Abstract
Summary
In this 4-year follow-up study including 1083 subjects (≥ 60 years), the prevalence of frailty was estimated to be 5.6%; osteoporosis was found to be significantly associated with frailty. Moreover, the presence of both osteoporosis and sarcopenia increased the risk of frailty compared to the presence of osteoporosis or sarcopenia alone.
Introduction
This study aims to examine the contribution of sarcopenia and osteoporosis to the occurrence of frailty using 4-year follow-up information of a population-based cohort study.
Methods
The second survey of the Research on Osteoarthritis/Osteoporosis Against Disability (ROAD) study was conducted between 2008 and 2010; 1083 subjects (aged ≥ 60 years, 372 men, 711 women) completed all examinations on frailty, sarcopenia, and osteoporosis, which were defined using Fried’s definition, Asian Working Group for Sarcopenia criteria, and WHO criteria, respectively. The third survey was conducted between 2012 and 2013; 749 of 1083 individuals enrolled from the second survey (69.2%, 248 men, 501 women) completed assessments identical to those in the second survey.
Results
The prevalence of frailty in the second survey was 5.6% (men, 3.8%; women, 6.6%). The cumulative incidence of frailty was 1.2%/year (men, 0.8%/year; women, 1.3%/year). After adjustment for confounding factors, logistic regression analysis indicated that osteoporosis was significantly associated with the occurrence of frailty (odds ratio, 3.07; 95% confidence interval, 1.26–7.36; p = 0.012). Moreover, the occurrence of frailty significantly increased according to the presence of osteoporosis and sarcopenia (odds ratio vs. neither osteoporosis nor sarcopenia: osteoporosis alone, 2.50; osteoporosis and sarcopenia, 5.80).
Conclusions
Preventing osteoporosis and coexistence of osteoporosis and sarcopenia may help reduce the risk of frailty.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
According to the most recent National Livelihood Survey by the Ministry of Health, Labour and Welfare in Japan, frailty is ranked third among conditions that cause disability and subsequently necessitate the requirement of support with regard to activities of daily living (ADL) [1]. Given the increasing proportion of older individuals, it has become urgent to prevent frailty, which impairs ADL and quality of life (QOL).
Moreover, with regard to the diagnostic criteria for frailty, Fried et al. first defined frailty as a clinical syndrome comprising five variables: unintentional weight loss, self-reported exhaustion, low physical activity, weakness (grip strength), and slowness (slow walking speed) [2]. Older persons with three or more of the five factors are considered frail, those with one or two factors as pre-frail, and those with no factors as non-frail or robust.
Conversely, sarcopenia (SP) is characterized by the generalized loss of skeletal muscle mass and muscle strength and/or function, causing multiple adverse health outcomes, including physical disability, poor QOL, and death [3,4,5,6,7,8]. The European Working Group on Sarcopenia in Older People (EWGSOP) developed a practical clinical definition and diagnostic criteria for SP in 2010 [8]. After the publication of the EWGSOP consensus criteria, the Asian Working Group for Sarcopenia (AWGS) announced appropriate diagnostic cutoff values for Asian populations [9]. Two of the three SP criteria, namely muscle strength and function, overlap with two of the five criteria of frailty, which may suggest that SP and frailty are strongly associated.
Furthermore, the abovementioned National Livelihood Survey performed by the Ministry of Health, Labour and Welfare in Japan found that falls and osteoporotic fractures are ranked fourth among the causes of disabilities requiring support and long-term care [1]. Therefore, the prevention of osteoporosis (OP) is also important with respect to disability in the older population. With regard to the association between SP and OP, we found that a large proportion of SP subjects also had OP using the follow-up information of the population-based cohort [10]. In addition, we estimated the incidence of SP and found that the presence of OP significantly increased the risk of SP [10]. However, no report has clarified the prevalence of frailty, coexisting proportions of frailty, SP, and OP and whether SP and/or OP significantly contributes to the subsequent occurrence of frailty in the general population.
Using the information collected in the 4-year follow-up of the population-based cohort study, we aimed to clarify the prevalence of frailty and the proportion of coexisting frailty in SP and OP. Then, we estimated the cumulative incidence of frailty. Finally, we assessed whether SP and/or OP significantly contributed to the occurrence of frailty.
Methods
Study participants
The present study was performed using the ROAD study cohorts that were established in 2005. The ROAD study is a national, prospective study of osteoarthritis that consists of population-based cohorts from several communities in Japan. Details of the cohort profiles have been reported elsewhere [11, 12]. Briefly, between 2005 and 2007, a baseline database was created that included the clinical and genetic information of 3040 residents {1061 men and 1979 women with a mean age of 70.3 (standard deviation [SD], 11.0) years; 71.0 (10.7) years, men; 69.9 (11.2) years, women}. The subjects were recruited from resident registration listings in three communities with different characteristics: 1350 subjects from an urban region in Itabashi, Tokyo; 864 subjects from a mountainous region in Hidakagawa, Wakayama; and 826 subjects from a coastal region in Taiji, Wakayama.
