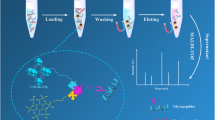
Abstract
A glutathione (GSH)-functionalized silica material was prepared using divinyl sulfone activation chemistry (named SiO2-DVS-GSH). The successful synthesis of the SiO2-DVS-GSH material was confirmed by FT-IR, elemental analysis, and zeta potential analysis. The effects of water content, pH value, and salt concentration in the mobile phase on the model compound (uracil, uridine, cytosine, cytidine, guanosine, xanthosine, orotic acid) retention was studied, and a hydrophilic interaction liquid chromatography (HILIC) retention feature together with electrostatic interaction of the SiO2-DVS-GSH material was observed. The prepared stationary phase was further applied for the separation of oligosaccharide. In addition, the SiO2-DVS-GSH material displayed remarkable selectivity and specificity for the sialylated N-glycopeptides’ enrichment from bovine fetuin tryptic digests, even at a mass ratio of 1:1000 (w/w) to bovine serum albumin (BSA, non-glycosylated protein), showing superior performance compared to commercial ZIC-HILIC material. Moreover, the SiO2-DVS-GSH material behaved well in the N-glycopeptides’ enrichment from human serum, demonstrating its promising potential for glycoproteomics of complex biological samples.

A glutathione (GSH)-functionalized silica material was prepared using divinyl sulfone activation chemistry, and it shows remarkable selectivity and specificity for the sialylated N-glycopeptides’ enrichment.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Protein glycosylation is one of the most common and important post-translational modifications (PTMs) of proteins, and it plays essential roles in various biological processes, such as cell-cell communication, molecular recognition, protein folding, and immune response [1,2,3]. In addition, the progression of many diseases has been found to be associated with the changes in N-linked glycans [4, 5]. Up to date, high-performance liquid chromatography-tandem mass spectrometry (LC-MS/MS) has been widely used for the characterization of protein glycosylation [6]. However, the identification of protein glycosylation remains a challenge, owing to the heterogeneity of glycosylation site and the low abundance of glycopeptides [7, 8]. Furthermore, the signals of glycopeptides in MS analysis are always suppressed by the large amount of non-glycosylated peptides. Therefore, it is necessary to develop efficient materials and enrichment methods prior to mass spectrometry (MS) analysis.
Various technologies have been developed for glycopeptides’ enrichment, such as boronic acid affinity chromatography [9], lectin affinity chromatography [10], hydrazide chemistry [11], IMAC [12], and hydrophilic interaction liquid chromatography (HILIC) [13]. Among them, HILIC has been widely used as a distinctive technique for the separation of polar and hydrophilic compounds such as nucleosides, oligosaccharides, amino acids, and highly polar natural products [14, 15]. In recent years, HILIC attracts more and more attention owing to its unbiased selectivity to glycan structures, and it becomes a promising N-glycopeptides’ enrichment strategy.
A number of silica-based materials chemically functionalized with polar groups have been developed for HILIC, including diol, amide, amine, triazole, and zwitterionic function group [16]. Zwitterionic stationary phases, which contain both positive and negative charges, such as commercial ZIC-HILIC, and a series of zwitterionic stationary phases synthesized by “click chemistry,” have been found to be suitable for the enrichment of glycopeptides [17, 18]. However, the required anhydrous conditions and special control parameters during the silane synthesis challenge the surface modification, which is time consuming and laborious. Therefore, it is necessary to develop novel synthesis methods for HILIC material with different linker and functional groups, which will improve the selectivity and specificity to the N-glycopeptides.
Divinyl sulfone (DVS), known as an efficient and robust homobifunctional linker, has been widely used in bioconjugate chemistry [19,20,21]. The versatile reactivity of DVS permits it to react conveniently either with amino, hydroxyl, or sulphydryl groups via 1,4-Michael addition [22, 23]. In addition, vinyl sulfone–based chemistry has been used for biomolecule microarray fabrications and study of carbohydrate-lectin interactions [24]. However, there is seldom report on DVS-based chemistry in the synthesis of silica-based stationary phase for HILIC applications. Glutathione (GSH) is a tripeptide, and the zwitterionic property allows it to be a potential candidate for the hydrophilic ligand [25]. Thus, it is possible to design a material functionalized with GSH using DVS as a bridge to prepare hydrophilic stationary phase.
Herein, DVS chemistry was used for the preparation of silica-based HILIC material with GSH as the hydrophilic group (named SiO2-DVS-GSH). The properties of the material were characterized by elemental analysis, FT-IR, and zeta potential analysis. And the retention properties of the new material were investigated under different water contents, pH values, and salt concentrations. In HILIC mode, oligosaccharide was employed to evaluate the separation performance of the new material. Furthermore, the SiO2-DVS-GSH material was applied to enrich sialylated N-glycopeptides from tryptic digests of standard bovine fetuin and biological complex sample.
