstone and Klein 1993; Walker et al 1995). This implies that activity associated with a gap (fixation cell disengagement, preparatory effects) does have asymmetrical properties, or that once alerted, attentional processes (other than disengagement) may then influence saccade latencies.
The finding that SI directional biases were uncorrelated with the voluntary saccade biases indicates that the underlying cause of SI is unlikely to be related to an inherent bias within the circuitry of the saccadic system. However, the observation that voluntary saccade latency biases are not consistent implies that there are likely to be a number of governing factors. For example, biases may depend on the particular task; for example, a static fixation task where no response is required may evoke more of a baseline bias than an active saccade task, where prior knowledge activates mechanisms involved in prediction and selection that override any baseline asymmetries. In such a case, SI and voluntary saccade asymmetries may be under the influence of the same system (attention) but represent different modes of that system.
Neural substrate of SI
Our studies suggest that an imbalance between saccade initiating and saccade inhibiting processes does not appear to be responsible for SI production. It is well established that interactions between the SC fixation, build-up and burst cells determine saccade initiation and latency (Gandhi and Keller 1999; Munoz and Wurtz 1993a, 1993b, 1995a, 1995b) with higher levels of pre-saccadic burst and build-up cell activity being correlated with express saccade and antisaccade error formation (Dorris et al 1997; Edelman and Keller 1996; Everling et al 1998a; Gomez et al 1994; Suzuki and Hirai 2000). If a local reduction in SC fixation cell activity or increase in burst/build-up cell excitement was responsible for SI production we would have expected shorter voluntary saccade latencies, an increased number of express saccades (particularly on the synchrony and overlap saccade tasks), and more frequent antisaccade errors to be associated with higher SI frequencies. As our findings are of a behavioural nature, we cannot conclusively rule out a direct contribution of the SC to SI production, but would suggest that although the SC may well be activated during SI production, the original SI signal is probably not generated within the circuitry of this structure.
It also appears unlikely that higher cortical areas such as the dorsolateral prefrontal cortex that are involved in saccade inhibition are responsible for SI generation. SI could arise if the inhibitory influence of the dorsolateral prefrontal cortex over the SC was reduced, facilitating saccade generation. This is reflected in the difficulty that patients with dorsolateral prefrontal cortex lesions have in suppressing inappropriate saccades (Braun et al 1992; Gaymard et al 2003; Pierrot-Deseilligny et al 1991, 2003) and the increase in express saccade number when the dorsolateral prefrontal cortex is inhibited (Muri et al 1999). Reduction in inhibitory signals from the substantia nigra pars reticulata has also been shown to lead to the production of square wave intrusion-like interruptions of fixation in monkeys (Hikosaka and Wurtz 1985). However, if these two areas were involved in physiological SI production, an increase in express saccade and antisaccade error rate would be expected to correlate with SI frequency. Additionally, in those subjects with higher SI frequencies, the normal latencies and express saccade percentage found on the synchrony, gap, overlap and antisaccade tasks in conjunction with the ability to correct for antisaccade errors indicates the normality of the frontal eye field that is involved in intentional saccade triggering and fixation disengagement (Dias and Bruce 1994; Friedman et al 1997; Gaymard et al 1999; Pierrot-Deseilligny et al 2003; Sommer and Wurtz 2000).
In summary, our results suggest that SI are not the result of an underlying change in the excitability of the saccadic system. It is possible that they are a consequence of descending commands related to attentional shifts arising from areas such as the parietal cortex, anterior cingulate, thalamus, basal ganglia or cerebellum (Coull et al 2000; Nobre et al 2000; Posner et al 1984; Powell et al 1999; Rafal 1999; Rafal and Henik 1994; Rosen et al 1999; Walker and Findlay 1996). Activity from these areas may stimulate the SC build-up cells that have been proposed to be possible neurophysiological correlates of attention allocation, resulting in a saccade (Kustov and Robinson 1996). This would suggest that the fixation system, rather than existing as a separate entity or as a part of another system such as smooth pursuit, is comprised of a network of interacting areas such as the dorsolateral prefrontal cortex, frontal eye fields, basal ganglia and SC that are governed by a supramodal attention system and task demands.
