Abstract
In real life situations large gaze saccades may involve rotations of the trunk, as well as the eyes and head. When this happens the rotation of the head-in-space is similar whether or not the trunk is also rotating. However, the rotation of the head on the trunk (i.e. the neck movement) is very different in the two circumstances. For similar head-in-space rotations to occur, the neck and trunk movements cannot simply add independently: they must be coordinated. It is argued that this is achieved via a feedback loop in which the semi-circular canals monitor the rotation of the head-in-space, and the neck is driven by an error signal representing the difference between the intended head-in-space trajectory and the actual trajectory. This mechanism, which is essentially the same as the vestibulo-collic reflex, nulls out disturbances to the head-in-space trajectory, whether these are caused by active or passive trunk rotation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Most everyday activities, for example food preparation or carpentry, involve shifts of the point of gaze to the site of each new action (Ballard et al. 1993; Land et al. 1999; Hayhoe 2000; Johansson et al. 2001). Small shifts (<10°) may be made with eye saccades alone, larger shifts are made with eyes and head, and very large ones—for example from one work surface to another—may require trunk movements that can involve a few stepping movements. Often several eye saccades accompany these large turns, but frequently they are made with a single large eye saccade augmented by the head and trunk movements. These large eye-head-trunk saccades raise the question of how the three components of the movement are coordinated. A typical situation is represented in Fig. 1a. Eye, head and trunk start rotating more or less together (1 and 2), then when gaze reaches the target the vestibulo-ocular reflex (VOR) is switched on (Guitton et al. 2003), counter-rotating the eye and thus maintaining fixation (3 and 4). During the last phase (4 and 5) the target is in line with both eye and head, but the body continues to rotate. This means that the head must counter-rotate on the trunk until the trunk too reaches a direction more in line with the target. Figure 1b shows a real example of this sequence.
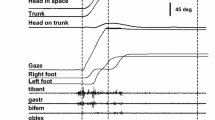
Stages in a large saccadic turn involving eye-in-head, head-on-trunk and trunk-in-space movements. 1–2 eyes, head and trunk all turn together; 2–3 eyes approach their target and VOR commences; 3–4 head approaches its final direction; 4–5 trunk continues to turn, with the neck counter-rotating, and finally stops. In this case eyes, head and body finish in line. b Five frames from the turn detailed in the first example of Fig. 2, taken at intervals of approximately 0.2 s. They correspond roughly to the five stages depicted in Fig. 1a
The coordination of large head and eye saccades was explored by Guitton and Volle (1987). For eye-head saccades of 10° or less Morasso et al. (1973) had shown that the trajectory of gaze is the sum of the saccadic eye movements that would have been made with the head still, and a signal from the semi-circular canals that is the opposite of the rotation made by the head (VOR). In this way the extent and time course of the gaze saccade is the same whatever the head does. However, for saccades beyond the oculomotor range (±50º) this will not work, because the eye cannot make the whole saccade on its own. Guitton and Volle (1987) showed that in these large saccades VOR is suspended, so that for at least part of the saccade gaze rotation results almost entirely from rotation of the head. When gaze reaches the target, VOR is re-instated, and fixation is maintained as the head completes its movement. The end-point of the saccade (VOR switched on) is reached when a pre-specified amount of gaze rotation is achieved, as determined by internal signals that monitor head rotation (particularly the semicircular canals, but possibly also neck proprioceptors or efference copy) and eye rotation (efference copy or muscle proprioceptors). Their sum gives gaze rotation (e.g. Guitton 1992; Guitton et al. 2003).
In this study I compare large gaze saccades made under natural conditions in two different situations. (1) Gaze saccades made by drivers at road intersections. These gaze saccades can be as large as 180°, and are made with the head free but the trunk fixed by a seat belt, and the car stationary. (2) Gaze saccades made during food preparation in a kitchen, where the subject turns from one work surface to another. Here both trunk and head are free to move. Both sets of recordings were made for other purposes, and at the time neither the subjects nor the experimenter were aware that they would be used to study the coordination of turns. Although consciously controlled head saccades of the same size can vary in velocity over a six-fold range (Guitton and Volle 1987), in the present study the corresponding range was less than 1.5-fold, presumably because without conscious intrusion a natural optimum velocity becomes apparent. (The term “saccade” is used to here mean an eye-in-head saccade. “Head saccade” is the rapid head movement that accompanies an eye saccade and “gaze saccade” is the change in gaze direction resulting from a combined eye and head saccade. Trunk rotations are less closely related to the eye saccades and the term saccade is not used for them).
