Abstract
Repetitive transcranial magnetic stimulation (rTMS) has been shown to induce adaptations in cortical neuronal circuitries. In the present study we investigated whether rTMS, through its effect on corticospinal pathways, also produces adaptations at the spinal level, and what the neuronal mechanisms involved in such changes are. rTMS (15 trains of 20 pulses at 5 Hz) was applied over the leg motor cortical area in ten healthy human subjects. At rest motor evoked potentials (MEPs) in the soleus and tibialis anterior muscles were facilitated by rTMS (at 1.2×MEP threshold). In contrast, the soleus H-reflex was depressed for 1 s at stimulus intensities from 0.92 to 1.2×MEP threshold. rTMS increased the size of the long-latency depression of the soleus H-reflex evoked by common peroneal nerve stimulation and decreased the femoral nerve facilitation of the soleus H-reflex. These observations suggest that the depression of the H-reflex by rTMS can be explained, at least partly, by an increased presynaptic inhibition of soleus Ia afferents. In contrast, rTMS had no effect on disynaptic reciprocal Ia inhibition from ankle dorsiflexors to plantarflexors. We conclude that a train of rTMS may modulate transmission in specific spinal circuitries through changes in corticospinal drive. This may be of relevance for future therapeutic strategies in patients with spasticity.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Repetitive transcranial magnetic stimulation (rTMS) is used to an increasing extent to study cognitive functions in human subjects (Amassian et al. 1989; Epstein et al. 1996; Walsh and Cowey 2000), and it may also be of therapeutic value in the treatment of depression (George et al. 2000; Iyer et al. 2003; Rohan et al. 2004) and motor disorders (Homberg et al. 1991; Ghabra et al. 1999; Siebner et al. 1999, 2000; Huang et al. 2004). It is of great importance to understand how rTMS interacts with the neuronal circuitries in order to elucidate the physiological mechanisms underlying these behavioral effects.
Several studies have documented that rTMS induces long lasting adaptations in cortical neuronal circuitries (for reviews, see Pascual-Leone et al. 1998; Peinemann et al. 2000, 2004; Fierro et al. 2001; Di Lazzaro et al. 2002; Gilio et al. 2003; Kobayashi et al. 2004). In humans and primates, corticospinal cells exert modulation over a large group of spinal interneurones (Jankowska 1992). Some of these interneurones mediate presynaptic inhibition of Ia afferents (Valls Sole et al. 1994; Iles 1996; Meunier and Pierrot–Deseilligny 1998) or reciprocal Ia inhibition (Hultborn et al. 1976; Jankowska et al. 1976; Nielsen et al. 1993). It is possible that activating corticospinal neurons by rTMS may also induce long-lasting changes in spinal neuronal circuitries.
H-reflex recording has been used in some studies as a control measure of spinal motoneuronal excitability following rTMS (Berardelli et al. 1998; Valero-Cabre et al. 2001; Modugno et al. 2001; Touge et al. 2001). No change, an increase, and a decrease have all been reported in these studies. This variability in the observed effects is likely due to differences in the intensity, frequency and duration of the stimulation protocols used. This illustrates that studies addressing directly whether and how rTMS interacts with the spinal neuronal circuitries, in addition to its well-documented effects at the cortical level, are required. Such studies would open new avenues for investigating the interaction and plastic adaptability of cortical and spinal control centers and could potentially lead to the development of new treatment strategies for the rehabilitation of subjects with motor disorders such as Parkinsonism and spasticity. The purpose of the present study was therefore to investigate whether rTMS produces any changes in the size of the soleus H-reflex, which outlast the duration of the stimulus train, and if so to test the effect of rTMS on spinal neuronal mechanisms that may underlie these effects, such as presynaptic and reciprocal Ia inhibition.
Methods
Subjects
Ten healthy volunteers (six female, four male) with an average age of 29±8 years took part in the study. Not all of the subjects participated in every experiment. All subjects gave their informed consent to the experimental procedure, which was approved by the local ethics committee. The study was performed in accordance with the Declaration of Helsinki. Subjects were seated in an armchair with the examined left leg flexed in the hip (120°), the knee (120°) and the ankle in 110° of plantarflexion. The foot was attached to a plate, which was connected to a torque meter.
Data recording
Bipolar surface electrodes (interelectrode distance, 2 cm) were used to record motor evoked potentials (MEPs) and H-reflexes from the soleus (SOL) and tibialis anterior (TA) muscles. The amplified EMG signals were filtered (band-pass, 25 Hz to 1 kHz), sampled at 2 kHz, and stored on a PC for off-line analysis (CED 1401+ with Signal software, Cambridge Electronic Devices, Cambridge, UK).
Experimental design
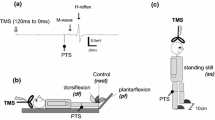
The experimental design is illustrated in Fig. 1. In all experiments we investigated the effect of rTMS over the leg cortical area on MEPs or spinal circuitries. rTMS was applied by a magnetic stimulator (Magstim super rapid stimulator, Magstim company, Dyfed, UK) with the capacity to deliver a magnetic field of 2 T for 100 μs through a figure-of-eight coil (loop diameter, 9 cm; type no.: 8809). The coil was positioned with the center of the coil 2 cm to the right of the vertex and secured by straps to ensure that the same area of the cortex was stimulated during the testing. rTMS consisted of 15 trains of 20 pulses at 5 Hz separated by inter-train intervals of 10 s. MEPs and H-reflexes were evoked in separate trials at different intervals (200 ms to 3 s) after each train of rTMS. This sequence was repeated every 2 min in the experiments where multiple intensities and conditioning-test intervals were tested (see below). In all experiments the stimulus intensity for the rTMS was adjusted in relation to the threshold of the TA MEP as the SOL muscle usually has a higher threshold, and at that stimulus intensities leg movements were elicited. Since cortical excitability increases during rTMS (Berardelli et al. 1998), MEPs were nevertheless often elicited by the last pulses in the train even when the intensity of the stimulation had been adjusted to be below MEP threshold following single pulses. In such cases MEP threshold was determined as the intensity where no MEPs were observed during rTMS. All measurements were performed at rest.
