Abstract
The present study observed significant effects of whole-body vibration (WBV) on bone mineral density (BMD) in postmenopausal women, with high-quality evidence for high-frequency, low-magnitude, and high-cumulative-dose use. The aim was to update a previous systematic review with meta-analysis to observe the effects of WBV on BMD in postmenopausal women. For the meta-analysis, the weighted mean difference between WBV and control groups, or WBV and conventional exercise, was used for the area of bone mineral density (aBMD) of the lumbar spine, femoral neck, total hip, trochanter, intertrochanter, and Ward’s area, or volumetric trabecular bone mineral density (vBMDt) of the radius and tibia. Methodological quality was assessed using the PEDro scale and the quality of evidence using the GRADE system. In total, 23 studies were included in the systematic review and 20 in the meta-analysis. Thirteen studies showed high methodological quality. WBV compared with control groups showed significant effects on aBMD in the primary analysis (lumbar spine and trochanter), sensitivity (lumbar spine), side-alternating vibration (lumbar spine and trochanter), synchronous vibration (lumbar spine), low frequency and high magnitude (lumbar spine and trochanter), high frequency and low magnitude (lumbar spine), high frequency and high magnitude (lumbar spine, trochanter, and Ward’s area), high cumulative dose and low magnitude (lumbar spine), low cumulative dose and high magnitude (lumbar spine and trochanter), and positioning with semi-flexed knees (trochanter). Of these results, only high frequency associated with low magnitude and high cumulative dose with low magnitude showed high-quality evidence. At this time, considering the high quality of evidence, it is possible to recommend WBV using high frequency (≈ 30 Hz), low magnitude (≈ 0.3 g), and high cumulative dose (≈ 7000 min) to improve lumbar spine aBMD in postmenopausal women. Other parameters, although promising, need to be better investigated, considering, when applicable, the safety of the participants, especially in vibrations with higher magnitudes (≥ 1 g).
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Randomized controlled trials (RCTs) have been carried out to verify the effects of whole-body vibration (WBV) on bone mineral density (BMD) in postmenopausal women since 2003 [1,2,3,4,5,6,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23]. The first systematic review study with meta-analysis on the subject was published in 2010 by Slatkovska et al. [24], with an update proposed by our research group in 2016 [25]. Since then, new RCTs have emerged [1,2,3,4,5,6,7], thus necessitating a new update.
Postmenopausal women suffer from a number of negative health factors, due to the hormonal alterations typical of this period of life, such as the decrease in estrogen levels, with the decrease in BMD being one of the main factors, increasing the risk of fracture and its consequences, which can lead to hospitalization and death [26]. Drug treatment to attenuate or reverse the loss of BMD, despite being efficient, promotes side effects and is restricted to limited periods of time [27]. On the other hand, the systematic practice of physical exercises involving muscle resistance promotes mechanical stimuli capable of increasing BMD and presents few adverse events [28].
However, exercises that involve muscle resistance may have low adherence, especially by older people [29]. Interventions with WBV [30] are a relatively safe practice that requires little effort and low exposure time, has few adverse events, and has shown significant effects on BMD [1,2,3,4,5, 12, 13, 17, 20, 21]. In this form of intervention, the individual normally stands on a vibrating platform that transmits mechanical stimuli throughout the body, requiring a greater response from muscle and bone tissues to absorb and dampen the energy caused by the oscillatory actions [31].
In this sense, mechanical stimuli made possible by WBV seem to be capable of providing osteogenic effects: (a) directly, such as through osteocytes and Wnt–β-catenin signaling, and (b) indirectly, through skeletal muscle activation, resulting from the stretch reflex [32]. Even so, the exact mechanism by which WBV would potentially increase BMD remains under discussion, mainly because different factors can influence this outcome.
In animal studies, it has recently been shown that low frequencies can induce greater BMD than higher frequencies [33,34,35]. However, in humans, these same parameters present contradictory results [25]. One of the hypotheses for the contradictions reported is that the vibration parameters (frequency, amplitude, and magnitude) can vary widely [1,2,3,4,5,6,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23]. In addition, different factors can influence the results observed, such as the type of vibration (synchronous or side-alternating), the position on the platform (e.g., standing with knees extended, knees semi-flexed, or performing squat exercises), and the cumulative dose of WBV, which takes into account session duration, weekly frequency, and total intervention duration [25].
With this issue in mind, the aim of the current study was to update the previously published systematic review and meta-analysis [25], in order to observe the effects of WBV vs. no intervention, minimal intervention, and other forms of exercise, considering the different parameters that can impact BMD gain in postmenopausal women.
Methods
This study was prospectively registered in PROSPERO (CRD42021288930). For reporting, the recommendations of the PRISMA checklist [36] were followed and for methodological questions the Cochrane Handbook for Systematic Reviews of Interventions [37].
Inclusion/exclusion criteria
Inclusion criteria
RCTs, which investigated the effects of WBV on BMD on postmenopausal women (there was no restriction on ethnicity or level of physical activity); studies with an intervention period of 6 months or more; studies in which WBV training occurred through a sinusoidal oscillation platform.
Exclusion criteria
Studies with duplicate information in another RCT; non-use of WBV training on sinusoidal oscillation platforms (e.g., localized mechanical vibration or electrical stimulation); studies in which the volunteers were not standing on the vibrating platform (e.g., sitting or lying down); studies in which the volunteers who used medication to improve BMD were not evenly divided between the vibration and control groups; studies that associated WBV with another form of exercise and did not have a comparison group that performed the same form of exercise.
Databases and search strategy
The search was performed in the following databases: EMBASE, PubMed, CENTRAL, CINHAL, Web of Science, LILACS, PEDro, and SportDiscus, without using a filter for publication date or language. Two clinical trial registry databases were consulted (clinicaltrials.gov and https://www.who.int) to identify any potential unpublished studies. The searches took place in December 2021.
The following keywords were used in the search strategy: (“whole body vibration” OR “WBV” OR “vibration” OR “vibrations” OR “vibratory” OR “vibration therapy” OR “vibration training” OR “oscillating platforms” OR “vibration plate”) AND (“bone mineral density” OR “bone density” OR “bone” OR “bones” OR “bone mass” OR “bone mineral content” OR “bone quality” OR “BMC” OR “BMD” OR “bone strength” OR “osteoporosis” OR “osteopenia”) AND (“postmenopausal” OR “menopause” OR “perimenopause” OR “climacteric” OR “postmenopausal women” OR “aged” OR “aging” OR “ageing” OR “elderly” OR “older people” OR “older adults” OR “older adult” OR “older women” OR “geriatric” OR “geriatrics”).
Selection of studies
One reviewer performed the initial search strategy in the databases, extracting titles and abstracts. Subsequently, the selection of studies, evaluation, and data extraction were independently conducted by two authors. First, titles and abstracts were read. Subsequently, potentially eligible studies were read in full. For eligible articles, a manual search was performed in the reference lists, to identify any further studies. Differences, when not resolved between the two researchers, were passed on to a third researcher who decided on the issue. The same data extraction form was used by both authors.
The PICO method [37] was used to structure the bibliographic search and data extraction: P (population) = postmenopausal women; I (intervention) = WBV; C (comparison) = no intervention, or other forms of intervention; O (outcome) = BMD.