The velocity profile of the head in large (trunk-fixed) head saccades is closely related to their size. Zangemeister et al. (1981), and many authors since, have shown that when subjects are asked to rotate the head fast between two targets the trajectory is sigmoid, the peak velocity being an almost linear function of size. Unlike eye saccades, the duration of head saccades was almost independent of size, at about 400 ms. Thus, although head movements of almost any form can be made at will, and the coupling between head and eye may vary with the task (for example, Kowler et al. (1992) found that during reading head and eye could move in opposite directions), it seems that during large saccades there is a definite “default condition” in which head movements have a pre-planned time course predicted by gaze saccade size. The question I ask here is: how is this pattern of head movements modified when part of the head-in-space movement is achieved by rotation of the trunk?
There are a number of possible outcomes. Head saccades and trunk rotations could be “co-programmed” so that each performs, say, half the total turn; the head might make a complete turn (as in trunk-fixed circumstances) and when that was complete counter-rotate as the trunk caught up; the head might stay in line with the trunk and let the latter make the whole turn on its own; or the neck might receive a dual command, to make the turn (as in the trunk-fixed condition) and to have the contribution of the trunk subtracted from it continuously throughout the turn via a reflex mechanism. All the evidence in this paper suggests that the last of these suggestions is the correct one. The main result that leads to this conclusion is that the head-in-space trajectories are very similar whether or not the trunk is free to move, but the head-on-trunk trajectories are quite different. The origin of the compensatory command which subtracts the trunk movement is examined in the Discussion, and it is argued that it is achieved via the same vestibular feedback loop responsible for the vestibulo-collic reflex (Guitton et al. 1986)
Materials and methods
The results in this paper come from data obtained during a study of eye-movements and vision during tea-making (Land et al. 1999), and from a recent study of eye movements during urban driving (in preparation). Three subjects (ML male 55, SF female 28, JB male 46) made cups of tea in a small kitchen at the University of Sussex. They were equipped with a light-weight head-mounted eye tracker which provided a video record of the view from the head, and in the bottom of the same image a view of the left eye in its socket. The video was processed frame-by-frame to extract the position of the iris. This was done by matching the image of the eye to a rotatable computer model of the eye, which provided coordinates of eye direction via a calibration routine. These coordinates were then used to position a dot, corresponding to foveal direction, on the scene part of the video, which was then re-recorded. The accuracy was about 1°. Eye movements during blinks were determined approximately by tracking the corneal bulge, and by interpolation, but cannot be considered accurate. In addition, head-in-space records were obtained from the movement of objects across the field of view of the head-mounted camera, as the head rotated. These movements were again measured to an accuracy of about one degree. In principle a correction is required to compensate for translational movements, but significant translation rarely occurred until rotation was almost complete, and no correction was applied. Both eye and head records had a time resolution of 20 ms. See Land (1993) or Land et al. (1999) for further details.
In addition to the eye-camera records, a second tripod-mounted video camera in the corner of the room monitored the actions of the subjects. It was these videos that provided information about trunk-in-space orientation. A doll model of the subject (Barbie or Ken), equipped with a backpack like that worn by the subject, was mounted on a calibrated turntable in front of the screen used to view the video). The observer sat in a standard position and rotated the model so that its orientation matched that of the subject. This proved to be surprisingly easy and adequately accurate (mean absolute error was ±3.1°, based on the bearings of a magnetic compass attached to a subject rotating through 360° at 20° intervals). More elaborate methods were tried (for example, fitting a sine function to the width across the shoulders normalised for head height) but these were no more accurate than direct matching. The action video was synchronized with the eye-movement video, and trunk measurements were made every 0.1 s. Head-on-trunk records were obtained by subtracting the trunk-in-space from the head-in-space records, again with a time resolution of 0.1 s.
The three tea-making sessions yielded a total of about 800 saccades but the vast majority of these were small, less than 20 degrees. These were discarded, leaving a total of 58 larger saccades that were analysed for eye, head and gaze movements. All of these were accompanied by trunk movements, but in only 32 could the trunk movements be measured reliably. The remainder were not used either because the scene camera moved to track the subject, or the trunk movement was partially out of the field of view, or there was too much translational movement by the subject. They were, however, used for head velocity measurements. No attempt was made to distinguish trunk movements made by body twists as opposed to stepping. However the videos showed that turns of 90° or larger were usually produced by two to four foot movements, and many smaller turns involved two foot movements.