The above is highlighted by the findings of a recent study that examined the effect of increased task demand on SI during pursuit tracking, and found the SI rate to decrease as the speed and unpredictability of pursuit tracking increased as well as to a memorized rather than visual target during fixation (Shaffer et al 2003). Increased attention levels upon the task may have suppressed attention shifts that could give rise to SI. Indeed, attention and eye movements appear to share much neural circuitry (Hunt and Kingstone 2003; Nobre et al 2000; Smith et al 2004) and microsaccade characteristics are affected by the orientation of exogenous and endogenous attention (Engbert and Kliegl 2003; Galfano et al 2004; Hafed and Clark 2002). Furthermore, evidence for an asymmetry in the strength of attentional processes between the right and left hemispheres is suggested by imaging studies (Nobre et al 1997) and the fact that hemispatial neglect occurs more often and to a stronger extent with right-sided parietal lesions then left-sided lesions (Kastner and Ungerleider 2000; Posner et al 1984). This could explain the predominant rightward SI directional bias, and further investigation into the influence of attention on SI has yielded supportive results (Gowen et al, Cogn Brain Res, in review).
References
Abadi RV, Gowen E (2004) Characteristics of saccadic intrusions. Vision Res 44:2675–2690
Amador N, Schlag-Rey M, Schlag J (2004) Primate antisaccade. II. Supplementary eye field neuronal activity predicts correct performance. J Neurophysiol 91:1672–1689
Biscaldi M, Fischer B, Stuhr V (1996) Human express saccade makers are impaired at suppressing visually evoked saccades. J Neurophysiol 76:199–214
Braun D, Weber H, Mergner T, Schulte-Monting J (1992) Saccadic reaction times in patients with frontal and parietal lesions. Brain 115:1359–1386
Carpenter RHS (1988) Miniature eye movements. In: Carpenter RHS (ed) Movements of the eyes. Pion, London, Ch 6, pp 124–138
Coull JT, Frith CD, Buchel C, Nobre AC (2000) Orienting attention in time: behavioural and neuroanatomical distinction between exogenous and endogenous shifts. Neuropsychologia 38:808–819
Dias EC, Bruce CJ (1994) Physiological correlate of fixation disengagement in the primate’s frontal eye field. J Neurophysiol 72:2532–2537
Ditchburn RW (1973) Kinematic description of eye movements. In: Eye movements and visual perception. Clarendon, Oxford, Ch 4, pp 78–107
Dorris MC, Munoz DP (1995) A neural correlate for the gap effect on saccadic reaction times in monkey. J Neurophysiol 73:2558–2562
Dorris MC, Pare M, Munoz DP (1997) Neuronal activity in monkey superior colliculus related to the initiation of saccadic eye movements. J Neurosci 17:8566–8579
Doslak MJ, Dell’Osso LF, Daroff RB (1983) Multiple double saccadic pulses occurring with other saccadic intrusions and oscillations. Neuroophthalmology 3:109–116
Edelman JA, Keller EL (1996) Activity of visuomotor burst neurons in the superior colliculus accompanying express saccades. J Neurophysiol 76:908–926
Engbert R, Kliegl R (2003) Microsaccades uncover the orientation of covert attention. Vision Res 43:1035–1045
Everling S, Munoz DP (2000) Neuronal correlates for preparatory set associated with pro-saccades and anti-saccades in the primate frontal eye field. J Neurosci 20:387–400
Everling S, Dorris MC, Munoz DP (1998a) Reflex suppression in the antisaccade task is dependent on prestimulus neural processes. J Neurophysiol 80:1584–1589
Everling S, Spantekow A, Krappmann P, Flohr H (1998b) Event-related potentials associated with correct and incorrect responses in a cued antisaccade task. Exp Brain Res 118:27–34
Feldon SE, Langston JW (1977) Square wave jerks: a disorder of microsaccades? Neurology 27:278–281
Fischer B, Weber H (1992) Characteristics of “anti” saccades in man. Exp Brain Res 89:415–424
Fischer B, Weber H (1993) Express saccades and visual attention. Behav Brain Sci 16:553–610
Fischer B, Weber H (1997) Effects of stimulus condition on the performance of antisaccades in man. Exp Brain Res 116:191–200
Fischer B, Biscaldi M, Otto P (1993a) Saccadic eye movements of dyslexic adult subjects. Neuropsychologia 31:887–906
Fischer B, Weber H, Biscaldi M, Aiple F, Otto P, Stuhr V (1993b) Separate populations of visually guided saccades in humans: reaction times and amplitudes. Exp Brain Res 92:528–541