Materials and methods
Materials and chemicals
Spherical silica gel (5 μm, 100 Å, surface area 300 m2/g) was purchased from Fuji Silysia Chemical (Kasugai, Japan). 3-Aminopropyl-triethoxy silane (APTES), divinyl sulfone (DVS), glutathione (GSH), polar solutes orotic acid, uridine, uracil, cytosine, cytidine, and guanosine were from Acros (Cambridge, USA). Fructooligosaccharides were kindly donated by the Natural Products and Glycobiotechnology group (Dalian Institute of Chemical Physics, Chinese Academy of Sciences, Dalian, China). Bovine fetuin, bovine serum albumin (BSA), urea, dithiothreitol (DTT), iodoacetic acid (IAA), ammonium bicarbonate (NH4HCO3), formic acid (FA), and ammonium formate (NH4FA) were purchased from Sigma (St. Louis, MO). Trypsin was purchased from Promega (Madison, WI). ZIC Glycocapture Resin (denoted as commercial ZIC-HILIC material in this study) and chromatographic grade acetonitrile (ACN) were from Merck (Darmstadt, Germany). Geloader tips were purchased from Eppendorf (Hamburg, Germany). C18 material for desalting was from Acchrom Co. Ltd. (Beijing, China). Water was purified by the Milli-Q system (Milford, MA). Healthy human serum (n = 1, male, age 41) was provided by the Second Affiliated Hospital of Dalian Medical University (Dalian, China) and stored at − 80 °C until usage. All other reagents were of analytical grade.
Apparatus
All the chromatographic evaluation was performed on HPLC of Waters 2695-2489/2424 with a UV/visible detector or evaporative light scattering detector (ELSD) (Waters, Milford, MA, USA). Elemental content was measured by Elementar CHNS analyzer model Vario EL III (Elementar, Hanau, Germany). The ζ-potential measurements were performed on a Zetasizer Nano ZS90 system (Malvern, UK) according to the literature [26].
Preparation of the SiO2-DVS-GSH material
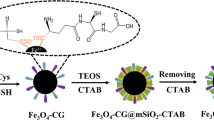
The synthetic procedure of SiO2-DVS-GSH was outlined in Scheme 1. Firstly, the silica gel was dried at 150 °C for 8 h before the reaction. At 0 °C, APTES (0.5 mL) in DMF (10 mL) and divinyl sulfone (250 mg) in DMF (10 mL) were added to 10 mL DMF. The reaction was kept at 0 °C for 2 h to generate the silane coupling reagent 2. The mixture was then warmed to room temperature, and a solution of silica gel (2.5 g) in 30 mL toluene was added. After stirring for 24 h at 45 °C, the mixture was filtered; washed with DMF, toluene, methanol, water, and methanol, successively; and then dried at 60 °C overnight to obtain divinyl sulfone–modified silica material 3 (SiO2-DVS).
Secondly, GSH (3.0 g) and methanol (0.5 mL) were added to a solution of SiO2-DVS (1.5 g) in methanol (20 mL). The mixture was stirred at room temperature for 24 h and filtered. The residue was thoroughly washed with methanol, water, and methanol, successively, and finally dried at 45 °C overnight to obtain GSH-functionalized silica material 4 (named SiO2-DVS-GSH).
The resulting SiO2-DVS-GSH (2.8 g) material was packed into a stainless steel column (4.6 mm i.d. × 150 mm) under a pressure of 40 MPa, with methanol as the slurry and propulsion solvent.
Chromatographic evaluation
For the chromatographic evaluations, all HPLC analyses were carried out at least three times and averaged. The flow rate was 1.0 mL/min, and the column temperature was 30 °C, with the detector wavelength of 260 nm. The dead time was 1.70 min, which was determined by injecting 2 μL acetone with the mobile phase of H2O/ACN at 20/80 (v/v).
Tryptic digestion of proteins and serum
One milligram protein (bovine fetuin or BSA) was denatured with 8 M urea in 50 mM NH4HCO3 for 3 h, and then reduced by DTT (50 mM, 8 μL) for 2 h at 37 °C. After adding IAA (50 mM, 10 μL) for alkylation in dark for 30 min, the solution was diluted tenfold with 50 mM NH4HCO3, then mixed with trypsin at an enzyme/protein ratio of 1:25 (w/w) and incubated for 16 h at 37 °C.
