Abstract
Summary
Exercise is recommended for people with osteoporosis, but the effect for people who have suffered vertebral fracture is uncertain. This study shows that a multicomponent exercise-program based on recommendations for people with osteoporosis improved muscle strength, balance, and fear of falling in older women with osteoporosis and vertebral fracture.
Introduction
Guidelines for exercise strongly recommend that older adults with osteoporosis or osteoporotic vertebral fracture should engage in a multicomponent exercise programme that includes resistance training in combination with balance training. Prior research is scarce and shows inconsistent findings. This study examines whether current exercise guidelines for osteoporosis, when applied to individuals with vertebral fractures, can improve health outcomes.
Methods
This single blinded randomized controlled trial included 149 older women diagnosed with osteoporosis and vertebral fracture, 65+ years. The intervention group performed a 12-week multicomponent exercise programme, the control group received usual care. Primary outcome was habitual walking speed, secondary outcomes were physical fitness (Senior Fitness Test, Functional Reach and Four Square Step Test), health-related quality of life and fear of falling. Descriptive data was reported as mean (standard deviation) and count (percent). Data were analyzed following intention to treat principle and per protocol. Between-group differences were assessed using linear regression models (ANCOVA analysis).
Results
No statistically significant difference between the groups were found on the primary outcome, walking speed (mean difference 0.04 m/s, 95% CI − 0.01–0.09, p = 0.132). Statistically significant between-group differences in favour of intervention were found on FSST (dynamic balance) (mean difference − 0.80 s, 95% CI − 1.57 to − 0.02, p = 0.044), arm curl (mean difference 1.55, 95% CI 0.49–2.61, p = 0.005) and 30-s STS (mean difference 1.85, 95% CI 1.04–2.67, p < 0.001), as well as fear of falling (mean difference − 1.45, 95% CI − 2.64 to − 0.26, p = 0.018). No statistically significant differences between the groups were found on health-related quality of life.
Conclusion
Twelve weeks of a supervised multicomponent resistance and balance exercise programme improves muscle strength and balance and reduces fear of falling, in women with osteoporosis and a history of vertebral fractures.
Trial registration
ClincialTrials.gov Identifier: NCT02781974. Registered 25.05.16. Retrospectively registered.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Osteoporosis represents a major health burden worldwide with fractures as the clinically significant consequence of the disease [1]. In the European Union, osteoporosis affects 21% of women and 6% of men aged 50–84 [2]. Vertebral fractures are among the most common fragility fractures caused by osteoporosis, prevalence increases with age, and are more prevalent in women than men [1, 3, 4]. An osteoporotic vertebral fracture can cause severe pain and disability or present itself with mild or no symptoms [3], and its presence elevates the risk of subsequent fractures, up to 5-fold for new vertebral fractures [5]. The consequent disability increases with number of fractures [3]. Vertebral fracture is associated with hyperkyphosis, back pain and reduced physical function (e.g. balance, mobility) [1]. In addition, people with vertebral fractures are more likely to experience falls [6], fear of falling, anxiety, depression and loss of social roles [7, 8]. Indeed, they often avoid physical activity through fear [9] and have a reduced health-related quality of life (HRQoL) [10], both in the short- and longer-term perspective [11] compared to those who have not experienced fractures.
Exercise is a non-pharmacological intervention that is recommended for people with osteoporosis to prevent bone loss, falls and fractures [3]. A recent Cochrane systematic review [12] concludes that for people with vertebral fracture, there is a moderate-quality evidence that exercise probably improves physical performance. No other definitive conclusions can be made regarding other outcomes such as incident fragility fractures, falls, adverse events and patient-reported outcomes (pain, quality of life) [12]. However, some individual trials did report benefit on minor outcomes such as balance, back extensor muscle strength, trunk muscle endurance and fear of falling, but there was heterogeneity across trials in the effects of exercise and in the risk of bias [12]. The trials included in the Cochrane review reported inconsistent findings, and more research is needed for better prescription of exercise for older people with osteoporosis and vertebral fracture [12]. Although exercise can reduce fear of falling in older adults [13], effects of exercise interventions on fear of falling, pain and HRQoL are heterogeneous [12]. More recently, Olsen and Bergland [14] concluded that a 12-week group-based exercise programme and an educational session had a positive and durable effect on fear of falling in community-dwelling elderly women with osteoporosis and a history of vertebral fracture.