Fischer B, Gezeck S, Huber W (1995) The three-loop model: a neural network for the generation of saccadic reaction times. Biol Cybern 72:185–196
Fischer B, Biscaldi M, Gezeck S (1997) On the development of voluntary and reflexive components in human saccade generation. Brain Res 754:285–297
Fischer B, Gezeck S, Hartnegg K (1997) The analysis of saccadic eye movements from gap and overlap paradigms. Brain Res Brain Res Protoc 2:47–52
Friedman HR, Burman DD, Russo GS, Dias EC, Shi D, Stanton DB, Bruce CJ (1997) Neuronal activity in primate frontal eye field during memory guided saccades. Soc Neurosci 23:844
Galfano G, Betta E, Turatto M (2004) Inhibition of return in microsaccades. Exp Brain Res 159:400–404
Gandhi NJ, Keller EL (1999) Comparison of saccades perturbed by stimulation of the rostral superior colliculus, the caudal superior colliculus and the omnipause neuron region. J Neurophysiol 82:3236–3253
Gaymard B, Rivaud S, Pierrot-Deseilligny C (1993) Role of the left and right supplementary motor areas in memory-guided saccade sequences. Ann Neurol 34:404–406
Gaymard B, Ploner CJ, Rivaud-Pechoux S, Pierrot-Deseilligny C (1999) The frontal eye field is involved in spatial short-term memory but not in reflexive saccade inhibition. Exp Brain Res 129:288–301
Gaymard B, Francois C, Ploner CJ, Condy C, Rivaud-Pechoux S (2003) A direct prefrontotectal tract against distractibility in the human brain. Ann Neurol 53:542–545
Gomez C, Atienza M, Vazquez M, Cantero JL (1994) Saccadic reaction times to fully predictive and random visual targets during gap and non-gap paradigms. In: Delgado-Garcia JM, Godaux E, Vidal PP (eds) Information processing underlying gaze control. Pergamon, Oxford, pp 109–115
Guitton D, Buchtel HA, Douglas RM (1985) Frontal lobe lesions in man cause difficulties in suppressing reflexive glances and in generating goal-directed saccades. Exp Brain Res 58:455–472
Hafed ZM, Clark JJ (2002) Microsaccades as an overt measure of covert attention shifts. Vision Res 42:2533–2545
Hikosaka O, Wurtz RH (1985) Modification of saccadic eye movements by GABA-related substances. II. Effects of muscimol in monkey substantia nigra pars reticulata. J Neurophysiol 53:292–308
Honda H (2002) Idiosyncratic left-right asymmetries of saccadic latencies: examination in a gap paradigm. Vision Res 42:1437–1445
Hunt AR, Kingstone A (2003) Covert and overt voluntary attention: linked or independent?. Brain Res Cogn Brain Res 18:102–105
Kastner S, Ungerleider LG (2000) Mechanisms of visual attention in the human cortex. Annu Rev Neurosci 23:315–341
Kingstone A, Klein RM (1993) Visual offsets facilitate saccadic latency: does predisengagement of visuospatial attention mediate this gap effect? J Exp Psychol Hum Percept Perform 19:1251–1265
Kowler E, Anderson E, Dosher B, Blaser E (1995) The role of attention in the programming of saccades. Vision Res 35:1897–1916
Kustov AA, Robinson DA (1996) Shared neural control of attentional shifts and eye movements. Nature 384:74–77
Lasker AG, Zee DS, Hain TC, Folstein SE, Singer HS (1987) Saccades in Huntington’s disease: initiation defects and distractibility. Neurology 37:364–370
Mayfrank L, Mobashery M, Kimmig H, Fischer B (1986) The role of fixation and visual attention in the occurrence of express saccades in man. Eur Arch Psychiatry Neurol Sci 235:269–275
Munoz DP, Wurtz RH (1992) Role of the rostral superior colliculus in active visual fixation and execution of express saccades. J Neurophysiol 67:1000–1002
Munoz DP, Wurtz RH (1993a) Fixation cells in monkey superior colliculus. 1. Characteristics of cell discharge. J Neurophysiol 70:559–575
Munoz DP, Wurtz RH (1993b) Fixation cells in monkey superior colliculus II. Reversible activation and deactivation. J Neurophysiol 70:576–589
Munoz DP, Wurtz RH (1995a) Saccade-related activity in monkey superior colliculus I. Characteristics of burst and buildup cells. J Neurophysiol 73:2313–2333
Munoz DP, Wurtz RH (1995b) Saccade-related activity in monkey superior colliculus II. Spread of activity during saccades. J Neurophysiol 73:2334–2348
Munoz DP, Broughton JR, Goldring JE, Armstong IT (1998) Age-related performance of human subjects on saccadic eye movement tasks. Exp Brain Res 121:391–400
