Abstract
Sodium silicate (Si) at 100 mM was used as a postharvest treatment agent of induction resistance on muskmelon (Cucumis melon L. cv. Yindi) to investigate the mechanism of controlling pink rot disease, which caused by Trichothecium roseum. Si treatment significantly reduced (P < 0.05) the lesion diameter of melons inoculated with T. roseum during storage. Si treatment increased the content of superoxide (O •−2 ) and could be further raised by challenged with T. roseum inoculation. The content of hydroxyl radical (·OH) in inoculated fruit was also increased. Both malondialdehyde (MDA) and hydrogen peroxide (H2O2) were also accumulated with Si treatment and challenged inoculation. Si treatment maintained membrane integrity in non-inoculated fruit, as compared to untreated control. Si treatment and challenge inoculation significantly (P < 0.05) increased the activity of superoxide dismutase (SOD), glutathione reductase (GR), peroxidase (POD), and polyphenoloxidase (PPO), while markedly decreased the activity of catalase (CAT) and ascorbic peroxidase (APX). Si treatment and challenge inoculation also enhanced the content of ascorbic acid (ASA) and glutathione (GSH). These findings suggested that the effects of sodium silicate on postharvest disease in muskmelon fruit may be associated with the elicitation of antioxidant defense system in fruit.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Muskmelon (Cucumis melo L.) is an economically important crop in northwest of China. The fruit are susceptible to postharvest decay caused by several pathogenic fungi. Pink rot, caused by Trichothecium roseum, was reported as one of the principal postharvest diseases of muskmelons. The rot can be controlled by the use of synthetic fungicides. However, due to problems related to fungicide toxicity, development of fungicide resistance by pathogens, and potential harmful effects on the environment and human health, new strategies for controlling postharvest diseases have been proposed.
Induction of fruit resistance to infection of pathogens has been evaluated as a promising approach for controlling postharvest diseases [1]. Resistance in fruits and vegetables can be induced through using chemical elicitors [2]. Silicon (Si) is the second most abundant element in the Earth’s crust [3]. The ability of soluble silicon to reduce the impact of fungal diseases has been amply described in the plant of wheat [4, 7], dicots [5], rice [6], cucumber [8], zucchini squash [9], and in the harvested crop of cherry [10], muskmelon [11], and potato [12]. Si is considered to be fungistatic in inhibiting the pathogens [10], and it is also considered to be biologically active and to trigger a faster and more extensive deployment of plant natural defenses [13]. It is reported that Si may act by eliciting biochemical defense reactions, including the accumulation of lignin, phenolic compounds, aconitate, methyl aconitate, and pathogenesis-related proteins in the infected plants [7, 8, 10].
Fawe et al. [5] suggested a model to explain how Si would play a role in induced resistance. According to their model, Si bioactivity was compared to that of known activators/secondary messengers of SAR whereby it would act as a modulator influencing the timing and extent of plant defense responses. Reactive oxygen species (ROS) have been found to play a critical role in activating defense mechanisms during plant and pathogen interactions [14]. Production of ROS has been considered a central event in the activation of disease resistance [15]. However, whether Si treatment activated antioxidant defense responses in plants and postharvested fruits was not found.
The objective of this study was to investigate whether Si is responsible for the elicitation of the oxidative burst during the storage after treatment and challenged by inoculation with T. roseum. In addition, the possible role of MDA content and membrane integrity involved in the resistance of “Yindi” muskmelons to this pathogen were also examined.
Materials and methods
Plant material
Muskmelon (Cucumis melo L. cv. Yindi) fruits were harvested at physiological maturity (45 days after full bloom) from Minqin County in Gansu Province, China. After harvest, fruit were sorted for uniform size and absence of obvious injuries, packed individually in 30-cm-long foam net bag, put in the standard melon-shipping boxes (six melons/box), transported to the laboratory arriving within 24 h of harvest, and stored at room temperature (22 ± 2 °C) with a relative humidity (RH) of 55–60%.
Pathogen source and inoculums production
T. roseum was isolated from infected melons and maintained on potato dextrose agar (PDA) at 4 °C. Conidia of the pathogens were obtained from 10-day-old PDA cultures incubated at 25 °C. To obtain conidia, the cultures were flooded with sterile water containing 0.05% Tween 80 and rubbed gently with a glass rod. The spore suspension was filtered through four layers of sterilized cheesecloth and adjusted to a concentration of 1 × 105 cfu/mL using a hemocytometer. Mycelium was removed from the suspensions by filtering through two layers of cheesecloth.
