Abstract
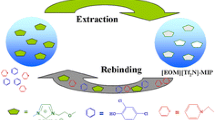
We aimed to improve the imprinting effect of ionic liquid molecularly imprinted polymers (MIPs) by use of a molecular crowding agent. The ionic liquid 1-vinyl-3-ethylimidazolium tetrafluoroborate ([VEIm][BF4]) was used as the functional monomer and aesculetin was used as the template molecule in a crowding environment, which was made up of a tetrahydrofuran solution of polystyrene. The ionic liquid MIPs that were prepared in the crowding environment displayed an enhanced imprinting effect. NMR peak shifts of active hydrogen of aesculetin suggested that interaction between the functional monomer and the template could be increased by the use of a crowding agent in the self-assembly process. The retention and selectivity of aesculetin were affected greatly by high molecular crowding, the amount of high molecular weight crowding agent, and the ratio of [VEIm][BF4] to aesculetin. The optimal MIPs were used as solid-phase extraction sorbents to extract aesculetin from Cichorium glandulosum. A calibration curve was obtained with aesculetin concentrations from 0.0005 to 0.05 mg mL−1 (correlation coefficient R 2 of 0.9999, y = 1519x + 0.0923). The limit of quantification was 0.12 μg mL−1, and the limit of detection was 0.05 μg mL−1. The absolute recovery of aesculetin was (80 ± 2)% (n = 3), and the purity of aesculetin was (92 ± 0.5)% (n = 5). As a conclusion, molecular crowding is an effective approach to obtain ionic liquid MIPs with high selectivity even in a polar solvent environment.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Molecular imprinting creates particular binding materials for a predetermined target molecule [1]. The resulting molecularly imprinted polymers (MIPs) are made by mixing of the template, functional molecule, and cross-linker monomers to form copolymers [2]. After removal of the template by extraction or chemical reaction, the binding sits are complementary to the template molecules in shape, size, and position of the functional groups. In addition to good physical and chemical performance, MIPs are cheap compared with natural antibodies. MIPs are largely used for biosensors [3, 4], mimicking materials [5], immunoassay [6], artificial enzyme catalysis [7, 8], and enrichment [9, 10].
Traditionally, methacrylic acid (MAA) or acrylamide has been used as a functional monomer to prepare most MIPs. When the template molecules are acidic, because of weak interaction with the functional monomer, stronger recognition ability was observed only when 4-vinylpyridine was used as the functional monomer because of electrostatic interaction and π–π stacking between the template and the monomer. However, use of 4-vinylpyridine as a functional monomer often leads to poor selectivity and a low imprinting factor (IF) because there are few types of intermolecular forces between the template and the monomer [11, 12].
Ionic liquids are either organic salts or mixtures consisting of at least one organic component with a low melting point (less than 100 °C), and have been the focus of many investigations because of their unique nature, such as chemical stability, low flammability, negligible vapor pressure, high ionic conductivity, and wide electrochemical window. Functionalization of polymers with some of the characteristics of ionic liquids has been pursued as a way of developing high-performance polymers because of multiple interactions, for example, ion–dipole electrostatic interaction, hydrogen bonding, π–π stacking, hydrophobic interaction, and anion exchange interaction [13, 14]. Therefore, polymerizable ionic liquids have been adopted as functional monomers to prepare MIPs [15,16,17], and an imprinting effect can be achieved even in water-based media [6, 18]. However, the imprinting effect of ionic liquid MIPs is limited, and a strategy for enhanced imprinting is needed.
With high concentrations of biological macromolecules, for instance, proteins, nucleic acids, and lipids, an unusual molecular environment can be caused in biological cells; this is called “biomolecular crowding.” Previous studies [19,20,21] proved that biopolymers’ stable high-order structures were affected by such molecular crowding, and the molecular crowding promotes the association of biomolecules. In recent years, Matsui et al. [22] showed that molecular crowding could enhance the interactions between MAA and the template. When high concentrations of linear polymers such as poly(methyl methacrylate) and polystyrene (PS) are used as the crowding agent in the synthesis, the equilibrium of template reactions with MAA or acrylamide is shifted to complex formation. Moreover, an increased number of electron affinity and ionization potentials or crowding-assisted MIP binding sites is noticed in many cases [22,23,24,25].