After the baseline study, a second survey was performed in the same communities from 2008 to 2010 [13], and the third survey was followed from 2012 to 2013. In the second and third surveys, all examinations for OP, SP, and frailty were performed only in the cohorts from the mountainous and coastal regions. Therefore, in the present study, among the 1551 participants (521 men; 1030 women) from such cohorts in the second survey, those aged ≥ 60 years were selected based on the AWGS criteria for SP [9]. Consequently, 1083 {372 men, 711 women; mean age, 72.1 (7.4) years (72.7 [7.5] years, men; 71.7 [7.3] years, women)} participants were recruited as eligible subjects for the follow-up. A flowchart of the subjects’ recruitment and follow-up, with reasons for dropout, is shown in Fig. 1.
All participants provided written informed consent, and the study was approved by the ethics committees of the University of Tokyo (No. 1264 and No. 1326) and Wakayama Medical University (No. 373).
Examinations of the second survey of the ROAD study
Interviewer-administered questionnaire
Participants completed an interviewer-administered questionnaire that comprised 200 questions related to lifestyle, including occupation, smoking habits, alcohol consumption, family history, medical history, physical activity, reproductive history, and health-related QOL.
Anthropometric measurements and medical history
Anthropometric measurements, including height and weight, were measured for all participants. The body mass index (BMI; weight [kg]/height [m2]) was calculated. Experienced orthopedic surgeons collected the medical information of all subjects.
Muscle strength and walking speed
To assess muscle strength, handgrip strength was measured using a handgrip dynamometer (Toei Light Co., Ltd., Saitama, Japan). Both hands were tested, and the larger value was used to determine the maximum muscle strength. As an index of muscle function, the usual walking speed was measured. The time (s) to walk 6 m at a normal walking speed in a hallway was taken manually using a stopwatch, and the usual gait speed was calculated.
Assessment of frailty
We defined frailty according to Fried’s definition [2]. Unintentional weight loss and self-reported exhaustion were defined using the Kihon Checklist (KCL), which is a questionnaire comprising 25 yes/no questions, and created and validated by the Ministry of Health, Labour and Welfare in Japan to identify older adults at a higher risk of requiring long-term healthcare [14]. Among the KCL questions, the presence of unintentional weight loss was defined as a positive answer to question 11: “Have you lost 2 kg or more in the past 6 months?” The presence of self-reported exhaustion was defined as a positive answer to question 25: “In the last 2 weeks have you felt tired without a reason?” The presence of low physical activity was defined as a negative answer to question 1: “Do you go out at least once a week?”
To measure weakness, low handgrip strength was established according to a sex-specific cutoff of the maximum muscle strength of the subject according to the AWGS criteria (< 26 kg, men; < 18 kg, women) [9]. To measure slowness, each participant’s 6-m normal walking speed (m/s) was calculated, and a slow walk was defined as ≤ 0.8 m/s according to the AWGS criteria [9]. Those with three or more and two of the five factors were diagnosed with frailty and pre-frailty, respectively.
Skeletal muscle mass
Skeletal muscle mass was measured with bioimpedance analysis [15,16,17,18,19] using the Body Composition Analyzer MC-190 (Tanita Corp., Tokyo, Japan). The protocol was described by Tanimoto et al. [20, 21] and has been validated by Nemoto et al. [22]. Appendicular skeletal muscle mass (ASM) was derived as the sum of the muscle mass of the arms and legs. Absolute ASM was converted to an appendicular muscle mass index (SMI) by dividing ASM by height in meters squared (kg/m2).
Bone mineral density examination
Lumbar spine and proximal femur bone mineral density (BMD) values were determined using dual-energy X-ray absorptiometry (DXA; Hologic Discovery C; Hologic, Waltham, MA, USA). To control DXA precision, the equipment was checked every examination during the second and third surveys using the same phantom. The BMD of the phantom was regulated within ± 1.5% in all examinations. In addition, the same physician (N.Y.) examined all participants to control interobserver variability. Intraobserver DXA variability using the Lunar DPX in vitro and in vivo had been measured by the same physician (N.Y.) in another study [23], and the coefficient of variance for L2–4 in vitro was 0.35%. The coefficients of variance for L2–4, proximal femur, Ward’s triangle, and trochanter examined in vivo in five male volunteers were 0.61–0.90, 1.02–2.57, 1.97–5.45, and 1.77–4.17%, respectively.