Enrichment of N-glycopeptides using SiO2-DVS-GSH and commercial ZIC-HILIC
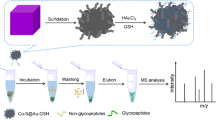
For the enrichment of N-glycopeptides from standard protein digests, 1 mg SiO2-DVS-GSH was suspended in 500 μL ACN/H2O/FA (80:19:1, v/v/v, Buffer A) and mixed with 5 μL (1 mg/mL) bovine fetuin digests. The end volume was 1 mL by adding Buffer A. The obtained solution was gently shaken for 20 min at room temperature and centrifuged at 10,000 rpm for 5 min. The supernatant was removed and the beads were resuspended in 1 mL Buffer A to wash off non-glycosylated peptides. The wash step with Buffer A was repeated three times; then, the non-glycosylated peptides were further washed with 1 mL ACN/H2O/FA (75:24:1, v/v/v) and the beads were transferred to the GELoader tip. At last, 20 μL of ACN/H2O/FA (40:59:1, v/v/v) was added to elute the N-glycopeptides from the SiO2-DVS-GSH material.
For the enrichment of N-glycopeptides from the mixture of tryptic digests of bovine fetuin and BSA, 1 mg SiO2-DVS-GSH material was suspended in Buffer A and then mixed with the tryptic digests of bovine fetuin and BSA sample (the digests of BSA were desalted with C18 material and eluted with Buffer A). The digests of bovine fetuin were kept at 5 μg. The other washing and elution steps were the same as for the standard protein.
One milligram commercial ZIC-HILIC material was mixed with the digests of standard protein bovine fetuin or the mixture of bovine fetuin and BSA (the digests of BSA were desalted with C18 material and eluted with Buffer A) in ACN/H2O/FA (80:19:1, v/v/v). The obtained solution was gently shaken for 20 min at room temperature. After washing with ACN/H2O/FA (80:19:1, v/v/v) for three times, the beads were transferred to the GELoader tip and then eluted with 20 μL of ACN/H2O/FA (40:59:1, v/v/v) to obtain the N-glycopeptides.
For the enrichment of N-glycopeptides from the digests of human serum, 1 mg SiO2-DVS-GSH material was mixed with the tryptic digests of human serum (according to 5 μL human serum), and the end volume was 5 mL by adding Buffer A. The other processes were the same as for the mixture of bovine fetuin and BSA described above.
Deglycosylation of N-glycopeptides by PNGase F
After the enrichment of human serum digests with the SiO2-DVS-GSH material, the N-glycopeptides were dried and redissolved in 50 mM NH4HCO3, followed by the addition of PNGase F (1 U). The solution was incubated for 24 h at 37 °C and then quenched by adding 10 μL formic acid.
Mass spectrometric analysis
The enriched N-glycopeptides were analyzed on a Waters Acquity nano-LC system (Milford, MA, USA) coupled with nano-electrospray ionization quadrupole time-of-flight (ESI-Q-TOF) mass spectrometer (Waters, Manchester, UK). The MS analysis was carried out under positive ion mode. Full-scan MS data were obtained at m/z 600–2000.
For characterization of the enriched N-glycopeptides and N-glycosylation sites from human serum, the deglycosylated peptide samples were analyzed on the LTQ-Orbitrap Velos instrument with an Accela 600 HPLC system (Thermo Fisher Scientific, San Jose, CA). The deglycosylated peptides were redissolved in H2O/FA (100:0.1, v/v) and loaded on a trap column (200 μm i.d. × 3 cm), then separated by a capillary analysis column (75 μm i.d. × 12 cm) packed with C18-AQ 3 μm resin. Mobile phases for online analysis were H2O/FA (100:0.1, v/v, LC Buffer A) and ACN/FA (100:0.1, v/v, LC Buffer B). The mobile phase gradient was as follows: 0 to 5 min, 0 to 5% LC Buffer B; 5 to 90 min, 5 to 35% LC Buffer B; 90 to 105 min, 35 to 80% LC Buffer B; 105 to 115 min, 80% LC Buffer B. MS/MS spectra were acquired under data-dependent CID mode. Full mass scan was acquired from m/z 400 to 2000 with resolution of 60,000. The 20 most intense ions with charge state ≥ 2 and above an intensity threshold of 300 from the full scan were selected for fragmentation in the ion trap. The dynamic exclusion was set as follows: repeat count, 1; duration, 30 s; exclusion list size, 500; exclusion duration, 90 s. System control and data collection were carried out by Xcalibur software version 2.1 (Thermo).
Database searching
The mass spectrometric raw data were analyzed with the MaxQuant software (version 1.3.0.5) against a database (database of IPI human v3.80). Trypsin restriction was set with two missed cleavages. The mass tolerances were 10 ppm for initial precursor ions and 0.05 Da for fragment ions. The cutoff false discovery rate for peptide identifications was controlled to less than 1%. Peptides with N-X-S/T (X≠P) were considered as acceptable results.