In the car study 55 saccades of various sizes were obtained at four intersections on suburban roads in Sussex, UK, by the same three subjects as in the tea-making study. The car was either stationary at the time or moving slowly forwards. Eye movements were monitored using the same device as for tea-making, and head movements were monitored by measuring the changing positions of small pieces of tape attached to the windscreen, which give head-in-car (i.e. neck) movements (see Land 1992; Land and Lee 1994). For the observations in which the car was progressing round a corner, head-in-space rotations were also monitored by measuring the changing positions of distant objects in the direction of the car’s heading (this avoids contamination by translational movements). The difference between head-in-space and head-in-car measurements gives car-in-space rotation (Land and Tatler 2001).
Statistical methods used for testing the significance of differences in regression, correlation and variance were those given by Bailey (1959).
Results
The behaviour of eyes, head and trunk
Figure 2 shows three examples of gaze saccades of about 130° made by three subjects in turns from one work surface to another in a kitchen. All three involve rotations of the trunk in which the feet move (two to four foot movements). In all three cases the principal gaze saccade landed close to an object involved in the task, which was then fixated by small secondary saccades (in the first two examples). In all three the main saccade was accompanied by a blink (shaded). Figure 1b illustrates five phases of the first turn shown in Fig. 2.
Records of three large combined eye-head-trunk saccades made by different subjects when turning between work-surfaces in a kitchen. Eye-in-head and head-in-space records were both obtained from the eye-tracker video, and the gaze record is their sum. The trunk-in-space record was obtained from a separate synchronised video. The head-on-trunk record was obtained by subtracting the trunk-in-space record from the head-in-space record. In record SF tea-bags are moved from one bench to another; in JB an kettle is moved from a counter to the sink; and in ML a teapot is moved to the sink. Further detail in text
The eye movements in Fig. 2 are very much as described by Guitton and Volle (1987) . The eyes begin to move about 200 ms later than the head, and travel for about 40° before reaching a plateau position, which is maintained for 100–200 ms. They then abruptly reverse their initial direction (as the vestibulo-ocular reflex—VOR—is turned on, indicated by the arrow) and move back towards the primary position at a velocity similar to and opposite in direction to the movement of the head-in-space. The result is that gaze is held almost stationary.
Head-in-space movements follow a smooth sigmoid trajectory in all three examples. The maximum velocity is in the range 250–350°/s and occurs about 350 ms from the start of the head movement, and about 150 ms from the beginning of the eye saccade. In the third example, where there are no secondary saccades, the sigmoid is symmetrical, and the whole head movement has a duration of about 650 ms. These numbers are consistent with medium-speed head saccades recorded by others in controlled conditions (e.g. Guitton et al. 2003; Fig. 2).
Trunk-in-space movements are more variable, with amplitudes of 135, 105 and 50° in the three examples. Maximum velocities range from 100–250°/s, but the velocity varies throughout the movement as the subject makes stepping movements. Head-on-trunk (neck) rotations are also variable (geometrically, they are the difference between head-in-space and trunk-in-space movements). In the first two examples the neck first rotates in the direction of the overall gaze saccade, and then starts to counter-rotate (see Fig. 1) soon after the initiation of VOR. The neck then rotates towards the primary position (head in line with trunk; 0° on ordinate). The third example is different as there is no counter-rotation of head on trunk. This is because the head starts from a position 60° out of line with the trunk, against the direction of the turn as a whole. This initial bias means that the neck movements made during the saccade bring the head approximately into line with the trunk without the necessity for counter-rotation. Not all turns end with the head more or less in line with the trunk, as in the three examples here. There were several cases where a subject turned to open a door, finishing with the trunk at 90° to the door but with the head facing towards it.
The observations in this section suggest that whereas head-in-space rotations for gaze saccades of a given size vary rather little in their trajectories, the corresponding head-on-trunk rotations are very diverse and depend to some extent on the task that the trunk is engaged in at the time.