Postharvest treatment
Fruit were dipped for 10 min in sodium silicate solution at 100 mM and then air-dried in the shade. Control fruits were similarly dipped in water. The treated fruit were packed individually in net bags of foam plastic, put in the standard melon-shipping boxes, and stored at room temperature (22 ± 2 °C, RH 55–60%) for inoculation and sampling.
In vivo assays
Fruits were sampled after 48 h of treatment with Si. Ten spots (approximately 1 cm in diameter) were disinfected with 70% ethanol around the equator of each melon. Wounds were made on the disinfected spots with a sterile needle (3 mm deep by 3 mm diameter). Spore suspensions (20 μL, 1 × 105 CFU/mL) of T. roseum were pipetted into each wound. After drying for approximately 1 h in air, the melons were putted into boxes, covered with plastic film to increase RH to 85–90%, and kept at 22 ± 2 °C. Lesion diameter was measured at 6, 8, and 10 days after inoculation. Each treatment contained three replicates of 12 fruits, and the entire experiment was repeated twice.
Preparation of the tissue samples from fruit
Accurately 3 g of tissue was removed from 0.5 to 1.5 cm below the skin and 1 cm from the edge of the wound with a stainless steel cork borer from 12 fruit of each treatment at 0, 2, 4, 6, 8, and 10 days after inoculation. Treated fruit without inoculation were removed at 0, 2, 4, 6, 8, and 10 days after treatment. Water treatment was as the control. Each sample was packed, frozen in liquid nitrogen, and kept at −80 °C until enzyme extraction.
Assays of lipid peroxidation determination
The malondialdehyde (MDA) content and membrane integrity were measured by the methods of Hodges [16] and Song [17], respectively.
The membrane integrity, expressed by percentage of electrolyte leakage, was determined by the method of Song [17]. Thirty discs, 10 mm diameter and 4 mm thick, from 12 fruits at 0, 2, 4, 6, 8, and 10 days after inoculation were washed three times in deionized water for about 1 min each time, dried with filter paper, and put together into 50-mL conical flasks containing 40 mL of deionized water. Thirty discs were rinsed twice and incubated in 25 mL of 0.3 M mannitol solution at 25 °C, followed by shaking for 30 min. Electrolyte leakage was determined with a conductivity meter (Model DDS-307A, Shanghai Scientific Instruments). Total electrolyte leakage was determined after boiling another batch of 30 discs for 15 min and cooling rapidly to 25 °C (total electrolytes). Relative leakage was expressed as a percentage of total electrolyte leakage.
Assays of active oxygen determination
Hydrogen peroxide (H2O2), hydroxyl radicals (HO), and superoxide anion (O •−2 ) were measured according to the method of Prochazkova [18], vonTiedemann [19], and Wang and Luo [20], respectively.
Assays of antioxidase activities
Activity of catalase (CAT) and superoxide dismutase (SOD) was measured according to the method of Wang [21], while activity of ascorbic peroxidase (APX), glutathione reductase (GR), peroxidase (POD), and polyphenoloxidase (PPO) was determined by employing the methods of Yazici [22], Yazici [22], Yao and Tian [23], and Zhang [24], respectively.
Protein content of enzyme extracts was measured according to the method of Bradford [25], using bovine serum albumin (BSA) as the standard protein. The specific activity of enzyme was expressed as U mg−1 protein.
Assays of antioxidants
The concentration of ascorbic acid (ASA) and glutathione (GSH) concentration was measured according to the method of Law [26] and Guri [27], respectively.
Statistical analysis
All experiments were performed in triplicate, and statistical analysis of variance analysis was conducted with SAS software version 9.1 (SAS software Institute, Cary, NC, USA).
Results
Effects of Si treatment on lesion diameter of inoculated melons
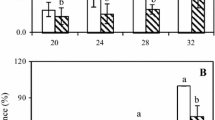
Si treatment significantly reduced (P < 0.05) the lesion diameter of melons inoculated with T. roseum during storage. The lesion diameter decreased by 22.8 and 12.4% after 8 and 10 day of storage after treatment compared with the control (Fig. 1).