Results and discussion
Synthesis and characterization of the SiO2-DVS-GSH material
The novel SiO2-DVS-GSH material was prepared by DVS chemistry. Firstly, the amino propyl silane reagent was reacted with DVS via 1,4-addition reaction to generate the DVS-linked silane reagent. Secondly, the DVS-functionalized silica material was prepared. Finally, the SiO2-DVS-GSH material was obtained via the Michael addition between alkenyl and mercapto groups of GSH molecules. The introduction of imine and sulfonyl groups would affect the hydrophilicity and the charge distribution of the material. Moreover, the reagents for DVS chemistry were accessible, and the synthesis process was simple and easy to be controlled.
The SiO2-DVS-GSH material was characterized by FT-IR and elemental analysis. As shown in Electronic Supplementary Material (ESM) Fig. S1, the peak at 2927 cm−1 was ascribed to the characteristic stretching frequency of –CH2 vibration, and the peaks at 1994 cm−1 and 1876 cm−1 were attributed to –NH. The one at 1666 cm−1 was ascribed to –NH2, and the peaks at 1310–1350 cm−1 and 1160–1120 cm−1 were attributed to –O=S=O. All these characteristics suggested the successful synthesis of the SiO2-DVS-GSH material. The elemental analysis data (shown in ESM Table S1) also demonstrated the presence of DVS and GSH groups on the surface of silica gel after the reaction. In addition, zeta potential at different pH values (pH 2.7–10.9) was measured to understand the charge properties on the material surface. As shown in Fig. 1, positive zeta potential at pH range from 2.7 to 7.1 was observed, indicating that the surface of SiO2-DVS-GSH was positively charged. When the pH was higher than 7.1, the zeta potential became negative. The surface of the material exhibited neutral charge property near pH 7.0. This behavior was different from the typical zwitterionic HILIC (sulfobetaine) [26] and our previous prepared GSH material through “thiol-ene” click chemistry, in which the surface charge was negative when the pH value was higher than 2.7 [27]. This may be attributed to the existence of linker imine groups in this study that counteract the charge effect resulted from the silanol groups of the gel and the carboxyl groups of GSH molecules. This result demonstrated the zwitterion characteristics of the SiO2-DVS-GSH material.
Chromatographic evaluation of typical compounds on the SiO2-DVS-GSH material
Effect of water content
To investigate the effect of water content in the mobile phase on the retention behavior of the SiO2-DVS-GSH material, six nucleosides were selected as test compounds. The structures and pKa of the tested compounds are shown in ESM Fig. S2 and Table S2, respectively. The water content varied from 15 to 40% in volume ratio and the results are shown in Fig. 2a. It can be seen that with increasing water content in the mobile phase, the compound retention displayed a decreasing trend, indicating a typical HILIC retention characteristics of the SiO2-DVS-GSH material.
Effect of factors in mobile phase for nucleoside retention on SiO2-DVS-GSH material, flow rate, 1 mL min−1; column temperature, 30 °C; UV detection, 265 nm. a Effect of water concentration, mobile phase A: ACN; B: H2O; C: 100 mM NH4FA (pH 3.3), 10% C was constant. b Effect of pH value, 100 mM NH4FA 10%, ACN 85%, H2O 5%. c Effect of buffer concentration, pH 3.3, ACN 85%. d Typical chromatogram of nucleoside separation. Mobile phase A: ACN; B: H2O; C: 100 mM NH4FA (pH 3.2). 0–15 min, 85% A/5% B/10% C
Effect of pH
The selectivity and retention of compounds in HILIC mode are usually affected by the pH value in the mobile phase, especially for ionic compounds. Thus, six compounds, including neutral (uracil and uridine), weak basic (cytosine, cytidine, and guanosine), and acidic (orotic acid) species were selected to investigate the retention property on the SiO2-DVS-GSH material at different pH values. The solutions of 100 mM NH4FA with pH in the range of 3.3 to 6.0 were freshly prepared, and the mobile phase ratio of 10% NH4FA was kept constant. As shown in Fig. 2b, for the neutral compounds uracil and uridine, the retention factors were almost unchanged in the studied pH range. For weak basic compounds of cytosine, cytidine, and guanosine, the retention decreased slightly when the pH was varied from 3.3 to 5.0, and remained unchanged from pH 5.0 to 6.0. This was probably due to the distinct ionization degree of weak basic analytes at different pH values. The basic analytes ionized at low pH conditions and exhibited better hydrophilicity. When the pH was increased, the ionization of the weak basic analytes was suppressed, and the hydrophilicity of the molecular analytes was decreased, leading to the decreased retention on the stationary phase. For the acidic compound orotic acid, the electrostatic attractive interaction existed between the stationary phase and the analyte. When the pH was increased, the positive charges on the SiO2-DVS-GSH material decreased which in turn led to the reduced electrostatic attraction and thus decreased the retention.