In consideration of these facts, we investigated whether macromolecular crowding can be used to prepare ionic liquid MIPs to increase the interaction between the ionic liquid monomer and the template. PS was selected in the presence of tetrahydrofuran (THF) as the porogenic agent and crowding agent. Aesculetin, a potential antitumor drug [26, 27], was chosen as the template. Previously, aesculetin was isolated by capillary electrophoresis or solid-phase extraction (SPE) [28,29,30], such as with silica gel, macroporous resin, or C18. The principle of separating aesculetin is the interaction between these materials and target compounds, which occurs through hydrophobic, anion exchange, and hydrogen bonding. Nevertheless, these materials cannot separate aesculetin effectively from the complex matrix. To address this issue, materials with multiple interactions have been proposed as alternative stationary phases or sorbents. The effects of polymerization factors such as the concentration and molecular weight of PS and the monomer-to-template ratio on the imprinting effect were investigated. Further, the resulting aesculetin-based MIP was used as the SPE absorbent for real samples.
Experimental
Reagents and chemicals
Aesculetin (98%), epigallocatechin gallate (98%), aesculin (98%), apigenin (98%), isorhamnetin (98%), imidacloprid (99%), and triclosan (99%) were supplied by Shifeng Biological Technology Co. (Shanghai, China). 1-Vinyl-3-ethylimidazolium tetrafluoroborate ([VEIm][BF4]) of analytical grade was from Aladdin Industrial Corporation (Shanghai, China). Ethylene glycol dimethacrylate (EDMA; 98%) and PS (analytical grade, average M w = 10,000, 30,000, 350,000, and 1,000,000) were purchased from Sigma (St Louis, MO, USA). 2,2-Azobis(2-methylpropionitrile) (AIBN) of analytical grade was purchased from the Special Chemical Reagent Factory of Nankai University (Tianjin, China). Acetonitrile and methanol [high-performance liquid chromatography (HPLC) grade)] were obtained from Merck (Darmstadt, Germany). All other reagents were analytical grade.
Preparation of MIPs in crowding environments
The preparation of aesculetin-imprinted monoliths was conducted in a 100-mm stainless steel tube with an inner diameter of 4.6 mm. Aesculetin was dissolved in PS–THF (40 mg mL-1, 2060 μL), and then a solution of [VEIm][BF4] in methanol (200 μL, 0.418 mg mL-1), EDMA (565 μL), and AIBN (40 mg) were added. Other solvents were added to the mixture as described in Table 1. The prepolymerization mixture was sonicated for 10 min and nitrogen-stream purge was used for 15 min to eliminate oxygen. Both ends of the tube were rapidly sealed, and the tube was placed in a water bath for 24 h (55 °C). Then the MIP monolith obtained was flushed with THF and methanol by use of an HPLC pump until a stable baseline was obtained. The unreacted reagents, PS, and the template were removed in this step. The nonimprinted polymers (NIPs) were prepared in the same way but with the template omitted.
Chromatographic assessment
The chromatographic assessment was performed with an UltiMate 3000 HPLC system (Thermo Scientific, Sunnyvale, CA, USA) comprising an UltiMate 3000 pump, an UltiMate 3000 variable-wavelength detector, and an UltiMate 3000 autosampler injector with a 20-μL loop. The data were processed by Chromeleon 7. The optimal mobile phase for MIP assessment was acetonitrile and sodium acetate buffer, pH 3.6 (93:7, v/v). The detection wavelength was 349 nm, and the column temperature was 30 °C. The retention factor was calculated with the equation k = (t R − t 0)/t 0, where t R is the retention time of the analyte and t 0 is the void time marked by acetone. The IF was determined by the equation IF = k MIP/k NIP, where k MIP and k NIP are the retention factors for the MIP and the NIP, respectively.