Guidelines for exercise from an expert panel strongly recommend that older adults with osteoporosis or osteoporotic vertebral fracture should engage in a multicomponent exercise programme that includes resistance training in combination with balance training [15]. In addition, people with vertebral fracture should seek assistance with a physical therapist to ensure safe and appropriate exercise. However, the guidelines were based on evidence in people with osteoporosis and expert opinion, as there were less data to draw on for people with vertebral fractures. Prior work by our team [16] and others [12] has combined exercise with education, and the exercise interventions may not align with recent guidelines that advise weight bearing impact exercise, balance training and progressive resistance training of adequate dose to improve muscle strength. Therefore, we propose to build on our prior work and examine whether current exercise guidelines for osteoporosis, when applied to individuals with vertebral fractures, can improve health outcomes.
The primary aim of this study was to assess the effect of a 12-week supervised multicomponent resistance and balance exercise programme on habitual walking speed among older women with osteoporosis and a history of vertebral fracture. Walking speed is indicative of an individual’s functional capacity and general health status and also a measure predictive of a range of outcomes including rehabilitation response, frailty and mobility disability [17]. The secondary aims were to assess the effects of the same programme on physical fitness, HRQoL and fear of falling.
Hypotheses
An intervention consisting of a 12-week supervised group exercise programme will improve habitual walking speed as well as physical fitness, HRQoL and fear of falling in older women with osteoporosis and a history of vertebral fracture.
Methods
Study design
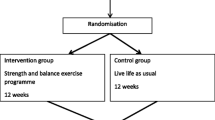
The study is a parallel-group, single-blinded randomized controlled trial with two arms. The outcome assessors were blinded for group allocation of the participants. Participants in the intervention group participated in a 12-week resistance and balance exercise programme, and participants in the control group were instructed to “live life as usual”. Reporting follows the CONSORT 2010 Statement [18].
Setting and participants
The intervention was carried out at OsloMet—Oslo Metropolitan University—and at a physiotherapy clinic in the area around Oslo, Norway. Participants were recruited from a specialty outpatient clinic for osteoporosis in Oslo, and from two outpatient clinics at hospitals in and around Oslo. We included women aged 65+ years, diagnosed with osteoporosis with a T-score − 2.5 standard deviation (SD) or less at the femoral neck or lumbar spine, verified by dual X-ray absorptiometry (DXA) and at least one vertebral fracture classified grade 1, 2 or 3 [5] verified by DXA-based vertebral fracture assessment or X-ray by medical doctors in a clinical setting. The women were community-dwelling and able to walk independently with or without a walking aid and able to speak and understand the Norwegian language. We excluded individuals with known medical contraindications for exercising [19], such as severe lung diseases or progressive neurological disorders. In cases of uncertainty, the women had to consult their physician to make sure that it was safe for them to exercise.
Intervention
The intervention was a resistance and balance exercise programme informed by exercise recommendations for people with osteoporosis and vertebral fracture [15], which recommend progressive resistance training for all major muscle groups in combination with balance training. In addition, recommendations for fall prevention exercises, guidelines for treatment in postmenopausal and age-related osteoporosis, as well as guidelines from a position stand on exercise and physical activity for older adults [13, 19, 20] were also taken into consideration when designing the exercise programme. The intervention was organized as a group-based stationary circuit exercise programme consisting of eight different exercises performed in a circuit; squats, step ups and sideway step-ups for lower limb strength and dynamic balance; upright row for upper back strength and posture; diagonal-lift for lower back strength; chest press and biceps curl for upper limb strength and exercises on balance pad for balance. The group was led by an experienced physiotherapist, with up to eight participants in each group [21]. Safety cautions were considered when designing the programme, like modifying or avoiding rapid, repetitive, weighted and sustained or end-range flexion or twisting of the spine [15, 22, 23], as well as safe transition between the stations. Participants attended the exercise-group twice a week for 12 weeks, with each session lasting 1 h. The physiotherapist leading the group-sessions was responsible for individual tailoring, suitable dosage and progression of the exercises. Different resistance bands, free weights, weight belts with adjustable load and steps in different heights were used in the exercises. The participants worked for 1.5 min at each station aiming to complete 8–12 repetitions of each exercise, followed by a break of 30 s for rest and transition, and they performed two rounds of the eight exercises. Participants were encouraged to work up to volitional fatigue during the 1.5 min with a target intensity corresponding to a level of 13–14 on a scale from 6 to 20 on the Borg Rating of Perceived Exertion Scale [24]. This corresponds to moderate intensity. Adjustment in the intervention were made for those experiencing muscle soreness and joint pain. Progression was made when the perceived level of intensity was lower than 13–14 on the Borg Rating of Perceived Exertion Scale [24] and the physiotherapist observed correct technique. The exercise programme is described in detail in the study protocol [25] and in additional file 1, adhering to the CERT-recommendations [26].