Data extraction
The following variables were extracted from each study: (a) name of the first author and year of publication; (b) number of participants allocated to each group and geographic region where the study was conducted; (c) mean and standard deviation of age in each group; (d) time exposed to vibration (months, weekly frequency, daily minutes of vibration); (e) vibration frequency, peak-to-peak displacement, and/or vibration magnitude and type (synchronous or side-alternating); (f) positioning of the body or activity performed on the vibrating platform; (g) activities carried out by other intervention groups; (h) activities of the control group; (i) BMD assessment instrument and assessed region; (j) condition of the participants regarding alterations in BMD (no change, osteopenic, or osteoporotic); (k) use of calcium, vitamin D, or medication; (l) results reported for BMD; (m) percentage of volunteers who completed the WBV program; and (n) adverse events resulting from WBV.
Assessment of the methodological quality of the studies
Methodological quality was assessed using the PEDro (Physiotherapy Evidence Database) scale [38,39,40]. Whenever possible, the scores were extracted from the PEDro platform database (https://pedro.org.au/). When studies were not found in the PEDro database, two independent trained reviewers rated the article using the PEDro scale. This scale takes into account the internal validity and sufficiency of statistical information of the studies, presenting 11 questions, with three items from the Jadad scale [41] and nine items from the Delphi list [42]. The first question is not scored (related to the external validity of the study). Each item that meets the required criteria receives one point, making it possible to classify each study as high (score ≥ 6) or low (score < 6) quality. Maher et al. [39] demonstrated good inter-rater reliability, with an intra-class correlation coefficient of 0.68 when using consensus ratings, generated by two or three independent raters on the PEDro scale.
Definition of WBV
WBV was defined as mechanical vibrations provided by a sinusoidal oscillation platform that transmits mechanical stimuli to the human body standing on it. Two main types of vibrating platforms are marketed: (a) synchronous vibration and (b) side-alternating vibration. In the first, vibration occurs in a predominantly vertical direction, synchronously across the base of the oscillating platform, while in the second, the vibration occurs through an anteroposterior axis, causing the right and left sides to alternate horizontally [31].
Essentially, vibrating platforms make it possible to configure two parameters that affect the intensity of the vibration: frequency expressed in hertz (Hz) and peak-to-peak displacement in millimeters (mm), which determine the magnitude of the acceleration due to gravity, expressed in grams (g) or meters per second squared (m/s2). The acceleration can be obtained by an accelerometer or estimated using the formula: m/s2 = 2.π2.f2.m, where f is the frequency in Hz and m is the peak-to-peak displacement expressed in meters (gravitational acceleration: 1 g = 9.8 m/s2) [31]. Regarding the classification of these parameters, the magnitude is considered high when ≥ 1 g [30] and the frequency, when > 20 Hz [12, 22].
External factors also determine the vibration intensity, such as the cumulative dose of WBV (obtained by multiplying the time of each session, the weekly frequency, and the total intervention period) [24, 25] and the body position on the oscillation platform, which typically occurs while standing with the knees extended, semi-bent, or performing muscle strengthening exercises during WBV [25].
Synthesis of results
For the meta-analysis, the measure of effect was a weighted mean difference between the WBV vs. control, or WBV vs. conventional exercises, in absolute change in bone mineral density area (aBMD) between pre- and post-intervention, measured by dual-energy x-ray absorptiometry (DXA) expressed in g/cm2, for the following body segments: lumbar spine, total hip, femoral neck, trochanter, intertrochanter, and Ward’s area; or trabecular volumetric bone mineral density (vBMDt), measured by peripheral quantitative computed tomography (pQCT) expressed in mg/cm3, for the following body segments: tibia and radius.
The Cochrane Q test for heterogeneity was performed and considered statistically significant if p ≤ 0.10. Heterogeneity was also quantified with the I2 statistic, where 0–40% may not be important, 30–60% may represent moderate heterogeneity, 50–90% may represent high heterogeneity, and 75–100% is defined as considerable heterogeneity [37]. Fixed effects models were used when there was no statistically significant heterogeneity; otherwise, random effects models were used. The values referring to the treatment effect were only considered statistically significant when p < 0.05. To assess the risk of publication bias, a funnel plot was used when there were ≥ 10 trials in a meta-analysis. All analyses were performed using Review Manager (RevMan) [Computer program], version 5.4, Copenhagen: the Nordic Cochrane Centre, Cochrane Collaboration. As this is a systematic review update study, we adopted the same procedures for statistical manipulation of BMD data as previously described [25].
The overall quality of evidence in each meta-analysis was rated according to the Grading of Recommendations, Assessment, Development, and Evaluation (GRADE) [43], by two independent reviewers in a blinded manner, with disagreements being resolved by consensus. GRADE has domains to establish the quality of evidence: (a) limitations in study design or execution (risk of bias), (b) inconsistency of results, (c) indirectness of evidence, (d) imprecision, and (e) other factors (publication bias, dose–response gradient, effect magnitude, and confounding factors).
The GRADE approach contemplates reasons to decrease or increase the quality of evidence in each meta-analysis. It is therefore possible, for each analysis performed, to classify the degree of quality of the evidence, as (a) high — in this case, further research is unlikely to change the estimate or confidence in the results; (b) moderate — it is likely that new research will have an impact on the confidence in the estimate of the effect, and may even modify the estimate; (c) low — future research is likely to have a significant impact on the confidence in the effect estimate and change the estimate; and (d) very low — results are highly uncertain.
Results
Qualitative synthesis of studies
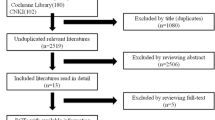
It was possible to identify 3098 potentially relevant reports in the databases, in addition to 24 clinical trial records. After removing the duplicates, 1727 titles and abstracts were read, of which 1669 were excluded because they did not meet the inclusion/exclusion criteria. Of the remaining 56 reports, one was not found. Therefore, fifty-five reports were accessed in full, of which thirty-two did not meet the eligibility criteria (a complete list of studies excluded at this stage is available in Supplementary Table 1). The reasons for exclusion were (a) not being an RCT (eleven studies), (b) intervention less than 6 months (six studies), (c) not evaluating BMD (five studies), (d) other populations (five studies), and (e) no WBV intervention (four studies). One study [44] included in the previous version of the current review was retracted in the year 2019 [45] so it was excluded from this update. Thus, twenty-three reports were included in the systematic review, comprising twenty-one studies (the reports by Marín-Cascales et al. [6] and Marín-Cascales [7] make up the same study, as well as the reports by Slatkovska et al. [11] and Slatkovska et al. [15]) (Fig. 1). Of these, twenty studies included enough information to compose the meta-analysis [2,3,4,5,6, 8,9,10,11,12,13,14,15,16,17,18, 20,21,22,23].
The RCTs included in this systematic review (Table 1 [1,2,3,4,5,6,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23]) were published between the years 2003 and 2021, and the total number of participants was 2089 (ranging from 28 [12, 20] to 596 women [10]). The groups in each study ranged from two [1, 3, 5, 8,9,10, 12, 13, 20, 22, 23] to four [16] and the mean age of participants ranged from 53 [14] to 82.3 [8] years. The intervention time ranged from six [2,3,4, 6, 7, 12, 14, 16, 21, 23] to 18 months [10, 18]. The weekly frequency of WBV ranged from two [3, 8, 9, 13, 18, 23] to seven times [1, 11, 15, 22] and the exposure time between 1 min (at a given time in the periodization) [16] to 30 min [21]. The mean cumulative dose of WBV ranged from 208 min [23] to 7800 min [10]. Regarding the WBV parameters, the frequency ranged from 12 [23] to 90 Hz [11, 15]; the peak-to-peak amplitude ranged from 1 mm [5] to 12 mm [17]; and the magnitude ranged from 0.1 g [23] to 12.9 g [6, 7]. The type of vibration was synchronous in 16 studies [1,2,3, 5,6,7,8, 10, 11, 14,15,16,17,18, 21, 22] and side-alternating in 8 studies [4, 9, 12, 13, 17, 19, 20, 23].