Comparison of natural head movements with trunk either free to turn or immobile
Figure 3 shows the head-in-space trajectories made by each subject for three saccades of different but roughly corresponding sizes, made in the car and in the kitchen. For comparison, the head-on-trunk (neck) rotations are also shown for the same gaze saccades made in the kitchen. It is clear that, qualitatively at least, the head-in-space trajectories in the two situations are very similar, and follow a consistent pattern; as others have found, the peak velocities of the trajectories increase with gaze saccade size (see Fig. 5), but the durations vary rather little. However, the neck movements (head-on-trunk) are totally different in the two situations. In the car they are practically the same as the head-in-space movements because the driver’s trunk is belted to the seat, but in the kitchen they bear little resemblance to the head-in-space trajectories. As in the examples in Fig. 2, they mostly begin by rotating in the direction of the saccade, but then either counter-rotate, or flatten off. Neither their starting and finishing positions, nor their amplitudes, are related in any obvious way to the magnitudes of the head-in-space movements. The tentative conclusion is that head-in-space trajectories are controlled by the same process in both situations, but that the control of the neck itself is different.
Records of head-in-space saccades of three different amplitudes made at road intersections in a car, with the trunk fixed by a seat-belt (left), and three similar sized saccades made in the kitchen with the trunk free to move (centre). On the right are shown the head-on-trunk movements corresponding to the head-in-space rotations in the kitchen. The vertical lines mark the beginning of the eye (and gaze) saccade, and in all cases the head movements begin 100–200 ms earlier than the eye movements. In the head-on-trunk records the zero ordinate is the condition with head and trunk aligned, so in some the head lags the trunk and in others it leads. Same three subjects in both recordings. Note the similarities of the head-in-space trajectories, and the variability of the head-on-trunk trajectories
The head-in-space trajectories, however, are not quite the same in the two situations. Those made in the kitchen have, in general, a more drawn out final phase than those made in the car. One reason for this is that in the car a head saccade in one direction is usually followed by one in the opposite direction, the beginning of which brings the first head saccade to rapid halt (Fig. 4). In the kitchen, on the other hand, a large saccade is almost invariably followed by a secondary saccade in the same direction (first two examples in Fig. 2). The end of the first head saccade merges with the beginning on the next so that, in this case, the head trajectory does not flatten off (see, however, the third example in Fig. 3).
Eye head and gaze saccades made at an intersection. In the two large saccades between seconds 1 and 2 the head saccades are curtailed by the beginning of the following saccade, whereas the final large saccade is prolonged by the small saccade that follows it. It is rare, in either the car and kitchen, to get single head saccades that begin and end perfectly cleanly. The slope of the gaze fixations between saccades is due to the car moving slowly forwards, resulting in some smooth tracking of fixated objects to the side. Note the reciprocity of eye and head records (VOR) during the gaze fixations
The second difference is that the head-in-space saccades made in the car appear to be slightly faster than those made in the kitchen. Figure 5 compares the maximum velocities of all the head saccades made in the car (filled circles) with those in the kitchen (open circles), as a function of gaze saccade size, for all three subjects. In both situations there is a very high correlation (r ranges from 0.93 to 0.98) between maximum velocity and gaze saccade size. For trunk-fixed head saccades, this has been a consistent finding by all authors since Zangemeister et al. (1981). However, the slopes of the regression lines for the head saccades made in the car are greater than those made in the kitchen in all three cases. The differences are small, and the situation is complicated by the fact that the regression lines cross, but at least for subject JB the difference is significant (p<0.05). Gaze saccades at intersections are made under greater time pressure than those in the kitchen, and one would imagine that this might affect the speed of their execution. What is perhaps surprising is that the difference is as small as it is: a ratio of less than 1.3:1, compared with about 6:1 for deliberately made head saccades (Guitton and Volle 1987)
Maximum head-in-space angular velocities for saccades of different sizes, for three subjects. The maximum occurs approximately 150 ms after the start of the eye-saccades (see Figs. 2, 3). Closed circles saccades made in the car; open circles saccades made in the kitchen. All distributions show a high correlation (r between 0.93 and 0.98) and have similar slopes, although the difference in slope between car and kitchen is significant for JB (p<0.05). Most of the scatter is due to real differences in speed, rather than measurement error
Neck movements during different phases of a turn
In the head-on-trunk records in Figs. 2 and 3 there are two phases. During the first phase, which lasts about 0.3–0.4 s, the neck rotates in the direction of the turn. This is followed by a second phase in which the neck either counter-rotates or does not rotate further. During the second phase the head-in-space rotation is slowing down or has stopped, meaning that the action of the neck is almost entirely to compensate for the continued rotation of the trunk, which in most cases continues to turn. I shall return to origins of the compensatory signal in the Discussion. The question here is whether the compensatory command is present throughout both phases of the turn, or only during the second part.
