Abstract
The gross chemical composition and functional properties (solubility, emulsifying and foaming properties) of different amaranth protein preparations were studied in model systems and were compared to those of casein and soy protein isolates. Preparations of alkaline-soluble total protein, albumin, globulin, and glutelin-like alkaline-soluble residual protein were produced from two different types of defatted amaranth meals by extraction and fractionation. Although similarity can be shown between protein patterns of legumes (including soy) and amaranth, the emulsifying and foaming properties of amaranth protein preparations are relatively poor in comparison to the reference proteins, except foaming properties of albumin preparations. Nevertheless, taking in mind that these properties depend on interactions with other food components and textural requirements of individual food products, the amaranth protein preparations may be treated as potential protein sources and food ingredients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The rapidly increasing demand for food-grade proteins has raised the cost of traditionally used animal protein sources such as proteins of milk, egg, meat and fish. However, the requirement and the regulation concerning quality of animal proteins has become more strict, most likely due to the food safety problems connected to BSE, dioxine crisis, avian influenza, etc. Therefore, it is understandable that the interest in potential use of alternative protein sources of plant origin is growing, although problems relating to questionable acceptance of GMOs by consumer exists. According to some perspectives, seeds of amaranth may play a significant role as a potential source of food protein.
The interest in this crop is developing due to its several advantageous properties. The nutritionists find that the grain has higher nutritional value than conventional cereals due to the higher protein content, more valuable amino acid composition and some functional components like dietary fibre, squalene [1–3]. At present, the seeds of some species of grain amaranth are mainly popped to make different products, milled for baking products use and also as ingredients of alimentary pasta production [4–6]. The amaranth proteins have good digestibility, and majority of proteins belong to the group of water-soluble albumin and salt-soluble globulins [3, 7, 8]. Although, the properties of amaranth protein isolates and concentrates were studied by several researchers [12–14], very little data are available in the literature on functional properties of different protein fractions of this crop[4, 9–11 ].In addition different methods were used under different conditions which makes the comparison and evaluation of research data difficult. The aim of research presented in this paper was to study functional properties of different amaranth protein preparations applying the same methodology.
Materials and methods
Materials
Two Hungarian types of amaranth seed samples were studied. The first one is an Amaranthus hypochondriacus cultivar (Edit), produced by Klorofill Ltd, Kecskemét, Hungary. The second, is a mixture of Amaranthus hybridus and Amaranthus cruentus, was purchased from Botanical Garden, Vácrátót, Hungary.
Methods
Preparation of defatted amaranth flours
The whole seeds were ground in a laboratory mill (Chemotec, LabMill, Tecator AB, Sweden). The flours (granularity: 95% of particles smaller than 1 mm) were defatted by shaking with hexane at a ratio of 3:1 solvent/flour. The extraction was repeated 3 times and hexane was evaporated under vacuum. The defatted flour contained 0.7–0.9% fat.
Extraction of alkali soluble protein isolates
One part of defatted flour of both samples was suspended in distilled water (water:flour ratio was 8:1) and the pH of suspension was adjusted to pH 9.5 by adding 1 M sodium hydroxide. The suspension was stirred for 60 min and centrifuged for 20 min (rpm 3,500). The supernatant was decanted and the procedure was repeated. The two supernatants were pooled and the pH adjusted to pH 4.5 in order to precipitate the proteins. After centrifugation (30 min), the precipitate was separated and freeze-dried.
Preparation of protein fractions
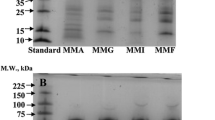
The other part of the samples was used for preparation of protein fractions, like albumin, globulin and glutelin-type alkali soluble proteins. The proteins were extracted according to the modified Osborne’s procedure [15]. All protein preparations were freeze-dried (Fig. 1). Alkali soluble total proteins [ASTP(ed) and ASTP (mix)], albumins [ALB(ed) and ALB(mix)], globulins [GLB(ed) and GLB(mix)] and alkali soluble residue proteins [ASRP (ed) and ASRP(mix)] were obtained from the two seed types.
Chemical composition
Macro-components (moisture, protein, oil, fibre and ash contents) of samples were determined according to AOAC methods [16].
Determination of functional properties
Emulsifying properties
Emulsifying Activity Index (EAI) and Emulsifying Stability Index (ESI) were determined. In both cases 0.2% (m/v) protein solutions were prepared in 0.05 M of phosphate buffer (pH 8.1). For preparing the emulsions 12 ml of protein solution and 4 ml of sunflower oil (commercial grade) was used. The homogenisation was carried out with Ultra Turrax T25 laboratory homogenizer.
EAI was determined by turbidimetric method of Pearce and Kinsella [17]. For calculation of EAI the following equation was used:
where T = turbidity, c = protein concentration (g/cm3), in this case 0.002, and v = ratio of oil volume to total volume, in this case 0.25.
