Abstract
An analytical study was carried out on phenols in two fig cultivars (Ficus carica L.), one black and one green. Fresh fruits were peeled and phenols were extracted separately from the peel and the pulp. The extracts were subjected to HPLC-DAD analysis, by monitoring at four wavelengths, 280, 316, 365 and 520 nm for catechins and benzoic acids, hydroxycinnamic acids, flavonols and anthocyanins, respectively. Results showed that phenols are concentrated almost exclusively in the peel, with the black cultivar having the highest content. In particular, the peel was rich in rutin, with amounts from 527 to 1,071 mg/kg (fresh basis) for green and black figs, respectively. The peel of black figs showed an appreciable content of cyanidin 3-O-rutinoside. The pulp of both green and black cultivars contained only cyanidin 3-O-rutinoside, while the cyanidin 3-O-glucoside was found only in the peel of the black figs. Chlorogenic acid and another cinnamic acid were detected in the peel of both cultivars, while benzoic acids and catechins were not present.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Phenolic compounds are ubiquitously distributed in fruit and vegetables, where they exert specific functions and are very important for sensory properties (flavor and color). Moreover, phenolic compounds have become popular among scientists and consumers for their health promoting properties, namely their antioxidant activity. There is plenty of literature on the antioxidant activity of polyphenols, including references to many fruits and vegetables [1–7]. The antioxidant activity of polyphenols may give rise to a number of biological functions such as antimutagenicity, anticarcinogenicity and antiaging [8–14]. Many phenolics, moreover, seem to have a stronger antioxidant activity than that exerted by vitamins [15]. Nonetheless, reports on the polyphenolic composition of fig fruits (Ficus carica L.) are very scarce and incomplete. Lugasi and Hóvári [16] report on the determination of flavonoid-aglycons in 40 fruit species, including dried fruits, where, in any case, they did not find the presence of these phenolic classes. Vinson [17] and Vinson et al. [5] analyzed colorimetrically the total phenol content of fig fruits, but they do not give any information on the content of the various classes. More precisely, some authors [18] made a complete flavonoid identification in fig fruits of different origin, while others [19] made a qualitative (by spectrophotometry) characterization of anthocyanins in peel and drupelets of California fig cultivars. Very recently, Solomon et al. [20] discussed for the first time the differences in polyphenol composition between pulp and peel of six commercial fig varieties. Moreover, in their work a correlation between each polyphenol class and the antioxidant activity was calculated.
Anyway, there are no reports so far on differences in single polyphenols in peel and pulp of green and black figs. Moreover, no data are available for catechins, benzoic and cinnamic acids. Thus, this paper was mainly aimed to give information on the amounts of the main polyphenolic compounds as well as their classes both in peel and pulp of two cultivars of Italian fresh figs. For this purpose we extracted and analyzed by HPLC-DAD polyphenols from the peel and pulp of both a black and a green skinned cultivar.
Materials and methods
Plant material
Fruits of two local cultivars were used. “Mattalona” black and “San Pietro” green first crop figs were harvested at commercial maturity and homogenous size in a specialized orchard, packaged in cardboard boxes (in monolayer), placed in a 4 °C refrigerated box and transported within 1 h to our lab, where they were immediately subjected to analysis. A sample of 30 undamaged fruits for each cultivar was selected for phenolic analysis, which was done on both the peel and peeled pulp. Fruits were peeled manually with a knife, paying attention not to include the fruit pulp, which was saved for further extraction.
Chemicals
HPLC grade methanol and RPE ACS-ISO methanol were purchased from Carlo Erba Reagenti (Rodano, Milano, Italy); chlorogenic acid was purchased from Fluka Chemical Corp. (Buchs, Switzerland); cyanidin 3-O-rutinoside, cyanidin 3-O-glucoside and rutin were obtained from Extrasynthese (B.P. 62, Genay, France).