Concerning the first phase there are two possibilities. If trunk-in-space and head-on-trunk rotations are controlled by independent mechanisms, then the head-in-space rotation will simply be the sum of the contributions of the two rotations (the additive condition). However, if the head-in-space rotation is compensated for the rotation of the trunk, by subtraction of the consequences of the commands for trunk-in-space rotation from the head-on-trunk rotation, then head-in-space rotation should be independent of trunk-in-space rotation (the subtractive condition). Figure 6 shows that head-in-space, head-on-trunk and trunk-in-space angular velocities, measured at the time of the maximum head-in-space velocity, all correlate to different degrees with the size of the saccadic gaze change. Both the slopes of the regression lines and the correlation coefficients decrease in the order head-in-space, head-on-trunk and trunk-in-space. The fact that head-in-space velocities are similar whether or not the trunk rotates (Fig. 5) suggests that head-in-space rotation is independent of trunk-in-space rotation (subtractive condition), but we do not know for sure what the slope of the regression line for head-in-space vs. gaze change would be if the trunk were immobile in the kitchen situation. It is still possible that the head-on-trunk velocity commands in this situation are considerably slower than in the car, and that when the trunk-in-space velocities are added this brings them up to those seen in the car. For this reason the slope of the head-in-space vs. gaze saccade size distributions cannot be used directly as evidence for or against compensation.
Relations between gaze saccade size and head-in-space, head-on-trunk and trunk-in-space velocities, all measured in a 200 ms period around the time of maximum head-in-space velocity, approximately 150 ms after the start of the gaze saccade. Combined data from all three subjects, using all 32 saccades for which reliable trunk-in-space data was obtained. The head-in-space distribution has the lowest scatter, and the head-on-trunk the highest. This is inconsistent with a model in which the head-in-space velocity is simply the sum of the other two, which is what simple geometry suggests. See text for further details
The variation about the regression lines in Fig. 6 is more informative. If head-in-space is not compensated (additive condition), then its residual variance (after the main gaze change regression has been removed) should be the sum of the residual variances of the head-on-trunk and trunk-in-space distributions, and it should thus be more variable than the head-on-trunk distribution. On the other hand, if head-in-space rotation is compensated, by having an estimate of the trunk-in-space rotation subtracted from the head-on-trunk rotation (subtractive condition), then the residual variance of the head-in-space distribution will not contain that of the trunk-in-space distribution. This will, instead, be added to the head-on-trunk distribution (although this is a subtraction, the variances add). Since the head-on-trunk variance now contains the residual variances of both the head-in-space and the trunk-in-space distributions, it should be approximately equal to the sum of the residual variances of both distributions. In this case the head-in-space distribution should show less scatter than the head-on-trunk distribution. Figure 8 shows that this is the case. There is clearly more scatter in the head-on-trunk than the head-in-space distribution, and this is reflected in the correlation coefficients. The residual variance of the head-on-trunk distribution is close to the sum of the other two distributions. The difference in the residual variances between head-in-space and head-on-trunk is significant (p<0.05). Furthermore, two of the points in the head-on-trunk plot are actually negative, i.e. the neck is turning in the opposite direction to the gaze movement, and this could not arise if the neck only received a command to turn in the direction of gaze rotation. These two points are associated with high rates of turn by the trunk, resulting in appropriate compensation in the opposite direction.
A slight complication is that trunk-in-space (and hence head-on-trunk) data contain additional measurement error not present in the head-in-space data. The extent of this error was estimated from repeated measurements of rotational velocity over the 200 ms period centred on the maximum head-in-space velocity. The standard deviations of these measurements averaged 16°/s, which introduces a variance of approximately 10% to the residual variances of the distributions themselves. This has a negligible effect on the statistics, and does not affect the conclusions given in the previous section.
It is concluded that the neck muscles compensate for rotations of the trunk not only at the end of the movement, when the head has stabilized but the trunk continues to rotate, but also during the initial active phase of the turn, when eyes, body and trunk are all rotating in the same direction.