Effect of Si treatment on MDA content and membrane integrity in melons challenged by T. roseum
The membrane integrity gradually decreased in all muskmelon fruit during storage, and this decrease was remarkably accelerated by Si treatment. The maximum membrane integrity was found at the 4th day of storage after treatment. A more significant decrease in membrane integrity of the control fruits was found after challenged with T. roseum, however, failed to in treated fruits. The membrane integrity percentage in fruits of Si-treated and control was 44.5 and 21.5% on day 8 of experiment, respectively (Fig. 2a). As a result, Si treatment delayed the velocity of membrane integrity decrease in treated fruit inoculated with T. roseum.
Si treatment significantly increased MDA content in fruit during storage. The MDA content in treated fruit was 72.6 and 37.7% higher than that in control fruit after 4 and 8 days of treatment, respectively. Si caused a progressive and significant increase in MDA content in fruit challenged by T. roseum. The maximum content of MDA was found after 8 days of storage after treatment, with 45.9% higher in Si-treated fruit than that in the control (Fig. 2b).
Effect of Si treatment on levels of H2O2, O •−2 , and ·OH generation
Hydrogen peroxide concentration in the muskmelon significantly increased as a result of sodium silicate treatment, and the increased H2O2 concentration persisted during the whole experiment. The most difference in H2O2 level between control and Si treatment appeared at 6th day after treatment, Si-treated fruit was 43.6% higher than the control fruit. T. roseum challenge inoculation provoked rapid H2O2 accumulation in both control and Si-treated fruits from the 4th day, and the H2O2 content in sodium silicate treatment fruits was greater than that in control, the maximum difference appeared at 6th day, which was 42.6% higher than control (Fig. 3a).
Some small increase in superoxide anion generation was observed in the muskmelon fruits as a result of sodium silicate treatment during the studied period, and its production rate increased gradually with the extension of experiment time. In addition, O •−2 concentrations did not change significantly as a result of sodium silicate treatment. However, inoculation of fruit with T. roseum resulted in the increase of O •−2 content, but failed to increase in control fruit. O •−2 content in T. roseum-inoculated Si-treated fruit was 33.8% higher than that in the controls on the day 8 (Fig. 3b).
Sodium silicate treatment markedly increased hydroxyl radicals content in muskmelon fruits at the first 2 days, but its content has no significantly difference between Si treatment and control at the following period during the whole experiment. Challenge inoculation with T. roseum significantly increased the content of ·OH in Si-treated fruit, while failed to control. The maximum content of ·OH was found at the 8th day of storage after treatment, with the contents 1.04 times higher in Si-treated fruit than those in the control at the same time (Fig. 3c).
Effect of Si treatment on antioxidant enzymes activities
Sodium silicate treatment markedly decreased CAT activity in muskmelon fruits during the whole experiment period. The maximum difference of CAT activity was found at the 2nd day of storage after treatment, with the activity in fruit treated with Si was 34.8% lower than that in control at the same time. CAT activity in control fruit was significantly increased after challenge inoculation with T. roseum, while CAT activity in Si-treated fruit was maintained at a lower level after challenge (Fig. 4a).
The fruit treated with sodium silicate presented higher SOD activity during experiment in comparison with the control. SOD activity in the treated fruit increased at the beginning of storage time and reached maximum activity at 4th day of storage after treatment and then markedly declined. After inoculation with T. roseum, a more progressive and significant increase in SOD activity was found both in control and in Si-treated fruits during experiment, and the fruit treated with Si presented relatively higher activity in comparison with the control (Fig. 4b).
APX activity was significantly inhibited after treatment with sodium silicate. APX activity in control fruits increased during storage time and reached a maximum level at the 6th day of storage after treatment, while it remained lower activity in Si-treated fruits compared to control. After challenge inoculation with T. roseum, APX activity increased in both control and Si-treated fruits, and the control was more effective than Si treatment. The maximum APX activity was found at 6th day after inoculation in the control fruit, APX activity in water-treated (control) fruit was 34.2% higher than that in the Si-treated fruits at the same storage time (Fig. 4c).
Treatment of sodium silicate significantly induced the activity of GR in muskmelon fruit from 6 days after treatment, when it declined in control fruit. GR activity reached its highest value at 6th day in Si-treated fruit, and the level was almost 4.4 times higher than that in control fruit. GR activity significantly increased in both Si-treated and control fruits after challenge inoculation with T. roseum, but GR activity in Si-treated fruit was higher than that in control fruit. GR activity in Si-treated fruit was 44 and 45% higher than that in the controls on days 6 and 8, respectively, after challenge inoculation (Fig. 4d).