Experimental set-up. A Diagram of the experimental set-up. B Effect of rTMS (20 pulses at 5 Hz at a conditioning-test interval of 200 ms) on the SOL H-reflex in a single subject. C Time course of the long-latency depression of the SOL H-reflex (D2 inhibition), femoral nerve facilitation of the SOL H-reflex (the arrow indicates the time at which FN facilitation was measured) and the short-latency depression of the SOL H-reflex (disynaptic reciprocal Ia inhibition) in three different subjects. The ordinate shows the size of the conditioned SOL H-reflex (as percentage of the control reflex size). The abscissa shows conditioning-test intervals, positive values indicate that the conditioning pulse preceded the control pulse and negative values indicate that the control pulse preceded the conditioning pulse. At each interval, 15 control and 15 conditioned H-reflexes were averaged. Bars indicate standard errors. TMS transcranial magnetic stimulation; FN femoral nerve; PTN posterior tibial nerve; CPN common peroneal nerve
In different sessions, separated by at least two days, we assessed the effect of rTMS on: A) SOL and TA MEPs, B) SOL H-reflex, C) presynaptic inhibition of SOL Ia afferents evoked by stimulation of the common peroneal nerve, D) the size of the monosynaptic femoral nerve facilitation of the SOL H-reflex, which may also be used as a measure of presynaptic inhibition of Ia afferents, E) disynaptic reciprocal Ia inhibition of the SOL H-reflex evoked by stimulation of the common peroneal nerve, and F) motor evoked responses elicited by transmastoid electrical stimulation.
A: SOL and TA MEPs
In six subjects we investigated the effect of rTMS delivered to the leg motor cortex on the size of the TA and SOL MEPs. The MEP threshold was determined at rest as the lowest intensity of magnetic stimulation required to evoke MEPs of 50 μV in peak-to-peak amplitude in at least three of five consecutive trials in the TA and SOL muscles (Rossini et al. 1994). The stimulation intensity for the train of pulses was adjusted to 1.2×MEP threshold for each muscle, and the conditioning-test intervals tested were 200 and 2000 ms. A computer program automatically delivered rTMS and a single pulse (control MEP) at the desired intensities and conditioning-test intervals through the same coil. The program did not allow us to test the effect of rTMS at different stimulus intensities on the MEPs, through the same coil, when the difference in intensity between the rTMS train of pulses and the control MEP was larger than 5%. The control MEP for both muscles was adjusted to obtain an MEP about 0.5 mV in size. In all experiments the MEPs were evoked at an interval of 10 s.
B: SOL H-reflex
In six subjects we investigated the effect of rTMS delivered to the leg motor cortex on the size of the SOL H-reflex. We measured the effect of different stimulus intensities ranging from 0.75 to 1.2×MEP threshold, and different conditioning-test intervals ranging from 200 to 3000 ms. The SOL H-reflex was evoked by stimulating the posterior tibial nerve through a monopolar stimulating electrode (1 ms rectangular pulse) in the popliteal fossa. The reflex response was measured as peak-to-peak amplitude of the non-rectified reflex response, and recorded by surface electrodes placed over the SOL muscle. The sensitivity of the H-reflex to facilitatory or inhibitory conditioning effects has been shown to depend crucially on its size (Crone et al. 1990). During measurements of the effect of a conditioning stimulus, the size of the SOL control H-reflex was therefore maintained at 20–25% of the maximal motor response (M-max). To measure the SOL M-max the posterior tibial nerve was stimulated with supramaximal intensity at the beginning of each session. In all experiments the SOL H-reflexes were evoked at an interval of 10 s.
C–E: Presynaptic inhibition and reciprocal Ia inhibition
In six subjects we investigated the effect of rTMS on the short and long latency depression of the SOL H-reflex and the size of the monosynaptic femoral nerve facilitation of the SOL H-reflex. The SOL H-reflex was conditioned by stimulation of the common peroneal nerve (CPN) and the femoral nerve (FN). The CPN was stimulated (1 ms rectangular pulse) by bipolar surface electrodes placed distal to the neck of the fibula. Electrodes were placed in a position to evoke a threshold motor response in the TA muscle without activation of a motor response in the peroneal muscles. The stimulus strength was expressed in multiples of the motor threshold (MT) in the TA muscle. CPN stimulation elicits a significant depression of the SOL H-reflex at conditioning-test intervals of 2–3 ms (Crone et al. 1987). This depression has been shown in all likelihood to be mediated by the disynaptic reciprocal Ia inhibitory pathway (Crone et al. 1987). In order to investigate the effect of rTMS on this pathway we selected a conditioning-test interval at which the largest amount of inhibition was observed. At longer conditioning-test intervals of 60 to 80 ms, CPN stimulation elicits a depression of the SOL H-reflex, which has been termed D2 inhibition (Mizuno et al. 1971). This inhibition is believed to be caused by presynaptic inhibition of the terminals of Ia afferents on SOL motoneurones.
The FN was stimulated through a monopolar ball electrode placed over the femoral triangle. The indifferent electrode was placed just below the gluteus maximus muscle. The intensity for stimulating the FN was above the threshold for a motor response in the quadriceps muscle (1.2×MT). In each experiment the time course of the effect of the FN facilitation was recorded for every subject. FN elicits a significant facilitation of the SOL H-reflex at conditioning-test intervals of −8 to −4.5 ms (the negative values indicate that stimulation to the FN was given after stimulation to the posterior tibial nerve; Hultborn et al. 1987a, 1987b). The onset of facilitation was considered to be the earliest conditioning-test interval at which the conditioned reflex was 10% larger than the control reflex. The interval used during the testing was 0.5–1 ms longer than this interval. The size of the facilitation measured at this conditioning-test interval reflects the size of the monosynaptic excitatory postsynaptic potential in the SOL motoneurones evoked by the activation of Ia afferents from the quadriceps muscle (Meunier et al. 1993), and changes in its size are considered to indicate changes in presynaptic inhibition of these afferents (Hultborn et al. 1987a, 1987b). It is considered that presynaptic inhibition of Ia afferents from the SOL and quadriceps are controlled in parallel (Hultborn et al. 1987a, 1987b). We measured the effect of rTMS on both D2 inhibition and FN facilitation because both measurements provide independent information about presynaptic inhibition and help us to exclude changes in recruitment gain in the SOL motoneurones (Nielsen and Kagamihara 1993). D2 inhibition reflects the level of presynaptic inhibition evoked by peripheral nerve stimulation and FN facilitation reflects the level of ongoing presynaptic inhibition of SOL Ia afferents.