Discussion
The coupling of head and trunk movements
During natural tasks the head-in-space movements made during a gaze saccade of a given size are very similar, qualitatively and quantitatively, whether or not the trunk is also contributing to the overall rotation (Figs. 3, 5). However, the neck movements (head-on-trunk) are very different in the two conditions. With the trunk fixed the head-on-trunk and head-in-space movements are necessarily the same, and show the same sigmoid trajectory as described by previous authors. When the trunk is free to move, however, the neck initially rotates the head in the same direction as the overall turn, but then it either counter-rotates, or stops rotating. The fact that head-in-space movements are almost the same under both circumstances implies that the movements of the neck are in some way modified to take into account the movements of the trunk.
Where does the compensatory signal to the neck come from?
There seem to be four possible ways that a signal compensating for trunk movement might be derived. (1) The rotations of the trunk and neck might be co-programmed, in the sense that both neck and trunk effectors receive pre-determined commands to make complementary contributions to the turn. (2) The neck muscles might receive a signal derived from the actual motion of the trunk, coming either from proprioceptors in the trunk and legs, of from efference copies of the commands to the trunk and legs. (3) The neck might be controlled via a feedback system which monitors head position in space. The natural sensor for this would be the vestibular system. (4) Alternatively, vision itself could provide the feedback required to control head position.
Co-programming
In principle, head and trunk movements could each have pre-computed trajectories that operate throughout a turn of a given size. The main objection to this is that the trunk movements for gaze saccades of similar magnitudes can be very variable, and so inevitably are the neck movements (Figs. 2, 4). An examination of all turns in the range 90–130° showed that the starting and finishing positions of the head relative to the trunk, and the velocity profile of the neck during the turn are all highly variable, even though the head-in-space trajectories are very similar. The strong impression is that the head-in-space and trunk-in-space systems have different agendas, and the trunk-in-space trajectories depend on what has to be accomplished as a result of the turn. Co-programming, in the sense that there is a constant contribution of the two systems to a turn of a given size, is clearly not the answer.
Proprioception and efference copy
Mechanically, the head and trunk movements are related by:
This means that the only way for head-in-space movements to be the same whether or not the trunk also rotates is for the head-on-trunk movements to be composed of two components. One is the intended trajectory of the head-in-space, and the other is a compensatory rotation that is equal and opposite to the trunk-in-space rotation. Hence:
where the asterisk indicates that these are internally generated commands. If the trunk does not move, then the neck simply executes the intended head-in-space trajectory, but if the trunk does rotate the neck reduces its own rotation by an amount equal to the trunk rotation.
The compensatory signal (trunk-in-space*) that ensures that the head-in-space trajectories are similar, no matter how the trunk moves during the turn, could be derived either from proprioceptors in the trunk and legs that monitor their movements, or alternatively from copies of the commands to move the trunk and leg muscles.
Some combination of proprioception and efference copy signals is also possible. There is nothing in the present study that rules out these possibilities. However, some evidence against both proprioception and efference copy emerged from a parallel study of the behaviour of drivers steering round right angle bends (Fig. 7). Experienced drivers, when steering round a corner, first direct their gaze into the bend, with a large head movement. Then, about halfway through the bend the head begins to straighten up, with the neck counter-rotating, and the head-in-space staying more or less steady as the neck compensates for the continued rotation of the car (Fig. 7b). This behaviour was always observed (four corners taken by four drivers). It has remarkable similarities to the last phase of large trunk-free saccadic turns (Fig. 7a), where head-in-space is held almost still by neck movements that compensate for trunk rotation. If the compensatory mechanism is the same in the two cases, then this tends to rule out either proprioception or efference copy, since the rotation of the car provides neither. (Although the operation of the steering wheel might provide some proprioceptive feedback or indeed efference copy information, the wheel is not actually rotating during most of the compensatory phase of the manoeuvre.)
Records of a large saccadic turn made during tea-making (top), and a record of driver’s head movements whilst turning a right angle corner in a car (below). From about 1.5 s in the tea-making record trunk-in-space and head-on-trunk records show near perfect reciprocity, indicating that neck movements compensate for trunk movements. In the car record, after 3 s, the car-in-space record (effectively the driver’s trunk) and the head-in-car record (neck) show very similar reciprocity, resulting in near-stabilization of the head in space. This demonstrates that compensation by the neck is unlikely to depend on proprioceptive feedback or efference copy from the trunk
Vestibular feedback control
Both vestibular and visual feedback mechanisms for stabilizing the head against rotations of the body are known. The vestibular mechanism is the vestibulo-collic reflex (VCR), in which signals from the vestibular system, indicating departures of the head from zero velocity, are fed back negatively to the neck muscles, via the vestibulo-spinal and vestibulo-reticulo-spinal pathways (Wilson and Melvill Jones 1979). Guitton et al. (1986) investigated the ability of subjects to stabilize their heads on a rotating platform with and without visual input. Their principal conclusion was that visual fixation and the vestibular feedback system play roughly equal roles in stabilizing the head. A subject with bilateral vestibular loss was unable to stabilize the head when blindfolded. Interestingly, performing mental arithmetic almost abolished the vestibular component of the stabilization, implying that this is under a degree of conscious control.