SPE by aesculetin-based MIP
To apply the aesculetin-based MIP to SPE, the MIP (P3) was pumped out by high pressure, and then dried at 40 °C for 24 h. The MIP was ground, the powder was collected and passed through a 43-μm sieve. Then 1.20 g MIP powder was loaded to establish an SPE column (75 mm × 15 mm) with a slurry packing method. Cichorium glandulosum (10.0 mg) was dissolved in 10 mL methanol, which was used as the appropriate solvent for Cichorium glandulosum extraction. Acetonitrile (18 mL) was used to wash the impurities from the column. Then methanol (18 mL) was used as the eluent to remove aesculetin from the column, and ultimately impurities. The fraction of crude extract, washing and eluent were collected respectively and injected into a Waters XBridge C18 column (5 μm, 250 mm × 4.6-mm inner diameter) for identification and quantification, with use of a mobile phase of acetonitrile and 0.2 vol% aqueous H3PO4, with 15% acetonitrile from 0 to 40 min. The flow rate was 1.0 mL min−1, and the injected volume was 20 μL. The column temperature was 30 °C, and the detection wavelength was 349 nm.
Result and discussion
Preparation of ionic liquid MIP with PS
It has been shown that PS is an effective crowding agent for MIPs, and can increase the capacity and selectivity of MIPs [22]. Typically, PS is dissolved in chloroform [31]. However, the resulting polymers are too soft to be evaluated by HPLC. It was also found that the MIPs were soft when they were prepared with use of ionic liquids as the functional monomer, especially in the in situ polymerization process. Recently, THF–PS has been used as a new system of crowding agent to prepare MIP monoliths [32]. In this work, use of THF–PS as the porogenic solvent led to a gel-like polymer, and thus high backpressure. Fortunately, the addition of macromolecular crowding agents can enhance the rigidity of the resulting ionic liquid MIPs. When a mixture of THF–PS–methanol was used as a ternary porogen, MIP monoliths with good permeability and imprinting effect were obtained. In contrast to the NIP monoliths without recognition ability (Fig. 1a), the addition of PS to the polymerization mixture caused greater retention of aesculetin on the ionic liquid MIP (Fig. 1b). The MIP prepared in the presence of PS gave rise to an IF of 11.83, approximately four times as large as that of the MIP prepared in a noncrowding environment. To ensure the reproducibility of the materials, the synthesized materials were prepared four times at the same time. The relative standard of the IFs for all the materials was less than 4.1%.
Chromatograms of aesculetin on a molecularly imprinted polymer (MIP) monolith (P3) at flow rate of 3.0 mL min−1 and nonimprinted polymer (NIP) monoliths (N1 and P6) at flow rate of 1.0 mL min−1 showing the imprinting effect. The mobile phase was acetonitrile and sodium acetate buffer (200 mmol L−1, pH 3.6; 93:7, v/v), the detection wavelength was 349 nm, injected volume was 20 μL, and the temperature was 30 °C. PS polystyrene. a The MIP means that there are template molecule and PS in the column and NIP means that there is PS in the culumn but no template molecule. b P6 means there is template molecule in the column but no PS P3 means that there are template molecule and PS in the column
At the prepolymerization stage, it was expected that complexation of aesculetin and [VEIm][BF4] would be promoted by a high concentration of PS. To verify this hypothesis, a pseudo-prepolymerization mixture of aesculetin, [VEIm][BF4], and PS were subjected to an NMR study. Because of its insignificant role, the initiator AIBN was omitted in the NMR study. EDMA was also omitted because of the carbonyl group, which may form hydrogen bonds with aesculetin and/or [VEIm][BF4] and make the system too complicated to be observed. The concentrations of aesculetin, [VEIm][BF4], and PS were the same as the concentration of P3. When [VEIm][BF4] was added to the solution of aesculetin, the peaks derived from the hydroxy hydrogen protons (8.237 ppm, 9.104 ppm) of aesculetin showed a downfield shift (9.114 ppm, 9.375 ppm). This suggested the formation of hydrogen bonds between the ionic liquid monomer and aesculetin [33,34,35,36]. When PS was added to the pseudo-prepolymerization mixture, a further shift of the hydroxy hydrogen protons of aesculetin downfield was observed (Fig. 2, spectrum c). The NMR data were in agreement with the chromatography data, showing that there were a lot of binding sites in the crowding-assisted polymer [22]. The results indicate that the reason for the enhanced retention of aesculetin was the equilibrium shift to the complexes.