Participants allocated to the control group were instructed to live life as usual and maintain their usual activities and physical activity level.
Sociodemographic and descriptive variables
We used a questionnaire to collect socio-demographic and descriptive information regarding age, education, smoking status, medication, comorbidities, living alone or not, afraid of falling or not, have you fallen last year, injuries caused by falls, taking analgesics (yes/no) and pain level last week by score from 0 to 10 on a Numeric Rating Scale (NRS). Height and weight were measured to calculate body mass index (BMI).
Outcome measures
Outcome measures were carried out at baseline and after 3 months (end of intervention). Physiotherapists blinded to participants’ group allocation performed the outcome assessments. Prior to study start, the assessors went through an educational programme regarding testing procedures to ensure consistency in performing the tests and to ensure that the protocol was standardized.
Primary outcome measure
The primary outcome was 10-m habitual walking speed. The participants were instructed to walk 10 m on a straight path at the speed they normally choose when walking from one point to another. Participants who used walking aids were asked to use the same walking aid at the 3-month assessment.
Secondary measures
Secondary outcomes were physical fitness/function, HRQoL, Fall-Efficacy Scale International (FES-I) and International Physical Activity Questionnaire short form (IPAQ-SF) and adverse events.
Physical fitness
Dynamic and static balance was assessed by Four Square Step Test (FSST) [27] and Functional Reach (FR) [28]. Grip strength was measured with a hydraulic handheld dynamometer [21]. Lower extremity leg strength was measured by 30-s sit to stand (30-s STS), mobility by 2.45 m up and go, upper arm strength by number of arm curls in 30 s with a 5 pounds manual and functional endurance by 6-min walk test (6MWT), as suggested in the Senior Fitness test [29].
HRQoL
HRQoL was measured using the generic Short Form 36 Health Survey (SF-36) [30] and the disease-specific Quality of Life Questionnaire of the European Foundation for Osteoporosis (QUALEFFO-41) [31]. SF-36 is divided into eight subscales: physical functioning (PF), role limitations due to physical limitations (RP), bodily pain (BP), general health (GH), vitality (VT) social function (SF), role limitations-emotional (RE) and mental health (MH). The instrument has no overall summary score, but is divided into a physical component score (PCS) and a mental component score (MCS). The scores range from 0 to 100 (worst-best) [30]. QUALEFFO-41 contains 41 questions in five subscales: pain, physical function, score leisure and social activities, views about health in general and score mood. These subscales can be evaluated in a total score. The scores ranges from 0 to 100 (best-worst) [31].
Other outcome measures
Fear of falling was assessed using the Norwegian version of the FES-I, measuring fear of falling in 16 different daily activities [32]. Physical activity was measured by International Physical Activity Questionnaire short form (IPAQ-SF) [33]. The physiotherapist leading the intervention group recorded any adverse events such as falls, pain, fracture and joint pain or other events in a log during the training sessions. The participants in the intervention group were instructed to report any adverse events outside the exercise session to the instructor of the group. Adverse events in the control group were not recorded. Adherence to exercise was recorded in an attendance scheme at every session by the instructing physiotherapist.
Sample size
We calculated the sample size based on a substantial meaningful change in 10-m habitual walking speed. Perera [34] defined a substantial meaningful change as 0.10 m/s with an expected standard deviation of 0.2 m/s. This estimate required 128 patients, 64 in each group to obtain 80% statistical power with 5% significance level for an independent sample t test. We aimed to recruit 150 to allow for drop-outs.
Randomization
Participants were randomly assigned on a 1:1 ratio to the intervention group and control group after baseline assessment. We used a computer-generated permuted block randomization scheme to allocate the participants, block sizes varied from 4 to 8. A person not involved in testing or in contact with the participants kept the allocation scheme and administered the allocation. The allocation scheme was not accessible to others and was generated prior to recruiting and randomization by a person not involved in the allocation.