Activity of POD in fruit was notably enhanced by sodium silicate treatment. POD activity in Si-treated fruit showed a sustained increase before 8 days of storage and then declined, and the POD activity in Si-treated fruit was 81.6 and 96.9% higher than those in the control on days 4 and 8, respectively. After T. roseum inoculation, POD activity increased in both the control and Si-treated fruits during the whole experiment, the increase was greater for Si-treated fruit than for control fruit. The maximum POD was found at 8th day after inoculation, which was over 2.36 times higher in the fruit treated with sodium silicate than that in the control at the same of storage time (Fig. 4e).
Treatment of sodium silicate caused a significant increase in PPO activity in the muskmelon fruit. The relative maximum PPO activity in Si-treated fruit was found at 4th day of storage after treatment, with the activities in the fruit treated with sodium silicate were 2.96 and 2.92 times higher than that in the control on the days 4 and 8, respectively. A more progressive increase in PPO activity was observed in both Si-treated and control fruits after challenged by T. roseum, and Si-treated fruit was more effective than control. PPO activity in Si-treated fruit was 5.7 and 3.4 times higher than that in the control on days 4 and 8, respectively, after challenge inoculation (Fig. 4f).
Effect of Si treatment on antioxidant contents
Treatment of sodium silicate significantly induced an accumulation of the ASA in fruits. The content of total ASA in Si-treated fruit increased quickly and reached a peak at the 6th day after treatment, and the level was about 1.7-fold higher than that of control. Challenge inoculation with T. roseum enhanced the ASA content in both Si-treated and non-treated fruits, and it was higher in the latter. A slight increase in ASA content was noted in inoculated relative to non-inoculated control fruit, increasing with storage time, whereas in Si-treated fruit, inoculation caused a rapid decline, which was maintained from 6 days after inoculation, at a level above the non-inoculated control (Fig. 5a).
Sodium silicate markedly increased the contents of GSH. The maximum content of GSH was found at 6th and 8th day after treatment, with the contents were 3.8 times and 2.9 times higher in Si-treated fruit than those in the control, respectively. Challenge inoculation with T. roseum caused a more progressive increase in GSH in Si-treated fruit. However, the content of GSH in the control fruit was higher only on day 6 after T. roseum inoculation. Si-treated fruit was more effective than control. The maximum content of GSH was found at the 6th day of storage after treatment, with the content was 4.19 times higher in Si-treated fruit than that in the control (Fig. 5b).
Discussion
In this study, we found that postharvest dipping with sodium silicate at 100 mM significantly reduced lesion diameter of muskmelon fruit inoculated with T. roseum. The effect of Si treatment for inhibiting decay was related with involvement of induced resistance in oxidative stress. The ability of Si to elicit an oxidative burst and inducement of its scavenging system have already been demonstrated in some crops, such as wheat [28] and spinach [29]. However, this is the first report that Si participate in the induction of an oxidative burst in postharvest fruit.
ROS has been suggested to be the first defense line against pathogen invasion either directly killing the pathogen or slowing down its ingress due to the rapidity of production and cytotoxicity [30]. ROS also participates in other resistance mechanisms, e.g. reinforcement of the plant cell wall (lignification, cross-linking of cell wall structural proteins), phytoalexin production, the hypersensitive response, and in SAR [31]. The excess H2O2 production has been related to lipid peroxidation in plant tissue [32]. In our study, some positive relationships were found between Si-induced resistance and higher generation of ROS in the muskmelon fruits. Treatment of the muskmelon fruit with sodium silicate caused the increases in ROS generation, including H2O2, ·OH and O •−2 . Rapid accumulation of ROS in Si-treated fruits as well as pathogen-challenged fruits may be a critical step in the containment of the pathogen. This period immediately after the pathogen attack might be important in determining whether protection will be successful in induced fruits. The increase in ROS generation seemed to be an important element in muskmelon fruit defense system and may play a role in restricting the development of disease symptoms infected with the T. roseum.
Meanwhile, ROS can seriously disrupt normal metabolism through oxidative damage to membrane lipids, proteins, and nucleic acids [33]. It is already known that free radical induced peroxidation of lipid membranes. MDA, a decomposition product of polyunsaturated fatty acids hydroperoxides, has been utilized as a suitable biomarker for lipid peroxidation [34], which has an effect of oxidative damage at the cellular level [35]. In the present work, MDA content was significantly increased in Si-treated and pathogen-challenged fruits, while the membrane integrity decrease was remarkably restrained by sodium silicate treatment, which suggested a better protection from oxidative damage.