Figure 8a illustrates the basic mechanism of the vestibulo-collic reflex. As emphasised by Outerbridge and Melvill Jones (1971), who first studied the dynamics of the reflex in humans, this is a feedback loop in which the compensatory movement of the head cancels out the error signal generated by the semi-circular canals. It is thus quite unlike the vestibulo-ocular reflex (VOR) which is an open (feed-forward) system in which the movements of the eyes themselves have no effect on the sensor—the semi-circular canals (Carpenter 1988) . In a simple stabilizing version of the loop the reference input is zero velocity, the aim of the loop being to keep the head still Fig. 8a). However, this need not be the case. In the version of the loop proposed here (Fig. 8b) the input is not zero, but a command to move the head with a sigmoid trajectory, whose amplitude depends on the size of the gaze saccade to be made. The input to the neck muscles then corresponds to the difference between this command and the signal coming back from the semi-circular canals indicating the progress of the trajectory. In this scheme the rotation of the head caused by rotation of the trunk is simply a disturbance to the loop, and if the loop gain is adequate it will be removed. The loop thus acts as a follow-up servo, obeying the input command and opposing disturbances.
Feedback diagrams showing a the basic organization of the vestibulo-collic reflex, seen as a loop for preventing rotation of the head in spite of disturbance from trunk-in-space movements, and b the same loop seen as a follow-up servo, where the reference input to the loop is the planned trajectory of the head-in-space. See text
The equivalence of this arrangement to one in which compensation is provided by a direct signal concerning trunk rotation can be seen by determining the output of the feedback loop. In Fig. 8b the input to the loop is the planned head-in-space trajectory (H/S*), where the asterisk denotes that this is the desired state. The neck muscles receive an error signal equal to H/S* minus the estimate by the semi-circular canals of the current head-in-space position (I assume for convenience that the system works on positions but it could equally well work with velocities). This in turn is equal to the head-on-trunk (H/T) position + the trunk-in-space position (T/S). Thus the error signal to the to the neck muscles is:
The output from the neck is this multiplied by the gain of the neck plant g, which we assume to be large, and this produces H/T. Hence:
This simplifies to:
And if g is large (g/(1+g)) tends to 1, so that:
Thus the neck produces an output that is the desired head-in-space trajectory with the contribution of the trunk subtracted from it.
Visual feedback control
The role of vision is harder to assess. Because the eyes are mobile in the head (unlike the semi-circular canals) it is not clear that a fixation mechanism would produce the same result as vestibular feedback. Gaze fixation would not necessarily mean head fixation. There are numerous instances, both in the kitchen and the car records, where small fixation shifts are made during the stabilized final phase of a turn without this affecting the reciprocal relationship between trunk-in-space and head-on-trunk trajectories. This rules out stabilization by the use of simple fixation on a single target. A stronger argument against vision having the central role in compensation for trunk rotation is that compensation also occurs during the early part of each turn (Fig. 6) when, in gaze saccades larger than about 60°, the eyes are usually engaged in a blink (Fig. 2). However, as Guitton et al. (1986) found in their study, vision may still augment the vestibular mechanism, in a manner that has yet to be determined.