The activities of the groups that performed WBV were strengthening exercises for the lower limbs during vibration [2, 3, 6,7,8, 13, 16,17,18, 21]; remaining with the knees extended [1, 10,11,12, 15, 19, 22]; or semi-flexion of the knees [4, 5, 9, 14, 19, 20, 23]. In three studies, vibration training was associated with conventional muscle strengthening exercise [2, 13, 18]. In seven studies, there was a specific conventional exercise group [4, 6, 7, 13, 14, 18, 21], and only one study [13] did not include a no-intervention or minimal intervention group.
BMD assessment was performed using DXA [1,2,3,4,5,6,7,8,9,10, 12, 14,15,16,17,18,19,20,21,22], pQCT [1, 5, 9, 13, 15, 23], or quantitative ultrasound (QUS) [11]. Regarding BMD classification, the studies recruited participants in the following conditions: unaltered, osteopenic, or osteoporotic [4, 8, 12]; without alteration or osteopenic [1, 21]; osteopenic or osteoporotic [3, 11, 14, 15]; only osteopenic [9, 13]; only osteoporotic [2, 5, 22]; and the other studies did not report the BMD classification. Regarding vitamin D and calcium supplementation, eight studies administered daily doses [2, 3, 11, 15,16,17,18, 23] and only one administered Teriparatide [5].
Of the 23 RCTs included in this systematic review, nine did not observe that WBV training provides significant improvement in intra- or intergroup BMD (reported in the original publication) [7,8,9,10,11, 15, 19, 22, 23]; 10 studies observed significant intragroup improvement (pre- vs. post-intervention) for the lower back [2,3,4,5,6, 12, 18], total hip [2, 16, 21], femoral neck [2, 3], trochanter [3, 4], Ward’s area [3], and radius and tibia [13]; and 10 studies found a significant improvement in intergroup BMD in favor of WBV over time (vibration vs. control), for the lumbar regions [2,3,4,5, 12, 17], total hip [2, 21], femoral neck [2, 3, 20], trochanter [3, 4], Ward’s area [3], radius [13], and tibia [1]. When reported, mean compliance to WBV programs was 84.2%, ranging from 66% [10] to 97.2% [6], similar to what was found in the conventional exercise groups, where the mean was 85.9%, ranging from 75% [18] to 95.8% [6, 7] (Supplementary Table 2).
Methodological quality of the studies
Table 2 demonstrates the methodological quality of the studies, with a mean of 6.0 ± 1.5 points (range of 4 to 10 points). Of the 23 studies included in the systematic review, 13 [1, 3,4,5,6, 10, 11, 15,16,17,18,19, 22] had high methodological quality (PEDro score ≥ 6).
Quantitative synthesis of the studies (meta-analysis)
For each meta-analysis, we analyzed the quality of the evidence using GRADE (Supplementary Table 3), with very low to low-quality evidence for most analyses (87%). The main problems were linked to the risk of bias, inconsistency, and imprecision.
Primary and sensitivity analyses
Table 3 and Figs. 2 and 3 present primary and sensitivity analyses for aBMD. In the primary analysis, in which all studies were included, there was a significant effect in the comparison between WBV and control groups for aBMD of the lumbar spine and trochanter, with evidence of low (downgraded by risk of bias and inconsistency) and very low (downgraded by risk of bias, inconsistency, and imprecision) quality, respectively. In the sensitivity analyses, in which studies of lower methodological quality were excluded (score < 6 on the PEDro scale), there was a significant effect for WBV compared with control groups only for aBMD of the lumbar spine, with moderate quality evidence (downgraded by inconsistency). No effect was observed for vBMDt of the radius and tibia (Supplementary Figs. 1 and 2).
Sensitivity analyses of whole-body vibration effect on areal bone mineral density (g/cm2) in postmenopausal women — excluding clinical trials with more bias (score < 6 on the Physiotherapy Evidence Database — PEDro scale). a Lumbar spine. b Femoral neck. c Total hip. d Trochanter. e Intertrochanter. f Ward’s area
Side-alternating vibration
In the analysis of subgroups involving side-alternating vibration, there was a significant difference in favor of WBV when compared with the control groups, for the aBMD of the lumbar spine (0.017 g/cm2 [95% confidence interval (CI), 0.012 to 0.022] p < 0.00001, n = 152, I2 = 34%, studies = 4) and trochanter (0.020 g/cm2 [95% confidence interval (CI), 0.012 to 0.029] p < 0.00001, n = 62, I2 = 0%, studies = 2), both with low-quality evidence (downgraded by risk of bias and imprecision). No significant effect was observed for other bone regions (Supplementary Fig. 3).
Synchronous vibration
When only studies that performed synchronous vibration were grouped together, there was a significant difference in favor of WBV when compared with the control groups, only for aBMD of the lumbar spine (0.007 g/cm2 [95% confidence interval (CI), 0.000 to 0.013] p = 0.04, n = 1261, I2 = 75%, studies = 11), with low quality of evidence (downgraded by risk of bias and inconsistency). No significant effect was observed for other bone regions (Supplementary Fig. 4).
Low frequency and high magnitude
In the subgroup analysis, in which we included only studies that used low frequency (≤ 20 Hz) and high magnitude (≥ 1 g), significant effects comparing WBV with control groups were observed for aBMD of the lumbar spine (0.014 g/cm2 [95% confidence interval (CI), 0.005 to 0.023] p = 0.004, n = 124, I2 = 53%, studies = 3) and trochanter (0.019 g/cm2 [95% confidence interval (CI), 0.012 to 0.027] p < 0.00001, n = 99, I2 = 0%, studies = 3), with evidence of very low (downgraded by risk of bias, inconsistency, and imprecision) and low (downgraded by risk of bias and imprecision) quality, respectively. No significant effect was observed for other bone regions (Supplementary Fig. 5).
High frequency and low magnitude
When we grouped studies that used low frequency (> 20 Hz) and low magnitude (< 1 g), significant effects comparing WBV with control groups were observed for aBMD of the lumbar spine (0.004 g/cm2 [95% confidence interval (CI), 0.000 to 0.007] p = 0.03, n = 868, I2 = 22%, studies = 3), with high-quality evidence. No significant effect was observed for other bone regions (Supplementary Fig. 6).
High frequency and high magnitude
In the subgroup analysis in which we included only studies that used high frequency (> 20 Hz) and high magnitude (≥ 1 g), significant effects comparing WBV with control groups were observed for aBMD of the lumbar spine (0.012 g/cm2 [95% confidence interval (CI), 0.002 to 0.021] p = 0.02, n = 392, I2 = 78%, studies = 9), with very low quality of evidence (downgraded by risk of bias, inconsistency, and imprecision), trochanter (0.040 g/cm2 [95% confidence interval (CI), 0.017 to 0.064] p = 0.001, n = 43, I2 = not applicable, studies = 1), and Ward’s area (0.140 g/cm2 [95% confidence interval (CI), 0.081 to 0.199] p < 0.00001, n = 43, I2 = not applicable, studies = 1), both with low-quality evidence (downgraded by inconsistency and imprecision). No significant effect was observed for other bone regions (Supplementary Fig. 7).
High cumulative dose and high magnitude
For subgroup analysis in which we included only studies with a high cumulative dose (> 822 min) and high magnitude (≥ 1 g), no significant difference was observed between WBV and control groups for BMD (Supplementary Fig. 8).