To further verify the validity of the macromolecular crowding agent with regard to the formation of ionic liquid MIPs, the environmental pollutants triclosan (antibacterial and antifungal agent) and imidacloprid (pesticide) were selected as templates (Table 2, Fig. S5). When triclosan was used as the template, the IF of the MIP was 3.95 in the crowded environment but was 0.97 in the absence of the crowded environment. When imidacloprid was used as the template, the IF of the MIP made in the presence of PS was 2.70 but that of the MIP made without PS was 1.78. Thus, the experimental data prove that macromolecular crowding agents can increase the IF of ionic liquid MIPs.
Morphological characterization of imprinted monoliths
The scanning electron micrographs (Fig. 3) show a lot of channels and macropores in the imprinted monolith, and thus chromatography at a high flow rate is possible because of low flow resistance. For instance, at a flow rate of 3.0 mL min-1, the backpressure of the imprinted monolith prepared with PS was only 0.21 MPa. In contrast, the backpressure of the imprinted monolith prepared without PS (P6) was much higher over the whole range of flow rates.
To investigate the differences between MIPs and NIPs made in the presence and absence of PS, the mesopores of the aesculetin-imprinted monoliths were studied by N2 adsorption experiments (Table S1). Data for four samples—MIPs made with the template (P3, P6) and NIPs made without the template (N1, N6)—were obtained. According to IUPAC nomenclature [37], incomplete N2 adsorption–desorption isotherms of type II was obtained with type H2 hysteresis loops (Fig. 4). A difference in the isotherms for the MIPs synthesized in the absence or presence of PS as the porogen was observed [38]. This suggests that there are pores of ill-defined shape and size in the solid monolith materials that were prepared with PS. The Brunauer–Emmett–Teller surface area was 400.28 m2 g−1 for MIP P3 (Table S1) and 427.93 m2 g−1 for NIP N1. The pore diameter of MIP P3 and NIP N1 was 2.25 nm (Fig. S1). Thus, the contribution to the imprinting effect induced by the pore structure and morphology of the PS-based ionic liquid MIPs with aesculetin imprints was insignificant relative to that from the interaction between [VEIm][BF4] and aesculetin. In contrast, the pores of the MIP (P6) and NIP (N6) prepared in the absence of PS had larger average diameter and more cumulative volume than those of the MIP (P3) and NIP (N1) prepared in the presence of PS.
Effect of molar ratio of [VEIm][BF4] to aesculetin
The selectivity of the MIP monolith was affected by the template-to-monomer molar ratio under crowding conditions, and was further studied. The IF and retention factor were investigated by our varying the molar ratio of [VEIm][BF4] to aesculetin in the range from 8:1 to 2:1. As shown in Fig. 5, the selectivity of the MIP increased with increasing concentration of aesculetin. This phenomenon may be due to the increasing concentration of the prepolymerization complex when the molar ratio of [VEIm][BF4] to aesculetin was decreased [39]. However, further increase in the template-to-monomer molar ratio caused the retention factor to decrease. The optimal MIP was P3, which was made with an [VEIm][BF4]-to-aesculetin ratio of 4.8:1.
Effect of different ratios of functional monomer to template on the imprinting factor of aesculetin-imprinted monoliths. The mobile phase was acetonitrile and sodium acetate buffer (200 mmol L−1, pH 3.6; 93:7, v/v), the flow rate was 1.0 mL min−1, the detection wavelength was 349 nm, the injected volume was 20 μL, and the temperature was 30 °C
Effect of PS concentration
The dependence of the IF on the PS concentration in the prepolymerization mixture was studied by our changing the concentration of PS from 10 to 50 mg mL−1 (Fig. 6) (P5, P7, P8, P9, and P10). Higher or lower levels of PS can cause too low or too high backpressure of the MIP monolith, which is difficult to use in HPLC. The tendency of the retention factor with regard to aesculetin and the NIP was the same as for the MIP. In contrast, the IF increased with increased concentration of PS, revealing that the IF can be enhanced by the presence of the crowding agent. Higher PS concentration (40 mg mL−1) led to the MIP with the greatest IF (P3). However, further increase in the PS concentration resulted in a decline of the IF. This shows that the magnitude of the IF can be controlled by a change in the concentration of the macromolecular crowding agent. Thus, it is important to balance the ratio of the template and the functional monomer to obtain the desired performance of the MIP [40].