Muri RM, Rivaud S, Gaymard B, Ploner CJ, Vermersch AI, Hess CW, Pierrot-Deseilligny C (1999) Role of the prefrontal cortex in the control of express saccades. A transcranial magnetic stimulation study. Neuropsychologia 37:199–206
Nobre AC, Sebestyen GN, Gitelman DR, Mesulam MM, Frackowiak RSJ, Frith CD (1997) Functional localization of the system for visuospatial attention using positron emission tomography. Brain 120:515–533
Nobre AC, Gitelman DR, Dias EC, Mesulam MM (2000) Covert visual spatial orienting and saccades: overlapping neural systems. Neuroimage 11:210–216
Ohtsuka K, Mukuno K, Ukai K, Ishikawa S (1986) The origin of square wave jerks: conditions of fixation and microsaccades. Jpn J Ophthalmol 30:209–215
Pierrot-Deseilligny C, Rivaud S, Gaymard B, Agid Y (1991) Cortical control of reflexive visually-guided saccades. Brain 114:1473–1485
Pierrot-Deseilligny C, Muri RM, Ploner CJ, Gaymard B, Demeret S, Rivaud-Pechoux S (2003) Decisional role of the dorsolateral prefrontal cortex in ocular motor behaviour. Brain 126:1460–1473
Pierrot-Deseilligny C, Milea D, Muri RM (2004) Eye movement control by the cerebral cortex. Curr Opin Neurol 17:17–25
Posner JB, Walker JA, Friedrich FJ, Rafal R (1984) Effects of parietal injury on covert orienting of attention. J Neurosci 4:1863–1874
Powell KD, Colby CL, Gottlieb J, Kusunoki M, Goldberg ME (1999) Space and salience in parietal cortex. In: Becker W, Deubel H, Mergner T (eds) Current oculomotor research. Physiological and psychological aspects. Kluwer/Plenum, New York, pp 25–35
Rafal R (1999) Neglect. In: Parasuraman R (ed) The attentive brain. MIT Press, Cambridge, MA, pp 489–525
Rafal R, Henik A (1994) The neurology of inhibition integrating controlled and automatic processes. In: Dagenbach D, Carr TH (eds) Inhibitory processes in attention, memory and language. Academic, San Diego, CA, pp 1–51
Rosen AC, Rao SM, Caffarra P, Scaglioni A, Bobholz JA, Woodley SJ, Hammeke TA, Cunningham JM, Prieto TE, Binder JR (1999) Neural basis of endogenous and exogenous spatial orienting. A functional MRI study. J Cogn Neurosci 11:135–152
Schiller PH, Sandell JH, Maunsell JHR (1987) The effect of frontal eye field and superior colliculus lesions on saccadic latencies in the rhesus monkey. J Neurophysiol 57:1033–1049
Schlag-Rey M, Amador N, Sanchez H, Schlag J (1997) Antisaccade performance predicted by neuronal activity in the supplementary eye field. Nature 390:398–401
Shaffer DM, Krisky CM, Sweeney JA (2003) Frequency and metrics of square wave jerks: influences of task-demand characteristics. Invest Ophthalmol Vis Sci 44:1082–1087
Shepherd M, Findlay JM, Hockey R (1986) The relationship between eye movements and spatial attention. Q J Exp Psychol 38A:475–491
Smith DT, Rorden C, Jackson SR (2004) Exogenous orienting of attention depends upon the ability to execute eye movements. Curr Biol 14:792–795
Sommer MA, Wurtz RH (2000) Composition and topographic organization of signals sent from the frontal eye field to the superior colliculus. J Neurophysiol 83:1979–2001
Suzuki DA, Hirai N (2000) The role of fixation point and subjects’ readiness in the occurrence of express saccades as revealed by the self initiation paradigm. Neurosci Res 36:235–244
Walker R, Findlay JM (1996) Saccadic eye movement programming in unilateral neglect. Neuropsychologia 34:493–508
Walker R, Kentridge RW, Findlay JM (1995) Independent contributions of the orienting of attention, fixation offset and bilateral stimulation on human saccadic latencies. Exp Brain Res 103:294–310
Weber H, Fischer B (1995) Gap duration and location of attention focus modulate the occurrence of left/right asymmetries in the saccadic reaction times of human subjects. Vision Res 35:987–998
Weber H, Aiple F, Fischer B, Latanov A (1992) Dead zone for express saccades. Exp Brain Res 89:214–222
Wenban-Smith MG, Findlay JM (1991) Express saccades: is there a separate population in humans?. Exp Brain Res 87:218–222
Zee DS, Robinson DA (1979) A hypothetical explanation of saccadic oscillations. Ann Neurol 5:405–414
Acknowledgements
Emma Gowen was in receipt of a UMIST scholarship.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gowen, E., Abadi, R.V. Saccadic instabilities and voluntary saccadic behaviour. Exp Brain Res 164, 29–40 (2005). https://doi.org/10.1007/s00221-004-2209-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-004-2209-2