Definition of SP, OP, and osteosarcopenia (OSP)
For the definition of SP, the following recommended cutoff values of skeletal muscle mass were used according to the AGWS report [9]:
-
1.
Age 60 or 65 years as the age for SP diagnosis according to the definitions of elderly in each country
-
2.
Low appendicular skeletal muscle mass, 7.0 kg/m2 for men and 5.7 kg/m2 for women using bioimpedance analysis
-
3.
Low handgrip strength, < 26 kg for men and < 18 kg for women
-
4.
Low gait speed: usual gait speed ≤ 0.8 m/s
-
5.
In the present study, we considered subjects aged ≥ 60 years old as having SP if they had a low skeletal muscle mass and either criterion (c or d), such as low handgrip strength or low gait speed.
OP was defined according to the World Health Organization criteria; OP was diagnosed when BMD T-scores were lower than the peak bone mass by 2.5 SD [24, 25]. The mean L2–4 BMD in young adult men and women, as measured by the Hologic DXA in Japan, is 1.011 (0.119) g/cm2 [26]. Therefore, OP of the lumbar spine was defined as an L2–4 BMD of < 0.714 g/cm2. The mean BMD of the femoral neck in young adult men and women is 0.863 (0.127) and 0.787 (0.109) g/cm2, respectively [26]. Moreover, OP at the femoral neck in men and women was defined as a femoral neck BMD of < 0.546 and < 0.515 g/cm2, respectively.
In the present study, OSP was defined as the presence of both SP and OP, using the abovementioned definition.
Four-year follow-up (ROAD third survey) and definition of cumulative incidence
Between 2012 and 2013, the 1551 individuals who completed the measurements in the second survey were invited to participate in the 4-year follow-up of the ROAD study (third survey). In this third survey, the same examinations in the second survey were repeated.
The cumulative incidence of frailty during these 4 years was determined based on changes in measurements of completers of both the second and third surveys. A new case of frailty was defined as an individual without frailty in the second survey but with frailty in the third survey; such cases were categorized using the frailty criteria.
Statistical analysis
All statistical analyses were performed using Stata statistical software (StataCorp., College Station, Texas, USA). Differences in proportions were compared using the chi-square test. Continuous variables were compared using the analysis of variance for multiple groups or Scheffé’s least-significant-difference test for pairs of groups.
Logistic regression analysis was used to evaluate the association between SP, OP at L2–4 and/or OP at the femoral neck, and OSP, and frailty. In the analysis, we used the occurrence of frailty as the objective variable and the presence of SP, OP at L2–4 and/or OP at the femoral neck, or OSP as the explanatory variable, after adjusting for age, gender, and unconfirmed confounding factors. The unconfirmed risk factors used for adjustment in the multivariate logistic analysis are described in the “Results” section.
Results
Prevalence of frailty and pre-frailty and their characteristics
As noted in the “Methods” section, 1083 subjects were included in the present study (Table 1), and 8.4% of the whole population was considered to be emaciated. The smoking rate was 18.9% in men and 3.0% in women, and the alcohol-drinking rate was 59.1% in men and 16.1% in women.
The prevalence of frailty and pre-frailty for the population aged ≥ 60 years was 5.6% (men, 3.8%; women, 6.6%) and 11.5% (men, 9.4%; women, 12.5%), respectively. The prevalence of frailty in the 60–64-, 65–69-, 70–74-, 75–79-, and ≥ 80-year age groups was 1.0, 0.0, 3.2, 8.0, and 17.6%, respectively, and that of pre-frailty was 5.9, 3.6, 8.7, 16.9, and 23.9%, respectively. Figure 2 shows the age- and gender-stratified prevalence of frailty and pre-frailty at the second survey. In both men and women, the prevalence of frailty was lower among the 65–69-year age group than the 60–64-year age group but thereafter increased with age, and a significant difference in prevalence existed between both genders (p > 0.05). The prevalence of pre-frailty was also lower among the 65–69-year age group than the 60–64-year age group and thereafter increased with age in both men and women, but there was no significant difference in the prevalence between men and women.
Comparison of the characteristics of individuals with frailty and pre-frailty to non-frail subjects
Table 1 shows a comparison of patient background characteristics with respect to frailty status. Subjects with frailty tended to be older, female, shorter, lighter, and more emaciated and tended to drink less than the non-frail. In addition, among the subjects with frailty, 44.3% had a concomitant diagnosis of SP, a significantly higher proportion than in pre-frail and non-frail subjects (pre-frail, 26.6%, non-frail, 3.1%; p < 0.001). Similarly, among subjects with frailty, 50.8% had a concomitant diagnosis of OP, which was a significantly greater proportion compared to the pre-frail and non-frail subjects (pre-frail, 38.7%, non-frail, 21.3%; p < 0.001).