Effect of buffer concentration
In order to further investigate the surface charge property of the SiO2-DVS-GSH material, the retention of the above six model compounds under different buffer concentration was studied. The concentration of NH4FA in the mobile phase was in the range of 5 to 25 mM at pH 3.3. The plot of NH4FA concentration to the retention factor is shown in Fig. 2c. In HILIC mode, the main retention mechanism is partitioning between a water-rich layer associated with the polar stationary phase surface and the less polar mobile phase, and the salt concentration affects the thickness of the water-enriched layer [28]. When the salt concentration increased, the high organic content in the mobile phase (ACN in this study) would drive more salt ions into the water-enriched layer; thereby, the hydrophilicity of the surface was increased. At pH 3.3, the surface of the SiO2-DVS-GSH material exhibited positive charges. For the tested compounds, the retention profiles were different with varying salt concentrations. For the neutral compounds, the retention increased slightly with increasing NH4FA concentration. For the weak basic compounds, the retention increased obviously with the increase of salt concentration. This result can be ascribed to the decrease of electrostatic repulsive interactions between the cationic compounds and the positively charged surface of the SiO2-DVS-GSH material, as the surface of the stationary phase was shielded by the increasing salt concentration in the mobile phase. For similar reasons but with opposite effects, when the salt concentration of NH4FA was increased, the retention of orotic acid decreased. This can be attributed to the reduced electrostatic attractive interactions between positively charged surface of the SiO2-DVS-GSH material and anionic orotic acid resulted from the shielding effect of salts.
According to the above results, it can be seen that the retention on the SiO2-DVS-GSH material involved both partitioning interaction and electrostatic interaction. A typical chromatogram for the studied nucleoside separation on the SiO2-DVS-GSH material is shown in Fig. 2d. Good baseline separation and peak shapes were observed for all compounds tested in the HILIC mode with 85% ACN as the mobile phase.
Separation of oligosaccharide in HILIC mode
Carbohydrate, a kind of polar compounds, has gained increasing attention in recent years. However, it is poorly retained on traditional reversed-phase liquid chromatography (RPLC). In order to investigate the applicability of the SiO2-DVS-GSH material for carbohydrate separation, fructooligosaccharides were selected as a typical compound and the result is shown in Fig. 3. The fructooligosaccharides with the degree of polymerizations (DPs) ranging from 2 to 26 were well separated with good peak shapes. As the main retention mechanism in HILIC mode is partitioning interaction [29], with the increase of DP, the interaction between the analyte and the water-enriched layer on the SiO2-DVS-GSH surface was enhanced; therefore, the fructooligosaccharides with higher DP were eluted later.
Enrichment of N-glycopeptides
Considering that the SiO2-DVS-GSH material displayed both hydrophilic and charge properties, it was supposed to be a solid-phase extraction adsorbent to enrich N-glycopeptides before MS detection. Herein, 5 μg tryptic bovine fetuin digests was used as the model sample to investigate the specificity and selectivity of the SiO2-DVS-GSH material (1 mg) for sialylated N-glycopeptides’ enrichment. As shown in Fig. 4a, the N-glycopeptides could not be detected only after desalting by C18 without any enrichment, as the signals of glycopeptides were completely suppressed by the non-glycosylated peptides. In contrast, most of the dominant non-glycosylated peptides were removed effectively after enrichment with the SiO2-DVS-GSH material, and the signals of the glycopeptides were markedly improved. Thirty-seven N-glycopeptides were identified in the fraction enriched from the same digests of bovine fetuin (Fig. 4b). Even low-abundant N-glycopeptides, such as m/z 1085.8086 (3+), 1110.4299 (4+), and 1151.1912 (4+), terminated with one, two, and three sialic acids, respectively, were observed due to the effective removal of non-glycosylated peptides. The proposed structures of the detected N-glycopeptides are shown in ESM Table S3. In order to further investigate the enrichment selectivity of the SiO2-DVS-GSH material to sialylated N-glycopeptides, a mixture of the tryptic digests of bovine fetuin and BSA in a ratio of 1:1, 1:100, 1:500 (w/w) (ESM Fig. S3), and 1:1000 (w/w) was carried out, respectively. As shown in Fig. 4c, even up to the ratio of 1:1000 (w/w), the SiO2-DVS-GSH material still displayed high selectivity, with 13 sialylated N-glycopeptides’ identification from bovine fetuin. In addition, the baseline of the MS spectrum was still flat. The highest abundant peaks in Fig. 4c with red triangle were N-glycopeptides derived from impurity glycoproteins in BSA sample. The m/z 1204.4329 (3+), 1214.4535 (3+), 1268.1489 (3+), and 1485.1366 (2+) were selected for MS/MS analysis, and the fragment ion of m/z 204 (HexNAc + H) +, 274 (NeuAc – H2O + H) +, 292 (NeuAc + H) +, and 366 (HexNAc + Hex +H) + verified that the selected peaks were sialylated N-glycopeptides (ESM Fig. S4). This observation is consistent with our previous study [30]. For comparison, commercial ZIC-HILIC material was also used for the enrichment of glycopeptides in the mixture of tryptic digests of bovine fetuin and BSA in 1:200 (w/w) (Fig. 4d). With commercial ZIC-HILIC enrichment, only 3 sialylated N-glycopeptides were detected after enrichment with low abundance. The higher abundance of m/z 1129.2811 (4+), 1171.2314 (3+), 1246.6147 (2+), and 1306.5686 (2+) were further analyzed by MS/MS, respectively (ESM Fig. S5), and these selected ions were non-glycosylated peptides. These results demonstrated that SiO2-DVS-GSH material not only has excellent anti-interferential abilities but also shows superior selectivity and specificity to the sialylated N-glycopeptides in complex mixtures.