High cumulative dose and low magnitude
When we grouped studies with high cumulative dose (> 822 min) and low magnitude (< 1 g), a significant effect comparing WBV with control groups was observed only for lumbar spine aBMD (0.004 g/cm2 [95% confidence interval (CI), 0.000 to 0.007] p = 0.03, n = 868, I2 = 22%, studies = 3), with high-quality evidence. No significant effect was observed for other bone regions (Supplementary Fig. 9).
Low cumulative dose and high magnitude
In the analysis involving studies with low cumulative dose (< 822 min) and high magnitude (≥ 1 g), significant effects comparing WBV with control groups were observed for aBMD of the lumbar spine (0.022 g/cm2 [95% confidence interval (CI), 0.013 to 0.031] p < 0.00001, n = 185, I2 = 58%, studies = 6), with very low quality of evidence (downgraded by risk of bias, inconsistency, and imprecision), and trochanter (0.023 g/cm2 [95% confidence interval (CI), 0.012 to 0.035] p < 0.00001, n = 114, I2 = 32%, studies = 3), with low quality of evidence (downgraded by risk of bias and imprecision). No significant effect was observed for other bone regions (Supplementary Fig. 10).
Knees extended during the WBV
For the positioning of the volunteers with knees extended, no significant difference was observed in BMD comparing the WBV with the control groups (Supplementary Fig. 11).
Semi-flexed knees during the WBV
When we grouped the studies in which the participants’ body positioning during vibration was with the knees semi-flexed, significant effects comparing WBV with the control groups were observed for aBMD of the trochanter (0.020 g/cm2 [95% confidence interval (CI), 0.012 to 0.028] p < 0.00001, n = 62, I2 = 0%, studies = 2), with low quality of evidence (downgraded by risk of bias and imprecision). However, the analysis involving total hip showed a favorable effect for the control group (− 0.011 g/cm2 [95% confidence interval (CI), − 0.020 to − 0.001] p = 0.03, n = 67, I2 = 0%, studies = 2), with moderate quality of evidence (downgraded by imprecision). No significant effect was observed for other bone regions (Supplementary Fig. 12).
Performing exercises during the WBV
Regarding the performance of exercises on the vibrating platform, no significant effect was observed for BMD, comparing WBV with the control groups (Supplementary Fig. 13).
WBV vs. conventional exercises
When comparing WBV with conventional exercises, no significant effect was observed for BMD (Supplementary Fig. 14).
Analysis per-protocol
Since our analyses prioritized data by intention-to-treat, we performed meta-analyses considering only studies that had available data per-protocol, that is, from subjects who actually completed the intervention program (Supplementary Fig. 15). In this case, we identified that overall, the primary analysis data did not change, with two exceptions: (a) we had identified statistical significance for trochanter aBMD in favor of WBV vs. control, which in the per-protocol analysis was not observed; (b) a significant effect in favor of the WBV vs. control was observed in the per-protocol analysis for total hip aBMD, when in the previous analysis, no effect had been observed. For vBMDt of the radius and tibia, the data in the per-protocol analysis did not change (Supplementary Fig. 16).
Adverse events
Of the 2089 volunteers included in this systematic review, 59 (2.8%) reported having adverse events possibly associated with WBV training, and of these, six gave up the intervention. Five clinical trials did not report adverse events in the text [2, 3, 13, 14, 21]. In eight studies, the authors stated that there were no adverse events related to WBV [1, 5, 6, 9, 12, 17,18,19,20, 22].
In two studies [8, 10], a total of six volunteers reported having back pain, and of these, two gave up the intervention due to pain [8]; Leung et al. [10] also stated that nine volunteers felt pain in their legs (three reported that the pain occurred within the first month of intervention, and six that the pain appeared between 3 and 17 months after the beginning of the intervention); five participants reported feelings of dizziness when performing the WBV training (two volunteers were in the first month of intervention and three were between 6 and 18 months of intervention); eight volunteers had worsening of hypertension after WBV training.
Slatkovska et al. [11, 15] identified that three participants discontinued WBV therapy within the first 2 months of intervention (due to dizziness at night, leg pain, or pain in the sole of the foot); another ten participants reported mild, transient symptoms, such as pain, numbness, or weakness, at various sites in the leg. Other symptoms were nausea (two participants), exacerbation of headaches (one participant), bladder discomfort (one participant), inner ear tenderness (one participant), and sore throat (one participant).
In the study by Russo et al. [23], two overweight participants with pre-existing osteoarthritis in the knee, reported moderate pain in this joint, which decreased after a few days of rest (one participant dropped out of the study due to pain); another six participants reported redness and itching in the lower limbs (during the first three intervention sessions). Oliveira et al. [4] observed the following adverse events associated with WBV: 60% of participants had delayed onset muscle soreness, in addition to muscle spasms and cramps that occurred sporadically.
Discussion
In this systematic review update, we included seven new studies published between 2017 and 2021 [1,2,3,4,5,6,7]. Of these new studies, only one did not present data that would allow inclusion in the meta-analysis [1]. We emphasize that, unlike the meta-analysis previously published by our research group [25], in the current study, we performed an analysis of the quality of the evidence using GRADE. This now allows us to describe with what certainty the results of each meta-analysis can be recommended for decision-making by health professionals. In the previous meta-analysis [25], we did not find any significant results in the primary analyses. In the present review, we identified significant results for aBMD of the lumbar spine and trochanter regions; however, the quality of evidence was low and very low, respectively. This demonstrates that the true effect can be substantially different from the found effect. To increase the quality of the evidence, we excluded studies of low methodological quality (sensitivity analysis). In this case, only the analysis involving the lumbar spine continued to demonstrate a significant effect, with moderate quality evidence. Still, it is possible that subsequent studies could have a significant impact on our confidence in the effect estimate. This specific analysis was downgraded due to inconsistency, which demonstrates the need for more robust RCTs, with an adequate sample size.
For subgroup analysis involving side-alternating vibration, in the current study, we found significant effects for aBMD of the lumbar spine and trochanter, coinciding with the findings of our previous study [25]; however, in the present study, we also identified a significant effect for synchronous vibration in the lumbar spine region, which we had not observed in the previous study. In principle, it is possible to consider that side-alternating vibration led to an effect on two bone regions (lumbar spine and trochanter), while synchronous vibration modified only one region (lumbar spine), plus the larger effect size for the lumbar spine for side-alternating vibration compared to synchronous vibration (0.017 g/cm2 vs. 0.007 g/cm2). However, these analyses had a low quality of evidence. Therefore, it is very likely that subsequent studies will change our confidence in estimating the effect.
An important consideration to be made is that in side-alternating vibration, the magnitude (G-force) is strongly dependent on the position of the foot, in relation to the center of the sway plate, since these devices have a rotation axis positioned in the anteroposterior direction, causing the sway plate to move like a seesaw. Typically, the peak-to-peak displacement in these devices is equal to zero at the center of the sway plate and close to 5 mm at the edge of the plate. Studies that used this type of device monitored the positioning of the feet, aiming to generate the desired magnitude, which should be considered when prescribing WBV on platforms with side-alternating vibration.
In the analyses involving low frequency and high magnitude, we verified significant effects for the lumbar spine and trochanter, while for high frequency and low magnitude, only for the lumbar spine, coinciding with the previous meta-analysis [25]. Recently, animal model studies have demonstrated anabolic effects on bone tissue in interventions with low frequency [33,34,35]. In the present meta-analysis, comparing effect sizes for the lumbar spine, low frequency and high magnitude (0.014 g/cm2) provided a substantially larger effect size when compared to high frequency and low magnitude (0.004 g/cm2). However, it should be considered that for analysis involving low frequency and high magnitude, the evidence was of low quality. On the other hand, for high frequency and low magnitude, the evidence was of high quality, with high confidence that we are close to the true effect. The studies that integrated this specific analysis used frequency ≈ 30 Hz and magnitude ≈ 0.3 g [10, 15, 22].