Coexistence of frailty, SP, and OP
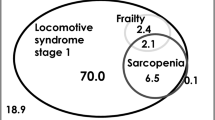
Figure 3 shows a Venn diagram showing the overlap of frailty, SP, and OP in the population aged ≥ 60 years. It reveals that the coexistence of all frailty, SP, and OP was observed in 1.7% of this population; 0.8% had SP and frailty, and 1.2% had OP and frailty. Frailty, SP, and OP, each alone, comprised 1.9, 2.6, and 19.0% of the population, respectively. The remaining 69.5% had no frailty, SP, or OP (Fig. 2).
Eligible subjects to estimate the cumulative incidence of frailty
Among the 1083 participants aged ≥ 60 years in the second survey for the assessment of frailty, 749 (69.2%, 248 men, 501 women) completed all examinations for frailty, SP, and OP (Fig. 1). Among the 749 completers, 20 (4 men, 16 women) had already been diagnosed with frailty at the second survey. Therefore, the number of participants at risk of frailty was 729 (244 men, 485 women).
Cumulative incidence of frailty
The cumulative incidence of frailty during the 4-year period between the second and third surveys was 1.2%/year (men, 0.8%/year; women, 1.3%/year). The cumulative incidence of SP for the total population at risk in the 60–64-, 65–69-, 70–74-, 75–79-, and ≥ 80-year age groups was 0.3, 0.4, 1.5, 2.1, and 2.5%/year, respectively (men, 0.0, 0.5, 0.9, 1.0, and 2.3%/year, respectively; women, 0.4, 0.4, 1.8, 2.9, and 2.8%/year, respectively). The incidence increased with age for all subjects (p < 0.01). There was no significant difference in incidence between men and women (p = 0.21).
Evaluation of the contribution of SP and OP to the occurrence of frailty
Table 2 shows the comparison of subjects’ background characteristics based on the occurrence or non-occurrence of OP during the 4-year follow-up. Subjects with subsequent frailty tended to be older and shorter at the baseline than those without frailty. Moreover, among subjects with subsequent frailty, 11.8% had SP at baseline (p < 0.01, compared to non-frail subjects). Similarly, among subjects with subsequent frailty, 44.1% had OP at baseline (p < 0.001, compared to non-frail subjects).
Logistic regression analysis, using the occurrence of frailty as the objective variable and the presence of SP and OP as the explanatory variables, was performed after adjustment for age, sex (0, men; 1, women), regional differences (0, mountainous area; 1, coastal area), BMI, current smoking habit (0, ex or never smoker; 1, current smoker), and alcohol drinking habit (0, ex or never drinker; 1, current drinker). The analysis showed that the presence of OP was a significant predictive factor for the occurrence of frailty in the following 4 years (odds ratio [OR], 3.07; 95% confidence interval [95% CI], 1.26–7.36; p = 0.012). However, the significant association between SP and the occurrence of frailty was not retained (Table 3).
We assessed the development of frailty with regard to the baseline status of SP and OP. Among 729 subjects in the population at risk, 3.3% of subjects with neither SP nor OP (n = 580) developed frailty during the 4-year observation. Among OP-only subjects (n = 125), 8.8% developed frailty, but among SP-only subjects (n = 7), no individual developed frailty. Finally, of subjects with both SP and OP (n = 17), 23.5% developed frailty during this period. Logistic regression analysis using the occurrence of frailty as the objective variable and the presence of OP alone as the explanatory variable was performed after adjustment for the factors in the abovementioned analysis. The analysis revealed that the risk of frailty significantly increased with the presence of OP and both OP and SP (vs. neither OP nor SP: OP alone; OR, 2.50; 95% CI, 1.04–6.03; p = 0.041; OP and SP; OR, 5.80; 95% CI, 1.38–24.4; p = 0.017). The presence of both SP and OP raised the risk of frailty to a much higher degree, compared to OP alone.
Discussion
In the present study, using information from the second and third surveys of the population-based cohort ROAD, we clarified the prevalence and cumulative incidence of frailty. In addition, we found that the coexistence of SP and OP in subjects with frailty was significantly higher than in those without frailty. In the follow-up for 4 years from the second and third surveys, we estimated the cumulative incidence of frailty. Further, we found that the presence of OP significantly increased the risk of frailty. Although SP did not significantly affect the risk of frailty, the presence of both OP and SP increased the risk of frailty to a much higher degree compared to the presence of OP and SP alone.