The accumulation of ROS resulted from altered balance between ROS production and capacity of scavenging for them [36]. While plants have evolved a complex antioxidant system to protect cellular membranes and organelles from damaging effects of ROS, the major ROS-scavenging activities include complex non-enzymatic antioxidant molecules, such as ASA, GSH, and antioxidant enzymes including CAT, APX, GR, SOD, and POD [37]. In the present work, we observed increased activities of SOD, GR, POD, and PPO, and decreased activities of CAT and APX after Si treatment and challenge inoculation. Besides, the content of ASA and GSH was increased as well after treatment. CAT is one of the main H2O2-scavenging enzymes that dismutates H2O2 into water and O2 [38]. SOD catalyzes the disproportion of superoxide radical to molecular oxygen and H2O2 [39]. However, H2O2 would be scavenged by CAT, APX, and different classes of POD [40], which decreased the risk of hydroxyl radical formation from superoxide via the Haber–Weiss-type reaction. APX activity plays a major role in maintaining the balance between free radical production and elimination. GR is known to act in conjunction with APX to metabolize H2O2 to water through an ASA–GSH cycle, and APX catalyzes the breakdown of H2O2 at the expense of oxidizing ascorbate to MDA [41]. High levels of O •−2 disumutation should be followed by the peroxidation of H2O2 catalyzed by APX activity. PPO catalyzes the O •−2 -dependent oxidation of phenolics to quinones, has been proposed as a component of elaborate plant defense mechanisms, and plays a critical role in disease resistance [42]. The ascorbate plays a crucial role in complex antioxidation processes [43]. Ascorbate is also an important cosubstrate of many enzymes; it has the capacity to directly eliminate several different ROS, including singlet oxygen, superoxide, and hydroxyl radicals [41]. GSH is often the first line of defense against oxidative stress; however, a severe oxidative stress may suppress GSH levels due to the loss of adaptive mechanisms and the oxidation of GSH to its oxidized form, GSSG [17].
Interestingly, our results show that the sodium silicate treatment increased the levels of MDA and decelerated velocity of integrity decline (Fig. 3), and MDA is the oxidative enzymic product of the membrane, so this seemingly to be contradictory, that might increase antioxidant enzyme activity. In our experiments, sodium silicate treatment and challenge inoculation lowered CAT and APX activities. However, a higher level of H2O2 was found in Si-treated fruit; it is the result of dual function of H2O2, and H2O2 is a signal molecule in the environment stress response, while excess H2O2 production related to lipid peroxidation. The disease resistance will be enhanced after sodium silicate treatment, and the ROS content should be maintained at a certain level. In general, an appropriate intracellular balance between ROS generation and scavenging exists in all cells in any case. The production and scavenging of activated oxygen species are interactive, which maintain the plants in a relatively stable state.
Results of current study seemingly showed that both Si treatment and pathogen challenge have the same effect to enhance resistant reaction. In fact, compared to the latter which was just a kind of compatible interaction between pathogen and host, only the former involved in induce resistance through eliciting the fruit against pathogen. It can be elucidated that Si treatment not only disrupts the infection through directly effect on pathogen, but also promotes host to enhance resistance. Moreover, it could be proved by significantly augmenting the effective of induce resistance in the fruit if it was treated by sodium silicon treatment combine with pathogen inoculation. However, the exact mechanisms involved in how to produce the synergistic reaction keep unclear and need further study.
Conclusions
Postharvest treatment with sodium silicate could reduce the decay caused by T. roseum in muskmelon fruit. It may be due to increase the content of ROS and stimulation of the activities of antioxidant system, including enzymatic and non-enzymatic antioxidant molecules. Reactive oxygen species play a key role in disease resistance, at the same time, and an effective defense system has developed to control its content to avoid harmful effects on muskmelon fruit. These results suggested that antioxidants system is important in defense response to pathogen infection and resistance inducing. Future study is necessary to investigate the molecular biology mechanism of the role of sodium silicate-induced resistance in postharvest fruits.