ESI was measured by the modified conductometric procedure [18]. The results were calculated by the graphical integration of area between the registered conductometric curves and lines of minimal and final conductance as shown in Fig. 2. The ESI was expressed in (ms min) units.
Foaming properties
For the determination of FPI and foam stability index (FSI), Kato’s conductometric method was applied with a slight modification [19]. The protein solution was prepared in the same way as determination of ESI, and a similar instrument was used for conductivity measurement. The foaming activity (FPI) was calculated as the ratio of maximal conductivity to time corresponding to this value:
The calculation of stability (FSI) is demonstrated in Fig. 3 and expressed in ms min units.
Protein solubility index
For the determination of protein solubility index (PSI) Sörrensen buffer solutions of pH = 1.15; 2.92; 3.53; 3.95; 4.45; 5.02; 6.98; 9.01; 11.07 and 12.7 were prepared. Five millilitres of buffer solution was added to each sample containing 50 mg of protein and mixed for 30 min. The suspension was centrifuged at 4,500rpm and the protein content of supernatant was determined by the Lowry method, which was automated with flow injection apparatus (Fiastar 5012, Tecator AB, Sweden). To calibrate the method casein was used. PSI was calculated as follows:
where c pr = protein concentration (g/L), V sol = volume of the protein solution (0.005l), m 0 = quantity of the protein preparation (g).
Statistics
The average of three parallel measurements were calculated in every case. The probability level (p values) at 0.05 level was considered to determine the degree of significance between results.
Results and discussion
Chemical composition
There were no significant differences in the chemical composition of the two raw materials (Table 1) and protein preparations produced from them (Table 2). Both basic materials have a typical chemical composition with ∼15% of protein and 5.0–5.5% of oil contents. With the modified Osborne extraction, relatively pure protein preparations were produced. The protein contents of ASTPs, GLBs and ASRPs are about 80%, which is comparable to the commercially used soy protein isolates. Only the albumin fractions have lower protein content, mainly due to relatively high carbohydrate and ash content. The lipid content of ASTPs, GLBs and ASPRs ranged from 0.2 to 3.0%. This may be explained with lipoproteins of germ and mechanical binding by proteins. In the studies of functional properties, the oil moiety of these protein fractions was removed by the same procedure described in chapter “Materials and methods”.
Emulsifying properties
The results of determination of EAI and ESI values of protein samples studied are collected in Fig. 4. As seen from the figure, the EAI values are significantly lower when compared with the EAI values of casein, particularly in the case of globulin fraction the difference is considerably high. The statements made above are valid for both amaranth samples. The differences between the two amaranth samples are not significant. Our results are partly different from those published by Marcone and Kakuda [9] evaluating the higher functional properties of amaranth globulin preparation. However, the measured differences in favor of amaranth globulin are considerable only in the region near the isoelectric point. It may be also noted that the differences in extraction procedure and method of measurement could explain partly the differences in results.
A similar conclusion may be made, concerning the ESI values. The protein preparations from amaranth may be evaluated as poor from the stability point of view; only the albumin fraction has an acceptable ability—however, significantly lower—to stabilize the emulsions. It is interesting, based on results of some preliminary experiments, that a treatment with beta-mercaptoethanol improves the emulsifying properties of alkali-extracted protein preparations. This is may be attributed to a decrease of average molecular weight and increased solubility and flexibility of protein molecules.
Foaming properties
The results of measurements of foaming properties of protein preparations are summarized in Fig. 5. Significant differences were observed between the preparations studied. Albumin preparations showed the best foaming power and foam stability, which corresponded practically to the functional properties of casein and soy, based on available data from our earlier investigations [18, 19]. Excellent foaming properties of albumin fraction from Mexican amaranth samples were reported also by Silva-Sanches et al. [10]. Poor foaming characteristics were measured in the case of globulin- and residue protein preparations. The total alkaline-extractable protein samples showed values between those of casein and residual protein samples. No significant differences were observed between preparations from Amaranthus hypochondriacus and the commercial amaranthus sample except in the case of albumin preparations.
Solubility
The solubility profiles of protein samples studied are presented in Fig. 6. The general shape of solubility curves corresponds to the typical solubility profiles of proteins. Thus a minimum may be observed in the region near the isoelectric point of proteins and an increase of solubility both in acidic and alkaline region. As expected, albumin and globulin fractions have the highest solubility and the residue protein, significantly, the lowest one. The solubility of proteins extracted in alkaline pH (ASTP and ASRP) is relatively weak, the difference between these two preparations is not significant. It may be due to the denaturation and the structural changes of proteins during preparation.