Research ethics
The study is approved by The Regional Research Committee for Medical Research Ethics in South East Norway (ref 2014/2050). Written, informed consent was obtained from all participants included in the study. The study is conducted according to the World Medical Association Declaration of Helsinki.
Statistical methods
The statistical analysis was performed using IBM SPSS version 25 (SPSS Inc., Chicago, Illinois, USA) and Stata version 15. The level of significance was 5%. Descriptive data is reported as mean (standard deviation) and count (percent). We analyzed the data following the intention-to-treat (ITT) principle. In addition, we analyzed per protocol with respect to 80% adherence to the intervention. Between-group differences in primary and secondary outcomes at 3-month follow-up were assessed using linear regression models (ANCOVA analysis). Respective outcome at baseline was the covariate and the randomized group was the factor. For the ITT analysis, missing values at 3-month follow-up were substituted by multiple imputation using a predictive mean matching model with arm, age and baseline values of the imputed variable as predictors [35]. Floor and ceiling effects were considered when more than 20% of the participants achieved the lowest or highest possible score.
Results
Flow of participants
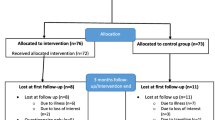
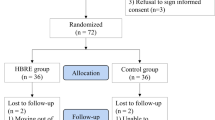
We recruited 149 women, in the period from January 2016 to April 2018. In total, 76 women were allocated to the intervention group, and 73 to the control group (additional file 2). At the 3-month follow-up, 63 participants were retained in the intervention group, and 59 in the control group (additional file 2). Three withdrew from the intervention because of increased back pain (sciatica and rheumatic pain). The overall adherence in the intervention group was 82.6% (24 sessions = 100%), and 57 of the women (75%) had an adherence of 80% or more (more than 19 sessions). Imputation for missing values was used for 18.1% of the participants.
Participant characteristics
The mean age of the women was 74.2 years, and 45.1% were living alone (Table 1). A total of 40.5% of the women reported comorbidity, defined as 4 or more self-reported diseases. The mean walking speed was 1.21 m/s (Table 1). The mean level of pain for the sample was 3.4 measured by NRS. Mean score of FES-I was 24.3.
Results of analysis
No statistically significant difference between the groups was found on the primary outcome: walking speed (0.13 m/s vs 0.08 m/s, mean difference 0.04, 95% CI − 0.01–0.09, p = 0.132) (Table 2). Further, sensitivity analysis showed no statistically significant association between mean difference on walking speed and number of attendance. On the secondary outcomes, we found statistically significant between-group differences in favor of intervention on FSST (dynamic balance) (mean difference − 0.80 s, 95% CI − 1.57 to − 0.02, p = 0.044), arm curl (arm strength) (mean difference 1.55, 95% CI 0.49–2.61, p = 0.005), and 30-s STS (leg strength) (mean difference 1.85, 95% CI 1.04–2.67, p < 0.001). In addition, we also found a statistically significant between-group difference in favor of the intervention group on FES-I (mean difference − 1.45, 95% CI − 2.64 to − 0.26, p = 0.018). No statistically significant differences between the groups were found in any of the subscales across the two HRQoL instruments SF-36 and QUALEFFO-41 (Table 2) or in 2.45 Up and Go, grip strength and 6 MWT (Table 2).
The per protocol analysis showed similar results with respect to between-group differences as the ITT analysis did (additional file 3).
Adverse events
No serious adverse events related to the intervention were reported. One participant had a fall at the facilities before the exercise session started, but with no consequences regarding pain or injuries. Two participants ended the intervention before 12 weeks, due to increasing sciatic back pain (3 weeks) and flare up of rheumatic pain (2 weeks).
Some participants in the intervention group experienced adverse events unrelated to the intervention: humerus fracture (n = 1), concussion after a fall (n = 1), pneumonia (n = 2), pelvic fracture (n = 1), falls (n = 7), flare up of rheumatic pain (n = 1).
Discussion
Our study demonstrates that a multicomponent resistance and balance exercise programme that is consistent with current guidelines improved physical fitness in terms of muscle strength and balance, as well as reducing fear of falling in older women with osteoporotic vertebral fractures. Results of this study add to previous research on this population, suggesting that exercise can be beneficial to improve outcomes as balance and fear of falling [16, 36, 37]. In addition, results show moderate effects on upper and lower limb muscle strength.