For high frequency and high magnitude, our previous study [25] did not present any results, while in the present meta-analysis we found significant effects for three bone regions: lumbar spine (0.012 g/cm2), trochanter (0.040 g/cm2), and Ward’s area (0.140 g/cm2). This can be explained by the fact that the majority of the studies [2, 3, 5,6,7] that were part of the update of this review used these parameters. However, these meta-analyses had very low or low quality of evidence, which makes these effect estimates uncertain. In this sense, future RCTs should better analyze the use of higher magnitudes, paying attention to safety issues, which will be discussed later in this manuscript.
Regarding the high cumulative dose and high magnitude, we did not find any results in the present meta-analysis, or in our previous study [25]. For high cumulative dose and low magnitude, both meta-analyses found significant results for the lumbar spine, as well as for low cumulative dose and high magnitude, in which both found significant results for the lumbar spine and trochanter. It should be considered that analyses involving high cumulative dose and low magnitude presented high quality of evidence for increased lumbar spine aBMD, while analyses involving low cumulative dose and high magnitude presented evidence of very low or low quality. In this case, an analysis that considered a high cumulative dose and low magnitude included studies [10, 15, 22] in which the total exercise dose throughout the intervention was more than 7000 min. This corresponds to daily 20-min sessions for approximately 1 year. In these studies, the frequency was ≈ 30 Hz and magnitude ≈ 0.3 g.
Regarding the cumulative dose, a previous study [46] also found that a higher dose of WBV (> 1000 min) may be more efficient on BMD in postmenopausal women; however, significant effects were observed only at magnitudes greater than 3 g. Probably, the divergence with the current study that verified results on aBMD in WBV of low magnitude is due to the fact that the systematic review in question [46] contemplated only nine studies, including non-randomized clinical trials.
Another issue that may still influence the results observed is body positioning during WBV. In this case, no results were observed for the positioning of extended knees, or when muscle strengthening exercises were performed during vibration, not differing from the results found previously [25]. For semi-flexed knees, significant effects were found for trochanter. However, an effect in favor of the control group was observed for total hip. In the previous review, we found significant effects for the lumbar spine, femoral neck, and trochanter. All analyses that took into account body positioning during WBV presented evidence of very low to moderate quality, demonstrating that the subject needs to be further explored by future RCTs.
The position with extended knees, in theory, should be the one that provides greater transmissibility of mechanical stimuli to the body, as demonstrated in previous research [47]. The studies that make up the present review that used extended knee, mainly [1, 10, 11, 15, 22] used low magnitude. This is justified by the fact that it is safe for the participants to maintain a position of extended knees in low-magnitude vibrations, in which the stimuli do not arrive prominently in the skull/brain. Regarding the performance of muscle strengthening exercises during WBV, such as squats, it is possible that alternating positions between extended knees and 90° flexion may compromise the transmissibility of the mechanical stimulus. The semi-flexed knee position, in turn, offers negligible loss of acceleration in the femur and spine up to frequencies of 30 Hz, indicating excellent transmissibility [48]. The studies that were part of the present systematic review and used the semi-flexed knee position mostly adopted frequencies of up to 30 Hz [4, 5, 9, 20, 23], and the only exception was the study of Karakiriou et al. [14].
When WBV was compared with conventional exercises (e.g., combined training, involving resistance and cardiorespiratory exercises; vertical jumps; and Pilates), no significant results were observed in the present meta-analysis, coinciding with results from our previous study [25]. Since WBV does not differ from the effects of conventional exercise on BMD in postmenopausal women, it is possible to consider the relevance of this type of intervention for this population. Conventional muscular resistance exercises, for example, typically require an intervention time of approximately 60 min, which requires motivation and individualized professional support to be performed. In contrast, WBV requires significantly less time in each intervention session (average of 10 min considering the studies in the present review) and little participant motivation. In addition, when using low magnitudes, individualized professional support is not required. It should also be taken into account that of the 2089 volunteers included in this systematic review, only 2.8% reported having adverse events possibly associated with WBV training.
Regarding the effect sizes observed in the different analyses of the present study, even when significant differences were found, the magnitudes of absolute change in aBMD were low, being around 0.010 g/cm2 for the lumbar spine and 0.020 g/cm2 for the trochanter. Still, it should be considered that other forms of intervention involving physical exercise [49], calcium and vitamin D supplementation [50], or medications [51] also found relatively low effect magnitudes.
A limiting aspect to be considered in the current study is the non-observance of the residual effects of vibration in the medium and long term. The RCTs included in the meta-analysis only observed immediate post-intervention effects. Therefore, at this time, it is not possible to determine whether the effectiveness of WBV on BMD of the lumbar spine and trochanter in postmenopausal women remains, for example, after 1–5 years of cessation of interventions. A meta-analysis study with physical exercise found that when significant differences in lumbar spine aBMD were found immediately after the intervention, they were maintained for 1 year but not after 5 years [49].
The current study could not verify in subgroup analyses the effects of WBV on BMD considering differences in age group, or time of menopause, as well as for the classification of BMD (no change, osteopenia, and osteoporosis). Future RCTs may consider performing subgroup analyses, aiming to identify differences between time in menopause (e.g., participants at the beginning of menopause vs. participants with menopause duration > 15 years) and BMD classification status, so that it is possible to determine the influence of these conditions on the effectiveness of the WBV.
Another relevant point that must be considered is the influence of body weight on BMD measurements as a result of WBV. The data available in the RCTs included in our systematic review did not allow a subgroup analysis for this factor. The study by Rubin et al. [22] identified that intervention with WBV in postmenopausal women may be more effective in lighter women (< 65 kg), especially for the lumbar spine region. Future studies may better explore this condition, considering subgroup analyses that take into account, for example, overweight postmenopausal women vs. eutrophic.
Furthermore, in general, the studies included in the present systematic review were limited to only reporting the mean compliance of participants during WBV interventions. Rubin et al. [22] demonstrated a strong correlation between greater use of the vibration device and increased bone mineral density. Likewise, future studies should strive to perform subgroup analyses considering compliance.
It should also be considered that few studies in the present systematic review used pQCT to analyze vBMDt. Our primary analyses could include only three studies to identify the efficacy of WBV on radio vBMDt and four studies for tibia. In addition, other variables that can be complementary as an indicator of bone strength and fracture risk, such as the trabecular bone score (TBS), can be explored in future studies that verify the effectiveness of WBV on BMD in postmenopausal women.
As can be seen, there are several issues that permeate the use of WBV to increase BMD. Understanding what is already known about the possible mechanisms of action of mechanical vibration on bone tissue can additionally contribute to the definition of intervention protocols. Bone tissue response to mechanical loads occurs through a complex interaction between stress parameters. For example, at low magnitudes, the mechanism of action in bone is unlikely to occur through tissue deformation, but rather through a series of parallel and serial pathways, such as the action of osteocytes, producing/downregulating soluble factors, inhibiting osteoclast formation [52].