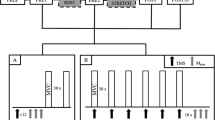
The following five conditions were randomly tested:
-
1.
H (control H-reflex)
-
2.
rTMS + H
-
3.
rTMS + H1 (control H-reflex elicited by a different stimulator)
-
4.
CPN (or FN) + H
-
5.
rTMS + CPN (or FN) + H1
Condition 1 corresponds to the control H-reflex, which was maintained at 20–25% of M-max. In condition 2, rTMS was applied prior to the control H-reflex at different conditioning-test intervals to assess the effect of rTMS on the SOL H-reflex size. H-reflexes of different sizes have different sensitivities to conditioning inputs (Crone et al. 1990). In order to evaluate the effect of rTMS on the femoral nerve facilitation, disynaptic reciprocal inhibition and D2 inhibition it was therefore essential to ensure that the H-reflex had the same size with and without rTMS. Since rTMS evoked a depression of the H-reflex (see the “Results” section), it was necessary to compensate for this depression by using a second stimulator set at a higher stimulation intensity to evoke the H-reflex (H1) in conditions 3 and 5. In this way the H-reflex conditioned by femoral nerve stimulation and peroneal nerve stimulation had the same size with and without rTMS (conditions 1 and 3) in all experiments.
F: Transmastoid electrical stimulation
We measured the effect of rTMS on SOL CMEPs (cervicomedullary motor evoked potentials) elicited by transmastoid electrical stimulation in a single subject. It was not possible to perform this experiment in more subjects because of the high threshold for evoking responses in the SOL muscle and the discomfort evoked by the stimulation. Transmastoid electrical stimulation was delivered by passing an electrical pulse (100 μs, D18 stimulator, Digitimer, Welwyn Garden City, UK) between Ag-AgCl surface electrodes fixed below the mastoids with the cathode on the right. Stimulus intensity was 20% of stimulator output (300–525 V) to produce control CMEPs with a size of 4–6% of the M-max. The size of the control SOL H-reflex was maintained 3–5% of the SOL M-max. The following four conditions were randomly tested:
-
1.
H (control H-reflex)
-
2.
rTMS + H
-
3.
CMEP (control CMEP evoked by transmastoid electrical stimulation)
-
4.
rTMS + CMEP
The intensity of the posterior tibial nerve stimulation was adjusted to evoke a control H-reflex with a similar magnitude to the control CMEP. To measure the effect of rTMS on the size of the H-reflex we compared conditions 1 and 2. To measure the effect of rTMS on the size of the CMEP we compared conditions 3 and 4.
Data analysis
During all measurements at least 30 peak-to-peak H-reflexes and MEPs were averaged during each trial (15 control and 15 conditioned responses). A repeated measures ANOVA test was used to determine the effect of rTMS on the amplitude of the SOL H-reflex at different intensities and conditioning-test intervals, MEP amplitudes, short and long latency depression of the SOL H-reflex and FN facilitation. Post hoc testing to determine significant comparisons was performed using a criterion of p<0.05. A paired t-test was used to compare the effect of rTMS on the CMEPs evoked by transmastoid electrical stimulation. The mean and the standard error of the mean were calculated for each condition.
Results
Modulation of MEPs by rTMS
First we wanted to confirm that rTMS increases the size of MEPs in leg muscles, as has been previously demonstrated for arm muscles (Berardelli et al. 1998). Figure 2 illustrates the effect of rTMS on the amplitude of TA and SOL MEPs at conditioning-test intervals of 200 and 2000 ms in six subjects. Repeated measures ANOVA showed a significant effect of rTMS on the peak-to-peak amplitude of the TA MEPs (ANOVA, F=5.25, p<0.05). Post hoc testing revealed that the TA MEPs were significantly increased at conditioning-test intervals of 200 (p<0.05) but not 2000 ms (p=0.9). We also observed a significant effect of rTMS on the peak-to-peak amplitude of the SOL MEPs (ANOVA, F=10.18, p<0.01) at 200 (p<0.05) but not 2000 ms (p=0.9) conditioning-test interval.
The effect of rTMS on MEPs. The graphs show the effect of rTMS on TA (A) and SOL (B) MEPs in six subjects. rTMS was given at 200 ms conditioning-test intervals and 1.2×MEP threshold. The ordinate shows the size of the conditioned MEP (as a % of the control MEP). The abscissa shows the control MEPs and the conditioning-test intervals. 15 control and 15 conditioned MEPs were averaged at each interval. Bars indicate standard errors (*p<0.05)
Modulation of the SOL H-reflex by rTMS
Figure 3A illustrates the effect of different intensities of rTMS on the amplitude of the SOL H-reflex (conditioning-test interval 200 ms) in six subjects. Repeated measures ANOVA showed a significant effect of the intensity of stimulation on the amplitude of the SOL H-reflex (ANOVA, F=20.80, p<0.001). From the figure, the inhibition appears to have a threshold at around 0.84×MEP threshold. Post hoc testing revealed that the SOL H-reflex was significantly depressed at 1.2 (p<0.001), 1.0 (p<0.001), 0.97 (p<0.001) and 0.92×MEP threshold (p<0.01).