Contrary to our hypothesis, there was no statistically significant between-group differences on the primary outcome, habitual walking speed. Both the intervention and the control group improved in mean walking speed from baseline to 3-month follow-up. Improvements in controls have been shown in previous studies [35], as has contamination of the control group [38]. Participating in a research study might partly explain this result. Habitual walking speed is considered a sensitive measure of change in physical function, but there is a ceiling effect once the individual has reached normal walking speed [39]. Mean walking speed at baseline was 1.20 m/s and 1.22 m/s for the intervention and control group, respectively, which corresponds to a relatively high level of physical functioning [17].
The statistically significant improvements in the intervention group on functional lower limb strength, as measured by 30-s STS and arm curl for upper limb strength, are of relevance for older women with osteoporosis and vertebral fracture. Poor performance on measures of lower limb functioning is known predictor of future fall, disability, hospitalization and mortality [40]. None of the studies included in the most recent Cochrane review of exercise in this population [12] had lower limb strength as an outcome, even though most of the exercise interventions applied across the studies included muscle strength training. However, improvements in lower limb strength have been found in a recent study on people with osteoporosis and vertebral fracture [38]. Upper limb strength starts to decline by the age of 40 both in men and women [41]. The statistically significant improvement in the intervention group regarding upper limb strength (arm curl) is of importance and suggests that regular resistance training may slow down age-related decline in upper limb strength. Results from our study show that the intervention improved dynamic balance measured by FSST. Our findings are consistent with those of Bergland et al. [16] and Evstigneeva et al. [36], as both studies reported improved balance for women with osteoporosis and vertebral fracture after the intervention. Improved dynamic balance measured by FSST has previously been seen following exercise in men and women with T-scores between − 1.0 and − 2.5 (osteopenia) [42], but comparison must be done with care, as the study population differs from the present study. Our results showed that the intervention group had statistically significantly lower scores on FES-I after the intervention. This corresponds with findings from a group-based exercise programme and an educational session [14]. Another study of balance training with multi-task exercises in older adults with osteoporosis found improved FES-I [37]. Interventions to reduce fear of falling are of importance as fear often leads to avoidance of activity and decreased physical function [9, 43].
In contrast to comparable trials on women with osteoporosis and vertebral fracture [16, 36] our study found no improvement in HRQoL scores across the two instruments. However, the participants in our study have scores indicating better HRQoL at baseline across all dimensions in the instrument compared to the participants in the studies by Bergland et al. [16] and Evstigneeva et al. [36]. This suggests that our participants experience relatively good HRQoL, and the scores may be harder to change. Indeed, two dimensions of SF-36 had ceiling effects, Social Functioning and Role Physical.
Strengths and limitations
The study has some strengths and limitations, which we enumerate here. One strength is the sufficient statistical power of this study. We included a relatively high number of participants compared to other studies on women with vertebral fractures and were sufficiently powered to show change and difference between the randomized groups in our primary outcome [16, 36]. The intervention emphasized weight-bearing exercises for muscle strength and dynamic balance, as well as strength exercises for the upper body and back exercises as recommended [15]. Supervision by experienced physiotherapists and the individual tailoring of intensity and progression may have contributed to the overall good adherence to the intervention. There were no adverse events related to the intervention during the study. Safety was a major concern both in designing the intervention programme and throughout the intervention, focusing on minimizing risk of falls and applying safe exercises for people with vertebral fracture. For limitations, the Hawthorne effect should be considered [44]. Exercise intervention studies appeal to healthier and better-motivated individuals [45]. Frail women in particular (in addition to having vertebral fractures) may not be able to attend community-based classes, even though they might benefit even more than healthy women from an exercise intervention. In addition, the follow-up period in this study is short. It might be questioned whether 3 months of intervention is enough time to observe the most benefit. It is unclear whether participants improved as much as possible over this time period, or whether the intervention ended before they had reached their full potential. Finally, we had no consistent registration of falls, adverse events in the control group or information about number of vertebral fractures and location of them were not available, which all could be valuable in the interpretation of the results.
Conclusion
In conclusion, a supervised multicomponent resistance and balance exercise programme for 12 weeks improved muscle strength and balance and reduced fear of falling in women with osteoporosis and a history of vertebral fractures. We observed no effect of exercise on walking speed or HRQoL in these community dwelling older women.