Few studies have estimated the prevalence of frailty in the Japanese population. One reason for this is the lack of an established definition of frailty. Therefore, in the present study, we used Fried’s definition of frailty, and the cutoff values of grip strength and walking speed were determined by the AWGS definition of SP [9]. With these criteria, we estimated that the prevalence of frailty in persons aged ≥ 60 years was 5.6% (men, 3.8%; women, 6.6%). Using the age-sex prevalence of frailty converted using the Japanese 2010 census [27], our results indicate that approximately 2,200,000 people (560,000 men and 1,640,000 women) aged ≥ 60 years are affected by frailty.
In the present study, we also clarified the proportion of the coexistence of frailty, SP, and OP in the ≥ 60-year-old population, and 5.6% had frailty, 8.2% had SP, and 24.9% had OP. Among the subjects with frailty, 30.4% had both SP and OP, 14.3% had SP alone, 21.4% had OP alone, and the remaining 33.9% had neither SP nor OP. In other words, among subjects with frailty, approximately one third had SP and OP, one third had SP or OP, and the remaining one third had neither SP nor OP. This reveals that individuals with frailty should be assessed for the coexistence of bone and/or muscle weakness.
Few studies have estimated the incidence of frailty not only in the Japanese population but also worldwide. In the present study, we demonstrated that the cumulative incidence of frailty in subjects aged ≥ 60 years in Japan was 1.2%/year. Using the age-sex incidence of frailty converted using the Japanese 2010 census [27], our results indicate that approximately 490,000 people (140,000 men and 350,000 women) aged ≥ 60 years are newly affected by frailty annually. Nevertheless, there was no significant difference between men and women in prevalence and incidence. However, the number of cases involving women was 2.5-fold greater than that of men.
The logistic regression analysis using the occurrence of frailty as an objective variable and the presence of SP and OP each as an explanatory variable, after adjustment for confounding factors, revealed that the presence of OP significantly increased the risk of frailty within 4 years (p < 0.05). We previously ascertained that the presence of OP could predict the occurrence of SP in the near future via an analysis of the same population [10]. It might not be a surprising result, because in our definition of frailty in the present study, two of Fried’s five items, namely grip strength and walking speed, were determined using the AWGS criteria. That is, two of the diagnostic criteria of frailty and SP overlapped, and all subjects with SP were categorized into the pre-frailty group. The present result might show that the presence of OP was associated with the two overlapping items of SP and frailty. Therefore, we examined the association between the presence of OP and decrease in grip strength and decrease in walking speed. The mean change of maximum grip strength during the 4-year observation period was − 1.29 (SD, 3.24) kg, and the mean change in usual walking speed was − 0.13 (SD, 0.23) m/s. Multiple regression analysis, using the change of grip strength as the objective variable and the presence of OP as the explanatory variable, was performed after adjustment for age, sex, regional differences, BMI, current smoking habit, and alcohol drinking habit, as used in the “Results” section. The analysis showed that the presence of OP was not significantly associated with the change in grip strength in the next 4 years (beta = 0.68, p = 0.098). Similarly, the multiple regression analysis, using the change in usual walking speed as the objective variable and the presence of OP as the explanatory variable, was also performed after adjustment for identical factors to the analysis of grip strength. There was no significant association between OP and neither the degree of change in walking speed during 4 years (beta = − 0.054, p = 0.185). These analyses showed that the presence of OP was not a significantly sensitive predictor for the change in grip strength and walking speed, which were common items of the criteria of SP and frailty. OP might not only influence the overlapped items of SP and frailty, or the 4-year follow-up period might be too short to clarify the association of OP and changes in grip strength and walking speed. Further, since frailty is a cumulation of physical, social, and psychological factors, OP might influence other unknown items which consisted of the social and/or psychological factors for frailty. Further observation should be required to clarify why OP was associated with the occurrence of not only SP, but also frailty.
Nevertheless, we could not find a significant association between the presence of SP and the occurrence of frailty. However, this does not suggest that SP treatment is not effective for the prevention of frailty. As described above, two of the diagnostic criteria of frailty and SP overlapped, which means all subjects with SP were categorized into the pre-frailty group. The logistic regression analysis using the occurrence of frailty as an objective factor and the presence of pre-frailty, replacing SP, as an explanatory variable, after adjustment for identical confounding factors, revealed that the presence of pre-frailty significantly increased the risk of frailty within 4 years (OR, 4.75; 95% CI, 1.99–11.3; p < 0.001). This suggests that among the diagnostic criteria of SP, common items such as grip strength and walking speed could be strong predictors for the development of frailty. The contribution of the remaining criterion, low muscle mass, to the occurrence of frailty might not be significant in the 4-year observation. To conclude on the contribution of SP, not pre-frailty, to the occurrence of frailty, a longer observation is required.