The effect of different intensities and conditioning-test intervals of rTMS on the amplitude of the SOL H-reflex. The graphs show the data from six subjects. A The effect of rTMS at stimulus intensities ranging from 0.75 to 1.2×MEP threshold (conditioning-test interval 200 ms). The ordinate shows the size of the conditioned SOL H-reflex (as a percentage of the control reflex size) and the abscissa shows the stimulus intensity expressed as a percentage of the MEP threshold. B The effect of rTMS (1.0×MEP threshold intensity) at conditioning-test intervals ranging from 200 to 3000 ms. The ordinate shows the size of the conditioned SOL H-reflex (as a percentage of the control reflex size) and the abscissa shows the conditioning-test interval. 15 control and 15 conditioned H-reflexes were averaged at each interval. Bars indicate standard errors (*p<0.05)
Figure 3B illustrates the effect of rTMS (1.0×MEP threshold intensity) at different conditioning-test intervals on the amplitude of the SOL H-reflex in six subjects. Repeated measures ANOVA showed a significant effect of the conditioning-test intervals on the amplitude of the SOL H-reflex (ANOVA, F=3.89, p<0.001). From the figure it appears that the SOL H-reflex was depressed at conditioning-test intervals up to 2000 ms. Post hoc testing revealed that the SOL H-reflex was significantly depressed at conditioning-test intervals of 200 (p<0.001), 400 (p<0.001), 600 (p<0.001), 800 (p<0.01) and 1000 ms (p<0.05). In order to test if the SOL H-reflex depression induced by rTMS was specific to stimulation of the leg motor cortical area, we investigated the effect of moving the coil lateral in two subjects. Whereas stimulation at 1.0×MEP threshold effectively depressed the SOL H-reflex when the coil was placed over the leg area, no depression was observed when the coil was moved 4 cm lateral.
Modulation of presynaptic inhibition of Ia afferents
Figure 4A illustrates the effect of rTMS on the long-latency depression of the SOL H-reflex (D2 inhibition) evoked by stimulation of the CPN at conditioning-test intervals of 60–80 ms in six subjects. A time course of the effect of CPN stimulation on the SOL H-reflex was obtained at the beginning of the experiment to determine a suitable interval for measurement of the D2 inhibition (Fig. 1C). As it was ensured that there was no difference between the size of H and H1, a comparison of the size of the D2 inhibition with and without rTMS was possible. Repeated measures ANOVA showed that the D2 inhibition was significantly increased 200 ms after rTMS (before rTMS=83% and after rTMS=57% of the control reflex size; p<0.05; see conditions CPN + H and rTMS + CPN + H1 in Fig. 4A). In the same testing paradigm, the SOL H-reflex was significantly depressed by rTMS (37% of the control reflex size, p<0.01; see conditions H and rTMS + H in Fig. 4A). Raw data from one of the subjects is provided in Fig. 4A. At an interval of 1000 ms after rTMS (not illustrated) there was still an increase in D2 inhibition in four of the subjects, but for the overall population of subjects there was no significant effect. D2 inhibition was also not increased at 2000 ms after rTMS.
The effect of rTMS on presynaptic inhibition and disynaptic reciprocal Ia inhibition. A The effect of rTMS (1.0×MEP threshold intensity, and conditioning-test interval 200 ms) on the long-latency depression of the SOL H-reflex (D2 inhibition, 60–80 ms conditioning-test interval). B FN facilitation of the SOL H-reflex (conditioning test interval −8 to −4.5 ms; the negative value indicates that the control pulse preceded the conditioning pulse). C Short-latency depression of the SOL H-reflex (2–3 ms conditioning-test interval) at 200 ms conditioning-test interval. The bar graphs show data from six subjects, and the traces are from three different subjects. The following conditions were tested: 1 H (control H-reflex), 2 rTMS + H, 3 rTMS + H1 (control H-reflex elicited by a different stimulator), 4 CPN (or FN) + H, 5 rTMS + CPN (or FN) + H1. The ordinate shows the size of the conditioned SOL H-reflex (as a percentage of the control reflex size), and the abscissa shows testing conditions from 1 to 5. 15 control and 15 conditioned SOL H-reflexes were averaged at each condition. Bars indicate standard errors (*p<0.05)
Figure 4B illustrates the effect of rTMS on the monosynaptic FN facilitation of the SOL H-reflex at conditioning-test intervals between −8 to −4.5 ms in six subjects. A time course was obtained at the beginning of the experiment to determine the interval for measurement of the FN facilitation (Fig. 1C). rTMS depressed the SOL H-reflex, but this was compensated for by increasing the intensity of the stimulator, which evoked the H-reflex in the conditioned situation. The effect of rTMS on the FN facilitation could therefore be evaluated. Repeated measures ANOVA showed that the facilitation of the SOL H-reflex was significantly decreased 200 ms after rTMS (before rTMS=132% and after rTMS=103% of the control reflex size; p<0.05; see conditions FN + H and rTMS + FN + H1 in Fig. 4B). In the same testing paradigm, the SOL H-reflex was significantly depressed by rTMS (55% of the control reflex size, p<0.001; see conditions H and rTMS + H in Fig. 4B). Raw data from one of the subjects is provided in Fig. 4B. The facilitation was also significantly (p<0.01) decreased 1000 ms after rTMS (not illustrated). No significant changes in the amount of facilitation were observed 2000 ms after rTMS.
Modulation of disynaptic reciprocal Ia inhibition
Figure 4C illustrates the effect of rTMS on disynaptic reciprocal Ia inhibition evoked by stimulation of the CPN at conditioning-test intervals of 2–3 ms in six subjects (stimulation to the CPN was given prior to stimulation of the posterior tibial nerve). A time course measured at the beginning of the experiment revealed the interval at which disynaptic reciprocal Ia inhibition was measured (Fig. 1C). Repeated measures ANOVA showed that the depression of the SOL H-reflex was unchanged 200 ms after rTMS (before rTMS=86% and after rTMS=82% of the control reflex size; p=0.6; see conditions CPN + H and rTMS + CPN + H1 in Fig. 4C). In the same testing paradigm, the SOL H-reflex was significantly depressed after rTMS (49% of the control reflex size, p<0.001; see conditions H and rTMS + H in Fig. 4C). Raw data from one of the subjects is provided in Fig. 4C.