Effect of different concentrations of polystyrene (PS) of molecular weight 350,000 on the imprinting factor (IF) of the aesculetin-imprinted and nonimprinted monoliths. The mobile phase was acetonitrile and sodium acetate buffer (200 mmol L−1, pH 3.6; 93:7, v/v), the flow rate was 1.0 mL min−1, the detection wavelength was 349 nm, the injected volume was 20 μL, and the temperature was 30 °C
Effect of molecular weight of PS
To study the effect of PS with different molecular weights on the affinity of the ionic liquid MIPs, several polymers (P11, N11, P12, N12, P13, N13, P14, and N14) were prepared (Table 1). Because the effect of the crowding agent on the activity coefficient of each macromolecular species increases significantly when the molecular weight is larger than 103 [41], PS with a molecular weight of 10,000 was chosen as the smallest molecule, but this led to a too rigid polymer monolith to remove the porogenic solvent. The minimum molecular weight of PS to prepare aesculetin-based MIPs is 150,000. The largest IF was achieved with PS with a molecular weight of 350,000 (IF = 11.83) with the optimum concentration of PS (40 mg mL−1) (P3). Further increase in the molecular weight of PS resulted in marginal effects on the resulting MIP, such as difficulty in removing polymer porogens (Fig. 7).
Effect of different molecular weights of polystyrene (PS) on the imprinting factor (IF) of aesculetin-imprinted and nonimprinted monoliths. The mobile phase was acetonitrile and sodium acetate buffer (200 mmol L−1, pH 3.6; 93:7, v/v), the flow rate was 1.0 mL min−1, the detection wavelength was 349 nm, the injected volume was 20 μL, and the temperature was 30 °C. MIP molecularly imprinted polymer, NIP nonimprinted polymer
Effect of composition of the organic phase on retention
The effect of the organic phase composition on the retention factor for aesculetin was investigated with P3 by use of a mixture of acetonitrile and sodium acetate buffer. When the ratio of acetonitrile to sodium acetate buffer was changed from 75% to 95% (Fig. 8), a similar trend of aesculetin retention was found. This proves that the hydrophobic interaction is nonspecific and insignificant in molecular recognition [42]. The interaction mechanism of aesculetin on the ionic liquid MIP seemed to be hydrogen bonding in consideration of the change in the IF observed with the change in the content of the organic modifier (Fig. 8).
Influence of the organic phase composition on the retention factor of aesculetin. The mobile phase was acetonitrile and sodium acetate buffer (200 mmol L−1, pH 3.6; 93:7, v/v), the flow rate was 1.0 mL min−1, the detection wavelength was 349 nm, the injected volume was 20 μL, and the temperature was 30 °C
The selectivity of aesculetin-based MIP
We investigated the selectivity of the aesculetin-based MIP by investigating the retention of aesculetin as well as epigallocatechin gallate, isorhamnetin, estradiol, and apigenin, its structural analogues (Fig. S2). On the ionic liquid MIP, much stronger retention of aesculetin compared with that of its analogues was observed (Fig. S3). In contrast, there is little selectivity for aesculetin and the structural analogues on the NIP (N1), because of the absence of specific cavities in the NIP. Thus, this suggests that highly selective cavities for molecular recognition were formed in the MIP.
Application of aesculetin-based MIP to Cichorium glandulosum extraction
The optimized aesculetin-based MIP (P3) was used for the extraction of Cichorium glandulosum. Because of the low column efficiency, aesculetin cannot be separated from the MIP monolith, and thus online extraction of Cichorium glandulosum was not performed. As a result, P3 was purged out and ground to fabricate an SPE column. The SPE conditions were optimized (Fig. S4) in terms of the extraction of the crude material, the washing solvent, and the eluent.