Mass spectra of the tryptic digests of bovine fetuin (5 μg) before and after enrichment with SiO2-DVS-GSH and ZIC-HILIC materials. a Desalting with C18 material. b Enrichment with SiO2-DVS-GSH. c Mixture of tryptic digests of bovine fetuin and BSA (1:1000, w/w) enriched with SiO2-DVS-GSH. d Mixture of tryptic digests of bovine fetuin and BSA (1:400, w/w) enriched with ZIC-HILIC. N-Glycopeptides from bovine fetuin are labeled with red stars. N-\glycopeptidederived from impurity glycoproteins in BSA sample are labeled with red triangles
The mechanism for the highly selective enrichment of glycopeptides by SiO2-DVS-GSH was proposed as follows. Most of the tryptic peptides carried a net charge of + 2 due to the positive charge at the C-terminal arginine/lysine and at their N-terminus [31]. In this study, 1% FA was added to the washing solution. At this pH value (pH 2.19), the positive charge on the surface of the SiO2-DVS-GSH material was strong; thus, the positively charged non-glycosylated peptides were eluted during the washing steps due to the electrostatic repulsive interaction. In contrast, on account of the negative charge from sialic acid, most mono-sialylated glycopeptides had a net charge of + 1 [32], and correspondingly, the electrostatic repulsive interaction between the SiO2-DVS-GSH material and mono-sialylated glycopeptides was weaker. Meanwhile, the hydrophilicity of the SiO2-DVS-GSH material increased the retention of N-glycopeptides, ensuring the retention of mono-sialylated glycopeptides on the SiO2-DVS-GSH material. Since most multi-sialylated glycopeptides were neutral or negatively charged, thus, they were retained on the SiO2-DVS-GSH material via electrostatic attractive interaction. The combination of hydrophilic and electrostatic interactions could explain the excellent selectivity of the SiO2-DVS-GSH material for the sialylated N-glycopeptides.
Applications of the SiO2-DVS-GSH material in the enrichment of N-glycopeptides from biological samples
In order to demonstrate the applicability of the SiO2-DVS-GSH material for complex samples, the enrichment of N-glycopeptides from human serum tryptic digests was investigated. The captured N-glycopeptides were analyzed by LC-MS/MS after deglycosylation. After database searching, 150 unique N-glycosylation sites assigned to 70 N-glycoproteins have been identified with the enrichment of SiO2-DVS-GSH material (ESM Table S4). The result is comparable to that of the recent report by using saccharide-based material [33]. The cellular component, molecular function, and biological process of the identified glycoproteins were summarized, according to the Gene Ontology database system (ESM Fig. S6). For the cellular component, 87% were distributed in extracellular region, 80% in extracellular exosome, and followed by those from plasma microparticle. This set of 70 proteins was found to be involved in various biological processes such as platelet degranulation, leukocyte-mediated immunity, and innate immune response. Furthermore, the enrichment specificity (the ratio of the number of glycopeptides to that of all identified peptides) of SiO2-DVS-GSH was 59.1%, which indicated the promising applicability of SiO2-DVS-GSH material in complex biological samples.
Conclusions
A novel glutathione-based hydrophilic interaction liquid chromatography (HILIC) stationary phase (SiO2-DVS-GSH) was facilely synthesized via divinyl sulfone (DVS) chemistry. The prepared SiO2-DVS-GSH material showed good hydrophilicity for the separation of nucleosides and oligosaccharide. With both hydrophilic interaction and electrostatic interaction, the SiO2-DVS-GSH material displayed high selectivity and specificity for the enrichment of sialylated N-glycopeptides, even in a mixture of 1:1000 (w/w) tryptic digests of bovine fetuin and bovine serum albumin (BSA). Furthermore, it also demonstrated the promising ability in glycoproteomics analysis of biological complex samples. This work is expected to extend the types of stationary phase via changing the hydrophilic group by DVS-based chemistry.