Effect of rTMS on CMEPs evoked by transmastoid stimulation
Figure 5 illustrates the effect of rTMS on the amplitude of the SOL CMEPs evoked by transmastoid electrical stimulation in one subject. A paired t-test showed that the SOL H-reflex was significantly decreased 200 ms after rTMS (60% of the control reflex size, p<0.01). In contrast, the SOL CMEP evoked by transmastoid electrical stimulation was significantly increased 200 ms after rTMS (100% of the control reflex size p<0.001).
The effect of rTMS on CMEPs. The graph shows the effect of rTMS (1.0×MEP threshold intensity, conditioning-test interval 200 ms) on CMEPs in a single subject. The ordinate shows the size of the responses expressed as a percentage of the SOL M-max. The abscissa shows the following testing conditions: 1 H (control H-reflex), 2 rTMS + H, 3 CMEP (control CMEP evoked by transmastoid electrical stimulation), and 4 rTMS + CMEP. Bars indicate standard errors (*p<0.05)
Discussion
In the present study we have demonstrated that: 1) rTMS induced a decrease in the SOL H-reflex at stimulus intensities ranging from 0.92 to 1.2×MEP threshold for up to 1 s; 2) rTMS increased the level of presynaptic inhibition at the terminals of Ia afferent fibers, and; 3) rTMS did not change the level of disynaptic reciprocal Ia inhibition. These results indicate that rTMS-induced changes in spinal cord excitability are, at least in part, mediated by changes in presynaptic inhibition at the terminals of the Ia afferent fibers.
The changes in SOL H-reflex excitability are related to rTMS intensity
Our finding that rTMS depressed the SOL H-reflex confirms earlier observations by Berardelli et al. (1998) for H-reflexes in the flexor carpi radialis muscle. In the study by Berardelli et al. (1998) a stimulation intensity of 1.3×MEP threshold was used. We investigated only stimulation intensities ranging from 0.75 to 1.2×MEP threshold, since stronger stimuli were too uncomfortable for most subjects. As there was a linear relation between the intensity of rTMS and the depression of the SOL H-reflex for the investigated stimulus intensity range (see Fig. 3A) it is likely that larger descending volleys to the spinal cord elicited by stronger stimuli would have produced an even larger depression of the SOL H-reflex.
A depression of the SOL H-reflex excitability could also be elicited by a train of rTMS pulses at intensities lower than the MEP threshold. Experiments in monkeys have shown that the threshold for a detectable MEP is higher than that of the direct (D) wave evoked by TMS (Baker et al. 1994). In humans, epidural recordings from the thoracic region have shown that D and I-wave generation using magnetic stimulation can be elicited at stimulus intensities at which no EMG activity is recorded from the TA muscle (Houlden et al. 1999; Di Lazzaro et al. 2001), suggesting that, as reported in the monkey, the threshold for a detectable MEP is higher than the threshold for descending D and I waves. Indirect evidence of this in humans is also obtained by the short-latency facilitatory effect of subthreshold TMS on the H-reflex (Nielsen et al. 1993). The most likely explanation of the depression of the SOL H-reflex by subthreshold rTMS is therefore that descending volleys were evoked by the rTMS train of pulses even when they were below the threshold for elicitation of the MEP.
In addition to the relation between the depression of the SOL H-reflex and rTMS intensity, our observations also suggest that the effects of rTMS are specific to the area of stimulation, since stimulation 4 cm lateral to the leg area had no effect. Previously, the specificity of the effects of rTMS has been reported in cortical neuronal circuits (Siebner and Rothwell 2003).
rTMS increases presynaptic inhibition but not disynaptic reciprocal Ia inhibition
Pyramidal tract neurons have widespread terminations in the spinal gray matter and control the motoneurones through non-monosynaptic connections in addition to the direct corticomotoneuronal connections in human subjects (for specific comments see Lundberg 1975; Petersen et al. 2003). Interaction between corticospinal activation and local interneurones in the spinal cord has been documented physiologically in the cat (Baldissera et al. 1981; Jankowska 1992), monkey (Jankowska et al. 1976; Alstermark et al. 1999) and humans (Iles and Pisini 1992; Nielsen et al. 1993; Iles 1996; Meunier and Pierrot-Deseilligny 1998). The rTMS-induced suppression of the SOL H-reflex may be mediated through such corticospinal projections to local inhibitory interneurones. We therefore investigated the effect of rTMS on the transmission in the pathways mediating presynaptic inhibition of SOL Ia afferents and disynaptic reciprocal Ia inhibition of SOL motoneurones from ankle dorsiflexors. We found that rTMS increased the inhibition of the SOL H-reflex evoked by CPN stimulation at conditioning-test intervals of 60–80 ms (D2 inhibition). The D2 inhibition is generally accepted to be mediated by presynaptic inhibition of the SOL Ia afferents (Mizuno et al. 1971). The increase of this inhibition at an interval of 200 ms following rTMS is therefore consistent with increased presynaptic inhibition of the Ia afferents. However, changes in motoneuronal excitability and recruitment gain within the motoneuronal pool induced by the conditioning rTMS stimulation may also explain the observed change in the D2 inhibition (Hultborn et al. 1987a, 1987b; Kernell and Hultborn 1990; Nielsen and Kagamihara 1993). We therefore also investigated the effect of rTMS on the monosynaptic Ia mediated FN facilitation of the SOL H-reflex. The size of this facilitation may be used as a measure of the level of presynaptic inhibition of femoral nerve Ia afferents on the SOL motoneurones (Hultborn et al. 1987a, 1987b). The FN facilitation of the SOL H-reflex was measured in addition to the D2 inhibition evoked by CPN stimulation because both measurements provide independent information about presynaptic inhibition. The D2 inhibition reflects the amount of presynaptic inhibition evoked by peripheral nerve stimulation, and FN facilitation reflects the level of ongoing presynaptic inhibition of SOL Ia afferents (Hultborn et al. 1987a, 1987b). Also, the assessment of both measurements will allow us to exclude the possibility that the depression of the SOL H-reflex was related to changes in recruitment gain in the SOL motoneurones (Nielsen and Kagamihara 1993). An increased D2 inhibition may reflect increased presynaptic inhibition or increased recruitment gain, and a decrease in FN facilitation might be explained by increased presynaptic inhibition or increased recruitment gain. Thus, it is only when changes in D2 inhibition and FN facilitation are observed in opposite directions, as in the present study, that the changes are likely to be explained by changes in presynaptic inhibition. As presynaptic inhibition appears generally to be controlled according to the motoneurones on which they terminate rather than according to their origin, our observation of decreased FN facilitation following rTMS strongly supports that rTMS induces increased presynaptic inhibition of SOL Ia afferents and that this is a main explanation of the decrease of the H-reflex. We also believe that this applies to the depression at 1000 ms after rTMS, although at this interval only the FN facilitation was significantly depressed, whereas there were no significant changes in the D2 inhibition for the population of subjects (although an increase was observed in four subjects). However, this lack of significant change is likely caused by the large intra- and interindividual variability in the D2 inhibition, and that presynaptic inhibition must be assumed to be only marginally increased by rTMS at the1000 ms interval as judged from the weak effect of rTMS on the H-reflex at this interval (see Fig. 3B).