Further, we found that the coexistence of SP and OP increased the risk of occurrence of frailty to a higher degree compared to OP alone. This result also supports the fact that SP management might help prevent frailty. The coexistence of both SP and OP is referred to as “OSP” [28] and has been described as a “hazardous duet,” adding the propensity of falls from SP to the vulnerability of the bones in OP [29]. Because the term OSP is relatively new, few studies have assessed its epidemiology. However, Wang et al., in a study of community-dwelling Chinese older persons aged > 65 years and comprising 164 men and 152 women, found a prevalence of “sarco-osteoporosis” in 10.4% of men and 15.1% of women [29]. They also described that the likelihood of being frail was substantially higher in the presence of sarco-osteoporosis (OR 4.16 in men; OR 4.67 in women) [30]. However, no report has elucidated the association between OSP and frailty. Our present result adds new evidence that the likelihood of becoming frail is substantially higher with OSP.
There are several limitations to this study. First, although the ROAD study includes a large number of participants, the participants in the present study (second survey, individuals from the mountainous and coastal regions alone) may not be representative of the general population. However, previously, we compared the anthropometric measurements and lifestyle factors, such as smoking and drinking habit, between the present study participants and the general Japanese population [10]. We found no significant differences between our participants in the second ROAD survey and the Japanese population, except for the proportion of current smokers and drinkers, which tended to be lower in our study population than in the general Japanese population, suggesting that the participants of the present study led healthier lifestyles. This selection bias should be taken into consideration when generalizing the results obtained from the present study. Second, in this study, the values of handgrip strength for both hands and the 6-m walking test for the calculation of walking speed were measured only once. Thus, we cannot exclude the effect of incidental changes in the participants’ performance around the examination date. To minimize the fluctuation of measurements, recurrent measurements might be taken into consideration. Finally, among 1083 participants in the second survey, because of the non-completion of the measurements regarding the assessments of frailty, SP, and OP in the third ROAD survey, we could only use the information of 749 individuals (69.2%, 248 men, 501 women). This withdrawal bias should also be considered when generalizing our results. Additionally, because the definition of frailty is unestablished, we defined frailty using Fried’s five measurable characteristics. However, the cutoff values of criteria such as walking speed and grip strength are currently undetermined, and we defined them according to the AWGS criteria [9]. A direct comparison seems impossible unless the definitions are unified internationally. We hope the definition of frailty, including the cutoff values, would be unified in the near future to aid studies aimed at preventing disability in older people.
In conclusion, the present study estimated the prevalence and cumulative incidence of frailty and indicated that the presence of baseline OP is significantly associated with the occurrence of frailty. Thus, OP prevention may be useful in reducing frailty risk, as well as osteoporotic fracture and SP risk. Moreover, OSP is predisposed to a higher risk of frailty than OP alone. Both OP and SP preventions may be the most useful means to reduce the development of frailty. To prevent the development of frailty, suitable management of OP or OSP should be established, which needs to be nutritional and physical activity based, considering as well the social and psychological components. We have already collected detailed information regarding the nutrition and physical activity and continue to observe the subjects at long term. In the near future, we would like to clarify the individual risk factors for frailty and contribute to its prevention.
References
Ministry of Health, Labour and Welfare. The outline of the results of National Livelihood Survey 2016. http://www.mhlw.go.jp/toukei/saikin/hw/k-tyosa/k-tyosa16/dl/16.pdf. Accessed 16 November 2017
Fried LP, Tangen CM, Walston J et al (2001) Cardiovascular Health Study Collaborative Research Group. Frailty in older adults: evidence for a phenotype. J Gerontol A Biol Sci Med Sci 256:M146–M156