Aesculetin was identified as corresponding to the peak at 19.317 min by comparison of the retention time with that of aesculetin standard on the C18 column (Fig. 9, chromatogram B). As aesculetin was strongly absorbed on the MIP column, acetonitrile was used as the washing solvent to thoroughly remove impurities. Aesculetin can be eluted by methanol (Fig. S2c), because methanol can break the hydrogen bond between aesculetin and the MIP. A calibration curve was obtained with aesculetin concentrations from 0.0005 to 0.05 mg mL−1 (correlation coefficient R 2 of 0.9999, y = 1519x + 0.0923). The limit of quantification was 0.12 μg mL−1, and the limit of detection was 0.05 μg mL−1.
Chromatograms of the aesculetin standard (A), Cichorium glandulosum extract (B), washing fraction (C), and elution fraction after solid-phase extraction (D). Peak 1 corresponds to aesculetin. The high-performance liquid chromatography conditions were as follows: Waters XBridge C18 column (5 μm, 250 mm × 4.6-mm inner diameter); mobile phase of methanol and 0.2 vol% aqueous H3PO4, 0–40 min 15% methanol; flow rate, 1.0 mL min−1; detection wavelength, 349 nm; injected volume, 20 μL; temperature, 30 °C
The absolute recovery of aesculetin was (80 ± 2)% (n = 3). When the target compound from the washing fraction and eluent fraction could not be detected, the target compound was thought to have been washed out thoroughly. Actually, in the washing process, some target compounds were lost. In contrast, in the elution process, the strong retention of target compounds on the SPE material may have resulted in relatively low absolute recoveries. The purity of aesculetin obtained from SPE was (92 ± 0.5)% (n = 5). Much more pure aesculetin in the elution extract than in the crude extract indicated the outstanding performance of the ionic liquid MIP to purify aesculetin from a complex matrix. The analogues of aesculetin were strongly adsorbed on the SPE column (Fig. S6). This may be due to poor solubility of the analogues in acetonitrile. Another reason may be the strong adsorption of the SPE materials. Figure S7 shows that when methanol was used as the eluent, some analogues were washed out. This can be attributed to a cross-reaction phenomenon [1]. Because of the main functional structure and steric configuration of the analogues, the force between the template and the material was similar, and thus similar chromatographic behavior of the analogues of aesculetin was observed.
Conclusions
By use of an ionic liquid as a functional monomer, a highly specific imprinted monolith for aesculetin was prepared with PS–THF–methanol as a ternary porogenic system and PS as a crowding agent. The results demonstrated that molecular crowding can be used to enhance the capacity and selectivity of a molecularly imprinted sorbent. The MIP prepared in the presence of PS gave rise to an IF approximately four times as large as that of the MIP prepared in a noncrowding environment. NMR studies showed that the presence of the crowding agent can promote an equilibrium shift in the direction of the formation of template–monomer complexes. The results indicate that the origin of the enhanced imprinting effect for the template presumably generates a lot of binding sites in a crowding-assisted environment. In conclusion, the ionic liquid can be used as a functional monomer for synthesis of MIP monoliths even in a polar solvent, and the presence of a molecular crowding agent can further enhance the imprinting effect.
References
Wulff G. Molecular imprinting in cross-linked materials with the aid of molecular templates—a way towards artificial antibodies. Angew Chem Int Ed Eng. 1995;34:1812–32.
Schillemans JP, Van CF. Molecularly imprinted polymer particles: synthetic receptors for future medicine. Nanomedicine. 2006;1:437–47.
Zhao P, Hao JC. 2,6-Diaminopyridine-imprinted polymer and its potency to hair-dye assay using graphene/ionic liquid electrochemical sensor. Biosens Bioelectron. 2015;64:277–84.
Graniczkowska K, Putz M, Hauser FM, De Saeger S, Beloglazova NV. Capacitive sensing of N–formylamphetamine based on immobilized molecular imprinted polymers. Biosens Bioelectron. 2017;92:741–7.
Zhang ZJ, Zhang XH, Liu BW, Liu JW. Molecular imprinting on inorganic nanozymes for hundred-fold enzyme specificity. J Am Chem Soc. 2017;139:5412–9.
Zhang XY, Zhang N, Du CB, Guan P, Gao XM, Wang CY, et al. Preparation of magnetic epitope imprinted polymer microspheres using cyclodextrin-based ionic liquids as functional monomer for highly selective and effective enrichment of cytochrome c. Chem Eng J. 2017;317:988–98.