The observation that the MEPs evoked by both TMS and transmastoid stimulation were increased following rTMS are most easily explained if the depression of the SOL H-reflex is caused by presynaptic inhibition of the Ia afferents rather than a decrease in the excitability of the motoneurones. Indeed, the observation that the MEPs evoked by transmastoid stimulation were facilitated by rTMS may suggest that the motoneuronal excitability was in fact increased following rTMS.
It is possible that the effects of rTMS on presynaptic inhibition are related to peripheral feedback from muscle twitches elicited during the rTMS train of pulses. However, the SOL H-reflex was strongly suppressed with subthreshold rTMS stimulus intensities during which muscle twitches are not evoked (see Fig. 3A). Furthermore, if significant changes in peripheral feedback had been evoked, it would be expected to result in post-activation depression of the SOL H-reflex, which lasts for a period of >8 s (Crone and Nielsen 1989). This is much longer than the effects observed in this study, which only lasted 1 s. Experiments in the cat have demonstrated that the classic GABAergic presynaptic inhibition of Ia afferents lasts for no more than a few hundred milliseconds (Eccles 1964). How do we then explain the longer duration of the suppression of the H-reflex? Previous studies have shown that a single TMS pulse depresses transmission in the pathways, which mediate presynaptic inhibition of Ia afferents (Valls-Sole et al. 1994; Iles 1996; Meunier and Pierrot-Deseilligny 1998). Also, in the cat, a single stimulation of the sensorimotor cortex decreases presynaptic inhibition evoked in Ia terminals by stimulation of group I fibers from flexor muscles (Lundberg and Vycklicky 1963; Rudomin et al. 1983). A possible explanation for the long lasting increase of presynaptic inhibition observed in our study may then be that rTMS has produced a decrease in the spontaneous activity of corticospinal neurons. This is supported by observations in the cat demonstrating that repetitive high frequency stimulation to the motor cortex caused a decrease in spontaneous corticospinal neuron activity (Phillips 1956; Branch and Martin 1958; Krnjevic et al. 1966). Possibly, this continuous background corticospinal discharge maintains a relatively low level of presynaptic inhibition of the Ia afferents, so that when it is removed transiently following rTMS, presynaptic inhibition is increased. However, we observed an increase in the amplitudes of both TA and SOL MEPs, which goes against this idea. This increase may be explained by changes in cortical, interneuronal and/or spinal motoneuronal excitability. Previous evidence from the arm suggests that a train of rTMS pulses at 5 Hz increases motor cortical excitability (Berardelli et al. 1998) and reduces the excitability of intracortical circuits (Pienemann et al. 2000; Fierro et al. 2001; Di Lazzaro et al. 2002). From the present data it is unclear whether cortical excitability changes are also responsible for the increased MEPs in leg muscles following rTMS, but based on the data from the arm this would not be unlikely and would point to a larger rather than smaller corticospinal activity following rTMS. On the other hand, the increase of the MEP evoked by transmastoid stimulation was similar to the increase of the MEPs evoked by TMS, which indicates that increased spinal motoneuronal excitability rather than increased cortical excitability may be involved.
Implications for investigations of neuronal plasticity and rehabilitation
The observations in the present study suggest that rTMS may modify transmission in neuronal circuitries not only in the cortex, but also in deeper lying structures such as the basal forebrain, basal ganglia, and spinal cord through modification of the cortical projections to these structures. Such modifications may be of importance for the clinical effect of rTMS on depression (George et al. 2000), Parkinsonism and other basal ganglia disorders (Homberg et al. 1991; Ghabra et al. 1999; Siebner et al. 1999, 2000), and potentially also motor disorders involving the spinal cord. In fact, it was recently demonstrated that a train of rTMS pulses at 1 Hz could temporarily modify transmission in spinal cord circuitries in patients with dystonia (Huang et al. 2004).
Patients with spasticity present large H-reflexes at rest and during voluntary movement (Morita et al. 2001; Crone et al. 2003), which may contribute to the abnormal muscle activation during motor behaviors such as locomotion (Fung and Barbeau 1989). One of the most effective antispastic drugs, diazepam, acts by increasing presynaptic inhibition (Abbruzzese 2002). Our observation that rTMS increases presynaptic inhibition and thereby decreases the SOL H-reflex suggests that rTMS may potentially be of value in the treatment of spasticity. The depression of the H-reflex, however, lasted only about one second after the end of the train. For rTMS to be of any therapeutic value, effects lasting considerably longer than this are necessary. Future studies need to address if long-lasting changes in spinal cord excitability can be induced by trains of rTMS with durations longer than the four second trains used in the present study.