Rosenberg I (1989) Summary comments: epidemiological and methodological problems in determining nutritional status of older persons. Am J Clin Nutr 50:1231–1233
Rosenberg IH (1997) Sarcopenia: origins and clinical relevance. J Nutr 127:990S–991S
Morley JE, Baumgartner RN, Roubenoff R, Mayer J, Nair KS (2001) Sarcopenia. J Lab Clin Med 137:231–243
Goodpaster BH, Park SW, Harris TB, Kritchevsky SB, Nevitt M, Schwartz AV, Simonsick EM, Tylavsky FA, Visser M, Newman AB, for the Health ABC Study (2006) The loss of skeletal muscle strength, mass, and quality in older adults: the health, aging and body composition study. J Gerontol A Biol Sci Med Sci 61:1059–1064
Delmonico MJ, Harris TB, Lee JS, Visser M, Nevitt M, Kritchevsky SB, Tylavsky FA, Newman AB, for the Health, Aging and Body Composition Study (2007) Alternative definitions of sarcopenia, lower extremity performance, and functional impairment with aging in older men and women. J Am Geriatr Soc 55:769–774
Cruz-Jentoft AJ, Baeyens JP, Bauer JM, Boirie Y, Cederholm T, Landi F, Martin FC, Michel JP, Rolland Y, Schneider SM, Topinková E, Vandewoude M, Zamboni M, European Working Group on Sarcopenia in Older People (2010) Sarcopenia: European consensus on definition and diagnosis: report of the European Working Group on Sarcopenia in Older People. Age Ageing 39:412–423
Chen LK, Liu LK, Woo J, Assantachai P, Auyeung TW, Bahyah KS, Chou MY, Chen LY, Hsu PS, Krairit O, Lee JSW, Lee WJ, Lee Y, Liang CK, Limpawattana P, Lin CS, Peng LN, Satake S, Suzuki T, Won CW, Wu CH, Wu SN, Zhang T, Zeng P, Akishita M, Arai H (2014) Sarcopenia in Asia: consensus report of the Asian Working Group for Sarcopenia. J Am Med Dir Assoc 15:95–101
Yoshimura N, Muraki S, Oka H, Iidaka T, Kodama R, Kawaguchi H, Nakamura K, Tanaka S, Akune T (2017) Is osteoporosis a predictor for future sarcopenia, or vice-versa? Four-year observations between the second and third ROAD study surveys. Osteoporos Int 28:189–199
Yoshimura N, Muraki S, Oka H, Kawaguchi H, Nakamura K, Akune T (2010) Cohort profile: research on osteoarthritis/osteoporosis against disability study. Int J Epidemiol 39:988–995
Yoshimura N, Muraki S, Oka H, Mabuchi A, En-Yo Y, Yoshida M, Saika A, Yoshida H, Suzuki T, Yamamoto S, Ishibashi H, Kawaguchi H, Nakamura K, Akune T (2009) Prevalence of knee osteoarthritis, lumbar spondylosis, and osteoporosis in Japanese men and women: the research on osteoarthritis/osteoporosis against disability study. J Bone Miner Metab 27:620–628
Yoshimura N, Oka H, Muraki S, Akune T, Hirabayashi N, Matsuda S, Nojiri T, Hatanaka K, Ishimoto Y, Nagata K, Yoshida M, Tokimura F, Kawaguchi H, Nakamura K (2011) Reference values for hand grip strength, muscle mass, walking time, and one-leg standing time as indices for locomotive syndrome and associated disability: the second survey of the ROAD study. J Orthop Sci 16:768–777
Arai H, Satake S (2015) English translation of the Kihon Checklist. Geriatr Gerontol Int 15:518–519
No authors listed (1996) Bioelectrical impedance analysis in body composition measurement: National Institutes of Health Technology Assessment Conference Statement. Am J Clin Nutr 64:524S–532S
Janssen I, Heymsfield SB, Baumgartner RN, Ross R (2000) Estimation of skeletal muscle mass by bioelectrical impedance analysis. J Appl Physiol (1985) 89:465–471
Kyle UG, Genton L, Slosman DO, Pichard C (2001) Fat-free and fat mass percentiles in 5225 healthy subjects aged 15 to 98 years. Nutrition 17:534–541
Kyle UG, Genton L, Karsegard L, Slosman DO, Pichard C (2001) Single prediction equation for bioelectrical impedance analysis in adults aged 20–94 years. Nutrition 17:248–253
Roubenoff R, Baumgartner RN, Harris TB et al (1997) Application of bioelectrical impedance analysis to elderly populations. J Gerontol A Biol Sci Med Sci 52:M129–M136
Tanimoto Y, Watanabe M, Sun W, Sugiura Y, Tsuda Y, Kimura M, Hayashida I, Kusabiraki T, Kono K (2012) Association between sarcopenia and higher-level functional capacity in daily living in community-dwelling elderly subjects in Japan. Arch Gerontol Geriatr 55:e9–e13
Tanimoto Y, Watanabe M, Sun W, Tanimoto K, Shishikura K, Sugiura Y, Kusabiraki T, Kono K (2013) Association of sarcopenia with functional decline in community-dwelling elderly subjects in Japan. Geriatr Gerontol Int 13:958–963
Nemoto M, Yasbushita N, Kim M et al (2012) Validity of the bioelectrical impedance method for assessing body composition in non-frail and pre-frail older adults. Int J Body Comps Res 10:225–262