Wang MF, Lv YQ, Liu XJ, Qi W, Su RX, He ZM. Enhancing the activity of peptide-based artificial hydrolase with catalytic Ser/His/Asp triad and molecular imprinting. ACS Appl Mater Interfaces. 2016;8:14133–41.
Gu Y, Yan XY, Li C, Zheng B, Li YR, Liu WL, et al. Biomimetic sensor based on molecularly imprinted polymer with nitroreductase-like activity for metronidazole detection. Biosens Bioelectron. 2016;77:393–9.
Ban L, Han X, Wang XH, Huang YP, Liu ZS. Carprofen-imprinted monolith prepared by reversible addition-fragmentation chain transfer polymerization in room temperature ionic liquids. Anal Bioanal Chem. 2013;405:8597–605.
Shi SY, Guo F, You QP, Chen XQ, Zhang YP. Selective and simultaneous extraction and determination of hydroxybenzoic acids in aqueous solution by magnetic molecularly imprinted polymers. Chem Eng J. 2014;243:485–93.
Fan DX, Jia L, Xiang HY, Peng MJ, Li H, Shi SY. Synthesis and characterization of hollow porous molecular imprinted polymers for the selective extraction and determination of caffeic acid in fruit samples. Food Chem. 2017;224:32–6.
Li J, Lu JX, Qiao XG, Xu ZX. A study on biomimetic immunoassay-capillary electrophoresis method based on molecularly imprinted polymer for determination of trace trichlorfon residue in vegetables. Food Chem. 2017;221:1285–90.
Bi W, Zhou J, Row KH. Solid phase extraction of lactic acid from fermentation broth by anion-exchangeable silica confined ionic liquids. Talanta. 2011;83(3):974–9.
Hou KX, Chen FL, Zu YG, Yang L. Ionic liquids-lithium salts pretreatment followed by ultrasound-assisted extraction of vitexin-4″-O-glucoside, vitexin-2″-O-rhamnoside and vitexin from Phyllostachys edulis leaves. J Chromatogra A. 2016;1431:17–26.
Du CB, Hu XL, Guan P, Guo LX, Qian LW, Song RY, et al. Water-compatible surface-imprinted microspheres for high adsorption and selective recognition of peptide drug from aqueous media. J Mater Chem B. 2016;3:3044–53.
Bi W, Tian M, Row KH. Separation of phenolic acids from nature plant extracts using molecularly imprinted anion-exchange polymer confined ionic liquids. J Chromatogr A. 2011;1232:37–42.
Guo L, Deng QL, Fang GZ, Gao W, Wang S. Preparation and evaluation of molecularly imprinted ionic liquids polymer as sorbent for on-line solid-phase extraction of chlorsulfuron in environmental water samples. J Chromatogr A. 2011;37:6217–77.
Xiang HY, Peng MJ, Li H, Peng S, Shi SY. High-capacity hollow porous dummy molecular imprinted polymers using ionic liquid as functional monomer for selective recognition of salicylic acid. J Pharm Biomed Anal. 2017;133:75–81.
Nakano S, Karimata H, Ohmichi T, Kawakami J, Sugimoto N. The effect of molecular crowding with nucleotide length and cosolute structure on DNA duplex stability. J Am Chem Soc. 2004;126:14330–1.
Minton AP. Molecular crowding: analysis of effects of high concentrations of inert cosolutes on biochemical equilibria and rates in terms of volume exclusion. Methods Enzymol. 1998;295:127–49.
Minton AP. The influence of macromolecular crowding and macromolecular confinement on biochemical reactions in physiological media. Biol Chem. 2001;276:10577–80.
Matsui J, Goji S, Murashima T, Miyoshi D, Komai S, Shigeyasu A, et al. Molecular imprinting under molecular crowding conditions: an aid to the synthesis of a high-capacity polymeric sorbent for triazine herbicides. Anal Chem. 2007;79:1749–57.
Li XX, Hao LF, Huang YP, Duan HQ, Liu ZS. Release evaluation of molecularly imprinted polymers prepared under molecular crowding conditions. Polym Eng Sci. 2012;52(7):1440–9.
Zong HY, Liu X, Liu ZS, Huang YP. Molecular crowding-based imprinted monolithic column for capillary electrochromatography. Electrophoresis. 2015;36:764–72.