References
Abbruzzese G (2002) The medical management of spasticity. Eur J Neurol (Suppl 1) 9:30–34
Alstermark B, Isa T, Ohki Y, Saito Y (1999) Disynaptic pyramidal excitation in forelimb motoneurons mediated via C(3)-C(4) propriospinal neurons in the Macaca fuscata. J Neurophysiol 82:3580–3585
Amassian VE, Cracco RQ, Maccabee PJ, Cracco JB, Rudell A, Eberle L (1989) Suppression of visual perception by magnetic coil stimulation of human occipital cortex. Electroen Clin Neuro 74:458–462
Baker SN, Olivier E, Lemon RN (1994) Recording an identified pyramidal volley evoked by transcranial magnetic stimulation in a conscious macaque monkey. Exp Brain Res 99:529–532
Baldissera F, Hultborn H, Illert M (1981) Integration in spinal neuronal system. In: Brooks VB (ed) Handbook of physiology. Section 1: The nervous system. Vol 2: Motor control. American Physiological Society, Bethesda, MD, USA, pp 509–595
Berardelli A, Inghilleri M, Rothwell JC, Romeo S, Curra A, Gilio F, Modugno N, Manfredi M (1998) Facilitation of muscle evoked responses after repetitive cortical stimulation in man. Exp Brain Res 122:79–84
Branch CL, Martin AR (1958) Inhibition of Betz cell activity by thalamic and cortical stimulation. J Neurophysiol 21:380–390
Crone C, Nielsen J (1989) Methodological implications of the post activation depression of the soleus H-reflex in man. Exp Brain Res 78:28–32
Crone C, Hultborn H, Jespersen B, Nielsen J (1987) Reciprocal Ia inhibition between ankle flexors and extensors in man. J Physiol 389:163–185
Crone C, Hultborn H, Mazieres L, Morin C, Nielsen J, Pierrot-Deseilligny E (1990) Sensitivity of monosynaptic test reflexes to facilitation and inhibition as a function of the test reflex size: a study in man and the cat. Exp Brain Res 81:35–45
Crone C, Johnsen LL, Biering-Sorensen F, Nielsen JB (2003) Appearance of reciprocal facilitation of ankle extensors from ankle flexors in patients with stroke or spinal cord injury. Brain 126:495–507
Di Lazzaro V, Oliviero A, Profice P, Meglio M, Cioni B, Tonali P, Rothwell JC (2001) Descending spinal cord volleys evoked by transcranial magnetic and electrical stimulation of the motor cortex leg area in conscious humans. J Physiol 537:1047–1058
Di Lazzaro V, Oliviero A, Mazzone P, Pilato F, Saturno E, Dileone M, Insola A, Tonali PA, Rothwell JC (2002) Short-term reduction of intracortical inhibition in the human motor cortex induced by repetitive transcranial magnetic stimulation. Exp Brain Res 147:108–113
Eccles JC (1964) The physiology of synapses. Springer, Berlin Heidelberg New York, pp 220
Epstein CM, Lah JJ, Meador K, Weissman JD, Gaitan LE, Dihenia B (1996) Optimum stimulus parameters for lateralized suppression of speech with magnetic brain stimulation. Neurology 47:1590–1593
Fierro B, Piazza A, Brighina F, La Bua V, Buffa D, Oliveri M (2001) Modulation of intracortical inhibition induced by low- and high-frequency repetitive transcranial magnetic stimulation. Exp Brain Res 138:452–457
Fung J, Barbeau H (1989) A dynamic EMG profile index to quantify muscular activation disorder in spastic paretic gait. Electroen Clin Neuro 73:233–244
George MS, Nahas Z, Molloy M, Speer AM, Oliver NC, Li XB, Arana GW, Risch SC, Ballenger JC (2000) A controlled trial of daily left prefrontal cortex TMS for treating depression. Biol Psychiat 48:962–970
Ghabra MB, Hallett M, Wassermann EM (1999) Simultaneous repetitive transcranial magnetic stimulation does not speed fine movement in PD. Neurology 52:768–770
Gilio F, Rizzo V, Siebner HR, Rothwell JC (2003) Effects on the right motor hand-area excitability produced by low-frequency rTMS over human contralateral homologous cortex. J Physiol 551:563–573
Homberg V, Stephan KM, Netz J (1991) Transcranial stimulation of motor cortex in upper motor neurone syndrome: its relation to the motor deficit. Electroen Clin Neuro 81:377–388
Houlden DA, Schwartz ML, Tator CH, Ashby P, MacKay WA (1999) Spinal cord-evoked potentials and muscle responses evoked by transcranial magnetic stimulation in 10 awake human subjects. J Neurosci 19:1855–1862
Huang YZ, Edwards MJ, Bhatia KP, Rothwell JC (2004) One-Hz repetitive transcranial magnetic stimulation of the premotor cortex alters reciprocal inhibition in DYT1 dystonia. Movement Disord 19:54–59
Hultborn H, Illert M, Santini M (1976) Convergence on interneurones mediating the reciprocal Ia inhibition of motoneurones. III. Effects from supraspinal pathways. Acta Physiol Scand 96:368–391
Hultborn H, Meunier S, Morin C, Pierrot-Deseilligny E (1987a) Assessing changes in presynaptic inhibition of I a fibres: a study in man and the cat. J Physiol 389:729–756
Hultborn H, Meunier S, Pierrot-Deseilligny E, Shindo M (1987b) Changes in presynaptic inhibition of Ia fibres at the onset of voluntary contraction in man. J Physiol 389:757–772
Iles JF (1996) Evidence for cutaneous and corticospinal modulation of presynaptic inhibition of Ia afferents from the human lower limb. J Physiol 491:197–207
Iles JF, Pisini JV (1992) Cortical modulation of transmission in spinal reflex pathways of man. J Physiol 455:425–446
Iyer MB, Schleper N, Wassermann EM (2003) Priming stimulation enhances the depressant effect of low-frequency repetitive transcranial magnetic stimulation. J Neurosci 23:10867–10872
Jankowska E (1992) Interneuronal relay in spinal pathways from proprioceptors. Prog Neurobiol 38:335–378
Jankowska E, Padel Y, Tanaka R (1976) Disynaptic inhibition of spinal motoneurones from the motor cortex in the monkey. J Physiol 258:467–487