Change history
27 April 2020
The original version of this article, published on 10 January 2020, contained a mistake. An author’s name was misspelled.
References
Cauley JA (2013) Public health impact of osteoporosis. J Gerontol A Biol Sci Med Sci 68(10):1243–1251. https://doi.org/10.1093/gerona/glt093
Hernlund E, Svedbom A, Ivergard M, Compston J, Cooper C, Stenmark J, McCloskey EV, Jonsson B, Kanis JA (2013) Osteoporosis in the European Union: medical management, epidemiology and economic burden. A report prepared in collaboration with the international Osteoporosis Foundation (IOF) and the European Federation of Pharmaceutical Industry Associations (EFPIA). Arch Osteoporos 8:136. https://doi.org/10.1007/s11657-013-0136-1
Kanis JA, Cooper C, Rizzoli R, Reginster JY (2019) European guidance for the diagnosis and management of osteoporosis in postmenopausal women. Osteoporos Int 30(1):3–44. https://doi.org/10.1007/s00198-018-4704-5
Pietschmann P, Rauner M, Sipos W, Kerschan-Schindl K (2009) Osteoporosis: an age-related and gender-specific disease--a mini-review. Gerontology 55(1):3–12. https://doi.org/10.1159/000166209
Kendler DL, Bauer DC, Davison KS, Dian L, Hanley DA, Harris ST, McClung MR, Miller PD, Schousboe JT, Yuen CK, Lewiecki EM (2016) Vertebral fractures: clinical importance and management. Am J Med 129(2):221.e221–221.e210. https://doi.org/10.1016/j.amjmed.2015.09.020
McDaniels-Davidson C, Davis A, Wing D, Macera C, Lindsay SP, Schousboe JT, Nichols JF, Kado DM (2018) Kyphosis and incident falls among community-dwelling older adults. Osteoporos Int 29(1):163–169. https://doi.org/10.1007/s00198-017-4253-3
Adachi JD, Adami S, Gehlbach S, Anderson FA Jr, Boonen S, Chapurlat RD, Compston JE, Cooper C, Delmas P, Diez-Perez A, Greenspan SL, Hooven FH, LaCroix AZ, Lindsay R, Netelenbos JC, Wu O, Pfeilschifter J, Roux C, Saag KG, Sambrook PN, Silverman S, Siris ES, Nika G, Watts NB (2010) Impact of prevalent fractures on quality of life: baseline results from the global longitudinal study of osteoporosis in women. Mayo Clin Proc 85(9):806–813. https://doi.org/10.4065/mcp.2010.0082
Gold DT (2001) The nonskeletal consequences of osteoporotic fractures. Psychologic and social outcomes. Rheum Dis Clin N Am 27(1):255–262
Reventlow SD (2007) Perceived risk of osteoporosis: restricted physical activities? Qualitative interview study with women in their sixties. Scand J Prim Health Care 25(3):160–165. https://doi.org/10.1080/02813430701305668
Lips P, van Schoor NM (2005) Quality of life in patients with osteoporosis. Osteoporos Int 16(5):447–455. https://doi.org/10.1007/s00198-004-1762-7
Hallberg I, Bachrach-Lindstrom M, Hammerby S, Toss G, Ek AC (2009) Health-related quality of life after vertebral or hip fracture: a seven-year follow-up study. BMC Musculoskelet Disord 10:135. https://doi.org/10.1186/1471-2474-10-135
Gibbs JC, MacIntyre NJ, Ponzano M, Templeton JA, Thabane L, Papaioannou A, Giangregorio LM (2019) Exercise for improving outcomes after osteoporotic vertebral fracture. Cochrane Database Syst Rev 7:Cd008618. https://doi.org/10.1002/14651858.CD008618.pub3
Vieira ER, Palmer RC, Chaves PH (2016) Prevention of falls in older people living in the community. BMJ (Clin Res ed) 353:i1419. https://doi.org/10.1136/bmj.i1419
Olsen CF, Bergland A (2014) The effect of exercise and education on fear of falling in elderly women with osteoporosis and a history of vertebral fracture: results of a randomized controlled trial. Osteoporos Int 25(8):2017–2025. https://doi.org/10.1007/s00198-014-2724-3
Giangregorio LM, Papaioannou A, Macintyre NJ, Ashe MC, Heinonen A, Shipp K, Wark J, McGill S, Keller H, Jain R, Laprade J, Cheung AM (2014) Too fit to fracture: exercise recommendations for individuals with osteoporosis or osteoporotic vertebral fracture. Osteoporos Int 25(3):821–835. https://doi.org/10.1007/s00198-013-2523-2