References
Helenius A, Aebi M. Intracellular functions of N-linked glycans. Science. 2001;291(5512):2364–9.
Huang KY, Su MG, Kao HJ, Hsieh YC, Jhong JH, Cheng KH, et al. dbPTM 2016: 10-year anniversary of a resource for post-translational modification of proteins. Nucleic Acids Res. 2016;44:435–46.
Kailemia MJ, Park D, Lebrilla CB. Glycans and glycoproteins as specific biomarkers for cancer. Anal Bioanal Chem. 2017;409(2):395–410.
Takahashi M, Kizuka Y, Ohtsubo K, Gu J, Taniguchi N. Disease-associated glycans on cell surface proteins. Mol Asp Med. 2016;51:56–70.
Sethi MK, Hancock WS, Fanayan S. Identifying N-glycan biomarkers in colorectal cancer by mass spectrometry. Acc Chem Res. 2016;49(10):2099–106.
Zhou S, Dong X, Veillon L, Huang Y, Mechref Y. LC-MS/MS analysis of permethylated N-glycans facilitating isomeric characterization. Anal Bioanal Chem. 2017;409(2):453–66.
Xiao H, Suttapitugsakul S, Sun F, Wu R. Mass spectrometry-based chemical and enzymatic methods for global analysis of protein glycosylation. Acc Chem Res. 2018;51(8):1796–806.
Khatri K, Klein JA, Zaia J. Use of an informed search space maximizes confidence of site-specific assignment of glycoprotein glycosylation. Anal Bioanal Chem. 2017;409(2):607–18.
Li D, Chen Y, Liu Z. Boronate affinity materials for separation and molecular recognition: structure, properties and applications. Chem Soc Rev. 2015;44(22):8097–123.
Liu Y, Fu D, Yu L, Xiao Y, Peng X, Liang X. Oxidized dextran facilitated synthesis of a silica-based concanavalin a material for lectin affinity enrichment of glycoproteins/glycopeptides. J Chromatogr A. 2016;1455:147–55.
Sajid MS, Jabeen F, Hussain D, Ashiq MN, Najam-ul-Haq M. Hydrazide-functionalized affinity on conventional support materials for glycopeptide enrichment. Anal Bioanal Chem. 2017;409(12):3135–43.
Glover MS, Yu Q, Chen Z, Shi X, Kent KC, Li L. Characterization of intact sialylated glycopeptides and phosphorylated glycopeptides from IMAC enriched samples by EThcD fragmentation: toward combining phosphoproteomics and glycoproteomics. Int J Mass Spectrom. 2018;427:35–42.
Yu L, Li X, Guo Z, Zhang X, Liang X. Hydrophilic interaction chromatography based enrichment of glycopeptides by using click maltose: a matrix with high selectivity and glycosylation heterogeneity coverage. Chem-Eur J. 2009;15(46):12618–26.
Buszewski B, Noga S. Hydrophilic interaction liquid chromatography (HILIC)-a powerful separation technique. Anal Bioanal Chem. 2012;402(1):231–47.
Rounova O, Demin P, Korotkov M, Malkova V, Ustinnikova O. Development of a hydrophilic interaction high-performance liquid chromatography method for the determination of glycine in formulations of therapeutic immunoglobulins. Anal Bioanal Chem. 2018;410(26):6935–42.
Taraji M, Haddad PR, Amos RIJ, Talebi M, Szucs R, Dolan JW, et al. Chemometric-assisted method development in hydrophilic interaction liquid chromatography: a review. Anal Chim Acta. 2018;1000:20–40.
Alagesan K, Khilji SK, Kolarich D. It is all about the solvent: on the importance of the mobile phase for ZIC-HILIC glycopeptide enrichment. Anal Bioanal Chem. 2017;409(2):529–38.
Xia CS, Jiao FL, Gao FY, Wang HP, Lv YY, Shen YH, et al. Two-dimensional MoS2-based zwitterionic hydrophilic interaction liquid chromatography material for the specific enrichment of glycopeptides. Anal Chem. 2018;90(11):6651–9.
Santos-Moriano P, Monsalve-Ledesma L, Ortega-Munoz M, Fernandez-Arrojo L, Ballesteros AO, Santoyo-Gonzalez F, et al. Vinyl sulfone-activated silica for efficient covalent immobilization of alkaline unstable enzymes: application to levansucrase for fructooligosaccharide synthesis. RSC Adv. 2016;6(69):64175–81.