Even recognizing that some intervention protocols with WBV can increase muscle action (high-magnitude utilization), there is evidence that bone responses can occur independently of the muscle response [53]. In this sense, if the objective of the intervention is exclusively linked to the improvement of BMD, it is likely that high vibration magnitudes can be avoided, increasing the safety of subjects during WBV. Our meta-analysis showed that high frequency (≈ 30 Hz) combined with low magnitude (≈ 0.3 g) enabled effects on lumbar spine aBMD. This was one of the few analyses where the quality of the evidence was high. In this sense, there is high confidence in the correlation between the effect estimated in the present study and the true effect.
WBV safety
An important issue that must be considered in WBV interventions is safety. Although few adverse events were reported in the RCTs of the present systematic review, it should be emphasized that 91% of the studies were carried out with an intervention time ≤ 1 year. Longer-term studies are needed to better determine safety aspects in the population of postmenopausal women. In this sense, although we have shown in our analyses that high magnitude allowed greater effects on aBMD, some aspects must be considered. First, that the studies that made use of high magnitudes worked under supervised conditions, that is, the subjects did not have a vibrating device at home. Second, that in most studies that used high magnitude, the participants’ knees were kept in semi-flexion, or performing squat exercises during WBV, which substantially decreases the transmission of vibratory stimuli to the upper body region, including the skull [47, 54].
Even so, possible risks to the knee joint should be considered when using high-magnitude WBV, especially in participants who present pre-existing pathological conditions in this joint, such as osteoarthritis [23]. For example, in the semi-flexion position of the knee, a vibration device, generating a vertical magnitude of 15 g in the oscillatory plate, delivered 7.3 g in the tibia region and 0.8 g in the cranial region [54]. In other words, the G-force dissipates through the human body, losing transmissibility, mainly along the joints, the knee joint being one of the ones that receives the greatest overload when attenuating the vibratory stimuli in the semi-flexion position [47]. In addition, high magnitude should be a concern when administering WBV to postmenopausal women of advanced age or with a diagnosis of pre-existing osteoporosis, given the imminent risk of fracture for this population.
The type of vibration should also be a factor to consider in relation to the transmissibility of vibration through the body. It has been shown that in devices configured to generate similar magnitudes, capable of generating high magnitudes, the transmissibility of vibratory stimuli from the foot to the head may be different in platforms that vibrate predominantly synchronously or side-alternating. While the first generated significantly greater mechanical stimuli in the thoracic spine at the T-10 level, the second enabled greater stimuli in the lower limbs, with no differences for the hip and lumbar spine regions [55]. This information should be considered when prescribing WBV in postmenopausal women using side-alternating vibration platforms, especially among those with a history of pathologies in the ankle and knee joints.
Considering the synchronous and side-alternating vibration platforms, capable of generating large magnitudes, in the current study, the average G-force generated was 5.1 (range between 1.9 and 11.4) and 3.9 (range between 1.0 and 7.4), respectively. On the other hand, low magnitude synchronous platforms (< 1 g), in the current study, generated G-force close to 0.3. The latter are the ones that offer greater safety to the participant, substantially reducing potential risks linked to high-magnitude vibrations. It is also noteworthy that for this type of vibration, our subgroup analysis showed a significant improvement for aBMD of the lumbar spine, being a safe and viable option to be administered in people diagnosed with osteopenia or osteoporosis, and it is even possible to provide the device for home use without supervision. This option can also be configured as an advantage when, for some reason, the participant has difficulty getting around for intervention in another location.
Quality of evidence
The twenty-three reports included in the current systematic review were from North America [1, 9, 11, 15, 22], Europe [5,6,7,8, 13, 14, 16,17,18, 20, 21, 23], Asia [2, 10, 12, 23], South America [4], Oceania [19], and Africa [3], with representation from all continents, however, with low representation of some regions, which limits generalizations for some locations. The analyses performed by the GRADE system showed that most of the effect estimates provided evidence of very low or low quality, which significantly reduces the confidence in the results observed for these analyses. The overall methodological quality of the studies included in the systematic review was good (mean PEDro score 6.0 ± 1.5), with most RCTs (56.5%) presenting a PEDro score ≥ 6 [1, 3,4,5,6, 10, 11, 15,16,17,18,19, 22]. Despite this, many analyses were conducted with a high risk of bias (> 25% of RCTs with low methodological quality). Still, several analyses showed high heterogeneity, which reduces the consistency of intervention effects. Another important limitation is that most RCTs used small sample sizes (only seven studies had a sample of over 30 participants in each group), with most having approximately 15 participants per group, which generated many analyses with imprecision. In addition, many studies have associated muscle strengthening exercises during WBV, which may have compromised the real effects of vibration in the primary and subgroup analyses, due to the loss of transmissibility of mechanical stimuli, considering movements such as squats, in that the knee is flexed around 90°. Finally, the bone regions considered by the different studies varied significantly, causing several analyses to have a reduced number of studies.
Potential biases in the review process
In the present review, only RCTs were included, which reduces the risk of bias. The majority of studies performed ITT analysis [2, 4, 5, 10, 11, 13, 15,16,17,18,19,20, 22]. Therefore, in our analyses, we used per-protocol results only when ITT was not available. Additionally, we performed meta-analyses with studies that had available per-protocol data and overall, the results did not change. However, most studies do not present per-protocol data, which limits verifying the results considering the subjects who effectively completed the interventions. For some studies [2, 3, 5, 6, 9, 10, 13, 16, 20, 22, 23], since we did not have the standard deviation of the difference, statistical manipulation was necessary (e.g., converting confidence intervals or standard errors into standard deviations), which may have reduced the precision of the data entered in the meta-analysis. Only two analyses enabled visual inspection in a funnel chart, in which it was not possible to identify any evident asymmetry (Supplementary Figs. 17 and 18). Finally, it is important to clarify that two of the authors of the present study (RGO and LCO) are authors of an RCT included in the systematic review and meta-analysis [4], which may, even if unintentionally, influence the interpretation of the results.
Conclusions
Implications for practice
Health professionals may recommend WBV training for postmenopausal women to increase BMD, especially in the lumbar spine region, using high frequency (≈ 30 Hz), low magnitude (≈ 0.3 g), and high cumulative dose (≈ 7000 min), in view of the high-quality evidence observed for these analyses. Other parameters, which despite having offered significant effects, need to be better investigated, given the low-quality evidence, such as vibrating platform type (side-alternating or synchronous), high magnitude, and standing position on the vibrating platform with the knees semi-flexed (this positioning is necessary in high-magnitude vibrations). Still, professionals should carefully analyze the use of high magnitude, in relation to possible health risks, given that data on adverse events regarding the long-term use (> 1 year) of WBV in high magnitudes are incipient. In this sense, we highlight some important precautions if professionals come to use WBV in high magnitude: constant supervision to investigate adverse events, to interrupt interventions if necessary; exposure time, which should be as little as possible; and mainly, not to be used in people with imminent risk of fracture, such as those diagnosed with osteopenia or osteoporosis.
Implications for research
Future RCTs aiming to verify the effects of WBV on BMD in postmenopausal women may compare the parameters indicated in this meta-analysis with other parameters, in order to confirm the findings elucidated here or indicate new possibilities. Three main points that deserve attention in future studies: (a) the sample size, since most studies used small samples; (b) analysis of the long-term residual effects of WBV; and (c) verification of adverse events in long-term studies, especially at higher magnitudes (> 1 g). Considering that most of the analyses had a small number of RCTs (< 10 studies) and many analyses showed low-quality evidence, it is possible that future meta-analysis studies could change the magnitude of the effect of WBV on BMD in postmenopausal women.