References
Terry LA, Joyce DC (2004) Elicitors of induced resistance in postharvest horticultural crops: a brief review. Postharvest Biol Technol 32:1–13
Bi Y, Li YC, Ge YH (2007) Induced resistance in postharvest fruits and vegetables by chemicals and its mechanism. Stewart Postharvest Rev 6:1–6
Ma JF (2006) Silicon and sodium. Encyclopedia Soil Sci 1:1568–1573
Bélanger RR, Benhamou N, Menzies JG (2003) Cytological evidence of an active role of silicon in wheat resistance to powdery mildew (Blumeria graminis f. sp. tritici). Phytopath 93:402–412
Fawe A, Menzies JG, Cherif M, Bélanger RR (2001) Silicon and disease resistance in dicotyledons. In: Datnoff LE, Snyder GH, Korndofer GHE (eds) Silicon in agriculture. Elsevier, Amsterdam, pp 159–170
Rodrigues FA, McNally DJ, Datnoff LE, Jones JB, Labbe C, Benhamou N (2004) Silicon enhances the accumulation of diterpenoid phytalexinsin rice: apotential mechanism for blast resistance. Phytopath 94:177–183
Rémus BW, Menzies JG, Bélangera RR (2009) Aconitate and methyl aconitate are modulated by silicon in powdery mildew–infected wheat plants. J Plant Physi 166:1413–1422
Chérif M, Benhamou N, Menzies JG, Bélanger RR (1992) Silicon-induced resistance in cucumber plants against Pythium ultimum. Physiol Mol Plant Pathol 41:411–425
Menzies JG, Bowen PA, Ehret DL (1992) Foliar application of potassium silicate reduce severity of powdery mildew on cucumber, muskmelon and zucchini squash. J Am Soc Hortic Sci 117:902–905
Qin GZ, Tian SP (2005) Enhancement of biocontrol activity of Cryptococcus laurentii by silicon and the possible mechanisms involved. Phytopath 95:69–75
Bi Y, Tian SP, Guo YR, Ge YH, Qin GZ (2006) Sodium Silicate reduces postharvest decay on hami melons: induced resistance and fungistatic effects. Plant Dis 3:279–283
Li YC, Bi Y, Ge YH, Sun XJ, Wang Y (2009) Antifungal activity of sodium silicate on Fusarium sulphureum and its effect on dry rot of potato tubers. J Food Sci 74:213–218
Fauteux F, Rémus-Borel W, Menzies JG, Bélanger RR (2005) Silicon and plant disease resistance against pathogenic fungi. FEMS Micro Lett 249:1–6
Kang NJ (2008) Inhibition of powdery mildew development and activation of antioxidant enzymes by induction of oxidative stress with foliar application of a mixture of riboflavin and methionine in cucumber. SCI Hortic (Amsterdam) 118:181–188
Urszula M, Sylwia R (2005) Nitric oxide and hydrogen peroxide in tomato resistance: Nitric oxide modulates hydrogen peroxide level in o-hydroxyethylorutin-induced resistance to Botrytis cinerea in tomato. Plant Phys Biochem 43:623–635
Hodges DM, DeLong JM, Forney CF, Prange RP (1999) Improving the thiobarbituric acid reactive-substance assay for estimating lipid peroxidation in plant tissues containing anthocyanin and other interfering compounds. Planta 207:604–611
Song LL, Gao HY, Chen HJ, Mao JL, Zhou YJ, Chen WX, Jiang YM (2009) Effects of short-term anoxic treatment on antioxidant ability and membrane integrity of postharvest kiwifruit during storage. Food Chem 114:1216–1221
Prochazkova D, Sairam RK, Srivastava GC, Singh DV (2001) Oxidative stress and antioxidant activity as the basis of senescence in maize leaves. Plant Sci 161:765–771
vonTiedemann A (1997) Evidence for a primary role of active oxygen species in induction of host cell death during infection of bean leaves with Botrytis cinerea. Physiol Mol Plant Pathol 50:151–166
Wang AG, Luo GH (1990) Quantitative relation between the reaction of hydroxylamine and superoxide anion radicals in plants. Plant Physiol Commun 6:55–57
Wang YS, Tian SP, Xu Y (2005) Effects of high oxygen concentration on pro- and anti-oxidant enzymes in peach fruits during postharvest periods. Food Chem 91:99–104
Yazici I, Türkan I, Sekmen AH, Demiral T (2007) Salinity tolerance of purslane (Portulaca oleracea L.) is achieved by enhanced antioxidative system, lower level of lipid peroxidation and proline accumulation. Environ Exp Bot 61:49–57