Stewart SA, Coulson MB, Zhou C, Burke NAD, Stover HDH. Synthetic hydrogels formed by thiol-ene crosslinking of vinyl sulfone-functional poly(methyl vinyl ether-alt-maleic acid) with alpha,omega-dithio-polyethyleneglycol. Soft Matter. 2018;14(41):8317–24.
dos Santos JCS, Rueda N, Barbosa O, Fernandez-Sanchez JF, Medina-Castillo AL, Ramon-Marquez T, et al. Characterization of supports activated with divinyl sulfone as a tool to immobilize and stabilize enzymes via multipoint covalent attachment. Application to chymotrypsin. RSC Adv. 2015;5(27):20639–49.
Cheng F, Shang J, Ratner DM. A versatile method for functionalizing surfaces with bioactive glycans. Bioconjug Chem. 2011;22(1):50–7.
Wang HQ, Cheng F, Li MY, Peng W, Qu JP. Reactivity and kinetics of vinyl sulfone-functionalized self-assembled monolayers for bioactive ligand immobilization. Langmuir. 2015;31(11):3413–21.
Lopez-Jaramillo FJ, Ortega-Munoz M, Megia-Fernandez A, Hernandez-Mateo F, Santoyo-Gonzalez F. Vinyl sulfone functionalization: a feasible approach for the study of the lectin-carbohydrate interactions. Bioconjug Chem. 2012;23(4):846–55.
Liu QJ, Deng CH, Sun NR. Hydrophilic tripeptide-functionalized magnetic metal-organic frameworks for the highly efficient enrichment of N-linked glycopeptides. Nanoscale. 2018;10(25):12149–55.
Jiang W, Fischer G, Girmay Y, Irgum K. Zwitterionic stationary phase with covalently bonded phosphorylcholine type polymer grafts and its applicability to separation of peptides in the hydrophilic interaction liquid chromatography mode. J Chromatogr A. 2006;1127(1–2):82–91.
Shen AJ, Guo ZM, Cai XM, Xue XY, Liang XM. Preparation and chromatographic evaluation of a cysteine-bonded zwitterionic hydrophilic interaction liquid chromatography stationary phase. J Chromatogr A. 2012;1228:175–82.
Hammer J, Haftka JJH, Scherpenisse P, Hermens JLM, de Voogt P. Investigating hydrophilic and electrostatic properties of surfactants using retention on two mixed-mode liquid chromatographic columns. J Chromatogr A. 2018;1571:185–92.
Qiao LZ, Shi XZ, Xu GW. Recent advances in development and characterization of stationary phases for hydrophilic interaction chromatography. Trac-Trend Anal Chem. 2016;81:23–33.
Dong XF, Qin HQ, Mao JW, Yu DP, Li XL, Shen AJ, et al. In-depth analysis of glycoprotein sialylation in serum using a dual-functional material with superior hydrophilicity and switchable surface charge. Anal Chem. 2017;89(7):3966–72.
Totten SM, Feasley CL, Bermudez A, Pitteri SJ. Parallel comparison of N-linked glycopeptide enrichment techniques reveals extensive glycoproteomic analysis of plasma enabled by SAX-ERLIC. J Proteome Res. 2017;16(3):1249–60.
Lewandrowski U, Zahedi RP, Moebius J, Walter U, Sickmann A. Enhanced N-glycosylation site analysis of sialoglycopeptides by strong cation exchange prefractionation applied to platelet plasma membranes. Mol Cell Proteomics. 2007;6(11):1933–41.
Chen LL, Ding D, Sheng QY, Yu L, Liu XP, Liang XM. Selective enrichment of N-linked glycopeptides and glycans by using a dextran-modified hydrophilic material. J Sep Sci. 2018;41(9):2003–11.
Funding
This study received funding from the National Natural Science Foundation of China (No. 21775148) and the Natural Science Foundation of Liaoning Province (No. 20180550134).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The use of serum sample in this experiment was approved by the Ethics Committee of the Second Affiliated Hospital of Dalian Medical University (Dalian, China).
Conflict of interest
The authors declare that they have no conflict of interest.
Informed consent
Informed consent was obtained from the participant in the experiment.
Additional information
Published in the topical collection New Insights into Analytical Science in China with guest editors Lihua Zhang, Hua Cui, and Qiankun Zhuang.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 840 kb)
Rights and permissions
About this article
Cite this article
Fu, D., Liu, Y., Shen, A. et al. Preparation of glutathione-functionalized zwitterionic silica material for efficient enrichment of sialylated N-glycopeptides. Anal Bioanal Chem 411, 4131–4140 (2019). https://doi.org/10.1007/s00216-019-01661-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-019-01661-0