References
Rajapakse CS, Johncola AJ, Batzdorf AS, Jones BC, Mukaddam MA, Sexton K (2021) Effect of low-intensity vibration on bone strength, microstructure, and adiposity in pre-osteoporotic postmenopausal women: a randomized placebo-controlled trial. J Bone Miner Res 36:673–684. https://doi.org/10.1002/jbmr.4229
Sen EI, Esmaeilzadeh S, Eskiyurt N (2020) Effects of whole-body vibration and high impact exercises on the bone metabolism and functional mobility in postmenopausal women. J Bone Miner Metab 38:392–404. https://doi.org/10.1007/s00774-019-01072-2
ElDeeb AM, Abdel-Aziem AA (2020) Effect of whole-body vibration exercise on power profile and bone mineral density in postmenopausal women with osteoporosis: a randomized controlled trial. J Manipulative Physiol Ther 43:384–393. https://doi.org/10.1016/j.jmpt.2019.12.003
Oliveira LC, Oliveira RG, Pires-Oliveira DAA (2019) Effects of whole-body vibration versus pilates exercise on bone mineral density in postmenopausal women: a randomized and controlled clinical trial. J Geriatr Phys Ther 42:E23–E31. https://doi.org/10.1519/JPT.0000000000000184
Jepsen DB, Ryg J, Hansen S, Jørgensen NR, Gram J, Masud T (2019) The combined effect of Parathyroid hormone (1–34) and whole-body Vibration exercise in the treatment of postmenopausal Osteoporosis (PaVOS study): a randomized controlled trial. Osteoporos Int 30:1827–1836. https://doi.org/10.1007/s00198-019-05029-z
Marín-Cascales E, Rubio-Arias JA, Alcaraz PE (2019) Effects of two different neuromuscular training protocols on regional bone mass in postmenopausal women: a randomized controlled trial. Front Physiol 10:846–854. https://doi.org/10.3389/fphys.2019.00846
Marín-Cascales E, Alcaraz PE, Rubio-Arias JA (2017) Effects of 24 weeks of whole body vibration versus multicomponent training on muscle strength and body composition in postmenopausal women: a randomized controlled trial. Rej Res 20:193–201. https://doi.org/10.1089/rej.2016.1877
Santin-Medeiros F, Santos-Lozano A, Rey-López JP, Garatachea N (2015) Effects of eight months of whole body vibration training on hip bone mass in older women. Nutr Hosp 31:1654–1659. https://doi.org/10.3305/nh.2015.31.4.8441
Liphardt AM, Schipilow J, Hanley DA, Boyd SK (2015) Bone quality in osteopenic postmenopausal women is not improved after 12 months of whole-body vibration training. Osteoporos Int 26:911–920. https://doi.org/10.1007/s00198-014-2995-8
Leung KS, Li CY, Tse YK, Choy TK, Leung PC, Hung VWY, Chan SY, Leung AHC, Cheung WH (2014) Effects of 18-month low-magnitude high-frequency vibration on fall rate and fracture risks in 710 community elderly - a cluster - randomized controlled trial. Osteoporos Int 25:1785–1795. https://doi.org/10.1007/s00198-014-2693-6
Slatkovska L, Beyene J, Alibhai SMH, Wong Q, Sohail QZ, Cheung AM (2014) Effect of whole-body vibration on calcaneal quantitative ultrasound measurements in postmenopausal women: a randomized controlled trial. Calcif Tissue Int 95:547–556. https://doi.org/10.1007/s00223-014-9920-1
Lai CL, Tseng SY, Chen CN, Liao WC, Wang CH, Lee MC, Hsu PS (2013) Effect of 6 months of whole body vibration on lumbar spine bone density in postmenopausal women: a randomized controlled trial. Clin Interv Aging 8:1603–1609. https://doi.org/10.2147/CIA.S53591
Stolzenberg N, Belavý DL, Beller G, Armbrecht G, Semler J, Felsenberg D (2013) Bone strength and density via pQCT in post-menopausal osteopenic women after 9 months resistive exercise with whole body vibration or proprioceptive exercise. J Musculoskelet Neuronal Interact 13:66–76
Karakiriou SK, Douda HT, Smilios IG, Volaklis KA, Tokmakidis SP (2012) Effects of vibration and exercise training on bone mineral density and muscle strength in post-menopausal women. Eur J Sport Sci 12:81–88. https://doi.org/10.1080/17461391.2010.536581
Slatkovska L, Alibhai SMH, Beyene J, Hu H, Demaras A, Cheung AM (2011) Effect of 12 months of whole-body vibration therapy on bone density and structure in postmenopausal women. A randomized trial Annals Intern Med 155:668–679. https://doi.org/10.7326/0003-4819-155-10-201111150-00005
Verschueren SMP, Bogaerts A, Delecluse C, Claessens AL, Haentjens P, Vanderschueren D, Boonen S (2011) The effects of whole-body vibration training and vitamin D supplementation on muscle strength, muscle mass, and bone density in institutionalized elderly women: a 6-month randomized, controlled trial. J Bone Miner Res 26:42–49. https://doi.org/10.1002/jbmr.181
Von Stengel S, Kemmler W, Bebenek M, Engelke K, Kalender WA (2011) Effects of whole-body vibration training on different devices on bone mineral density. ACSM 43:1071–1079. https://doi.org/10.1249/MSS.0b013e318202f3d3
Von Stengel S, Kemmler W, Engelke K, Kalender WA (2011) Effects of whole body vibration on bone mineral density and falls: results of the randomized controlled ELVIS study with postmenopausal women. Osteoporos Int 22:317–325. https://doi.org/10.1007/s00198-010-1215-4
Beck BR, Norling TL (2010) The effect of eight months of twice-weekly low or higher intensity whole body vibration on risk factors for postmenopausal hip fracture. Am J Phys Med Rehabil 89:997–1009. https://doi.org/10.1097/PHM.0b013e3181f71063
Gusi N, Raimundo A, Leal A (2006) Low-frequency vibratory exercise reduces the risk of bone fracture more than walking: a randomized controlled trial. BMC Musculoskelet Disord 7:92. https://doi.org/10.1186/1471-2474-7-9
Verschueren SMP, Roelants M, Delecluse C, Swinnen S, Vanderschueren D, Boonen S (2004) Effect of 6-month whole body vibration training on hip density, muscle strength, and postural control in postmenopausal women: a randomized controlled pilot study. J Bone Miner Res 19:352–359. https://doi.org/10.1359/JBMR.0301245
Rubin C, Recker R, Cullen D, Ryaby J, McCabe J, McLeod K (2004) Prevention of postmenopausal bone loss by a low-magnitude, high-frequency mechanical stimuli: a clinical trial assessing compliance, efficacy, and safety. J Bone Miner Res 19:343–351. https://doi.org/10.1359/JBMR.0301251
Russo CR, Lauretani F, Bandinelli S, Bartali B, Cavazzini C, Guralnik JM, Ferrucci L (2003) High-frequency vibration training increases muscle power in postmenopausal women. Arch Phys Med Rehabil 84:1854–1857. https://doi.org/10.1016/S0003-9993(03)00357-5
Slatkovska L, Alibhai SMH, Beyene J, Cheung AM (2010) Effect of whole-body vibration on BMD: a systematic review and meta-analysis. Osteoporos Int 21:1969–1980. https://doi.org/10.1007/s00198-010-1228-z
Oliveira LC, Oliveira RG, Pires-Oliveira DA (2016) Effects of whole body vibration on bone mineral density in postmenopausal women: a systematic review and meta-analysis. Osteoporos Int 27:2913–2933. https://doi.org/10.1007/s00198-016-3618-3
Teng GG, Curtis ER, Saag KG (2008) Mortality and osteoporotic fractures: is the link causal, and is it modifiable? Clin Exp Rheumatol 26:S125–S137