Kernell D, Hultborn H (1990) Synaptic effects on recruitment gain: a mechanism of importance for the input-output relations of motoneurone pools? Brain Res 507:176–179
Kobayashi M, Hutchinson S, Theoret H, Schlaug G, Pascual-Leone A (2004) Repetitive TMS of the motor cortex improves ipsilateral sequential simple finger movements. Neurology 62:91–98
Krnjevic K, Randic M, Straughan DW (1996) An inhibitory process in the cerebral cortex. J Physiol 184:16–48
Lundberg A (1975) Control of spinal mechanisms from the brain. In: Tower DB (ed) The nervous system. The basic neurosciences. Raven, New York, I:253–265
Lundberg A, Vyklicky L (1963) Inhibitory interaction between spinal reflexes to primary afferents. Experientia 19:247–248
Meunier S, Pierrot-Deseilligny E (1998) Cortical control of presynaptic inhibition of Ia afferents in humans. Exp Brain Res 119:415–426
Meunier S, Pierrot-Deseilligny E, Simonetta M (1993) Pattern of monosynaptic heteronymous Ia connections in the human lower limb. Exp Brain Res 96:534–544
Mizuno Y, Tanaka R, Yanagisawa N (1971) Reciprocal group I inhibition on triceps surae motoneurons in man. J Neurophysiol 34:1010–1017
Modugno N, Nakamura Y, MacKinnon CD, Filipovic SR, Bestmann S, Berardelli A, Rothwell JC (2001) Motor cortex excitability following short trains of repetitive magnetic stimuli. Exp Brain Res 140:453–459
Morita H, Crone C, Christenhuis D, Petersen NT, Nielsen JB (2001) Modulation of presynaptic inhibition and disynaptic reciprocal Ia inhibition during voluntary movement in spasticity. Brain 124:826–837
Nielsen J, Kagamihara Y (1993) Differential projection of the sural nerve to early and late recruited human tibialis anterior motor units: change of recruitment gain. Acta Physiol Scand 147:385–401
Nielsen J, Petersen N, Deuschl G, Ballegaard M (1993) Task-related changes in the effect of magnetic brain stimulation on spinal neurones in man. J Physiol 471:223–243
Pascual-Leone A, Tormos JM, Keenan J, Tarazona F, Canete C, Catala MD (1998) Study and modulation of human cortical excitability with transcranial magnetic stimulation. J Clin Neurophysiol 15:333–343
Peinemann A, Lehner C, Mentschel C, Munchau A, Conrad B, Siebner HR (2000) Subthreshold 5-Hz repetitive transcranial magnetic stimulation of the human primary motor cortex reduces intracortical paired-pulse inhibition. Neurosci Lett 296:21–24
Peinemann A, Reimer B, Loer C, Quartarone A, Munchau A, Conrad B, Siebner HR (2004) Long-lasting increase in corticospinal excitability after 1800 pulses of subthreshold 5 Hz repetitive TMS to the primary motor cortex. Clin Neurophysiol 115:1519–1526
Petersen NT, Pyndt HS, Nielsen JB (2003) Investigating human motor control by transcranial magnetic stimulation. Exp Brain Res 152:1–16
Phillips CG (1956) Cortical motor threshold and the thresholds and distribution of excited Betz cells in the cat. Q J Exp Physiol Cogn Med Sci 41:70–84
Rohan M, Parow A, Stoll AL, Demopulos C, Friedman S, Dager S, Hennen J, Cohen BM, Renshaw PF (2004) Low-field magnetic stimulation in bipolar depression using an MRI-based stimulator. Am J Psychiat 161:93–98
Rossini PM, Barker AT, Berardelli A, Caramia MD, Caruso G, Cracco RQ, Dimitrijevic MR, Hallett M, Katayama Y, Lucking CH et al. (1994) Non-invasive electrical and magnetic stimulation of the brain, spinal cord and roots: basic principles and procedures for routine clinical application. Report of an IFCN committee. Electroen Clin Neuro 91:79–92
Rudomin P, Jimenez I, Solodkin M, Duenas S (1983) Sites of action of segmental and descending control of transmission on pathways mediating PAD of Ia- and Ib-afferent fibers in cat spinal cord. J Neurophysiol 50:743–769
Siebner HR, Rothwell J (2003) Transcranial magnetic stimulation: new insights into representational cortical plasticity. Exp Brain Res 148:1–16
Siebner HR, Auer C, Ceballos-Baumann A, Conrad B (1999) Has repetitive transcranial magnetic stimulation of the primary motor hand area a therapeutic application in writer’s cramp? Electroencephalogr Clin Neurophysiol Suppl 51:265–275
Siebner HR, Rossmeier C, Mentschel C, Peinemann A, Conrad B (2000) Short-term motor improvement after sub-threshold 5-Hz repetitive transcranial magnetic stimulation of the primary motor hand area in Parkinson’s disease. J Neurol Sci 178:91–94
Touge T, Gerschlager W, Brown P, Rothwell JC (2001) Are the after-effects of low-frequency rTMS on motor cortex excitability due to changes in the efficacy of cortical synapses? Clin Neurophysiol 112:2138–2145
Valero-Cabre A, Oliveri M, Gangitano M, Pascual-Leone A (2001) Modulation of spinal cord excitability by subthreshold repetitive transcranial magnetic stimulation of the primary motor cortex in humans. Neuroreport 12:3845–3848
Valls-Sole J, Alvarez R, Tolosa ES (1994) Vibration-induced presynaptic inhibition of the soleus H reflex is temporarily reduced by cortical magnetic stimulation in human subjects. Neurosci Lett 170:149–152
Walsh V, Cowey A (2000) Transcranial magnetic stimulation and cognitive neuroscience. Nat Rev Neurosci 1:73–79
Acknowledgements
This work was supported by grants from the Danish Health Research Council, The Danish Society of Multiple Sclerosis, The Novo Nordisk Foundation and The Danish Ministry of Culture.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Perez, M.A., Lungholt, B.K.S. & Nielsen, J.B. Short-term adaptations in spinal cord circuits evoked by repetitive transcranial magnetic stimulation: possible underlying mechanisms. Exp Brain Res 162, 202–212 (2005). https://doi.org/10.1007/s00221-004-2144-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-004-2144-2