Bergland A, Thorsen H, Karesen R (2011) Effect of exercise on mobility, balance, and health-related quality of life in osteoporotic women with a history of vertebral fracture: a randomized, controlled trial. Osteoporos Int 22(6):1863–1871. https://doi.org/10.1007/s00198-010-1435-7
Middleton A, Fritz SL, Lusardi M (2015) Walking speed: the functional vital sign. J Aging Phys Act 23(2):314–322. https://doi.org/10.1123/japa.2013-0236
Schulz KF, Altman DG, Moher D (2010) CONSORT 2010 statement: updated guidelines for reporting parallel group randomised trials. BMC Med 8:18. https://doi.org/10.1186/1741-7015-8-18
Chodzko-Zajko WJ, Proctor DN, Fiatarone Singh MA, Minson CT, Nigg CR, Salem GJ, Skinner JS (2009) American College of Sports Medicine position stand. Exercise and physical activity for older adults. Med Sci Sports Exerc 41(7):1510–1530. https://doi.org/10.1249/MSS.0b013e3181a0c95c
Bonaiuti D, Arioli G, Diana G, Franchignoni F, Giustini A, Monticone M, Negrini S, Maini M (2005) SIMFER rehabilitation treatment guidelines in postmenopausal and senile osteoporosis. Eura Medicophys 41(4):315–337
Roberts HC, Denison HJ, Martin HJ, Patel HP, Syddall H, Cooper C, Sayer AA (2011) A review of the measurement of grip strength in clinical and epidemiological studies: towards a standardised approach. Age Ageing 40(4):423–429. https://doi.org/10.1093/ageing/afr051
Sinaki M (2012) Exercise for patients with osteoporosis: management of vertebral compression fractures and trunk strengthening for fall prevention. PM R 4(11):882–888. https://doi.org/10.1016/j.pmrj.2012.10.008
Kunutsor SK, Leyland S, Skelton DA, James L, Cox M, Gibbons N, Whitney J, Clark EM (2018) Adverse events and safety issues associated with physical activity and exercise for adults with osteoporosis and osteopenia: a systematic review of observational studies and an updated review of interventional studies. https://doi.org/10.22540/JFSF-03-155
Borg GA (1982) Psychophysical bases of perceived exertion. Med Sci Sports Exerc 14(5):377–381
Stanghelle B, Bentzen H, Giangregorio L, Pripp AH, Bergland A (2018) Effect of a resistance and balance exercise programme for women with osteoporosis and vertebral fracture: study protocol for a randomized controlled trial. BMC Musculoskelet Disord 19(1):100. https://doi.org/10.1186/s12891-018-2021-y
Slade SC, Dionne CE, Underwood M, Buchbinder R (2016) Consensus on exercise reporting template (CERT): explanation and elaboration statement. Br J Sports Med 50(23):1428–1437. https://doi.org/10.1136/bjsports-2016-096651
Dite W, Temple VA (2002) A clinical test of stepping and change of direction to identify multiple falling older adults. Arch Phys Med Rehabil 83(11):1566–1571
Duncan PW, Weiner DK, Chandler J, Studenski S (1990) Functional reach: a new clinical measure of balance. J Gerontol 45(6):M192–M197
Rikli RE, Jones CJ, Hanson T (2004) Senior fitness test : fysisk formåen hos ældre : manual og referenceværdier. Senior fitness test manual, human kinetics. FADL’s Forlag, København
Ware JE Jr (2000) SF-36 health survey update. Spine 25(24):3130–3139
Lips P, Cooper C, Agnusdei D, Caulin F, Egger P, Johnell O, Kanis JA, Kellingray S, Leplege A, Liberman UA, McCloskey E, Minne H, Reeve J, Reginster JY, Scholz M, Todd C, de Vernejoul MC, Wiklund I (1999) Quality of life in patients with vertebral fractures: validation of the Quality of Life Questionnaire of the European Foundation for Osteoporosis (QUALEFFO). Working Party for Quality of Life of the European Foundation for Osteoporosis. Osteoporos Int 10(2):150–160
Helbostad JL, Taraldsen K, Granbo R, Yardley L, Todd CJ, Sletvold O (2010) Validation of the falls efficacy scale-international in fall-prone older persons. Age Ageing 39(2):259. https://doi.org/10.1093/ageing/afp224
Craig CL, Marshall AL, Sjostrom M, Bauman AE, Booth ML, Ainsworth BE, Pratt M, Ekelund U, Yngve A, Sallis JF, Oja P (2003) International physical activity questionnaire: 12-country reliability and validity. Med Sci Sports Exerc 35(8):1381–1395. https://doi.org/10.1249/01.mss.0000078924.61453.fb