Rizzoli R (2018) Postmenopausal osteoporosis: assessment and management. Best Pract Res Clin Endocrinol Metab 32:739–757. https://doi.org/10.1016/j.beem.2018.09.005
Kemmler W, Shojaa M, Kohl M, Von Stengel S (2020) Effects of different types of exercise on bone mineral density in postmenopausal women: a systematic review and meta-analysis. Calcif Tissue Int 107:409–439. https://doi.org/10.1007/s00223-020-00744-w
Burton E, Hill AM, Pettigrew S, Lewin G, Bainbridge L, Farrier K, Airey P, Hill KD (2017) Why do seniors leave resistance training programs? Clin Interv Aging 12:585–592. https://doi.org/10.2147/CIA.S128324
Van Heuvelen M, Rittweger J, Judex S, Sañudo B, Seixas A, Fuermaier A, Tucha O, Nyakas C, Marín PJ, Taiar R, Stark C, Schoenau E, Sá-Caputo DC, Bernardo-Filho M, Van der Zee EA (2021) Reporting guidelines for whole-body vibration studies in humans, animals and cell cultures: a consensus statement from an international group of experts. Biology 10:965. https://doi.org/10.3390/biology10100965
Rauch F, Sievanen H, Boonen S, Cardinale M, Degens H, Felsenberg D, Roth J, Schoenau E, Verschueren S, Rittweger J (2010) Reporting whole-body vibration intervention studies: recommendations of the International Society of Musculoskeletal and Neuronal Interactions. J Musculoskelet Neuronal Interact 10:193–198
Ozcivici E, Luu YK, Adler B, Qin Y, Rubin J, Judex S, Rubin CT (2010) Mechanical signals as anabolic agents in bone. Nat Rev Rheumatol 6:50–59. https://doi.org/10.1038/nrrheum.2009.239
Chen J, Ruan H, Liu Y, Bao J, Xu H, Yao M et al (2018) Therapeutic effects of whole-body vibration on fracture healing in ovariectomized rats: a systematic review and meta-analysis. Menopause 26:677–686. https://doi.org/10.1097/GME.0000000000001285
Minematsu A, Nishii Y, Sakata S (2021) Effects of whole-body vibration on bone properties in aged rats. J Musculoskelet Neuronal Interact 21:287–297
Minematsu A, Nishii Y, Imagita H, Sakata S (2019) Whole body vibration at low-frequency can increase trabecular thickness and width in adult rats. J Musculoskelet Neuronal Interact 19:169–177
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD et al (2021) The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 372:n71. https://doi.org/10.1136/bmj.n71
Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA (editors) (2021) Cochrane handbook for systematic reviews of interventions version 6.2. Cochrane. Available from www.training.cochrane.org/handbook(updated February 2021)
Maher CG, Moseley AM, Sherrington C, Elkins MR, Herbert RD (2008) A description of the trials, reviews and practice guidelines indexed in the PEDro database. Phys Ther 88:1068–1077. https://doi.org/10.2522/ptj.20080002
Maher CG, Sherrington C, Herbert RD, Moseley AM, Elkins M (2003) Reliability of the PEDro scale for rating quality of randomized controlled trials. Phys Ther 83:713–721
The Centre of Evidence-Based Physiotherapy (2012) Sydney Available from: http://www.pedro.org.au
Jadad AR, Moore RA, Carroll D, Jenkinson C, Reynolds DJ, Gavaghan DJ, McQuay HJ (1996) Assessing the quality of reports of randomized clinical trials: is blinding necessary? Control Clin Trials 17:1–12. https://doi.org/10.1016/0197-2456(95)00134-4
Verhagen AP, de Vet HC, de Bie RA, Kessels AG, Boers M, Bouter LM, Knipschild PG (1998) The Delphi list: a criteria list for quality assessment of randomized clinical trials for conducting systematic reviews developed by Delphi consensus. J Clin Epidemiol 51:1235–1241. https://doi.org/10.1016/S0895-4356(98)00131-0
Schünemann H, Brożek J, Guyatt G, Oxman A (2013) GRADE handbook. Available from: https://gdt.gradepro.org/app/handbook/handbook.html
Iwamoto J, Takeda T, Sato Y, Uzawa M (2005) Effect of wholebody vibration exercise on lumbar bone mineral density, bone turnover, and chronic back pain in post-menopausal osteoporotic women treated with alendronate. Aging Clin Exp Res 17:157–163
Iwamoto J, Takeda T, Sato Y, Uzawa M (2019) Retraction note to: Effect of whole-body vibration exercise on lumbar bone mineral density, bone turnover, and chronic back pain in post-menopausal osteoporotic women treated with alendronate. Aging Clin Exp Res 31:1855. https://doi.org/10.1007/s40520-019-01373-6
Fratini A, Bonci T, Bull AM (2016) Whole body vibration treatments in postmenopausal women can improve bone mineral density: results of a stimulus focussed meta-analysis. PLoS ONE 11:e0166774. https://doi.org/10.1371/journal.pone.0166774
Harazin B, Grzesik J (1998) The transmission of vertical whole-body vibration to the body segments of standing subjects. J Sound Vib 215:775–787. https://doi.org/10.1006/jsvi.1998.1675
Judex S, Rubin CT (2010) Is bone formation induced by high frequency mechanical signals modulated by muscle activity? J Musculoskelet Neuronal Interact 10:3–11
Howe TE, Shea B, Dawson LJ, Downie F, Murray A, Ross C, Harbour RT, Caldwell LM, Creed G (2011) Exercise for preventing and treating osteoporosis in postmenopausal women. Cochrane Database Syst Rev 6:CD000333. https://doi.org/10.1002/14651858.CD000333.pub2
Tang BM, Eslick GD, Nowson C, Smith C, Bensoussan A (2007) Use of calcium or calcium in combination with vitamin D supplementation to prevent fractures and bone loss in people aged 50 years and older: a meta-analysis. Lancet 370:657–666. https://doi.org/10.1016/S0140-6736(07)61342-7
Cranney A, Guyatt G, Griffith L, Wells G, Tugwell P, Rosen C, Osteoporosis Methodology Group and The Osteoporosis Research Advisory Group (2002) Meta-analyses of therapies for postmenopausal osteoporosis. IX: summary of meta-analyses of therapies for postmenopausal osteoporosis. Endocr Rev 23:570–578. https://doi.org/10.1210/er.2001-9002
Beck BR (2015) Vibration therapy to prevent bone loss and falls: mechanisms and efficacy. Curr Osteoporos Rep 13:381–389. https://doi.org/10.1007/s11914-015-0294-8
Judex S, Rubin CT (2010) Is bone formation induced by high-frequency mechanical signals modulated by muscle activity? J Musculoskelet Neuronal Interact 10:3–11
Muir J, Kiel DP, Rubin CT (2013) Safety and severity of accelerations delivered from whole body vibration exercise devices to standing adults. J Sci Med Sport 16:526–531
Spain L, Yang L, Wilkinson JM, McCloskey E (2021) Transmission of whole body vibration - comparison of three vibration platforms in healthy subjects. Bone 144:115802
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
de Oliveira, R.D.J., de Oliveira, R.G., de Oliveira, L.C. et al. Effectiveness of whole-body vibration on bone mineral density in postmenopausal women: a systematic review and meta-analysis of randomized controlled trials. Osteoporos Int 34, 29–52 (2023). https://doi.org/10.1007/s00198-022-06556-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-022-06556-y