Tang L, Zhao CY, Wang XH, Li RS, Yang JR, Huang YP, et al. Macromolecular crowding of molecular imprinting: a facile pathway to produce drug delivery devices for zero-order sustained release. Int J Pharm. 2015;496:822–33.
Chu CY, Tsai YY, Wang CJ. Induction of apoptosis by esculetin in human leukemia cells. Eur J Pharmacol. 2001;416:25–32.
Wang CJ, Hsieh YJ, Chu CY. Inhibition of cell cycle progression in human leukemia HL-60 cells by esculetin. Cancer Lett. 2002;183:163–8.
Pincova L, Petru K, Pospisilova M, Polasek M. Micellar electrokinetic chromatography with short-end injection for rapid separation and simultaneous determination of aesculin, aesculetin, and phenylephrine in pharmaceutical preparations. Electrophoresis. 2015;36(21):2754–61.
Kubrak T, Dresler S, Szymczak G, Bogucka-kocka A. Rapid determination of coumarins in plants by capillary electrophoresis. Anal Lett. 2015;48:2819–32.
Tao YW, Xu WD, Tian Y, Shi JG. Steroids and aromatic derivatives from Euphorbia micractina. China J Chin Mater Med. 2015;40(23):4639–44.
Schmidt RH, Belmont AS, Haupt K. Porogen formulations for obtaining molecularly imprinted polymers with optimized binding properties. Anal Chim Acta. 2005;542:118–24.
Ban L, Zhao L, Deng BL, Huang YP, Liu ZS. Preparation and characterization of an imprinted monolith by atom transfer radical polymerization assisted by crowding agents. Anal Bioanal Chem. 2013;405:2245–53.
Quaglia M, Chenon K, Hall AJ, Lorenzi DE, Sellergren B. Target analogue imprinted polymers with affinity for folic acid and related compounds. J Am Chem Soc. 2001;123:2146–54.
Spivak D, Gilmore MA, Shea KJ. Evaluation of binding and origins of specificity of 9-ethyladenine imprinted polymers. J Am Chem Soc. 1997;119:4388–93.
Welhouse GJ, Bleam WF. Atrazine hydrogen-bonding potentials. Environ Sci Technol. 1993;27:494–500.
Sellergren B, Lepistoe M, Mosbach KJ. Highly enantioselective and substrate-selective polymers obtained by molecular imprinting utilizing noncovalent interaction–NMR and chromatographic studies on the nature of recognition. J Am Chem Soc. 1988;110:5853–60.
Sing KSW. Reporting physisorption data for gas/solid systems with special reference to the determination of surface area and porosity. Pure Appl Chem. 1982;54:2201–18.
Liu X, Wei ZH, Huang YP, Yang JR, Liu ZS. Molecularly imprinted nanoparticles with nontailing peaks in capillary electrochromatography. J Chromatogr A. 2012;1264:137–42.
Mosbach K, Ramström O. The emerging technique of molecular imprinting and its future impact on biotechnology. Biotechnology. 1996;14:163–70.
Li ZY, Liu ZS, Zhang QW, Duan HQ. Chiral separation by (S)-naproxen imprinted monolithic column with mixed functional monomers. Chin Chem Lett. 2007;18:322–4.
Ellis RJ. Macromolecular crowding: an important but neglected aspect of the intracellular environment. Curr Opin Struct Biol. 2001;11:114–9.
Kondo Y, Yoshikawa M. Effect of solvent composition on chiral recognition ability of molecularly imprinted DIDE derivatives. Analyst. 2001;126:781–3.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (grant no. U1303202), the Major Research Development Program of Xinjiang (grant no. 2016B03044-2), and the Urumqi Science and Technology Plan (Y141310027).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Electronic supplementary material
ESM 1
(PDF 1 mb)
Rights and permissions
About this article
Cite this article
Jia, M., Yang, J., Sun, Y.K. et al. Improvement of imprinting effect of ionic liquid molecularly imprinted polymers by use of a molecular crowding agent. Anal Bioanal Chem 410, 595–604 (2018). https://doi.org/10.1007/s00216-017-0760-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-017-0760-5