Yao HJ, Tian SP (2005) Effect of pre- and postharvest application of salicylic acid or methyl jasmonate on inducing disease resistance of sweet cherry fruit in storage. Postharvest Biol Technol 35:253–262
Zhang JF, Wang XR, Guo HY (2004) Effects of water-soluble fractions of diesel oil on the antioxidant defenses of the goldfish, Carassius auratus. Ecotoxic Environ Safety 58:110–116
Bradford MM (1976) A rapid and sensitive method for the quantitation of micrograms quantities of protein utilizing the principle of protein dye binding. Anal Biochem 72:248–254
Law MY, Charles SA, Halliwell B (1983) Glutathione and ascorbic acid in spinach (Spinacia oleracea) chloroplasts, the effect of hydrogen peroxide and of paraquat. Biochem J 210:899–903
Gur A, Can J (1983) Ozone effect on glutathine and ascorbic acid in beans. Plant Sci 63:733–736
Gong HJ, Zhu XY, Chen KM, Wang SM, Zhang CL (2005) Silicon alleviates oxidative damage of wheat plants in pots under drought. Plant Sci 169:313–321
Gunes A, Inal A, Bagci EG, Coban S, Pilbeam DJ (2007) Silicon mediates changes to some physiological and enzymatic parameters symptomatic for oxidative stress in spinach (Spinacia oleracea L.) grown under B toxicity soil. Sci Hortic 113:113–119
Peng M, Kuć J (1992) Peroxidase-generated hydrogen peroxide as a source of antifungal activity in vitro and on tobacco leaf disks. Phytopath 82:696–709
Maolepsza U (2006) Induction of disease resistance by acibenzolar-S-methyl ando-hydroxyethylorutin against Botrytis cinerea in tomato plants. Crop Prot 25:956–962
Larrigaudière C, Vilaplana R, Soria Y, Recasens I (2004) Oxidative behaviour of blanquilla pears treated with 1-methylcyclopropene during cold storage. J Sci Food Agric 84:1871–1877
Misra N, Gupta AK (2006) Effect of salinity and different nitrogen sources on the activity of antioxidant enzymes and indole alkaloid content in Catharanthus roseus seedlings. J Plant Phys 163:11–18
Bailly C, Benamar A, Corbineau F, Dome D (1996) Changes in malondialdehyde content anin superoxide dismutase, catalase and glutathione reductase activities in sunflower seed as related to deterioration during accelerated aging. Physiol Plant 97:104–110
Jain M, Mathur G, Koul S, Sarin NB (2001) Ameliorative effects of proline on salt stress induced lipid peroxidation in cell lines of groundnut (Arachis hypogea L.). Plant Cell Rep 20:463–468
Mittler R (2002) Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci 7:405–410
Lee DH, Lee CB (2000) Chilling stress-induced changes of antioxidant enzymes in the leaves of cucumber: In gel enzyme activity assays. Plant Sci 159:75–85
Corpas FJ, Palma JM, Sandalio LM, Lopez HE, Romero PMC, Barroso JB (1999) Purification of catalase from pea leaf peroxisomes: identification of five different isoforms. Free Radic Res 31:35–41
Scandalios JG (1993) Oxygen stress and superoxide dismutase. Plant Phys 101:7–12
Bowler C, Montagu M, Inze D (1992) Superoxide dismutase and stress tolerance. Annu Rev Plant Mol Biol 43:83–86
Gao Q, Zhang LX (2008) Ultraviolet-B-induced oxidative stress and antioxidant defense system responses in ascorbate-deficient vtc1 mutants of Arabidopsis thaliana. J Plant Phys 165:138–148
Mahanil S, Attajarusit Stout MJ, Thipyapong P (2008) Overexpression of tomato polyphenol oxidase increases resistance to common cutworm. Plant Sci 174:456–466
Conklin PL (2001) Recent advances in the role and biosynthesis of ascorbic acid in plants. Plant Cell Environ 24:383–394
Acknowledgments
This work was financially supported by National Natural Science Foundation of China (30671465 & 31071835). Thanks are also given to Mr Xu (president of muskmelon production and promotion association of Quanshan, Minqin County in Gansu Province, China) for his enthusiasm help to provide us testing materials.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, W., Bi, Y., Ge, Y. et al. Effects of postharvest sodium silicate treatment on pink rot disease and oxidative stress-antioxidative system in muskmelon fruit. Eur Food Res Technol 234, 137–145 (2012). https://doi.org/10.1007/s00217-011-1611-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00217-011-1611-9