Perera S, Mody SH, Woodman RC, Studenski SA (2006) Meaningful change and responsiveness in common physical performance measures in older adults. J Am Geriatr Soc 54(5):743–749. https://doi.org/10.1111/j.1532-5415.2006.00701.x
Bjerk M, Brovold T, Skelton DA, Liu-Ambrose T, Bergland A (2019) Effects of a falls prevention exercise programme on health-related quality of life in older home care recipients: a randomised controlled trial. Age Ageing. https://doi.org/10.1093/ageing/afy192
Evstigneeva L, Lesnyak O, Bultink IE, Lems WF, Kozhemyakina E, Negodaeva E, Guselnikova G, Belkin A (2016) Effect of twelve-month physical exercise program on patients with osteoporotic vertebral fractures: a randomized, controlled trial. Osteoporos Int 27(8):2515–2524. https://doi.org/10.1007/s00198-016-3560-4
Halvarsson A, Franzen E, Stahle A (2015) Balance training with multi-task exercises improves fall-related self-efficacy, gait, balance performance and physical function in older adults with osteoporosis: a randomized controlled trial. Clin Rehabil 29(4):365–375. https://doi.org/10.1177/0269215514544983
Gibbs JC (2018) Effect of home exercise on functional performance, posture, quality of life and pain in older women with vertebral fractures: a pilot feasibility trial. Paper presented at the Journal of bone and mineral research: JBMR
Wade DT (1992) Measurement in neurological rehabilitation. Oxford University Press, New York
Moreland JD, Richardson JA, Goldsmith CH, Clase CM (2004) Muscle weakness and falls in older adults: a systematic review and meta-analysis. J Am Geriatr Soc 52(7):1121–1129. https://doi.org/10.1111/j.1532-5415.2004.52310.x
Metter EJ, Conwit R, Tobin J, Fozard JL (1997) Age-associated loss of power and strength in the upper extremities in women and men. J Gerontol A Biol Sci Med Sci 52(5):B267–B276
Gianoudis J, Bailey CA, Ebeling PR, Nowson CA, Sanders KM, Hill K, Daly RM (2014) Effects of a targeted multimodal exercise program incorporating high-speed power training on falls and fracture risk factors in older adults: a community-based randomized controlled trial. J Bone Miner Res 29(1):182–191. https://doi.org/10.1002/jbmr.2014
Delbaere K, Crombez G, Vanderstraeten G, Willems T, Cambier D (2004) Fear-related avoidance of activities, falls and physical frailty. A prospective community-based cohort study. Age Ageing 33(4):368–373. https://doi.org/10.1093/ageing/afh106
De Amici D, Klersy C, Ramajoli F, Brustia L (2000) More about the Hawthorne effect. Anesth Analg 91(4):1043
Pacala JT, Judge JO, Boult C (1996) Factors affecting sample selection in a randomized trial of balance enhancement: the FICSIT study. J Am Geriatr Soc 44(4):377–382
Acknowledgements
We are grateful to the participants for their contribution to the study and to the physiotherapists involved in the intervention and testing of the participants.
Funding
The study received internal funding from OsloMet-Oslo Metropolitan University. The funding body has no role in the design of the study, writing the manuscript, collecting data, analysis and interpretation of data. The study has no external funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None.
Ethical approval
All procedures performed in the study involving human participants were in accordance with the ethical standards of national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The study has been approved by The Regional Committee for Medical Research Ethics in South East Norway (Ref. 2014/2050).
Informed consent
Written, informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Stanghelle, B., Bentzen, H., Giangregorio, L. et al. Effects of a resistance and balance exercise programme on physical fitness, health-related quality of life and fear of falling in older women with osteoporosis and vertebral fracture: a randomized controlled trial. Osteoporos Int 31, 1069–1078 (2020). https://doi.org/10.1007/s00198-019-05256-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-019-05256-4