Yoshimura N, Kakimoto T, Nishioka M et al (1997) Evaluation of reproducibility of bone mineral density measured by dual energy X-ray absorptiometry (Lunar DPX-L). J Wakayama Med Soc 48:461–466
World Health Organization (1994) Assessment of fracture risk and its application to screening for postmenopausal osteoporosis. WHO Technical Report Series 843. WHO, Geneva. 843:1–129
Kanis JA, Adachi JD, Cooper C et al (2013) Standardising the descriptive epidemiology of osteoporosis: recommendations from the Epidemiology and Quality of Life Working Group of IOF. Osteoporos Int 24:2763–2764
Orimo H, Hayashi Y, Fukunaga M, Sone T, Fujiwara S, Shiraki M, Kushida K, Miyamoto S, Soen S, Nishimura J, Oh-Hashi Y, Hosoi T, Gorai I, Tanaka H, Igai T, Kishimoto H, Osteoporosis Diagnostic Criteria Review Committee: Japanese Society for Bone and Mineral Research (2001) Diagnostic criteria for primary osteoporosis: year 2000 revision. J Bone Miner Metab 19:331–337
Portal site of Official Statistics of Japan (2010) Population Census 2010. http://www.e-stat.go.jp/SG1/estat/GL08020103.do?_toGL08020103_&tclassID=000001034991&cycleCode=0&requestSender=search [In Japanese]. Accessed on 8th May, 2018
Hirschfeld HP, Kinsella R, Duque G (2017) Osteosarcopenia: where bone, muscle, and fat collide. Osteoporos Int 28:2781–2790
Crepaldi G, Maggi S (2005) Sarcopenia and osteoporosis: a hazardous duet. J Endocrinol Investig 28:66–68
Wang YJ, Wang Y, Zhan JK et al (2015) Sarco-osteoporosis: prevalence and association with frailty in Chinese community-dwelling older adults. Int J Endocrinol 2015:482940
Acknowledgements
The authors wish to thank Dr. Naoki Hirabayashi of Kawakami Clinic, Hidakagawa Town; Mrs. Tomoko Takijiri, Mrs. Rie Takiguchi, Mrs. Kyoko Maeda, Ms. Ikuyo Ueyama, Mrs. Michiko Mori, Mrs. Hisayo Sugimoto, and other members of the public office in Hidakagawa Town; and Mrs. Tamako Tsutsumi, Mrs. Kanami Maeda, Mrs. Megumi Takino, Mrs. Shuko Okada, Mrs. Kazuyo Setoh, Mrs. Chise Ryouno, Mrs. Miki Shimosaki, Mrs. Chika Yamaguchi, Mrs. Yuki Shimoji, and other members of the public office in Taiji Town for their assistance in locating and scheduling participants for examinations. We would also like to thank Ms. Kyoko Yoshimura, Mrs. Toki Sakurai, Mrs. Saeko Sahara, and Mr. Noriyuki Oe for their assistance with data reduction and administration.
Conflicts of interest
None.
Funding
This work was supported by Grant-in-Aid funding from the Ministry of Health, Labour and Welfare: H17-Men-eki-009 (Director, Kozo Nakamura), H20-Choujyu-009 (Director, Noriko Yoshimura), H23-Choujyu-002 (Director, Toru Akune), H25-Choujyu-007 (Director, Noriko Yoshimura), and H25-Nanchitou (Men)-005 (Director, Sakae Tanaka). The study was also supported by Scientific Research grants B26293139, B23390172, and B20390182 and Challenging Exploratory Research grants 15K15219 and 24659317 to Noriko Yoshimura; Scientific Research grants B23390356 and C20591774 and Challenging Exploratory Research grant 23659580 to Shigeyuki Muraki; Challenging Exploratory Research grants 24659666 and 21659349 and Young Scientists grant A18689031 to Hiroyuki Oka; and Scientific Research grants B26293329, B23390357, and C20591737 and Challenging Exploratory Research grant 25670293 to Toru Akune and by Collaborating Research with NSF from the Ministry of Education, Culture, Sports, Science and Technology in Japan (08033011-00262; Director, Noriko Yoshimura). The study was partly supported by grants from the Japan Agency for Medical Research and Development (17gk0210007h0003; Director, Sakae Tanaka). Further, the study was partly supported by grants from the Japan Osteoporosis Society (Noriko Yoshimura, Shigeyuki Muraki, Hiroyuki Oka, and Toru Akune), Japan Osteoporosis Foundation (2015, Noriko Yoshimura), research aids from the Japanese Orthopaedic Association (JOA-Subsidized Science Project Research 2006-1 and 2010-2, Director, Hiroshi Kawaguchi; and 2014-1, Director, Kozo Nakamura), the Japanese Society for Musculoskeletal Medicine (2015, Director, Shigeyuki Muraki; and 2017, Director, Noriko Yoshimura), Mitsui Sumitomo Insurance Welfare Foundation (2016, Director, Noriko Yoshimura), and Japan Dairy Association (2017, Director, Noriko Yoshimura).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yoshimura, N., Muraki, S., Oka, H. et al. Do sarcopenia and/or osteoporosis increase the risk of frailty? A 4-year observation of the second and third ROAD study surveys. Osteoporos Int 29, 2181–2190 (2018). https://doi.org/10.1007/s00198-018-4596-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-018-4596-4