Conclusions
Proteins fractions of amaranth seed have relatively poor emulsifying and foaming properties when compared with casein and soy protein isolates. Some researchers [9, 10] reported relative good functional characteristics. This may be partly explained by the mode of preparation of proteins and the methods used. Nevertheless, the utilization of amaranth flour or their protein products is possible and could be useful, as food additives and ingredients. Optimization of the extraction procedures and modification of protein structure are essentially required. Some preliminary results of study of enzymatically or/and chemically modified amaranth protein suggest that an improvement of functional properties may be achieved. For finding the optimal utilization of amaranth or its components, the functional properties should be studied in real food systems also.
References
Bressani R, Gonzales JM, Zuniga J, Breuner M, Elias LG (1987) Yield, selected chemical composition, and nutritive value of 14 selections of amaranth grain representing four species. J Sci Food Agric 38:347–356
Escudero NL, Arellano ML, Luco JM, Gimenez MS, Mucciarelli SI (2004) Comparison of the chemical composition and nutritional value of Amaranthus cruentus flour and its protein concentrate. Plant Foods Hum Nutr 59(1):15–21
Gorinstein S, Pawelzik E, Delgado-Licon E, Haruenkit R, Weisz M, Trakhtenberg S (2002) Characterisation of pseudocereal and cereal proteins by protein and amino acid analyses. J Sci Food Agric 82:886–891
Saunders RM, Becker R (1984) Amaranthus: a potential food and feed resource. In: Pomeranz Y (ed) Advances in cereal science and technology, vol VI. AACC, St Paul, pp 357–396
Kovács ET, Maráz-Szabó L, Varga J (2001) Examination of the protein-emulsifyer-carbohydrate interactions in amaranth-based pasta products. Acta Alimentaria 30:173–187
Kuhn M, Goetz H (1999) Doughs and gluten in amaranth-wheat system. Getreide Mehl-und-Brot 53(6):326–333
Segura-Nieto M, Vazquez-Sanchez N, Rubio-Velasquez H, Olguin-Martinez LE, Rodriguez-Nester CE, Herrera-Estrella L (1992) Characterization of amaranth (Amaranthus hypochondriacus L.) seed proteins. J Agric Food Chem 40(9):1553–1558
Sanfeng Chen, Paredes-Lopez O (1997) Isolation and characterization of the11S globulin from amaranth seeds. J Food Biochem 21(1):53–65
Marcone MF, Kakuda Y (1999) A comparative study of the functional properties of amaranth and soybean globulin isolates, Nahrung 43(6):368–373
Silva-Sanchez C, Gonzales-Castaneda J, Leon-Rodrigez A, Barba-de-la-Rosa AP (2004) Functional and rheological properties of amaranth albumins extracted from two Mexican varieties. Plant Foods Hum Nutr 59(4):169–174
Tömösközi S, Bajkai T, Süle E, Nagy J, Popineau Y, Lásztity R (1996) Functional properties of plant protein isolates—a comparative study, In: Proceedings of conference on plant proteins from European crops, Nantes
Cordero-de-los-Santos MY, Osuna-Castro JA, Borodanenko A, Parades-Lopez O (2005) Physicochemical and functional characterisation of amaranth (Amaranthus hypochondriacus) protein isolates obtained by isoelectric precipitation and micellisation. Food Sci Technol Int 11:269–280
Fidantsi A, Doxastakis G (2001) Emulsifying and foaming properties of amaranth seed protein isolates. Colloids Surf B Biointerfaces 21:119–124
Scilingo AA, Ortiz SEM, Martinez EN, Anon MC (2002) Amaranth protein isolates modified by hydrolytic and thermal treatments. Relationship between structure and solubility. Food Res Int 35:855–862
Pomeranz Y (1970) Extraction of wheat germ proteins. Cereal Chem 47:373
Official Methods of Analysis of AOAC International (1998) 16th Edn, AOAC International
Pearce KN, Kinsella JE (1978) Emulsifying properties of proteins. Evaluation of turbidimetric technique. J.Agric Food Chem 26:716–718
Bajkai T, Popineau Y, Tömösközi S, Baniel A (1996) Determination of emulsifying properties by conductometric methods: comparison and evaluation. In: Proceedings of 75 years of cereal chemistry and food quality control, Jubilee Symposium of Department of Biochemistry and Food Technology, Technical University of Budapest, 06 17–06 18, Budapest. ISBN: 930–420–5070
Tömösközi S, Popineau Y, Bajkai T, Lásztity R (1994) Determination of foaming properties of food proteins by conductometric methods—a comparative study. In: Proceedings of the 1st international conference on food physics, journal of food physics supplement, pp 99–102
Acknowledgment
The research work was supported by OTKA (Hungarian National Scientific Research Foundation, project number: T-032650)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tömösközi, S., Gyenge, L., Pelcéder, Á. et al. Functional properties of protein preparations from amaranth seeds in model system. Eur Food Res Technol 226, 1343–1348 (2008). https://doi.org/10.1007/s00217-007-0663-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00217-007-0663-3