Conclusions
A role for the vestibular system in behaviour involving active body rotation has often been proposed before. In their study of the vestibulo-collic reflex Outerbridge and Melvill Jones (1971) remarked: “This suggests that during rapid angular movements of the body such as often occur in natural life, compensation through vestibularly controlled head movement may play an important role.” Berthoz (1991), in reviewing stabilization behaviour, especially during motion in the sagittal plane in humans, proposed “that the vestibular system is the fundamental egocentric (and geocentric) reference plane to which many if not all motor frames and the part of the visual system that deals with motion are spatially tuned.” This view is supported by the disorganization of head stabilization that occurs in patients with vestibular deficits (Pozzo et al. 1991). A complex stabilizing role of the vestibular system, involving both VCR and VOR, was demonstrated by Imai et al. (2001) in a study of eye, head and trunk rotations about all three axes during walking. They found that compensatory neck movements reduced the 3.5° of body yaw during each stride to less than 1° of head yaw, and (through VOR) to less than a quarter of a degree of gaze yaw. Head compensation also occurred during turns made on a 50 cm radius, similar to those described here. Imai et al. (2001) conclude: “Thus, while the movement of the legs and hips essentially governed the body, the head and eyes were governed by spatial maps....” That conclusion can also be drawn from the present study. It seems that whilst the vestibulo-ocular reflex emancipates the eyes from movements of the head, it is the vestibulo-collic reflex that liberates the head from the behaviour of the trunk. The results here also indicate that the vestibular system is concerned with the execution of head movements during large gaze changes, and not just with the stabilization of the head during the periods between them.
References
Bailey NTJ (1959) Statistical methods in biology. Hodder & Stoughton, London
Ballard DH, Hayhoe MM, Li F, Whitehead SD (1993) Hand-eye coordination during sequential tasks. Phil Trans R Soc Lond B 337:337–339
Berthoz A (1991) Reference frames for the perception and control of movement. In: Paillard J (ed) Brain and space. Oxford University Press, Oxford, pp 81–111
Carpenter RHS (1988) Movements of the eyes. Pion, London
Guitton D (1992) Control of eye-head coordination during orienting gaze shifts. Trends Neurosci 15:174–179
Guitton D, Volle M (1987) Gaze control in humans: eye-head coordination during orienting movements to targets within and beyond the oculomotor range. J Neurophysiol 58:427–459
Guitton D, Kearney RE, Wereley N, Peterson BW (1986) Visual, vestibular and voluntary contributions to human head stabilization. Exp Brain Res 64:59–69
Guitton D, Bergeron A, Choi WY, Matsuo S (2003) On the feedback control of orienting gaze shifts made with eye and head movements. Prog Brain Res 142:55–68
Hayhoe M (2000) Vision using routines: a functional account of vision. Visual Cognition 7:43–64
Imai T, Moore ST, Raphan T, Cohen B (2001) Interaction of the body, head, and eyes during walking and turning. Exp Brain Res 136:1–18
Johansson RS, Westling G, Bäckström A, Flanagan JR (2001) Eye-hand coordination in object manipulation. J Neurosci 21:6917–6932
Kowler E, Pizlo Z, Zhu G, Erlekens CJ, Steinman RM, Collewijn H (1992) Coordination of head and eyes during the performance of natural (and unnatural) visual tasks. In: Berthoz A, Graf W, Vidal PP (eds) The head-neck sensory-motor system, Chap 65. Oxford University Press, Oxford
Land MF (1992) Predictable eye-head coordination during driving. Nature 359:318–320
Land MF (1993) Eye-head coordination during driving. Proc IEEE Systems Man and Cybernetics Conf, Le Touquet, vol 3, pp 490–494
Land MF, Lee DN (1994) Where we look when we steer. Nature 369:742–744
Land MF, Tatler BW (2001) Steering with the head: the visual strategy of a racing driver. Curr Biol 11:1215–1220
Land MF, Mennie N, Rusted J (1999) The roles of vision and eye movements in the control of activities of daily living. Perception 28:1311–1328
Morasso P, Bizzi E, Dichgans J (1973) Adjustments of saccade characteristics during head movements Exp Brain Res 16:492–500
Outerbridge JS, Melvill Jones G (1971) Reflex vestibular control of head movement in man. Aerospace Med 42:935–940
Pozzo T, Berthoz A, Lefort L, Vitte E (1991) Head stabilization during various tasks in humans. II. Patients with bilateral vestibular deficits. Exp Brain Res 85:208–217
Wilson VJ, Melvill Jones G (1979) Mammalian vestibular physiology. Plenum Press, New York
Zangemeister WH, Jones A, Stark L (1981) Dynamics of head movement trajectories: main sequence relationship. Exp Neurol 71:76–91
Acknowledgements.
I thank Julia Horwood for technical assistance with the Video analysis. Ben Tatler read and made valuable comments on the manuscript. This study was supported by a grant from the EPSRC (UK).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Land, M.F. The coordination of rotations of the eyes, head and trunk in saccadic turns produced in natural situations. Exp Brain Res 159, 151–160 (2004). https://doi.org/10.1007/s00221-004-1951-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-004-1951-9