Abstract
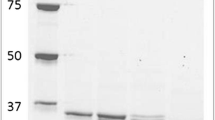
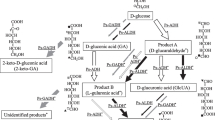
A novel C-2-specific sugar oxidoreductase, tentatively designated as pyranose 2-dehydrogenase, was purified 68-fold to apparent homogeneity (16.4 U/mg protein) from the mycelia of Agaricus bisporus, which expressed maximum activity of the enzyme during idiophasic growth in liquid media. Using 1,4-benzoquinone as an electron acceptor, pyranose 2-dehydrogenase oxidized d-glucose to d-arabino-2-hexosulose (2-dehydroglucose, 2-ketoglucose), which was identified spectroscopically through its N,N-diphenylhydrazone. The enzyme is highly nonspecific. d-,l-Arabinose, d-ribose, d-xylose, d-galactose, and several oligosaccharides and glycopyranosides were all converted to the corresponding 2-aldoketoses (aldosuloses) as indicated by TLC. d-Glucono-1,5-lactone, d-arabino-2-hexosulose, and l-sorbose were also oxidized at significant rates. UV/VIS spectrum of the native enzyme (λmax 274, 362, and 465 nm) was consistent with a flavin prosthetic group. In contrast to oligomeric intracellular pyranose 2-oxidase (EC 1.1.3.10), pyranose 2-dehydrogenase is a monomeric glycoprotein (pI 4.2) incapable of reducing O2 to H2O2 (> 5 × 104-fold lower rate using a standard pyranose oxidase assay); pyranose 2-dehydrogenase is actively secreted into the extracellular fluid (up to 0.5 U/ml culture filtrate). The dehydrogenase has a native molecular mass of ∼79 kDa as determined by gel filtration; its subunit molecular mass is ∼75 kDa as estimated by SDS-PAGE. Two pH optima of the enzyme were found, one alkaline at pH 9 (phosphate buffer) and the other acidic at pH 4 (acetate buffer). Ag+, Hg2+, Cu2+, and CN– (10 mM) were inhibitory, while 50 mM acetate had an activating effect.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Additional information
Received: 19 August 1996 / Accepted: 21 November 1996
Rights and permissions
About this article
Cite this article
Volc, J., Kubátová, E., Wood, D. et al. Pyranose 2-dehydrogenase, a novel sugar oxidoreductase from the basidiomycete fungus Agaricus bisporus. Arch Microbiol 167, 119–125 (1997). https://doi.org/10.1007/s002030050424
Issue Date:
DOI: https://doi.org/10.1007/s002030050424