Polyphenol extraction and analysis
The phenols were extracted and analyzed in HPLC according to the method described by other authors [21–23]. The peel or the pulp of 30 fruits were homogenized using a Waring-blender; 10 g of homogenized peel or pulp were extracted two times with 75 mL of CH3OH and a third time with 75 mL of CH3OH:H2O (80:20), centrifuged at 4,000 rcf at 15 °C for 5 min and evaporated under vacuum at a temperature below to 35 °C up to a volume of 50 mL. Then, the sample was diluted to 100 mL with MilliQ water and filtered through a 0.45 μm PTFE filter before the HPLC analysis. The column was a LiChrosphere C18, 4 × 250 mm, 5 μm; 20 μL loop; 0.5 mL/min flow; mobile phase: A = 50 mM NH4H2PO4 solution brought to 2.6 pH with H3PO4, B = 80% CH3CN and 20% phase A, C = 200 mM H3PO4; gradient was: 100% A at 5 min, 92% A and 8% B at 8 min, 14% B and 86% C at 20 min, 16.5% B and 83.5% C at 25 min, 21.5% B and 78.5% C at 35 min, 50% B and 50% C at 70 min; 100% A at 75 min, 100% A at 80 min. The phenols were monitored at four different wavelengths: 280 nm for catechins and benzoic acids, 316 nm for hydroxycinnamic acids, 365 nm for flavonols and 520 nm for anthocyanins. We decided to check for these five classes as they are the most representative in fruits. Quantitative analysis considered the sum of the areas of the main peaks, expressed as mg/kg after quantification by calibration with the following standards: chlorogenic acid (for hydroxycinnamic acids), rutin (for flavonols) and cyanidin 3-O-rutinoside and cyanidin 3-O-glucoside (for anthocyanins). Samples from each extraction were analyzed twice. Reproducibility was assessed by four extractions of the same figs used for this work. Every extraction was analyzed twice in HPLC. Quantitative data from each extraction was subjected to statistical analysis and the resulting coefficient of variation was ≤5% for anthocyanins and flavonols and ≤2% for hydroxycinnamic acids The % RSD for the HPLC analysis was ≤3% for anthocyanins and ≤1% for hydroxycinnamic acids and flavonols.
Results and discussion
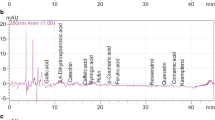
Representative chromatograms resulting from the HPLC analysis of the peel of black figs are shown in Fig. 1. It should be pointed out that few major peaks were detected and neither benzoic acids nor catechins were present. In particular, using HPLC-DAD we can attribute, by comparison with UV-spectra of authentic standards for each class, two peaks as hydroxycinnamic acids (Fig. 1a), three as flavonols (Fig. 1b) and two as anthocyanins (Fig. 1c). In particular, we can assign the peak 1 at 316 nm (Rt 31.125) as chlorogenic acid (Fig. 1a). Moreover, the peak 3 at 365 nm (Fig. 1b) matched the retention time and UV-spectra of quercetin 3-O-rutinoside (rutin), thus confirming previous results [18], while the peak 6 at 520 nm on the peel of black figs was that of cyanidin 3 O-monoglucoside, and the peak 7 was cyanidin 3-O-rutinoside (Fig. 1c), as reported by Solomon et al. [20]. Regarding the peak 2 at 316 nm (Fig. 1a) and the peak 4 and 5 at 365 nm (Fig. 1b), we can only make an assignation for the class by comparison with the UV-spectra. In fact, the UV-spectrum of peak 2 was very close to that of chlorogenic acid (Fig. 2), while the UV-spectra of peaks 4 and 5 fits with flavonols spectra as shown in Fig. 3. Thus, we decided to quantify peak 2 as equivalent to chlorogenic acid, and peak 4 and 5 as equivalents to rutin. Anyway, we have to highlight that the peaks 2, 4 and 5 represent only about the 10% of the polyphenols detected.
UV spectrum of chlorogenic acid (spectrum 1) and compound corresponding to peak 2 (spectrum 2) in Fig. 1a
Table 1 shows the amount of the single polyphenols above reported as well as the total for each class. Flavonols, with more than 1,450 mg/kg of fresh weight of rutin equivalent, were the main class detected, followed by anthocyanins and hydroxycinnamic acids, with up to 929 mg/kg of cyanidin 3-O-rutinoside equivalents and 154 mg/kg of chlorogenic acid equivalents, respectively. These data confirm those reported recently by Solomon et al. [20], even if we found in our samples a more than threefold higher concentration of flavonols and anthocyanins. The most representative compound was rutin, with 1071.41 mg/kg and the second one was cyanidin 3-O-rutinoside (901 mg/kg), both in the peel of black figs. Black figs had a significantly higher content of the three polyphenolic classes, compared with the green ones, moreover, it should be emphasized that almost all the polyphenols were found in the peel, thus confirming recent findings [20], with the only exception of cyanidin 3-O-rutinoside, which was found also in the pulp of black figs, while the other polyphenols were not found. This significant difference between peel and pulp content has also been recently found in a more widely consumed fruit, the apple [24]. However, our study showed a higher content of polyphenols in the peel of figs, compared to apples, which, in turn had a higher amount in the pulp than the figs.
Conclusions
The extraction procedure and the HPLC-DAD analysis performed gave chromatograms with high resolution of peaks, with evident major peaks and other minor ones. Results show that figs, at least the cultivars analyzed, are characterized by localization of the polyphenols in the peel; the pulp, on the other hand, has only a slight content of anthocyanins. As expected, black figs were significantly richer in polyphenols than the green ones. For the first time at our knowledge, this study reports the differences in the composition of single polyphenolic compounds between peel and pulp of figs, as well as the detection of cinnamic acids. Further studies are in progress in cooperation with other research groups to identify unknown peaks and to check for changes in polyphenols after fruit dehydration and storage, because figs are mostly consumed as dried.
References
Hertog MGL, Hollman PCH, Katan MB (1992) J Agric Food Chem 40:2379–2383. doi:10.1021/jf00024a012
Guo C, Cao G, Sofic E, Prior RL (1997) J Agric Food Chem 45:1787–1796. doi:10.1021/jf960786d
Heinonen IM, Meyer AS, Frankel EN (1998) J Agric Food Chem 46:4107–4112. doi:10.1021/jf980181c
Gil MI, Tomás-Barberán FA, Hess-Pierce B, Holcroft DM, Kader AA (2000) J Agric Food Chem 48:4581–4589. doi:10.1021/jf000404a
Vinson JA, Su X, Zubik L, Bose P (2001) J Agric Food Chem 49:5316–5321. doi:10.1021/jf0009293
Ismail A, Marjan ZM, Foong CW (2004) Food Chem 87:581–586. doi:10.1016/j.foodchem.2004.01.010
Bao JS, Cai YZ, Sun M, Wang GY, Corke H (2005) J Agric Food Chem 53:2327–2332. doi:10.1021/jf048312z
Huang MT, Ho CT, Lee CY (1992) ACS Symposium Series 507. Am Chem Soc, Washington DC, p 402
Cook NC, Sammam SC (1996) Nutr Biochem 7:66–76
Steinmetz KA, Potter JD (1996) Cancer Cause Control 2:325–351
Garcia-Closas R, Gonzalez CA, Agudo A, Riboli E (1999) Cancer Cause Control 10:71–75. doi:10.1023/A:1008867108960
Joseph JA, Shukit-Hale B, Denisova NA, Bielinski D, Martin A, McEwen JJ, Bickford PC (1999) J Neurosci 19:8114–8121
Dillard CJ, German JB (2000) J Sci Food Agric 80:1744–1756. doi:10.1002/1097-0010
Prior RL, Cao G (2000) Hort Sci 35:588–592
Vinson JA, Zubik L, Bose P, Sammam N, Proch J (2005) J Am Coll Nutr 1:44–50
Lugasi A, Hóvári J (2002) Acta Aliment Hung 31:63–71. doi:10.1556/AAlim.31.2002.1.9
Vinson JA (1999) Cereal Food World 44(2):82–87
Siewek F, Herrnann K, Gritjahn L, Victor W (1985) Z Naturforsch C 40:8–12
Puech AA, Rebeiz CA, Catlin PB, Crane JC (1975) J Food Sci 40:775–779
Solomon A, Golubowicz S, Yablowicz Z, Grossoman S, Bergman M, Gottlieb HE, Altman A, Kerem Z, Flaishman MA (2006) J Agric Food Chem 54:7717–7723. doi:10.1021/jf060497h
Donovan JL, Meyer AS, Waterhouse AL (1998) J Agric Food Chem 46:1247–1252. doi:10.1021/jf970831x
Nakatani N, Kajano S, Kikuzaki H, Sumino K, Katagiri K, Mitani T (2000) J Agric Food Chem 48:5512–5516. doi:10.1021/jf000422s
Piga A, Del Caro A, Corda G (2003) J Agric Food Chem 51:3675–3681. doi:10.1021/jf021207
Chinnici F, Bendini A, Gaiani A, Riponi C (2004) J Agric Food Chem 52:4684–4689. doi:10.1021/jf049770a
Author information
Authors and Affiliations
Corresponding author
Additional information
Alessandra Del Caro and Antonio Piga contributed equally to this study.
Rights and permissions
About this article
Cite this article
Del Caro, A., Piga, A. Polyphenol composition of peel and pulp of two Italian fresh fig fruits cultivars (Ficus carica L.). Eur Food Res Technol 226, 715–719 (2008). https://doi.org/10.1007/s00217-007-0581-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00217-007-0581-4